Iso Octanoic Acid: Past, Present, and Paths Forward
Historical Development
You can’t talk about Iso octanoic acid without digging into decades of organic chemistry research, not to mention the practical leaps made during the twentieth century. Long before fine-chemistry labs grew interested in branched-chain acids, early researchers tried to make sense of fatty acids from natural extracts. Chemistry’s old textbooks tell us that octanoic compounds didn’t get their starring role until petrochemical industries and synthetic labs kicked into gear. Once people figured out catalytic processes on branched hydrocarbons, iso octanoic acid moved from a mystery compound to a practical material. I’ve seen libraries stacked with journals logging its presence in countless reactions since the sixties. Each advance opened doors for more reliable preparation, tighter purity controls, and a surge in applications across fields like fragrance, lubricants, and specialty polymers.
Product Overview
Iso octanoic acid stands out as a branched carboxylic acid packed with potential in modern industry. You might catch it under names like isocaprylic acid or 6-methylheptanoic acid. What sets it apart isn’t just its eight carbons or carboxyl group. The unique structure delivers performance properties that straight-chain analogs just can't match. People working with lubricants, fragrances, or even medical intermediates favor these molecular quirks every day, looking for that balance of volatility, solvency, and manageable reactivity.
Physical & Chemical Properties
Iso octanoic acid usually shows up as a colorless to pale yellow liquid with a distinct, sharp scent much like other short-chain carboxylic acids, but with a little less punch thanks to branching. Boiling point tracks higher than more volatile acids, so handling rarely becomes an issue under normal use. Water solubility remains modest – not nothing, not overly troublesome. In the lab, the acid’s carboxyl group pulls its weight, making reactions like esterification or salt formation predictable and reliable. Having handled this acid, I noticed it sticks around on glassware unless cleaned immediately. With a melting point under room temperature, it remains liquid except under cold storage.
Technical Specifications & Labeling
Labs and suppliers sell iso octanoic acid at high purity, packaging it under both chemical and trade names. Labels feature CAS numbers and sometimes include alternate names, depending on the region or distributor. Quality control checks focus on purity percentage, acid value, and the absence of water or volatile impurities. Because impurities can tweak odor and reactivity, strict quality checks make a real difference in consistent performance. From my own work in specialty chemicals, proper documentation always spares users headaches when it comes to traceability or regulatory paperwork downstream.
Preparation Method
No one method dominates, but branched-octanoic acids often come from either controlled oxidation of petrochemical streams or through hydrocarboxylation of heptenes. Catalysts—traditionally cobalt or rhodium complexes—speed things up, letting chemists avoid the tedium of older methods that yielded low conversions or required brutal separations. Industrial-scale reactors handle the exothermic reactions with safety measures in place to prevent runaway heat. During my visits to pilot plants, the effort spent on cleanup steps always struck me—distillation, washing, and drying stacks up, but the payoff comes in reliable, nearly odorless product.
Chemical Reactions & Modifications
Iso octanoic acid steps up in a range of transformations. The classic reaction remains esterification, kicking off the production of esters used for flavors or as synthetic lubricants. Heat plus an alcohol, a dash of acid catalyst, and you end up with an ester that performs above straight-chain competitors due to branching—less oxidation, more stability, lower pour points. Conversion to acid chlorides opens a pathway into further organic synthesis—building blocks for surfactants, pharmaceutical intermediates, or even agricultural chemicals. Amidation with amines generates new compounds with surfactant tendencies. From my own experience modifying carboxylic acids, iso octanoic acid’s branched structure often means lower side-reaction rates and easier purification, especially when clean esters matter.
Synonyms & Product Names
Iso octanoic acid goes by many handles, depending on who’s speaking: you’ll see it called 6-methylheptanoic acid, isocaprylic acid, or even listed under various trade names in catalogs spanning lubricant additives to personal care. In my years combing through chemical catalogs, I learned to cross-reference names and identifiers just to dodge purchase mistakes.
