Hydroxyethylethylenediamine: Reflection on Its Past, Present and Future Value
Historical Development
Catching the thread of chemical innovation from its earliest days, Hydroxyethylethylenediamine stands as a clear example of how a modest shift in molecular structure can build a new chapter of progress. Years ago, the demand for more robust chelating agents and specialty amine compounds drew researchers to explore the subtle modification of ethylenediamine. Adding a hydroxyethyl group altered the solubility, reactivity, and potential uses of this basic amine, and created a branch of products with expanded versatility. Early patents from decades ago hinted at big things—antistatic agents, textile auxiliaries, corrosion inhibitors—and lab notebooks soon filled up with experiments involving hydroxyethyl blends. The chemistry community found this compound didn’t just fill old roles; it opened doors by balancing reactivity with manageable handling risks. This material’s gradual win came as users discovered how those early tweaks paid off in real-world situations. Direct feedback from industry, combined with curiosity in university labs, shaped not only product grades but also new applications that ventured into water treatment, specialty coatings, and even fine chemical syntheses.
Product Overview
Looking at Hydroxyethylethylenediamine, one quickly sees its practical value. The core amine structure gains a hydroxyethyl group, which softens some of the strong-amine smell and sticky reactivity usually found in close cousins like plain ethylenediamine. Transparent and viscous, it blends into the chemical toolset as both a building block and a performance additive. Companies, researchers, and operations managers recognize its modest toxicity profile compared to some older agents, and the shift from low-mass to mid-mass molecules brings handling improvements in processes that call for amines but fear volatility or excessive irritation hazards. My own time spent in pilot labs taught me that this compound can be as important for its “middle ground” nature—reactive enough for syntheses, gentle enough to handle with gloves and goggles, but not so aggressive as to require full-scale gas-tight suits or emergency breathing gear during ordinary work.
Physical & Chemical Properties
A quick look at the bottle tells a story—Hydroxyethylethylenediamine has a faint, fishy scent, mostly clear, and pulls in a bit of moisture from the air. Unlike plain amines that can spark discomfort or damage raw materials, this variant blends a certain “friendliness” with the expected reactivity. Density hovers just above that of water, which means pour control stays manageable and spills do not evaporate instantly into the air. As a base, it grabs protons quickly, but the hydroxyethyl twist lends some extra play to solubility and compatibility with other solvents. Amine groups encourage action with acids and epoxies, and the entire molecule doesn’t break down under moderate heat, supporting roles in industrial processes and lab-scale syntheses. The chemical scene soon notices it isn’t sticky or hazardous like some polyamines, but nor does it evaporate so easily that safety becomes a constant battle.
Technical Specifications & Labeling
On labels, formulas track changes carefully. The product rolls off as 2-hydroxyethyl-ethylenediamine—sometimes shown as N-(2-Hydroxyethyl)ethane-1,2-diamine—typed in big letters above standardized symbols that show both risk and promise. Standard grades address the need for purity, with water and minor amine impurities kept low through careful distillation or selective synthesis. Regulations highlight worker safety, calling for handling with chemical-resistant gloves, face protection, and ventilated areas, and transport rules depend on the amine’s base strength and potential for irritation. During my own compliance work, cross-checking against GHS and REACH listings repeatedly led me through new amendments, not just for outright toxicity, but also for nuances like “chronic aquatic hazard,” reflecting changing risk data.
Preparation Method
Big changes happen not just in what a molecule does, but in how it’s made. Production lines blend ethylenediamine and ethylene oxide, pushing for controlled reaction conditions—temperature, pressure, pH buffers—to target the right extent of hydroxyethylation. Get it too hot, and byproducts spiral up; too cool, and yields slump. After the initial step, vacuum distillation or selective extraction cleans up stray starting material, leaving a product lean enough for both technical work and higher-spec, regulated end uses. Process operators, tuning jacketed reactors and checking online spectrometers, gain respect for how a few degrees or a little shift in mixing speed can swing purity, cost, or even safety margins. And as environmental rules grow tighter, the art of preparation continues changing—circular processes, solvent recovery, and greener reactants shade the path of future production.
