Hydroxyamine Hydrochloride: Beyond Simple Chemistry
Historical Development
Few compounds have charted a more interesting course in the world of chemistry labs than Hydroxyamine Hydrochloride — or hydroxylamine hydrochloride if you prefer a spelling closer to its roots. Discovered in the mid-19th century, back when European chemists were racing to decipher the inner workings of molecules, this reagent grabbed attention not with any miracle cure or explosive display, but with its stubborn ability to mediate chemical change. As a writer who spent far too many late nights studying old patents and industrial blueprints, I've watched Hydroxyamine Hydrochloride shift from a rare curiosity to a staple in synthetic chemistry, especially during the years when the dyes and explosives industries were booming. Chemists eager for ways to reduce nitro compounds or synthesize new intermediates found that this humble reagent opened doors that seemed locked tight before. Over the decades, factories on both sides of the Atlantic crafted processes to make it reliably and cheaply, feeding the hunger of growing fields like pharmaceuticals, polymers, and even photography.
Product Overview and Properties
Hydroxyamine Hydrochloride, known among insiders as NH2OH·HCl, lands on the bench as a white, free-flowing crystalline powder. It dissolves in water without a fight but keeps to itself in organic solvents. What sets it apart isn’t just its looks; it brings a gentleness to chemical reductions that others can't match. With a melting point hovering around 155°C (with decomposition), this compound doesn’t like heat or rough handling, showing a finicky side that every lab worker quickly learns to respect. Its behavior springs from its peculiar structure — the interaction between a basic hydroxylamine and acidic hydrochloride gives it enough chemical punch for practical work while still avoiding the volatility that plain hydroxylamine sometimes delivers. Measuring the pH in solution tells another piece of the story: leave it too acidic and you lose its value as a nucleophile, go too basic and the risk of decomposition sparks concern.
Technical Details and Labeling
Every bottle of Hydroxyamine Hydrochloride rolls out of the supplier with specific labeling that’s really more than a formality. Anyone using it in research or manufacturing recognizes the familiar stamp of hazard warnings thanks to its potential to irritate skin, eyes, and respiratory tract. Manufacturers must list the CAS number, batch purity — often above 99% for lab-grade — and crucial safety information. Watching students in the lab, I’ve seen how easy it is to rely on a label, missing the deeper importance of those numbers. Consistent purity matters not only for the reaction outcome but for making sure scaled-up processes or sensitive applications don’t run into roadblocks or unintended hazards.
Preparation Method
Synthesizing Hydroxyamine Hydrochloride doesn’t require a wizard, but it does demand respect for the underlying chemistry. Typical production routes rely on the reduction of nitrite salts with sulfite, zinc, or iron in hydrochloric acid. Industrial plants, from Europe to Asia, embraced these reliable paths as they produce consistent yields without spawning too many by-products. In small labs, reactions run under cool temperatures, and precise acid control ensures the main product outcompetes numerous possible side reactions. Making the compound may sound straightforward but controlling exothermic steps and containing emissions pushes engineers and chemists into regular troubleshooting mode. Each batch made for pharmaceutical or food-contact applications ends up passing through more rigorous purification steps, often involving recrystallization to weed out the stubborn impurities that can slip past basic monitoring.
Chemical Reactions and Modifications
Chemists appreciate Hydroxyamine Hydrochloride for what it can change, not just what it is. Set it loose with aldehydes or ketones and it forms stable oximes, critical intermediates for further transformations like the Beckmann rearrangement — a cornerstone step for polyamide production, including nylon. Pharma labs use hydroxyamine salts to create hydrazones and other nitrogen-containing scaffolds. Up close, the molecule hands over its hydrogen atoms with surgical precision, reducing nitro groups to the corresponding amines without the unwanted mess common to many catalytic or metallic reductions. Each reaction this salt participates in leaves a distinct mark; it's rarely an anonymous bystander. Over time, researchers tuned its reactivity further by combining it with different acids or buffering systems, sometimes coaxing out new products by exploiting slight changes in local acidity or solvent choice.