Safety & Operational Standards
Handling any concentrated organic acid calls for sharp attention to material safety. Iso octanoic acid can irritate skin, eyes, and respiratory passages, with volatile vapors causing mild discomfort at low levels. Decent ventilation, gloves, and splash protection belong on every operator’s checklist. Industrial users store this acid in tight-sealed drums, away from bases and oxidizers to prevent unwanted reactions. In my workplace, regular training made sure spills never went ignored, and everyone knew how to keep acids from contaminating work areas or drainage. Regulations align with broader carboxylic acid standards—nothing exotic, but skipping precautions leads to predictable troubles.
Application Area
Iso octanoic acid finds favor where molecular quirks shape product performance. Lubricant makers look to it for oxidation-stable additives that last longer under stress. Fragrance chemists lean on this acid for specialty esters that bring out fruity or fatty notes, without the choking pungency seen in simpler acids. Some plasticizer manufacturers seek its branched backbone to make flexible, low-volatility compounds for specialty polymers. Personal experience in a plastics lab showed how tweaking acid structure could shift final resin performance. Researchers developing biocides and functional fluids harness iso octanoic derivatives when straight-chain versions deliver the wrong solubility or break down too quickly.
Research & Development
Across countless journals, you'll find ongoing work to upgrade preparation methods and expand iso octanoic acid’s applications. Catalytic process development stands out, with scientists sharpening selectivity and boosting yields by tuning ligands, metals, or even reactor design. Academic labs continue exploring new esters, amides, and acid chlorides of iso octanoic acid, searching for compounds with specialized antifungal, antibacterial, or lubricating properties. My own discussions with chemical engineers pointed to a rising interest in renewable feedstocks—upcycling waste hydrocarbons, using bio-based catalysts, or even harnessing fermentation routes to sidestep petrochemicals.
Toxicity Research
Most animal and cell studies suggest iso octanoic acid shares low acute toxicity and minimal chronic effects with other mid-length branched carboxylic acids. Nonetheless, careful review shows that concentrated doses could stress cell membranes or disrupt sensitive tissues. Occupational users get reminders to limit repeated skin and eye contact, and regulators keep an eye out for new data, especially as production volumes rise or applications spread into food, medical, or cosmetic products. As a chemist, I respect those guidelines because even familiar compounds can cause rare allergic, respiratory, or environmental effects when oversight slips.
Future Prospects
The road ahead for iso octanoic acid feels promising—growing demand for premium lubricants, advanced polymers, and eco-friendly additives lines up with the acid’s unique abilities. I noticed that more companies are casting around for bio-based sources, aiming to cut carbon footprints using green chemistry. By plugging into bio-refinery systems or engineered microbial pathways, iso octanoic acid could pivot away from fossil fuels and land in consumer products labeled as sustainable. In R&D circles, new ester and amide derivatives point toward fields like medical devices and electronics, where stability trumps basic function. After years watching specialty chemicals drift from lab innovation to industrial mainstay, I’ve learned that practical advances in process safety, purification, and performance testing will decide just how far iso octanoic acid travels in the years ahead.
What is Iso Octanoic Acid used for?
Everyday Products Rely on Iso Octanoic Acid
Most people never stop and think about what helps keep things working smoothly, smelling pleasant, or staying durable. Iso octanoic acid, a branched fatty acid with eight carbon atoms, pops up in a surprising number of products that millions of people use daily. From car engines to cosmetics, its impact reaches much further than a chemist’s counter.
Additives and Lubricants: Protecting What Matters
Take a look under the hood of an average car or a piece of heavy machinery. You’ll notice that clean, efficient operation isn’t just about good engineering; it’s also about the small things, like the right additives in fuels and lubricants. Iso octanoic acid acts as a corrosion inhibitor and lubricant booster. Tests have shown that its structure helps form strong protective layers on metal surfaces, cutting down rust and friction where those problems can chew through parts and cut the life of expensive equipment short. Factory workers and plant technicians count on these additives to keep production lines running day in and day out.
Personal Care: Smoother, Safer, Longer-Lasting
People looking for smoother skin or hair that shines often scan ingredient lists for familiar words. Iso octanoic acid sometimes appears under less technical names as an emollient and conditioning agent. It blends well into creams and lotions, creating that slick feeling people want from moisturizers and sunscreens. Dermatologists care about safety—so do users. Research into this acid points to a very low rate of skin irritation, setting it apart from harsher chemicals that can trigger flare-ups. Companies keep looking for alternatives to mineral oils and petroleum bases, driving up demand for ingredients like this one.