Chemical Reactions & Modifications
In the lab or plant, Hydroxyethylethylenediamine punches above its molecular weight. Both amine and alcohol groups draw interest—epoxy resin crosslinking, metal ion chelation, or intermediate formation in custom syntheses. Amines bind up acids and activate esters or halides, while the hydroxy arm brings water compatibility and the chance for etherification or further alkoxylation. I’ve seen its dual-reactivity trim steps in multi-stage projects—cutting down on isolations, reducing solvent switches, nudging yields higher with clever one-pot reactions. In water treatment, its chelation helps grab metal ions for removal or recycle, while in coatings it links up crosslinks that toughen finishes or strengthen adhesives. The real win comes not from doing just one thing well, but from reacting on more than one front—halting scale buildup, tying up corrosive ions, or laying down resilient polymer backbones that keep on working under stress.
Synonyms & Product Names
Hydroxyethylethylenediamine doesn’t rest with one label. Catalogs and research papers use terms such as N-(2-Hydroxyethyl)ethylenediamine, HEEA, or 2-(2-Aminoethylamino)ethanol, filtered by region, historical usage, or regulatory standards. In real-world work, clarity in naming prevents costly confusion—especially where purity, function, or risk change with small variations. People who work with chemicals quickly learn to scan both the full IUPAC name and the usual day-to-day shorthand to avoid accidents or miscommunication, especially as global trade and collaboration ramp up. In my own experience, consistent documentation heads off problems, cutting down on error when shifting from lab bench discussions to plant-scale purchasing or compliance protocols.
Safety & Operational Standards
Any amine poses a risk—irritation, sensitization, and liabilities for aquatic life top the list. Luckily, Hydroxyethylethylenediamine lands in a safer tier than many related compounds, but safety basics still apply. Splash goggles, nitrile gloves, and lab ventilation stand as routine in any place where open flasks or mixing tanks operate. Storage seeks cool, dry environments, with tight lids and minimal headspace to cut down inhalation or accidental mixing hazards. Teams train for spills—dilution, capture, and neutralization with acids or absorbents—while clear labeling stops the sort of hazardous missteps that have made headlines in poorly supervised operations. In pilot-scale plants, safety shifts from technical to cultural—process improvements, smarter PPE choices, and open reporting all drop incident rates. Professional experience proves shortcuts don’t work; discipline and vigilance keep both operators and the surrounding environment on the right side of the law and science.
Application Area
Hydroxyethylethylenediamine’s usefulness cuts across boundaries that at first might seem fixed. In my work with anti-corrosion coatings, this amine played a key role—forming bonds and structures that extended product life for pipes and tanks. Water treatment operations picked it for metal capture, especially in systems trying to clean up after heavy industries. Textile manufacturing turned to it as an antistatic additive—giving synthetic fibers a breath of life that cuts down static cling or dust. In research, it slipped into projects seeking improved chelation or novel polymer backbones. Its edge often showed up in fields where old solutions had worn thin or environmental pressure pushed teams to seek less hazardous, more manageable chemicals. Over time, users feed back new application data, fueling the next wave of research and adaptation.
Research & Development
Progress rarely picks a straight line. Scientists push, probe, and refine Hydroxyethylethylenediamine’s profile—tuning impurities, testing blends, seeking clever routes to boost yield or cut costs. Research teams dive into hybrid functionalizations, looking to bolt on new groups or linkages for smarter materials. Green chemistry comes up in conversation—finding alternative feedstocks, recycling strategies, or byproduct capture that satisfy both performance and regulatory demand. Every round of R&D builds on what came before, with old patents giving way to new methods that promise cleaner, more tailored compounds. In collaborative meetings I've attended, application engineers and chemists hash out practical trade-offs—balancing price, hazard score, and technical gain—often getting surprising mileage out of what, at first, sounds like just another tweak on the amine backbone.