Synonyms and Product Names
Ask ten chemists what they call Hydroxyamine Hydrochloride, expect at least ten answers. Some go for the old-school Hydroxylammonium Chloride, others abbreviate to HA·HCl or just “the oxime maker.” Across the pharmaceutical and industrial spectrum, product catalogs list synonyms ranging from “Oxyammonium Chloride” to “Aldoxime Reagent.” Regardless of the label, the molecule inside the bottle stands as an anchor in synthetic strategy, bridging tradition and modern advances.
Safety and Operational Standards
Nothing about Hydroxyamine Hydrochloride invites carelessness. Stories about lab accidents echo in the halls where it gets used. This compound can set off nasty skin and respiratory irritation, especially if handled without appropriate gloves and ventilation. Breathing its dust or getting it in the eyes delivers urgent reminders that even the most routine reagents deserve respect. Regulatory agencies enforce guidelines on storage, spill management, and personal protective equipment (PPE) — a lesson reinforced by hard experience in both academic and industrial settings. Facilities need reliable air handling and closed transfer techniques to manage larger quantities. On more than one occasion, I watched as spills led to weeks of paperwork and environmental reports, underlining how operational discipline heads off disasters large and small.
Application Areas
Not every reagent finds a second life outside the lab, but Hydroxyamine Hydrochloride stands apart. Chemical manufacturers drive up demand in textile dye synthesis, where its reduction powers unlock brilliant, durable colors. The pharmaceutical world leans on its ability to create specialized intermediates for antibiotics and anticancer drugs, all reliant on subtle modifications that depend on oxime formation or gentle reduction steps. Crop science draws on similar chemistry to shape modern pesticide molecules. Water treatment facilities use it in trace analysis of heavy metals, relying on its ability to form stable complexes. Even photo labs saw decades of use, where this compound played a quiet role in developing images on film — a detail remembered by anyone who spent their youth in a darkroom. Across these industries, changes in regulations, green chemistry pushes, and consumer safety concerns keep shifting how and where this chemical gets deployed. Today's demands edge toward lower emissions and biodegradable reagents, challenging suppliers to think beyond tradition.
Research and Development
R&D around Hydroxyamine Hydrochloride doesn’t just revolve around making more of the same. Academics and companies pour resources into finding safer variants, better catalysts, and greener synthesis pathways. Every major advance begins with understanding how its reactivity can cut down on waste, reduce toxicity, or make scaling up production more sustainable. In university research, groups double down on computational chemistry to predict what other reactions this molecule can mediate. I’ve seen firsthand how collaborations with toxicologists steer ongoing projects, asking tough questions about environmental persistence or unforeseen by-products. The dynamic between legislative shifts and scientific curiosity keeps this area moving, drawing in not just synthetic chemists but also environmental watchdogs and policy makers. New patents roll in each year aiming to tweak process efficiency, usually tied to energy savings, solvent reduction, or compatibility with renewable feedstocks.
Toxicity Research
Science doesn’t look away from the hazards tied to Hydroxyamine Hydrochloride. Studies show compelling evidence — skin sensitization, respiratory impact, and systemic toxicity at higher doses, urging every stakeholder to stay conscious of their handling protocols. Researchers map out the metabolic fate after exposure, narrowing down specific organ systems at risk and piecing together antidotes or medical responses for acute exposure. This line of investigation also affects regulatory stances on workplace exposure limits and informs the chemical labels you see in every lab. Across regulatory filings and scientific journals, the drumbeat is clear: minimize direct contact, control emissions, and implement engineering controls at every stage of the supply chain. Long-term toxicity data trigger debates about environmental discharge, pushing companies toward closed systems and improved waste processing technology.