Fragrances and Flavors: Subtle Roles, Big Impacts
I’ve always noticed how the smallest tweaks in a recipe can change everything—one drop of essence and suddenly a dish shifts. Iso octanoic acid works just like that in fragrances and flavorings, adding depth or rounding off sharp notes with just a small addition. Food scientists rely on its ability to enhance richness, and perfumers find it invaluable for creating balanced, inviting scents. Its molecular shape lets it bind with other aroma compounds, making a scent last longer or a flavor linger on the palate.
Polymer and Plastic Production: Building Better Materials
In materials science, controlling the quality of plastics and resins is a game-changer. Iso octanoic acid steps in as a monomer or modifier to improve flexibility, resistance to cracking, and workability. Manufacturers crafting automotive interiors, gadget casings, and specialty packaging benefit from its inclusion. Tough testing standards have shown that plastics modified this way stand up to heat and pressure, outperforming older alternatives. The growing push for durable, high-quality plastics in consumer products draws more attention to ingredients like iso octanoic acid.
Looking Ahead: Responsible Use and Environmental Impact
Heavy use of chemicals in manufacturing often stirs up questions about safety and environmental footprint. Researchers are already studying the breakdown products of iso octanoic acid in wastewater and soil, aiming to make sure its benefits don’t come with hidden risks. Responsible sourcing from renewable feedstocks and improved waste management take on fresh urgency as the chemical industry shifts away from fossil fuels, hoping to cut pollution and safeguard health. People who work in the labs and on the shop floor all know that the small molecules, used wisely, can shape a safer, cleaner future.
What are the main properties of Iso Octanoic Acid?
Understanding the Structure
Iso octanoic acid, sometimes called 6-methylheptanoic acid, turns up in the world of chemistry as a straight-forward, branched chain fatty acid. The formula goes by C8H16O2, and the structure throws in a methyl group on the sixth carbon. This little side branch changes the way it acts compared to the simple, straight-chain octanoic acid you might see elsewhere. As someone who's handled fatty acids in a lab, you can catch a slight, distinct smell as you open the bottle—reminiscent of the mix between old paint and faintly rancid butter.
Physical and Chemical Properties
Most of the properties come from its branched structure. You pour out a colorless to slightly yellowish liquid, depending on purity, which stays liquid at room temperature. Iso octanoic acid melts at a temperature lower than the straight kind, settling close to -30°C, and boils somewhere above 170°C. This change in melting point, mainly due to the branches, changes how the molecules pack together, so sheets of the stuff don’t freeze up as easily. It dissolves poorly in water but goes right into ether or alcohol, a fact that comes in handy if you ever need to use it as a solvent or reactant.
What Does This Mean for Industry?
The car industry often looks at iso octanoic acid as a blending agent for lubricants and coolants. That lower melting point prevents it from gumming up engines in cold weather. In paint manufacturing, it slips into alkyd resins—these resins form the backbone of quick-drying paints. From what I’ve seen on the job, replacing regular octanoic acid with the iso-form creates finishes that don’t yellow as quickly and stand up better to humidity. So, everyday products that get exposure to moisture last longer and look better.
Health and Environmental Concerns
Working with iso octanoic acid doesn’t require panic, but it pays to stay careful. Skin contact brings mild irritation after a short stint, so gloves go on right away. Inhalation, not so much; the smell itself warns you off, though good ventilation matters during large-scale processing. The chemical has a moderate tendency to stick around in the environment, and it resists breaking down in soil a bit more than simple organic acids. This can raise questions on waste management and disposal. As a solution, closed-loop systems and chemical recycling help lower the environmental risk. I’ve seen companies take waste streams of fatty acids and feed them into recovery plants, reusing the acids instead of burning them or dumping them as waste. That cuts pollution and shrinks costs.
