Hydroxyamine Sulfate: A Closer Look at a Quiet Workhorse
Historical Development
Chemistry often takes center stage in big discoveries, but quietly in the background, compounds like hydroxyamine sulfate keep the gears turning. Its story reaches back to the late 19th century, a period flush with rapid progress in synthetic chemistry. Scientists then, striving to harness oxidative and reductive power for new chemical reactions, discovered that hydroxylamine and its salts held the key to more controlled and safer processes than using more volatile or toxic chemicals. Unlike its predecessor, free hydroxylamine, which brought risks of instant decomposition and daunting toxicity, pairing it with sulfate made it stable enough for handling, measurement, and transport. The leap from unpredictable laboratory curiosity to solid industrial tool happened over decades, shaped by the need for scalable, reliable intermediates in everything from pharmaceuticals to explosives.
Product Overview
Today, hydroxyamine sulfate shows up everywhere outside the public eye. Look in chemical production lines, semiconductor labs, water treatment facilities, and textile plants, and it's often involved. As a white, crystalline solid with a slight acidic odor, it's shipped and stored in drums but rarely advertised. The real value comes from its dual power to reduce and react with a variety of functional groups, offering chemists a versatile and economical way to tweak molecular structures or create new ones from scratch. Over the years I've encountered it as an unsung helper in student organic labs, where it quietly enables reactions that would otherwise require far more sensitive setups or expensive reagents.
Physical & Chemical Properties
The properties carry a mix of opportunity and risk. Soluble in water, hydroxyamine sulfate gives a clear solution and maintains stability under reasonable temperatures and pressures, which allows safe measurement and manipulation. The substance tends to decompose slowly, releasing ammonia and nitrogen oxides, so good ventilation matters from the first day of storage. Its strong reducing capability lets it strip oxygen or nitrogen from many organic and inorganic molecules. Heat, acids, and bases can tip the balance toward rapid decomposition or even hazardous reactions, which means constant attention to temperature and pH. I’ve seen careless storage choices create needless hazards, emphasizing that familiarity with the compound shouldn't breed contempt.
Technical Specifications & Labeling
Labels on these drums have grown more detailed in step with tighter safety standards. They warn about the inhalation risks, detail the need for gloves, goggles, and fume hoods, and sometimes include QR codes linking straight to research or safety databases. Chemical manufacturers identify purity levels for industrial, laboratory, or technical grades, as even trace metal contamination can skew key reactions in pharmaceuticals or electronics. From a regulatory perspective, periodic reviews arise as new health effects or environmental persistence data emerge, triggering adjusted thresholds for workplace exposure and spill cleanup protocols.
Preparation Method
Making hydroxyamine sulfate starts with the reduction of nitrogen-containing compounds, a process tuned carefully to keep byproducts low. Industrial setups often use sodium bisulfite and hydroxyamine hydrochloride in controlled reactors, slowly adding sulfuric acid to capture the free base as a sulfate salt. Each change in process, whether in temperature, mixing rate, or acid strength, can bump yields up or down. Chemists learned that scale-up brings surprises, including heat surges and local acidities that threaten stability. My own hands-on experience confirmed that patience with process tuning wins out over shortcuts; impatience often leads to foaming, poor yield, or hazardous spills.
Chemical Reactions & Modifications
Reactivity sets hydroxyamine sulfate apart. In synthesis, it helps make oximes, which can then become useful building blocks for making antibiotics, dyes, and rubber additives. Its gentle reducing power grants more control than outright hydrogenation and works well in multi-step reactions where side-products are costly mistakes. Scientists continuously probe new modifications, tweaking the molecular structure or mixing with co-reagents to match specific reaction requirements. In my lab days, we pushed it to react with aldehydes and ketones—producing intermediates for more complex pharmaceuticals with fewer byproducts than older, dirtier methods.
Synonyms & Product Names
Across textbooks and warehouses, hydroxyamine sulfate answers to several names: oxammonium sulfate, carbohydroxylammonium sulfate, and simply hydroxylamine sulfate. Each label hints at its legacy—changing as its chemistry and applications grew. No matter the name, its chemical formula remains unchanged, but keeping track matters, since confusion between salts and related compounds in inventories can trip up projects and waste money. This kind of clarity only comes from hands-on experience; ignoring it courts expensive ordering mistakes and delays.