Future Prospects
Looking forward, Hydroxyamine Hydrochloride faces a fork in the road. Demand surges with every innovation in polymers, electronics, and specialty pharmaceuticals, but the shadow of tighter regulation lingers overhead. Trends lean toward replacing it with less hazardous analogues wherever possible, especially as consumer and environmental safety draw more attention. At the same time, there’s renewed interest in refining its manufacture and use — think continuous flow reactors, green chemistry strategies, and hybrid systems that capture fugitive emissions. The market won’t shift instantly, but with collaboration between chemists, engineers, and regulatory specialists, real gains stand within reach. The challenge now isn’t just making Hydroxyamine Hydrochloride in bulk; it’s about making it smarter, safer, and with a lighter footprint, matching the pace of industries that refuse to stand still.
What is Hydroxyamine Hydrochloride used for?
Why Chemists Value This Compound
Hydroxyamine hydrochloride doesn’t show up in the news like new cancer drugs or fertilizer breakthroughs, but people who work in labs wouldn’t ignore it. For years, I saw it sitting on the shelf in almost every chemistry lab I entered. The label isn’t flashy, but the chemical inside plays a big part in making other reactions work. Most often, hydroxyamine hydrochloride acts as a reducing agent. This means it helps turn more complicated molecules into ones that chemists can use for building new medicines, materials, or dyes.
Role in Medicine and Drug Research
Few realize how important it can be in pharmaceutical research. Drug makers run lots of experiments where tiny adjustments to a chemical’s structure make the difference between a medicine that heals and one that harms. Hydroxyamine hydrochloride lets researchers test out those variations by helping turn one type of chemical group into another. For example, it converts aldehydes or ketones into oximes, which gives drug developers new options for building blocks. That kind of transformation shows up in research on antibiotics, hormone treatments, and even cancer medications. Recognizing the importance of solid building blocks in these discoveries reminds me of how much careful, invisible work leads to every new medicine on the market.
Testing for Industrial and Environmental Use
Outside the lab, hydroxyamine hydrochloride proves its worth in quality control and safety. It’s essential for detecting certain metals or contaminants in water and industrial products. During my time doing water-quality checks, this compound often helped confirm the presence or absence of nitrogen-containing chemicals, or helped measure iron levels. Getting fast, clear results mattered because people downstream depended on those numbers remaining safe. Plenty of environmental labs lean on it because it reacts strongly in specific, useful ways—making it easier to spot trouble and fix it before small problems turn into big ones.
Production of Polymers and Dyes
Many industries that create synthetic fibers rely on hydroxyamine hydrochloride to shape polymer materials with predictable strength and flexibility. In textile dye production, this substance plays a crucial part in turning raw materials into brighter, longer-lasting color. Watching manufacturing processes in action, I saw how a chemical’s reliability—like the steady results from using hydroxyamine hydrochloride—makes the difference between a batch that sells and one that fails. Consistent chemistry drives business, especially when designers and buyers expect high performance every time.
The Safety Factor
Almost anyone handling chemicals must pay attention to safety. Hydroxyamine hydrochloride carries warning labels for a reason—it can irritate the skin and cause reactions if mixed carelessly. I remember the push for better training and safety gear in labs once stories spread about careless spills. Following safety protocols, using gloves, and storing it properly matters just as much as knowing what it’s supposed to do in a reaction.
Room for Improvement
People in research and industry keep asking for safer, more sustainable chemicals. Hydroxyamine hydrochloride isn’t perfect. Some companies want to find alternatives that produce less waste or risk. Supporting greener chemistry means looking for new ways to accomplish the same tasks with a lower environmental impact. By encouraging young chemists and supporting open science, better solutions become more likely. That’s why learning about where and how this compound gets used is more than an academic question—it points the way to safer, smarter practices for everyone.
What are the safety precautions when handling Hydroxyamine Hydrochloride?