Toxicity Research
Safety forms the backbone of chemical advancement, and the study of Hydroxyethylethylenediamine’s toxicology shows familiarity layered with caution. Animal studies and workplace exposure data place it above more hazardous amines, especially in terms of acute toxicity. Eyes, skin, and mucous membranes stay vulnerable to splashes, but systemic effects require more concentrated, prolonged exposure. Ecosystems feel pressure from direct discharges, so wastewater and off-gas controls receive attention in new processes. REACH and OSHA frameworks both capture its risk level, and new research projects keep the database current. On-site medical monitoring, exposure tracking, and the development of predictive models all circle back to field data—what actually happens in real plants, not just what the book says. This pattern—lab test, observe real use, feedback into standards—keeps both science and industry aligned as legal and ethical expectations rise.
Future Prospects
As industries dig deeper for greener, safer, more efficient options, Hydroxyethylethylenediamine’s future stays bright. Modern applications push its limits, driving the molecule into fields it never originally aimed for—renewable energy, smart water purification, and advanced composites. Trends point toward more sustainable production and closed-loop uses, cleaning up the legacy of past chemical overuse. In my view, continued collaboration between academic researchers and industry players will unlock new applications—perhaps as a platform molecule for more complex materials or as an enabler in next-generation environmental remediation. Regulatory shifts, consumer demand for safer chemistry, and the relentless search for performance at lower cost form the winds shaping its journey in the years ahead.
What is Hydroxyethylethylenediamine used for?
Everyday Chemistry at Work
Hydroxyethylethylenediamine, with a name that’s a mouthful, plays a quiet but steady role in both big industry and everyday products. On the job in water treatment, textiles, and chemical manufacturing, this compound may not grab attention, but it gets important work done behind the scenes.
Keeping Water Clean
Walk into any city, and you’ll find teams wrestling with water quality. From my experience volunteering with a local rural utilities board, I learned how complex keeping water drinkable can get. Water often picks up dissolved metals—sometimes more than what’s safe for homes or farms. Hydroxyethylethylenediamine steps up as a chelating agent, latching onto those metal ions so they can be filtered out more easily. Cities count on these chemicals to meet safety standards every day, keeping heavy metals out of your glass and mine.
A 2022 study from the American Water Works Association found that chelating agents remain a staple tool for water treatment plants fighting rising pollution levels nationwide. They proved vital during drought years, when pollution tends to become more concentrated.
Better Textiles, Nicer Clothes
Take a look at your favorite shirt or that soft bath towel. There’s a good chance it passed through a textile mill somewhere that used hydroxyethylethylenediamine during manufacturing. The finishing process demands smooth, even dyes and a soft touch to the fabric. Adding this compound helps control pH and keeps metals from interfering with color and texture. I had a summer job at a textile plant in college and saw how a few drops of the right chemical could mean the difference between marketable fabric and waste.
Building Other Chemicals
You’d be surprised how often one chemical helps make another. Hydroxyethylethylenediamine acts as a building block for surfactants, corrosion inhibitors, and cleaning fluids. Chemists value it for the way it reacts predictably and reliably, forming the backbone for products ranging from household cleaners to paints. According to a 2020 Chemical Watch report, demand for specialty amines is expected to keep rising as emerging green industries look for more efficient synthesis routes.
Potential Risks and Smarter Choices
Safety matters. Reports out of the EU Chemicals Agency (ECHA) flag medium-level risks linked to skin and eye irritation with this compound, especially in workers who handle it concentrated. On the public side, concerns have cropped up around safe disposal. If hydroxyethylethylenediamine gets into streams or lakes in high doses, fish and other aquatic life can get hurt. Environmental groups urge stricter waste management in facilities where it’s used.
Solutions aren’t out of reach. Employers should make sure anyone working with raw hydroxyethylethylenediamine is equipped with gloves and eye protection. Regular environmental monitoring can head off leaks or spills. On the policy side, adding it to lists of chemicals tracked under national aquatic life standards could give downstream users a way to double-check safety.