Safety & Operational Standards
Handling hydroxyamine sulfate calls for steady nerves and respect for its hazards. It irritates the skin and lungs, and labs keep antidotes and spill kits on hand. Protocols require double-gloving, eye protection, and never working alone with liters of solution. Industrial users rely on strict inventory controls, monitored venting systems, and regular air sampling. Guidance from OSHA and similar regulators adapts as new studies flag chronic exposure risks or environmental persistence. Accidents remind everyone that even a workhorse chemical must not be taken for granted. In my own career, I've seen smart people get careless after years of incident-free use, leading to costly shutdowns and investigations.
Application Area
Few see hydroxyamine sulfate’s reach. It shows up in the synthesis of pharmaceuticals like antibiotics and cancer drugs, shapes the properties of photographic film, cleans semiconductors for high-end electronics, and purifies wastewater by controlling harmful metals. Textile and rubber industries beat back unwanted color changes and extend product lifespans using it. Water treatment facilities appreciate its power to reduce residual chlorine and break down nitrogen compounds. Every industry where purity, reactivity, and affordable chemistry align tends to find a place for this salt, though rarely do they publicize it outside of regulatory discussions or process audits. In each new setting, workplace safety and adaptation to specific contamination risks become daily realities, not theoretical concerns.
Research & Development
Research teams today chase cleaner, safer, and more efficient ways to harness hydroxyamine sulfate. Many target the molecule’s role as a precursor in making active pharmaceutical ingredients, aiming for less toxic byproducts, quicker synthesis, or reactions in water instead of hazardous organic solvents. Sustainability drives new projects: can biocatalysts or electrochemical steps replace traditional chemical reduction? Can recycling recovery procedures handle spent sulfate solutions? Peer-reviewed studies track advances in reaction specificity and minimize side-reactions that generate regulatory headaches or expensive remediation. Investments pour into novel process controls, digital monitoring, and predictive maintenance for bulk storage and dosing equipment—turning lessons from past spills and leaks into smarter industrial management.
Toxicity Research
Toxicologists approach hydroxyamine sulfate with caution after repeated reports of mutagenic and cytotoxic effects in living systems. Even at low concentrations, chronic exposure in animal models produces hematological changes and organ toxicity. Plant and aquatic life also take a hit, making disposal and water treatment challenges for municipalities and manufacturers alike. Most alarming, studies continue to surface around impaired fetal development and neurotoxicity, pushing environmental agencies to keep their standards under review. Personal protective gear and air monitoring systems in laboratories—for all their inconvenience—save real lives as cumulative effects reveal themselves over time, often years after regulators first approved the compound’s use.
Future Prospects
Looking down the road, hydroxyamine sulfate stands at a crossroads. On one hand, industries bank on its continued service as chemistry and electronics demand higher purity, tighter margins, and nimble reaction conditions. On the other, sustainability pressures call for cleaner alternatives, lower-emission production cycles, and safer handling across the life cycle. New regulatory moves in Europe and North America could force process innovations or even gradual phase-outs in favor of bio-based or less toxic reagents. In research, rising interest in green chemistry creates funding streams for next-generation reductants and more efficient catalyst recycling, possibly shifting hydroxyamine sulfate’s primary territory from blockbuster manufacturing to specialized or legacy roles. One thing seems clear: as with so many chemicals that form the backbone of industry but fly under the consumer radar, its future will swing on a balance of innovation, vigilance, and an honest accounting of both benefits and risks.
What is Hydroxyamine Sulfate used for?
A Closer Look at Its Uses in Industry and Everyday Life
Walk into any chemistry lab or chemical plant, and you’ll likely stumble across hydroxyamine sulfate. This isn’t some fringe chemical, either. Factories rely on it to make things work behind the scenes, especially in textile, rubber, and pharmaceutical arenas. For anyone who has handled dirty laundry or gotten a blood test, this compound already plays a role in your day.
Textile and Dyeing Applications
Look at colored fabrics in clothing stores. Staying bright and holding color after multiple washes doesn’t just happen by accident. Hydroxyamine sulfate helps fix dyes onto fibers. Without this chemical, jeans and t-shirts would fade before seeing their third spin in the washer, and cloth manufacturers would face bigger headaches with consistency. This compound helps maintain the sharp, stable hues people want in their shirts, bedsheets, and towels.