Understanding the Risks
Working with chemicals like Hydroxyamine Hydrochloride doesn't leave much room for shortcuts. This compound sees regular use in laboratories, industrial cleaners, and sometimes in pharmaceuticals. The risks linked to it get real fast. Breathing the dust can irritate the lungs, and skin contact can set off nasty burns. No matter how many years someone spends in the lab or on the shop floor, accidents happen most often when people let their guard down or skip protection gear, maybe just for a minute.
Staying Safe Starts with Planning
Staying safe means respecting the substance and sticking to well-known safety routines. Prepare the workspace so Hydroxyamine Hydrochloride never ends up near food, clutter, or anything flammable. This compound combusts with some organic materials, and if heating equipment is running or other chemicals are open, trouble finds a way in. Reliable lab ventilation — not just a cracked window — limits inhalation risks. In crowded labs or small shops, a working chemical hood makes a difference. I still remember a colleague struggling for breath from a spill caught too late — the right hood and mask would have saved her a trip to the hospital.
Personal Protection Gear Makes a Difference
Anyone handling Hydroxyamine Hydrochloride should never ignore personal protective equipment. Gloves made from nitrile or neoprene, safety goggles, a lab coat, and closed shoes are the basics. The skin on the face and neck needs shielding just like hands — a face shield or mask steps up protection. Disposable gloves protect, but regular checks for holes or tears cut down the odds of an accident. Chemical-resistant aprons help when handling larger amounts, and a full-face respirator comes into play if dust or fumes might get in the air.
Storage and Handling: Respect the Rules
Hydroxyamine Hydrochloride should stay sealed in its original container, away from heat, light, and moisture. Too many stories circle around warehouses where this chemical ended up on the wrong shelf, close to strong oxidizers or acids. That mistake triggers dangerous reactions. Spill kits and showers need to sit within easy reach, never buried behind equipment or stacks of boxes. The people who run the cleanest labs I’ve seen always keep emergency gear visible, tested, and ready.
What To Do If an Accident Happens
Even with all precautions, spills can happen. Scrambling doesn’t fix things — calm, clear procedures cut down the harm. Sweep up the powder carefully with tools, not bare hands, and never let the dust fly. Dispose of it in containers labeled for hazardous chemicals. Anyone who gets the powder on their skin should hit the emergency shower and wash off for at least fifteen minutes. Eyes exposed to any amount need an eyewash station right away. Acting early limits the damage.
Training and Updates Matter
Shortcuts cost more than the time they save. Proper training and up-to-date safety sheets save lives. In my experience, the strongest safety culture grows in places where new staff get hands-on demonstrations, regular review sessions happen, and everyone trusts each other to speak up about risks. It only takes one forgetful moment for Hydroxyamine Hydrochloride to cause real harm. Reliable safety demands respect for the risks — every shift, every person, every time.
What is the chemical formula of Hydroxyamine Hydrochloride?
Getting to Know Hydroxyamine Hydrochloride
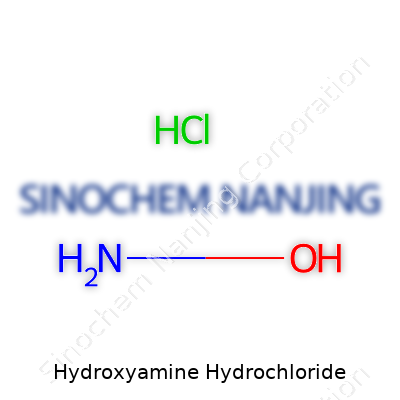
Hydroxyamine hydrochloride pops up often in laboratories, popping out of reagent bottles with the formula NH2OH·HCl. On paper, it combines hydroxylamine (NH2OH) and a molecule of hydrochloric acid (HCl) to create a white, crystalline solid. Put simply, it’s a salt, and that formula isn’t only for accuracy—it’s for safety, understanding, and smart decision-making behind the scenes.
