Hydrazine Hydrate: A Double-Edged Chemical
Historical Development
Hydrazine hydrate has a history full of turns, both fascinating and sobering. Chemists first cooked up hydrazine compounds in the 19th century, chasing new ideas about chemical structure and reactivity. Curiosity drove relentless experimentation, and chemists like Theodor Curtius pushed the boundaries in the late 1800s to isolate hydrazine itself. Innovation in industrial chemistry followed soon after, with new processes like the Raschig method transforming small-scale curiosity into practical production by the early to mid-20th century. When fuel demand for rockets and industrial propulsion surged, hydrazine hydrate transformed from laboratory oddity to an industrial backbone, helping to tilt the balance in world wars and later forming a foundation for the space race. The chemical’s journey, from a glass flask to a launch pad, shows how new molecules don't stay in the lab for long—industry grabs hold as soon as the application turns from theory to practice.
Product Overview
Despite its complicated chemistry, hydrazine hydrate looks simple enough: a colorless, fuming liquid with a sharp, ammonia-like odor that signals danger to anyone with a decent nose. On the molecular level, this compound holds two nitrogen atoms chained together, capped with hydrogens and balanced by just enough water to keep it stable. Its reactivity brings both promise and peril, letting it serve as a raw chemical, propellant, reducing agent, and corrosion fighter across diverse sectors—pharmaceuticals, water treatment, energy, and aerospace among them. For all the technical talk, people using hydrazine hydrate worry about more than reaction yields; the risks, handling, and constant need for vigilance hover above every step in its chain from factory to end-use.
Physical & Chemical Properties
Hydrazine hydrate boils above that of water but remains desperately eager to react. Its density and viscosity challenge engineers, who must factor in not just transport safety, but simple measurement and transfer. It forms hydrogen bonds, giving it a higher boiling point than hydrazine alone and making it less volatile, though not enough to let anyone drop their guard. It dissolves freely in water, which explains its uses in aqueous solution form, but this solubility also means that any spill can move quickly in the environment. The compound also catches fire easily, and once it does, burns with a nearly invisible flame. These traits demanded that labs and plants develop strict internal ground rules, lest a minor misstep snowball into a costly accident.
Technical Specifications & Labeling
Regulators and manufacturers shaped rigid technical standards to control hydrazine hydrate at every point. Purity levels may run as high as 80% or more for demanding industrial and research purposes. Labels feature not just concentration but hazard information in clear language—otherwise, the risk of accidental injury or contamination rises. Real-world mistakes remind people to pay attention; what’s on the label may be the only warning separating safe handling from disaster. Vessel design, corrosion resistance, and the constant threat of leaks force plants to invest in stainless steel or specialized coatings just to keep storage and movement under control. Ignoring a purity check or relaxing operational discipline costs more than time—it puts lives and assets on the line.
Preparation Method
Chemical process engineers use the Raschig method to crank out hydrazine hydrate at scale. The process hinges on ammonium hypochlorite and ammonia, reacting them under controlled conditions to birth hydrazine, which then picks up water to form the hydrate. Scaling up, plants must counter runaway reactions, deal with byproducts, and wrangle the waste streams. Theoretically, other routes may exist—dehydrogenating urea or using hydrogen peroxide in place of hypochlorite—but the classic route wins because it fits existing infrastructure. These choices matter, because each pathway shapes operational costs, energy demand, and the waste customers must eventually dispose of.
Chemical Reactions & Modifications
Hydrazine hydrate doesn’t sit still. Its strong reducing power lets it strip oxygen, convert metals, or neutralize potent chemicals, which turns it into a tool for synthesis, water treatment, and extraction. Chemists use it to crack open complex molecules or keep unwanted ions in check. Modifications, including turning it into derivatives like methylhydrazine or phenylhydrazine, open fresh doors—all while keeping the hazard profile in mind. In pharmaceuticals, for instance, its role in assembling anti-tubercular drugs stands as an example of how ancient reactivity found modern relevance. The trick lies in exploiting its gifts while building enough fail-safes that its presence doesn’t dominate risk assessments or emergency drills.
Synonyms & Product Names
Hydrazine hydrate shows up under a surprising variety of names, both in industry catalogs and regulatory circles: hydrazine monohydrate, hydrated hydrazine, and sometimes masked by trade names depending on the brand or distributor. This mix can trap newcomers. Failure to recognize these synonyms can land an order for the wrong grade or lead to unfortunate mixing mishaps, especially when someone assumes two products named differently hold different hazards. Clarity in communication and training beats out even the most comprehensive labeling standards on paper.