Pharmaceutical and Laboratory Work
Chemists use hydroxyamine sulfate to snap molecules together in the lab, making it an essential step in some drug manufacturing. The pharmaceutical industry leans on it to build medications that require special chemical structures. While some people hear the word “chemical” and think of things to avoid, here it becomes a backbone for life-saving molecules. As a reducing agent, hydroxyamine sulfate brings atoms to the right state, which sets up successful production for antibiotics, cancer therapies, and diagnostic kits.
Metal and Water Treatment
Rust never sleeps. Anyone who’s fought it on a bike chain or garden tool knows this well. Metal manufacturers count on hydroxyamine sulfate to clean surfaces and prepare them for coating. By stripping away lingering oxidation, it makes sure paints and other finishes hold fast. In water treatment plants, it helps keep water clear by taking unwanted oxygen out of the mix, which can otherwise lead to rust in pipes or discoloration.
Photography and Analytical Testing
Old-school photo buffs might not realize hydroxyamine sulfate played a part in developing film. Even now, in labs, this chemical comes into play when measuring certain substances or running blood tests. Medical technicians use it to make readings more precise, which helps doctors spot health changes early. Accuracy saves lives, and this compound keeps equipment running smoothly.
Safety and Environmental Concerns
Handling chemicals gets tricky. Hydroxyamine sulfate works best under controlled settings. Safety protocols keep workers healthy—gloves, masks, good ventilation, and strict rules are part of daily routines in labs and factories. Accidental spills can irritate skin, eyes, or lungs, so facilities pay close attention to storage and disposal. Environmental rules demand careful waste management, pushing companies to filter runoff and shrink their pollution footprint.
Thinking Ahead: Better Practices and Sustainability
Green chemistry gets more attention each year. Researchers hunt for safer alternatives and methods that use fewer resources or less toxic ingredients. Efforts to capture and reuse byproducts from processes involving hydroxyamine sulfate aim to reduce pollution and lower costs. By focusing on smarter handling and new technology, industries can keep reaping the benefits while staying responsible to the people who work inside their plants and the communities nearby.
Is Hydroxyamine Sulfate hazardous or toxic?
What is Hydroxyamine Sulfate?
Hydroxyamine sulfate shows up in a surprising number of industries, from pharmaceuticals and photography to water treatment. The substance looks like a white crystalline powder, but what matters more is how it acts and what risks exist.
Health Concerns Arise from Handling
Direct skin contact with hydroxyamine sulfate leads to irritation, redness, or even burns. I’ve seen cases where people missed a spot on their gloves, and that error leaves a mark. Inhalation exposes the lungs. Coughing, sore throat, and respiratory distress require real attention. Its corrosive nature doesn’t stop at the surface. The eyes see the worst if the dust goes airborne—tearing, severe pain, long-lasting damage if not flushed right away.
Routes of Exposure Show the Bigger Picture
The trouble starts at handling and storage. In labs, students and workers with little training throw on goggles and hope for the best. Safety sheets for hydroxyamine sulfate point out serious acute toxicity risk if swallowed, too. Even a small mouthful pushes the body into convulsions, vomiting, and in some historical reports, death. It disrupts enzymes by attacking cells in blood, liver, and kidneys.
Chemical Instability and Workplace Risk
With moisture, hydroxyamine sulfate starts to break down and form gases. If mixed with strong acids, bases, or metal salts, spontaneous reactions become possible, even violent. It doesn’t take much heat for the substance to release toxic fumes. The 2011 incident in a manufacturing plant that made headlines happened when a storage container leaked—inside, workers scrambled, eye protection foggy, masks restricting breath. Medical staff saw several exposed people with symptoms ranging from headaches and dizziness to a few with blistered arms.
Supporting Facts from Scientific Studies
Toxicologists categorize the compound as “harmful if swallowed, inhaled, or in contact with skin.” The National Institute for Occupational Safety and Health (NIOSH) sets strict exposure limits. Cancer research ties extended or repeated exposure to changes in cell structure and function, with some animal tests showing organ damage. Environmental impact studies show high reactivity with water sources, stressing aquatic life when released in runoff.