Why the Formula Matters in Real Labs
Every time I measured out hydroxyamine hydrochloride, that formula feels heavy with meaning. A formula can look like a jumble of letters and numbers, but it actually determines much more: reactivity, storage, and how you should treat your own skin and eyes around it. For hydroxyamine hydrochloride, the NH2OH section means you’re dealing with a powerful reducing agent—one that’s good at stripping away oxygen atoms in reactions, making it a favorite in organic synthesis, explosives research, and analysis of minerals.
Clinging onto that hydrochloride bit means increased stability and ease of handling compared to pure hydroxylamine, which in the wrong hands, or without the right humidity, can decompose violently. The addition of HCl tames the substance, ensuring it stays safer on storage shelves and in chemistry classrooms. Anyone working in a real lab learns fast that proper labeling, knowing the chemical formula, and proper storage conditions aren’t formalities—they prevent fires, injuries, or worse.
Real-World Impact on Industry and Medicine
Hydroxyamine hydrochloride hasn’t stayed stuck in the lab. In my own experience with water analysis, this compound helps neutralize chlorine, allowing for accurate readings. Across industries, it steps into the scene for polymer production, rubber processing, and even pharmaceuticals. The pharmaceutical world values it for its role as an intermediate, pulling together critical nitrogen atoms for certain medications.
The proper use of NH2OH·HCl means fewer impurities and purer end-products. So, the formula reaches beyond paperwork, shaping the outcomes in hospitals and factories alike. Easier synthesis of clinical compounds can mean safer, cheaper medicine, which millions rely on. Getting these details right impacts patient outcomes, manufacturing costs, and our ability to scale research.
Safe Handling—No Shortcuts Allowed
Back in my early days, I saw what sloppy chemical handling looks like. Hydroxyamine hydrochloride deserves respect because exposure can lead to skin irritation, respiratory issues, and, if left unsupervised, violent decomposition. The chemical formula NH2OH·HCl lets trained eyes immediately understand how to store and neutralize spills—use plenty of ventilation, avoid high heat, and keep water or acid solutions nearby in case of emergencies. A good lab culture doesn’t skimp on safety training. It relies on professionals knowing both the name and the composition of what they’re handling.
Training should focus on recognizing these formulas and responding properly. Labs should invest in clear, accessible labeling, and safety information. Working from a solid knowledge base—rooted in real formulas and not mere chemical names—helps everyone go home safe and healthy after each shift.
Building Trust Through Knowledge
Transparency and knowledge-backed practices build trust in the chemical supply chain and research community. Fact-checking each reagent and demanding correct formulae on labels help avoid costly and dangerous mistakes. Academic and industry work both rest on a foundation of accurate formulas and reliable handling. That’s the sort of practical wisdom that keeps labs—and people—safe.
How should Hydroxyamine Hydrochloride be stored?
A Closer Look at Safety in Daily Practice
In the world of chemical storage, a slip-up rarely stays harmless. Hydroxyamine hydrochloride offers no exception. Lab veterans remember stories of unplanned reactions sparked by a careless jar left open, or the humidity in a storage closet. No one wants chemicals triggering a fire alarm. I learned early on, you treat hydroxyamine hydrochloride with respect or pay the price—storing it away from sunlight, heat, and incompatible chemicals is not just a suggestion; it’s a rule of survival. That granular powder looks innocent, but under the wrong conditions, it can decompose or even explode. Heat speeds things up in the worst way. Sunlight also raises risks, so it belongs deep inside a cool, dry storage cabinet.
Choosing the Right Container
Most science folks agree the best place for hydroxyamine hydrochloride is inside a tightly sealed container, untouched by air and water. Even if you’re in a rush, screw the lid back on—every time. Humidity will clump the powder and open the door to chemical breakdown. In my experience, plastic or glass vials made for chemicals do the job best. Paper bags or leaky boxes won’t stand up to the challenge, and they never keep the contents safe from spills or reactions.