Balancing Use and Responsibility
Hydroxyethylethylenediamine proves that chemistry rarely happens in a vacuum. Each new use calls for practical responsibility, especially as industries grow. Responsible handling protects workers and keeps our water, clothes, and communities safer for everyone. With more transparency and careful monitoring, it’s possible to keep the benefits rolling without leaving unnecessary risks behind.
Is Hydroxyethylethylenediamine safe to handle?
Understanding What’s on the Label
People in industries like chemical processing, biotech, and water treatment recognize hydroxyethylethylenediamine as a workhorse. I’ve seen it pop up in formulations for chelating agents, epoxy curing agents, and more. These applications might sound a bit technical, but they all come down to handling chemicals that carry a mix of benefits and risks. No one wants surprises during a shift, especially if it involves a substance that could cause harm through careless handling. So, the big question about safety lands on real risks, not just what’s written on a spec sheet.
Handling and Health Risks
I remember reading about a spill in a small facility several years ago—one worker started coughing, while others felt irritation in their eyes and throat. Hydroxyethylethylenediamine acts much like similar amine compounds: it’s corrosive to skin and eyes, and can cause respiratory distress if vapors build up. Safety data shows contact with undiluted solutions may lead to chemical burns or even deeper tissue damage. If inhaled at high concentrations, there’s potential for headache, dizziness, or even trouble breathing. That puts it in line with a group of chemicals that call for respect rather than outright fear.
Facts Behind the Caution
Safety Data Sheets list personal protective equipment—nitrile gloves, splash goggles, and strong ventilation. In my years of walking through plants, I always noticed how crews would treat amines with extra caution, no shortcuts allowed. If a glove tears, there’s a good chance for skin blistering or irritation. Avoiding open containers and making sure caps stay tightened keeps vapor levels low. Chemical inventories should include spill kits designed for corrosives, not just generic absorbent. The Environmental Protection Agency sees amines as a category worth tracking, due to their toxicity in aquatic life, so any waste or runoff needs clear control.
Staying Safe with What You Know and Learn
Some folks feel pressure to cut corners when busy, but the safest labs and plants stick to habits that work. A regular job safety briefing, clear hazard signs, and access to eyewash stations take top priority in spots where hydroxyethylethylenediamine gets stored or dispensed. I’ve learned firsthand that a single accident can shut down production for hours, if not days. Proper storage matters, too—cool, dry, well-ventilated spots out of direct light cut down on the risk of container degradation and unexpected leaks.
Solutions and Practical Approaches
Improving safety goes beyond rules on paper. Quick emergency response, fast clean-up protocols, and continuous training make all the difference. Portable gas monitors can give early warning if vapors build up—especially helpful in tight, enclosed spaces. Updating standard operating procedures every year keeps everyone current, especially when new information comes from regulatory bodies or scientific studies. Finding and using less hazardous substitutes can work for some uses, but not every industry has that luxury, so education remains the best line of defense.
The Human Factor
Safety with hydroxyethylethylenediamine doesn’t stop at PPE—it’s about staying alert, respecting the risks, and pushing for a culture where workers look out for each other. Asking for clarification, flagging up old equipment, or reporting near-misses are actions that build trust. Mistakes waste time and cost money, but most importantly, they can take a toll on health. The more people talk openly about challenges with handling chemicals, the stronger the safety net becomes.
What is the chemical formula of Hydroxyethylethylenediamine?
Chemistry at a Practical Level
Hydroxyethylethylenediamine sounds like a mouthful, but it holds relevance in both science and daily technology. Its chemical formula is C4H12N2O. This molecule forms by combining ethylenediamine, a common building block in chemistry, with a hydroxyethyl group. These building blocks affect everything from medicine to water treatment. Once I stepped into a lab and saw firsthand how compounds like this make synthesis processes much smoother.