Responsible Use and Safer Alternatives
Factories and schools try to keep hydroxyamine sulfate in secure, labeled containers, away from sunlight, sparks, or moisture. Double-gloving and full-face shields protect users from dust and splash. Fume hoods suck up airborne particles. But errors still happen—sometimes from skipping a step, sometimes from broken equipment. Routine audits and real, hands-on training make a difference. Some research labs swap it out for less hazardous chemicals where possible, though that’s not always an option. Environmental agencies call for spill kits and emergency showers in any room storing this chemical.
What Can Help Keep People Safe?
Clear communication saves lives. I learned early to ask questions—even the simple ones about what’s in a bottle or which gloves to wear—before starting any experiment. Full disclosure on safety data sheets, regular gear checks, and backup protocols for spills reduce the odds of injury. Stronger rules for disposal and accidents cut down risks for workers and the environment alike.
How should Hydroxyamine Sulfate be stored?
Putting Safety Front and Center
Hydroxyamine sulfate isn’t the kind of chemical anyone wants leaking, overheating, or getting anywhere near the wrong container. My own lab experience taught me that a small error with tricky substances sometimes multiplies fast. Hydroxyamine sulfate scores high on the list of chemicals that deserve respect, not just during use but in storage. Improper storage can lead to fires, toxic release, or serious injuries. So, let’s talk about what works.
Controlling The Basics: Temperature, Light, and Moisture
What worries most people about hydroxyamine sulfate is its instability. This compound reacts badly with heat, sunlight, and moisture. Even small increases in temperature can speed up decomposition, releasing gasses and heat. Air humidity can turn a tidy powder or crystal into messy, sticky clumps, which could trigger unintended reactions. Cool and dry storage isn’t a luxury—it’s required for safety.
Every year, safety reports pile up about workplace accidents involving poorly stored chemicals. In 2022, a small university lab had to evacuate after improper storage of hydroxyamine sulfate led to a frightening gas release when a bottle’s seal failed. This chemical’s risk sits right at the intersection of heat, damp, and air exposure.
Material Matters
The container itself shapes safety outcomes. Hydroxyamine sulfate should live in non-reactive containers, with airtight seals that aren’t metal. Metal lids or shelving can corrode or start redox reactions if crystals leak or dust settles. I’ve seen plastic screw-cap jars work well, but it pays to check that none of the material will react over time.
If you toss a powerful oxidizer or reducing agent next to hydroxyamine sulfate, you create a ticking time bomb. Real discipline comes from segregated storage. Set aside shelves, lockable cabinets, and, in larger facilities, climate-controlled rooms. That simple physical separation can prevent headline-making accidents.
Labeling and Access
Sloppy labeling and loose keys invite accidents. Every container should display a clear label with both the chemical name and the date received or opened. No one wants to hunt through faded handwriting in an emergency. In labs where more than a few people work, limit access to those who truly know the risks. I’ve seen near-misses traced back to someone pulling a lookalike bottle who never got proper training. Training and oversight save lives.
Planning for Emergencies
Let’s not pretend nothing ever spills. Smart labs keep spill kits, eye wash stations, and ventilation systems in easy reach. In factories and bulk storage, safety showers and large-volume neutralizers stand by. Leaving these out or letting them lapse through lack of inspection can be catastrophic. Having phones charged and emergency procedures visible on the wall makes a difference. Employees need to know not only how to store hazardous chemicals, but what to do when something goes wrong.
Stronger Standards, Safer Communities
Complying with workplace safety laws cuts risk. The Occupational Safety and Health Administration (OSHA) recommends storing hydroxyamine sulfate below 20°C (68°F) in a dry environment, away from sunlight or incompatible chemicals. Regular audits encourage compliance. Strong safety cultures rest on clear rules, reliable supplies, and well-trained staff. Using hydroxyamine sulfate responsibly isn’t just about protecting those who handle it. Neighbors, emergency responders, and the environment also benefit.
What are the safety precautions when handling Hydroxyamine Sulfate?
Hydroxyamine Sulfate Isn’t Your Everyday Chemical
Most people haven’t heard of hydroxyamine sulfate. If you have, chances are you work in a lab or a plant that deals with tough industrial or research jobs. Hydroxyamine sulfate packs a punch—on one side, it helps with making pharmaceuticals and cleaning up, but it also brings some risks. I’ve spent years working in labs and I’ve seen what happens when folks overlook safety with chemicals like this. It’s not worth the gamble.