The Importance of Separation
Hydroxyamine hydrochloride doesn’t mix well with oxidizers or bases. Years ago, I saw a cabinet sorted by the color of the labels, not their chemical risk. Thankfully, the worst that week was a mess, but things could have gone wrong fast. Storing this compound means keeping it away from strong acids, metal powders, or bleach. In shared spaces, look for a clearly marked section just for reducing agents like this one. Chemical compatibility charts might seem boring, but they stop fires and hospital trips.
Ventilation’s Role in Prevention
A tight room with no airflow can turn a mild spill into a major hazard. I always choose a spot with steady ventilation. Some labs invest in dedicated chemical storage rooms lined with steel and fitted with vents. At home, that’s overkill, but even a simple fume hood or a closet with a vented fan can make a real difference. Leaked fumes stay away from your face, and the risk of inhalation drops. It may mean a little hassle upfront, but better safe than coughing up regrets.
Disposal: Don’t Cut Corners
Cling to the same habits for disposal. Half-used bottles are tempting to toss in the trash, especially during cleaning sprees. But these chemicals shouldn’t end up in the municipal bin or go down the drain. Where I worked, the chemical waste container became our best friend. Used up or out-of-date hydroxyamine hydrochloride belongs in a sealed bag within secondary containment, labeled for hazardous waste pickup. Regulations might differ by city, but safety never takes a holiday.
Why Training Matters
Every good habit with hydroxyamine hydrochloride comes from real stories and experienced professionals. Training is not just for show—newcomers miss risky details if left alone. I was lucky to learn from mentors who explained not only what to do, but why it must be done that way. A reliable safety culture grows in places where staff correct each other, and no one treats shortcuts as good practice.
People who treat storage as just another chore invite trouble. Hydroxyamine hydrochloride rewards careful attention, and having the right habits, containers, and respect for its risks keeps people and workplaces safe.
What are the potential health hazards of Hydroxyamine Hydrochloride?
What Happens When Hydroxyamine Hydrochloride Enters the Workplace
Hydroxyamine hydrochloride shows up in labs, factories, and sometimes even in places where people don’t fully appreciate its hazards. I’ve seen seasoned workers reach for this white, powdery stuff without a second thought. It looks harmless, but anyone handling it regularly starts to notice: this chemical stings the nose, dries the skin, and if it spills on a bench, it leaves an odd smell for hours. Breathing in its dust turns a routine into a coughing fit. Rubbing itchy eyes after a mess-up? Forget about enjoying the rest of your shift.
Direct Exposure Tells the Real Story
Growing up in a family with a history of chemical manufacturing, I remember my father explaining how quick contact with certain chemicals could ruin your day. Hydroxyamine hydrochloride gives little warning beyond a faint tingle or burning sensation as it soaks in. If it touches bare skin, that spot gets red, irritated, and sometimes blistered. Those working in environments without proper gloves or eye protection see rashes and swelling all too often. Splashes to the eyes not only feel like a bee sting, but can also blur vision for hours or even cause damage if ignored.
Inhaling or Swallowing Isn’t Just Unpleasant—It’s Dangerous
A friend who once mixed this material for a summer job admitted he underestimated its bite. He barely inhaled more than a few dusty particles scooping it from a drum, but his throat closed up, his breathing turned shallow, and panic followed almost instantly. Swallowing even a pinch can trigger vomiting, stomach cramps, or worse – damage to internal organs. Hydroxyamine hydrochloride doesn’t just harm on the surface; long-term exposure may chip away at red blood cells, leading to a rare but serious blood disorder called methemoglobinemia. This steals oxygen from your blood, leaving you dizzy, weak, and pale. If exposure lasts for years, health problems only pile up.
Fire and Chemical Instability Raise the Stakes
People who store or transport this material know that it gets jumpy when jarred or exposed to heat. Imagine a routine equipment clean-up turning ugly because a trace of residue explodes into flames. Hydroxyamine hydrochloride not only catches fire; it also reacts violently with strong oxidizers and some metals. Without careful storage in cool, dry spots, even a small spill can release toxic fumes.