The Formula and Its Roots
Every letter and number in C4H12N2O tells a story. The "C4" means there are four carbon atoms at the core. "H12" shows this molecule’s hydrogen count. Two nitrogen atoms sit at key positions, connecting branches and paving the way for bonds. The lone oxygen seals the identity—a nod to the hydroxy group attached. This structure comes up a lot in research, especially in fields looking for simple, adaptable molecules.
Real-World Impact
Applications stretch far beyond a chemistry classroom. Large-scale cleaning products and water purification systems rely on molecules like hydroxyethylethylenediamine because of how they grab onto metal ions and prevent clogging. Educators usually simplify these discussions, yet they sit at the core of real-life solutions. Once, working on a science outreach project, we demonstrated chelation in polluted water. Kids watched this compound trap traces of heavy metals, making water noticeably clearer. It suddenly became less about complicated science and more about safe drinking water.
Safety and Handling
With any chemical, safe handling remains a must. This one doesn’t explode on contact or corrode metal at the drop of a hat, but direct skin contact may trigger irritation. That’s why labs keep gloves and splash goggles close by. The Material Safety Data Sheet (MSDS) emphasizes storing it in sealed containers, away from acids and oxidizers. Many people take these rules for granted or skip reading the safety sheets, but accidents only need one lapse. Years in workspaces taught me caution always trumps carelessness—even with chemicals considered “low hazard.”
Challenges in Use
Though easy to dissolve in water, hydroxyethylethylenediamine’s chelating abilities don’t appear out of thin air. pH changes can stall its performance. So, water treatment plants test their systems often to keep balance and avoid fouling pipes or equipment. In my university days, we spent a lot of time tweaking formulas to get consistent results—and sometimes the smallest change ruined a whole batch.
Steps Toward Safer Science
Alongside practical measures, better education about compounds like C4H12N2O keeps everyone safer and more informed. Updating old procedures, using equipment that helps with precise dosing, or adopting greener synthesis methods ensures both results and responsibility. Today’s chemistry isn’t about rote memorization. It circles back to understanding the molecules, respecting their quirks, and finding ways to use them with as little waste as possible.
Looking Ahead
Chemical formulas don’t just belong on paper. They touch everything from environmental cleanup to industrial manufacturing. Focusing on both science and safety lifts the conversation beyond confusion and closer to progress.
How should Hydroxyethylethylenediamine be stored?
Understand What You’re Handling
Hydroxyethylethylenediamine isn’t something you want to treat like just another bottle on a shelf. Anyone who’s spent time in a lab or handling chemicals knows damage often comes from the small things we overlook. This chemical comes with risks: skin and eye irritation, and in some cases, strong fumes that nobody wants to breathe. Over the years, I’ve seen people run into trouble by skipping the basics, figuring nothing could possibly go wrong from a quick, careless pour or a slipup during cleanup. That sort of thinking almost always backfires.
Why Cool, Dry, and Ventilated Means Safer Work
Temperature and moisture matter. I remember one summer when a shipment sat near a sunny window. Condensation formed inside the lid, and the whole room smelled sharp before lunch. Heat and humidity get chemicals moving in ways you don’t expect—they leach out, sometimes react, or even corrode storage containers. Storage isn't only about where you put a drum; it’s also about keeping the air dry and moving.
Basements with strong dehumidifiers, metal lockers kept out of the sun, rooms with steady fans—these setups make a real difference. Don’t just shove a barrel in a corner and hope for the best. Fumes find their way out. Always pick a ventilated place, with doors or windows, so any sneaky vapor doesn’t build into a problem.
Containers Matter More Than Most People Think
Never heard of somebody losing half a drum because they skipped a lid or used a rusting cap? I have. Hydroxyethylethylenediamine eats right through some plastics, and it doesn’t play nice with buckets made for food or quick fixes. Go with the manufacturer’s advice—usually high-density polyethylene or stainless steel. Check seals and gaskets often. Small drips become giant headaches during busy days.