Scientific and Technical Importance
Researchers often use iso octanoic acid to explore how branching in molecules affects behavior, especially when modeling how different fatty acids perform in living systems. Protein-lipid interactions sometimes shift dramatically when you introduce a branch, and iso octanoic acid serves as a reference molecule for these experiments. In testing synthetic routes, chemists use it to probe selectivity in reactions—results with this acid sometimes point toward better catalysts or more efficient conversions among related compounds.
Looking Forward
The properties of iso octanoic acid give it a spot in chemical manufacturing, automotive applications, and even as a tool for researchers. To keep the benefits while reducing risks, the focus on better recycling, improved handling, and real-time environmental monitoring will probably only become stronger. From my experience, staying proactive on these fronts keeps operations safer, greener, and more profitable in the long run.
Is Iso Octanoic Acid safe to handle?
Understanding the Substance
Iso octanoic acid, also called isooctanoic acid, turns up in a lot of production environments. From making lubricants to tweaking the formula in some paints and coatings, this branched-chain fatty acid shows up more often than people realize. You’d think something that pops up in so many products would seem routine. Still, anyone who handles chemicals needs facts, not assumptions, about the risks.
Where Problems Can Start
On the surface, iso octanoic acid doesn’t set off alarms like sulfuric acid or liquid ammonia. It isn’t known for heavy fumes or explosive reactions. Its flashpoint is relatively high, and it doesn’t eat through clothing or skin in seconds. That level of familiarity has a downside. People tend to get loose with their safety habits around chemicals they don’t see as “really dangerous.” My time working on shop floors taught me to spot the slow-build dangers. People forget their gloves. Spills languish until someone steps in them. Goggle use falls by the wayside on hot days. But even mild-mannered chemicals leave a mark if they get onto skin or in the air.
Health Effects and Safety Data
Take a closer look at the material safety data sheets (MSDS) for iso octanoic acid. Touching the liquid often leads to skin irritation — redness, itching, a rash if left on too long. Eyes sting and water if exposed. Breathe in enough of the vapor, especially in closed rooms, and expect headaches or coughing. Chronic exposure, which comes from regular skin contact or poor ventilation, increases the risk of dermatitis. Scientists and safety engineers have found that repeated exposures leave behind more stubborn reactions. A younger version of myself learned this the hard way after cleaning up a spill barehanded. My hands burned for days.
What the Experts Say
The American Conference of Governmental Industrial Hygienists and similar groups generally place iso octanoic acid in the “irritant” category, not the “highly toxic” category. But “not highly toxic” doesn’t mean harmless. Low risk adds up when workspaces treat it casually. No one tracks the long-term exposure numbers as closely as with lead or benzene. That gap in data sometimes leads teams to skip over basics — such as eye protection with tight seals or checking ventilation flow rates.
Practical Steps for Safer Handling
I’m a firm believer that setting up the right habits beats trying to “be careful” on the fly. Rigid gloves, splash goggles, and chemical-resistant aprons should just sit at the door of any area where iso octanoic acid moves around, whether drum storage or a hobbyist’s bench. Labeling all storage containers and keeping spill kits in plain sight helps everyone catch problems sooner. Any workspace that works with iso octanoic acid needs enough air changes per hour to keep vapor levels from creeping up — a tip learned from seeing too many shops with tired old fans.Never pour down drains or leave open containers. Accidental mixing with incompatible chemicals rarely ends well, even with simple acids like this one. Training new staff with hands-on spill drills and always using MSDS sheets as reference material can reduce panic if something does go wrong.
Responsibility in Use
Risk in a work zone or garage rarely comes from a single event — it’s the repeated lapses that bite back. Personal protective equipment, good habits, and strong ventilation each thumb the scale back toward “safe.” Watching out for small burns or rashes might seem excessive for a routine chemical, but long experience says the effort pays off in fewer missed days and less drama. Those small steps build trust and help keep shops running without a hitch. That’s the real return on handling iso octanoic acid smartly.
What is the chemical formula of Iso Octanoic Acid?