Prevention Starts With Real Awareness and Simple Habits
Familiarity sometimes breeds carelessness. Frustrated workers skip the hassle of goggles or let their gloves tear, simply because it feels faster. I always stuck to one rule: never let your guard down, especially with unpredictable chemicals like hydroxyamine hydrochloride. Ventilation keeps air moving and dust down. Double-checking containers for leaks and sticking to regular clean-up routines cuts down on accidents. Spill kits, safety showers, and eyewash stations save lives more than any half-hearted warning ever could.
Educating and Empowering Every Worker
Hazards shrink as knowledge grows. Safety data sheets should live in plain sight, not locked in a binder under a pile of papers. Real training, not just boring slideshows, helps workers recognize early symptoms and respond before things spiral. Healthcare professionals also deserve support with up-to-date facts for proper first aid or clinical care, since most have never seen a hydroxyamine hydrochloride exposure.
Better Policy, Smarter Design
Putting safer systems in place needs honest investment from company leaders. Engineering controls that box in hazardous dust, safer dispensing systems, and better emergency protocols spell the difference between minor scares and real tragedy. Regulators need to keep workplace exposure limits updated, and facilities must audit compliance more than once a year. If you build a culture that values safety, the odds of long-term harm from hydroxyamine hydrochloride drop fast.
| Names | |
| Preferred IUPAC name | hydroxylammonium chloride |
| Other names |
Oxyammonium chloride Hydroxylammonium chloride Hydroxylamine hydrochloride Hydroxyammonium chloride |
| Pronunciation | /haɪˌdrɒk.si.əˈmiːn haɪˌdrɒx.ləˈraɪd/ |
| Identifiers | |
| CAS Number | 5470-11-1 |
| 3D model (JSmol) | `/ajax/mol/jsmol.php?c=Cl.NO&l=fa` |
| Beilstein Reference | 1209227 |
| ChEBI | CHEBI:62972 |
| ChEMBL | CHEMBL1239 |
| ChemSpider | 6769 |
| DrugBank | DB11305 |
| ECHA InfoCard | 100.017.757 |
| EC Number | EC 200-612-2 |
| Gmelin Reference | 63912 |
| KEGG | C00547 |
| MeSH | D006927 |
| PubChem CID | 24250 |
| RTECS number | MX1225000 |
| UNII | E9AJ84A30Y |
| UN number | UN3260 |
| Properties | |
| Chemical formula | NH2OH·HCl |
| Molar mass | 69.49 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.67 g/cm³ |
| Solubility in water | Very soluble in water |
| log P | -2.2 |
| Vapor pressure | <0.01 mmHg (20°C) |
| Acidity (pKa) | 5.96 |
| Basicity (pKb) | pKb: 7.96 |
| Magnetic susceptibility (χ) | -49.0×10⁻⁶ cm³/mol |
| Viscosity | Viscous liquid |
| Dipole moment | 2.4 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 137.0 J K⁻¹ mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -41.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -93.0 kJ/mol |
| Pharmacology | |
| ATC code | V03AB38 |
| Hazards | |
| Main hazards | May be harmful if swallowed, inhaled, or absorbed through skin; causes burns; may cause allergic skin reaction; strong oxidizer; may cause fire or explosion; toxic to aquatic life. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| Precautionary statements | P210, P220, P221, P234, P260, P264, P270, P271, P273, P280, P283, P301+P312, P302+P352, P304+P340, P305+P351+P338, P306+P360, P312, P330, P337+P313, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Autoignition temperature | 292 °C |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 (oral, rat): 141 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 141 mg/kg |
| NIOSH | NT8050000 |
| PEL (Permissible) | PEL = 1 mg/m3 |
| REL (Recommended) | 10-50 mg/L |
| IDLH (Immediate danger) | 300 mg/m3 |
| Related compounds | |
| Related compounds |
Hydroxylamine Hydroxylamine-O-sulfonic acid Hydrazine Ammonium chloride Nitroxyl |