Why Personal Protection Matters
One of the first things hammered into my head in training: gear up before you handle anything strong. For hydroxyamine sulfate, this means gloves that keep skin safe, long sleeves, and goggles. Latex or nitrile gloves work; don’t settle for the cheap kind you grab for food prep. The dust and liquid mix of this chemical can burn or irritate skin. If you get some on your skin, act right away—wash it off with lots of water. Once, I saw a co-worker skip gloves “just for a quick transfer.” His hands turned red and itchy in minutes.
Breathing Easy—Control the Air
Unlike some chemicals that odor lets you know to back away, hydroxyamine sulfate doesn’t provide that warning. Breathing in dust or fumes can mess with your lungs or eyes, so ventilation isn’t just nice to have; it’s essential. Using a fume hood or working in an open, vented area does the heavy lifting here. For big jobs, adding a dust mask or a respirator adds another layer of protection. Shortcuts in the name of saving time often land folks in the nurse’s office.
Never Mix and Match Chemicals Without Thinking
A rookie mistake in any setting: pouring or mixing chemicals without checking compatibility. Hydroxyamine sulfate reacts fast with oxidizers and certain metals. A spill or sloppy mixing can cause fumes or even small explosions. Labeled containers, a tidy workspace, and clear training all help prevent accidents. It doesn’t take much—a forgotten beaker can trip up an experiment or a whole production run.
Storage Rules Make All the Difference
Don’t shove hydroxyamine sulfate into any old cabinet. Dry, cool spots away from sunlight work best. Storing it near acids or oxidizing chemicals is just asking for trouble. I keep chemicals like this on a shelf labeled and locked up, with an eye-level sign that lists hazards. Even the janitorial crew needs to know what’s in that cabinet, since a mop bucket with the wrong cleaner can set off a chain reaction.
Spills, Disposals, and Doing the Job Right
Nobody wants to clean up a chemical spill, but it happens. Quick action keeps a small mess from turning into a disaster. Spill kits with neutralizing agent, absorbent pads, and disposal containers make the difference. Standard trash cans and sinks are not for dumping leftover hydroxyamine sulfate—hazardous waste bins and proper paperwork are the only way. Years ago, I saw a rush job lead to a backup in the drains and a day-long cleanup that could have shut down the whole lab.
Training: The Forgotten Step
Reading a safety sheet once doesn’t cut it with hydroxyamine sulfate. Real training means drills, practice, and staying up to date on what to do if something goes wrong. A good team won’t let a new person work alone or with a shortcut-memory of safety steps. Using a chemical like hydroxyamine sulfate isn’t just a technical challenge—it’s about showing respect for the risks, the people around you, and the progress you’re trying to make.
What is the chemical formula and molecular weight of Hydroxyamine Sulfate?
Understanding the Basics
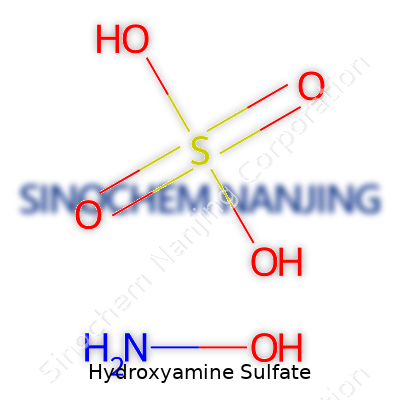
Hydroxyamine sulfate pops up a lot in labs, especially for people handling organic synthesis or analytical chemistry. This compound’s chemical formula stands as (NH2OH)2·H2SO4. Put differently, the formula shows two hydroxyamine molecules paired with one molecule of sulfuric acid. Its molecular weight is 164.14 grams per mole.
Why This Compound Matters
I recall my days as a lab assistant prepping solutions for qualitative analysis courses. The first time I handled hydroxyamine sulfate, I learned not only about its usefulness in reducing iron(III) to iron(II) but also about its quirks. Textbooks mention oxidizing properties, but seeing a pale color shift in a beaker for the first time locked the lesson in.
This salt shows up in more places than college labs. Industries use it in the synthesis of caprolactam, which eventually becomes nylon. A wrong calculation in mixing or in understanding its properties can throw off yields in a production batch. Getting the chemistry right requires practical knowledge—especially the ability to quickly recall that formula and molecular weight.