Label containers with bold, clear writing. Nobody wants a fire drill over an unlabeled jug. Hazmat teams and medical responders count on accurate identifiers. GHS pictograms, batch numbers, and warning tags all keep co-workers and the boss safe if something goes sideways. Years ago, I watched a close call unfold after someone dumped a clear liquid into the wrong tank. All because someone skipped a label.
Shared Spaces, Shared Responsibility
It’s easy to shrug and leave responsibility to the next shift. The truth is everybody has skin in the game if that chemical ends up on the floor. I once saw a janitor squeegee a puddle across the warehouse, thinking it was just water, only to discover the hard way that safety gear isn’t just a suggestion.
Keep spill kits visible and ready. People get lazy if they’re hidden in a locked cabinet, or worse, lost behind a pile of boxes. Eyewash and shower stations don’t belong at the other end of a building. Put up an SOP sheet right next to the entrance. Nobody should waste time searching for instructions in a pinch.
Regular Checks Make All the Difference
Small leaks come from forgetfulness. Caps crack, seals dry out, and before anyone knows it, vapor escapes. Once a month, walk the aisles. Touch every drum and bottle. Tighten, relabel, reorder. Use a log. It sounds simple, but routine checks prevent about 90% of storage mishaps I’ve witnessed.
All it takes is one accident to turn a routine week into nights filled with phone calls, paperwork, and lost trust. Spend a half hour on inspection. That half hour keeps your team healthy, your gear in top shape, and your workday moving forward without the drama.
What are the potential hazards of Hydroxyethylethylenediamine?
Understanding What We Are Handling
Working in chemical research taught me that even molecules with long, hard-to-pronounce names like hydroxyethylethylenediamine often land on the tables of local water plants or labs. You see a colorless liquid, but you’re looking at something with the power to change water chemistry, industrial processes, and sometimes the safety outlook for anyone nearby. Hydroxyethylethylenediamine shows up in water treatment, epoxy formulations, and as a complexing agent for metal recovery. Some labs rely on it for niche synthesis work, not always realizing the risks baked into each bottle.
The Immediate Risks: Skin, Eyes, and Airways
Splash a little hydroxyethylethylenediamine on your skin, and you’ll probably feel it fast. Chemical burns, itching, or lasting redness can follow direct contact. Once, a colleague accidentally flicked some on their arm, brushed it off, and then scratched their face. The reaction set in where the liquid hit, not where you’d expect. This stuff seeps through gloves that aren’t up to the job. Eye contact works quicker still—redness, pain, and potentially lasting vision trouble if you don’t flush right away. Inhaling its vapors can sting the nose and throat, bringing coughing or shortness of breath. The dangers go up in unventilated labs or closed rooms, where higher vapor concentrations build fast.
Longer-Term Concerns Worth Noting
Many solvents or agents leave traces inside the body if inhaled or absorbed repeatedly. Hydroxyethylethylenediamine is no different. Early occupational studies link ongoing exposure to problems with liver and kidney function, and in rare cases, suggest something might change in lung tissue after months or years. Direct links to cancer have yet to be pinned down, but the story rings familiar for anyone who’s seen older chemists retire with unexplained health issues. Sometimes, the best-sounding studies only turn hints into facts decades later.
Environmental Worries
Small spills might not seem urgent, but hydroxyethylethylenediamine washes quickly into groundwater or storm drains. It doesn’t always break down quickly in the environment. Local wildlife exposed to the chemical sometimes shows stunted growth or problems reproducing. Municipal waste stations keep an eye on this molecule because it can build up in rivers, landing inside fish or plants along the way. If your town relies on treated surface water, even slow accumulation matters. Stories from river cleanups show the long tail of industrial runoff, where small leaks turn into big headaches for decades.