Understanding Iso Octanoic Acid
Iso octanoic acid draws a lot of attention from both chemists and those working in manufacturing. Its chemical formula, C8H16O2, sounds simple, but this little chain brings much more to the table than just eight carbons, sixteen hydrogens, and two oxygens. My first run-in with iso octanoic acid happened during a project on specialty lubricants, where choosing the right chain length made all the difference in performance. An extra methyl group, the small branch that distinguishes the "iso" structure from normal octanoic acid, seems like a minor tweak, but it changes the way molecules pack and behave.
The Value Behind the Formula
Chemical structure and formula matter in the real world of manufacturing and product design. To someone mixing a batch of synthetic flavors or working on an engine oil, it means more than just textbook chemistry. Chemical suppliers make sure to specify "iso" or "normal" because before you know it, the smallest change in molecular layout alters the aroma in food, the solvency in paint, or how smoothly a machine runs in harsh conditions. It affected my project’s outcome when the wrong isomer slipped into a prototype—suddenly, lubricity dropped, and we had to trace it back to a mix-up in the acid type listed on the documentation.
Real-World Risks and the Need for Precision
Mixing up iso octanoic acid with its straight-chain cousin doesn't just mess with flavorings or machine oils. I once spoke with a colleague at a fragrance company who saw a whole product line pulled off shelves because mislabeling led to unintentional scent changes. It cost not just money but also eroded customer trust. These stories drive home the point: knowing the exact formula, C8H16O2, keeps the science on track and avoids costly surprises.
Safety and Handling
Behind that formula sits a real set of hazards. A strong, sour smell announces its carboxylic acid group, signaling the need for care in the lab. Any technician or student who’s splashed it on their skin knows the sting and quick scramble for the sink. OSHA and safety data sheets show that iso octanoic acid can be irritating if inhaled or touched. In my lab years, handling it meant gloves and eye protection, with a clear protocol for spills, not just to tick boxes, but to keep everyone healthy. That’s not just being cautious — it’s essential knowledge rooted in the exact identity of the compound we’re working with.
Building a Case for Chemistry Education
It’s easy to skim past chemical names and formulas, but each tells a story about the nature of a compound and its uses. Whether it’s about avoiding product recalls, keeping the lab safe, or pushing for new innovations, knowing the makeup of iso octanoic acid helps real people make real decisions. This kind of knowledge supports the integrity not only of science but also of the marketplace and public health. In every bottle, can, or drum that moves from lab to market, that explicit confidence in chemical identity protects brands, workers, and end users alike.
How should Iso Octanoic Acid be stored?
Lessons from Real-World Experience
Walking through the back room of a chemical warehouse, you quickly learn which acids demand more respect. Iso octanoic acid sits high on that list. Its strong odor and reactivity grab your attention fast, especially once you've seen drums start sweating from heat or watched the label print start to peel. People handling this chemical aren’t just following obscure rules. They’re protecting themselves, their co-workers, and the company’s bottom line.
The Risks Are Real, Not Theoretical
Iso octanoic acid doesn’t reach toxic status quite as fast as some industrial acids, but its low flash point and tendency to corrode many plastics and metals mean one careless storage decision can ruin valuable stock in just a few days. Cases exist where sloppy handling led to fire hazards or leaks that forced costly hazmat cleanups. No one working the loading dock forgets what it feels like to see a drum balloon out because of a temperature spike. Even a small spill leaves behind an unmistakable stench and a messy headache for the whole team.
Temperature Control Always Comes First
Storage temperature makes or breaks safety with iso octanoic acid. Once, a facility in the southwest kept drums outside during a hot summer. By noon, the acid’s low flash point—only 86°C—brought vapor concentrations near the explosive range, just from sitting in the sun. Keeping drums and containers in a shaded, temperature-controlled area isn’t optional. Even in temperate climates, fluctuating temperatures can weaken seals and allow vapors to escape inside a warehouse.
Choosing The Right Materials
Many make the mistake of pouring iso octanoic acid into random containers to save space. The reaction with mild steel or some types of HDPE eats away at the lining before you realize it. Stainless steel and specific grades of HDPE are trusted by professionals for a reason. Lab techs often spot early leaks and advise on the right container choice. In the field, you learn quickly that reusing drums without verifying compatibility turns into an expensive lesson. Don’t trust luck—double-check the drum specifications before pouring or storing a single liter.