Safety and Handling Challenges
Hydroxyamine itself poses risks; it decomposes easily and can release toxic fumes. Sulfate salts make it more stable, but you can’t relax around it. Minor spills can create dangerous situations. My former supervisor set rules that left no room for mistakes: always measure in a fume hood, never store near oxidizers, check expiration dates monthly.
The facts speak loudly. Between 2012 and 2020, chemical incident reports from the CDC featured hydroxyamine or its salts about 40 times, mainly due to improper storage or uncontrolled heating. The most serious involved burns and respiratory symptoms. I carry a healthy respect for the compound because I’ve come close to joining those statistics.
Improving Safe Use
At every company I’ve worked for, we tried something new to help people remember crucial data like formulas and weights, from large laminated signs near the chemical storage to QR code labels linking to an online database. None of these ideas replaced basic training, but every bit helped. The key has always been free-flowing communication, where no one feels shy asking for a refresher on chemical properties.
Regulatory bodies like OSHA and the European Chemicals Agency stress clear chemical labeling and up-to-date material safety data sheets. Short, practical safety meetings—never longer than the time it takes to drink a cup of coffee—made staff more comfortable handling hydroxyamine sulfate. Look at the formula. Check the bottle twice. Don’t just rely on memory.
Getting the Chemistry Right
If you look up the structure, you’ll see why accuracy counts. The right formula and molecular weight keep labs running smoothly, ensure production lines don’t halt, and prevent errors that could cost money and safety. Hydroxyamine sulfate isn’t some obscure chemical; it’s a workhorse behind plenty of industrial and research applications. Handling it well shows respect for chemistry and the people doing the work.
| Names | |
| Preferred IUPAC name | Oxidanium;sulfate |
| Other names |
Hydroxylammonium sulfate Hydroxylamine sulfate Dihydroxylammonium sulfate |
| Pronunciation | /haɪˌdrɒksi.əˈmiːn ˈsʌl.feɪt/ |
| Identifiers | |
| CAS Number | 10039-54-0 |
| 3D model (JSmol) | JSmol string for Hydroxylamine Sulfate (NH₂OH·H₂SO₄): ``` data "hydroxylamine sulfate" 3 Hydroxylamine sulfate N 0.000 0.000 0.000 O 1.250 0.000 0.000 S -1.500 0.000 0.000 end ``` |
| Beilstein Reference | 1690832 |
| ChEBI | CHEBI:60051 |
| ChEMBL | CHEMBL1236068 |
| ChemSpider | 21517 |
| DrugBank | DB11372 |
| ECHA InfoCard | 100.028.915 |
| EC Number | EC 226-798-2 |
| Gmelin Reference | 42101 |
| KEGG | C00029 |
| MeSH | D017250 |
| PubChem CID | 6115 |
| RTECS number | GR8050000 |
| UNII | K59037RCSU |
| UN number | UN2865 |
| Properties | |
| Chemical formula | (NH2OH)2·H2SO4 |
| Molar mass | 164.14 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.89 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -4.217 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 6.03 |
| Basicity (pKb) | 8.1 |
| Magnetic susceptibility (χ) | −53.0×10⁻⁶ cm³/mol |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 237.6 J·K⁻¹·mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -499 kJ/mol |
| Pharmacology | |
| ATC code | D06AX04 |
| Hazards | |
| Main hazards | Oxidizer, corrosive, harmful if swallowed, causes burns, may cause allergic skin reaction, decomposes on heating to release toxic fumes. |
| GHS labelling | GHS02, GHS05, GHS07, GHS09 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H318, H335 |
| Precautionary statements | P210, P220, P221, P264, P280, P302+P352, P305+P351+P338, P310, P370+P378, P402+P404, P501 |
| NFPA 704 (fire diamond) | 2 3 2 OX |
| Flash point | > 157°C (315°F) |
| Autoignition temperature | > 80°C (176°F) |
| Lethal dose or concentration | LD50 oral rat 65 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 65 mg/kg |
| NIOSH | RN 10039 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Hydroxyamine Sulfate: 1 mg/m³ |
| REL (Recommended) | 200g |
| IDLH (Immediate danger) | 300 mg/m³ |
| Related compounds | |
| Related compounds |
hydroxylamine hydroxylammonium nitrate hydroxylammonium chloride hydrazine ammonium sulfate |