Managing the Hazards—What Actually Helps
Everyone in the industry recognizes that training and protection gear count, but the real protection comes from culture and routine checks. In every solid lab or plant job I’ve seen, labeling stands out as the difference-maker: clear bottles, direct instructions, and workers that actually stop to read them. Switching to gloves and goggles rated for amines blocks the most direct risks. Fume hoods matter as much as fire extinguishers here. I learned that fast after a simple pipetting mistake filled our workspace with a strong, acrid smell.
The greatest fix, though, involves process changes. Closed systems, spill-proof batch tanks, and solid waste treatment trap most of what escapes. Local disposal rules work best when paired with real oversight. Whistleblowers, regular inspections, and workers trained to notice odd smells or skin symptoms can head off bigger health and environment explosions.
Hydroxyethylethylenediamine will stick around in industry and science circles because it works for tricky synthesis jobs, but ignoring its hazards simply multiplies the price everyone pays down the line. Taking it seriously doesn’t slow progress—it protects the people who keep the whole operation moving.
| Names | |
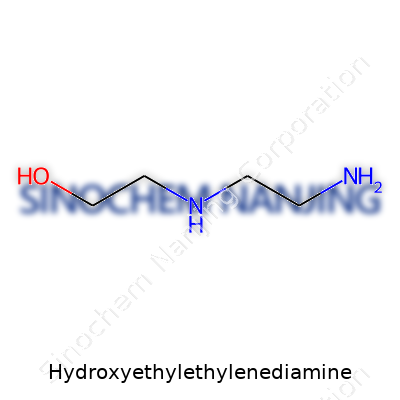
| Preferred IUPAC name | 2-(2-Aminoethylamino)ethan-1-ol |
| Other names |
N-(2-Hydroxyethyl)ethylenediamine 2-(2-Aminoethylamino)ethanol HEEDA AEEA 2-Hydroxyethyl-1,2-ethanediamine |
| Pronunciation | /ˌhaɪ.drɒk.siˌɛθ.ɪlˌɛθ.ɪˌliːn.daɪˈæm.iːn/ |
| Identifiers | |
| CAS Number | 111-41-1 |
| Beilstein Reference | 2811223 |
| ChEBI | CHEBI:16627 |
| ChEMBL | CHEMBL127967 |
| ChemSpider | 58483 |
| DrugBank | DB14161 |
| ECHA InfoCard | 18a070d7-7638-4273-9653-472a28a33143 |
| EC Number | 200-741-1 |
| Gmelin Reference | 153923 |
| KEGG | C00946 |
| MeSH | D03.438.460.890.213 |
| PubChem CID | 16715 |
| RTECS number | KK3675000 |
| UNII | Q4R7L4Q8E4 |
| UN number | UN3267 |
| CompTox Dashboard (EPA) | DTXSID7036793 |
| Properties | |
| Chemical formula | C4H12N2O |
| Molar mass | 90.121 g/mol |
| Appearance | Colorless to pale yellow transparent liquid |
| Odor | Ammonia-like |
| Density | 1.054 g/cm3 |
| Solubility in water | miscible |
| log P | -2.14 |
| Vapor pressure | <0.01 mmHg (20°C) |
| Acidity (pKa) | 8.1 |
| Basicity (pKb) | 5.75 |
| Magnetic susceptibility (χ) | -5.0E-6 cm³/mol |
| Refractive index (nD) | 1.486 |
| Viscosity | 500 mPa·s |
| Dipole moment | 3.23 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 319.27 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -152.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3569 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | C01EB09 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. |
| GHS labelling | GHS07, GHS05 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H412 |
| Precautionary statements | P280, P305+P351+P338, P310, P303+P361+P353, P363, P304+P340, P312 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | > 143°C |
| Autoignition temperature | 930°F (498.89°C) |
| Lethal dose or concentration | LD50 (oral, rat): 2520 mg/kg |
| LD50 (median dose) | LD50 (median dose): 910 mg/kg (oral, rat) |
| NIOSH | WE1575000 |
| PEL (Permissible) | Not established |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Ethylenediamine Diethanolamine Aminoethylethanolamine Triethanolamine N-Methylethanolamine |