Ventilation Matters As Much As The Drum
A slot in a warehouse’s corner doesn’t count as adequate storage for iso octanoic acid. Good airflow keeps dangerous vapors from accumulating, especially if a seal starts to fail. In some shops, workers tape notes over vents to block dust, only for fumes to gather and trigger alarms. Investing in mechanical ventilation—not just a cracked window—pays off every year in avoided sick days and fire marshal visits.
Labeling and Emergency Plans Count Daily
No one in any serious operation dismisses labeling. Emergency crews, employees, and auditors rely on those details. Too many skip full hazard labels, figuring everyone knows what’s inside. But real-world confusion leads to bigger errors. Having spill kits within reach, PPE at the ready, and exits clearly marked saves real lives every year. Posting emergency numbers and outlining procedures right by the storage site means no one faces a crisis alone or unprepared.
Treat Iso Octanoic Acid Like A Priority
Keeping iso octanoic acid in top condition takes more than meeting minimum code. From the long hours in shipping to a lab bench, those who work with this acid daily insist on strict storage rules. Respect for the material keeps small mistakes from becoming expensive accidents. Seasoned staff don’t just follow best practices—they enforce them. That’s what keeps people safe.
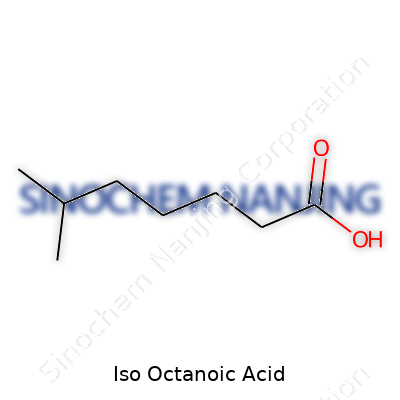
| Names | |
| Preferred IUPAC name | 2,2,4-Trimethylpentanoic acid |
| Other names |
Isooctanoic acid Isooctylic acid 7-Methylheptanoic acid 2-Ethylhexanoic acid |
| Pronunciation | /ˈaɪsoʊ ɑkˈteɪnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 25103-52-0 |
| 3D model (JSmol) | `Iso Octanoic Acid` JSmol 3D model string: ``` CCCC(C)CC(=O)O ``` |
| Beilstein Reference | 1909980 |
| ChEBI | CHEBI:30775 |
| ChEMBL | CHEMBL502380 |
| ChemSpider | 18304 |
| DrugBank | DB14185 |
| ECHA InfoCard | ECHA InfoCard: 03-2119457554-34-XXXX |
| EC Number | EC 203-051-9 |
| Gmelin Reference | 10417 |
| KEGG | C08360 |
| MeSH | D008426 |
| PubChem CID | 10469 |
| RTECS number | RH0700000 |
| UNII | 0YT8W1826I |
| UN number | UN3265 |
| Properties | |
| Chemical formula | C8H16O2 |
| Molar mass | 130.19 g/mol |
| Appearance | Clear, colorless to pale yellow liquid |
| Odor | Pungent |
| Density | 0.92 g/cm³ |
| Solubility in water | Insoluble |
| log P | 3.05 |
| Vapor pressure | 0.013 mmHg (25°C) |
| Acidity (pKa) | 4.85 |
| Basicity (pKb) | 9.31 |
| Magnetic susceptibility (χ) | -7.36 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.415 |
| Viscosity | 30 mPa·s (25°C) |
| Dipole moment | 1.689 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -530.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5100 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | J63AX15 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P264, P270, P280, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 1-2-0-腐 (Acid) |
| Flash point | 112 °C |
| Autoignition temperature | 420 °C |
| Lethal dose or concentration | LD50 (oral, rat): 2900 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3600 mg/kg (oral, rat) |
| NIOSH | DGK |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Iso Octanoic Acid: Not established. |
| REL (Recommended) | 410 mg/m³ |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
Formic acid Acetic acid Propionic acid Butyric acid Valeric acid Caproic acid Enanthic acid Caprylic acid Pelargonic acid Capric acid |