Safety & Operational Standards
Every engineer and lab worker who has had a spill or near-miss with hydrazine hydrate remembers the lesson. Its acute toxicity sinks in fast, causing skin burns and lung damage within minutes if not handled right. Chronic exposures have long-term health implications, including proven carcinogenicity. That knowledge drove industry adoption of strict safety standards—full-face shields, chemical-resistant gloves, and positive air pressure suits in high risk environments. Automated transfer systems, remote monitoring, and exhaustive ventilation systems all count as must-haves, not luxuries. Auditors check not just paperwork, but real conditions in storage and use areas, because weak safety cultures signal inevitable accidents. Training and accountability prove just as critical as hardware, especially with personnel turnover and tight schedules tempting shortcuts.
Application Area
Hydrazine hydrate finds work in some of the highest-stakes sectors. As a propellant ingredient, it has fueled everything from fighter jets to satellites; as a treatment chemical, it strips dissolved oxygen from water in power plants, protecting boilers and turbines from corrosion. Chemical manufacturers lean on it as a reducing agent for pharmaceutical synthesis, photography processing, and even in agriculture for fungicide production. Wastewater plants appreciate its reactivity for breaking down pollutants. Each of these uses brings checks and balances: industrial hygiene protocols and strict disposal regulations replace the early days of casual handling. People remember both market opportunity and public safety, since one accident can damage public trust much faster than any marketing campaign can rebuild it.
Research & Development
Innovation in hydrazine hydrate chemistry has not slowed. Scientists and engineers keep searching for greener synthesis routes with less waste and safer alternatives that might someday replace hydrazine in sensitive roles like propellants. Computational chemistry speeds up the hunt for derivatives with lower toxicity or improved energy density, especially for next-generation fuel cells. Research groups at major chemical institutes and aerospace agencies keep testing boundaries, tweaking the molecule’s structure and testing new blends. Results don’t follow a neatly predictable path; each positive step forces careful study of down-stream consequences, whether environmental, economic, or regulatory. Many labs limit access to hydrazine hydrate because risks outweigh potential gains in less-experienced hands.
Toxicity Research
No responsible commentary on hydrazine hydrate skips its toxicity. Decades of animal studies and accident reports catalog the risks: skin burns, respiratory harm, liver and kidney injury, DNA damage, and carcinogenicity. Regulatory bodies name it as a probable human carcinogen and enforce strict exposure limits in the workplace. People exposed during accidents remember the vomiting, headaches, and rashes long after cleanup. Modern toxicity research focuses not just on direct health effects, but secondary impacts, such as the compound’s breakdown products or persistence in ecosystems. Surveillance data, case reports, and controlled exposure trials all drive policymakers to keep tightening rules. Industry must stay nimble, since changes to hazard classifications force redesign of safety systems and retraining—sometimes at substantial cost but always justified by the potential for catastrophic health outcomes.
Future Prospects
As long as the world demands high-performance fuels, fast-reacting reducing agents, and robust water treatment solutions, hydrazine hydrate will stick around. Regulatory scrutiny grows tighter every year, with new rules forcing companies to invest in safety or look for alternatives. The push for greener chemistry and sustainable industrial practices presses the sector to develop less toxic replacements—sometimes borrowing from the same spirit of discovery that made hydrazine hydrate a staple a century ago. Advances in automation, continuous monitoring, and protective equipment raise the bar for safe handling, making accidents less common but not yet impossible. My experience in industrial labs says risk never leaves—people must keep teaching new generations to respect both the power and peril of hydrazine hydrate, balancing innovation with caution at every turn. If the sector rises to that challenge, the chemical’s controversial legacy will sit as a warning, not a verdict.
What is Hydrazine Hydrate used for?
A Chemical With Serious Reach
Hydrazine hydrate is not the sort of compound you hear about all the time. Yet, every time I walk past a rocket launch broadcast on TV, I’m reminded of its significance. Hydrazine hydrate plays a critical part in fuel technology. It’s powerful, reactive, and definitely not the safest thing to handle. Still, it drives some of the most complicated machines ever built.
Space Exploration: More Than Just Thrust
Most people know hydrazine hydrate as a key ingredient in rocket fuel. I remember first learning that spacecraft, from satellites to Mars rovers, rely on hydrazine for propulsion and maneuvering. Nothing else offers that same punch in such a compact form. NASA has leaned on hydrazine to correct satellite orbits and to get probes into the precise spots needed to explore other planets.
Looking closer, it’s clear why hydrazine remains popular among aerospace engineers. This liquid handles low temperatures well, stores efficiently, and, more importantly, doesn’t wait for special ignition systems to react—just an oxidizer. I’ve read about the careful protocols developed to store and transport the stuff because, left unchecked, it’s toxic and can pose a real safety threat to anyone nearby.
Keeping Industry Running
Outside space, hydrazine hydrate keeps a low but steady profile in several essential industries. Take water treatment, for example. Power plants need clean water running in their boilers, or they risk expensive repairs and loss of output. Hydrazine hydrate’s role as an oxygen scavenger helps stop corrosion in those high-pressure environments. There aren’t too many chemicals out there that can snatch up dissolved oxygen as cleanly or quickly.
I’ve also seen hydrazine hydrate at work in pharmaceuticals. Some blood pressure drugs and tuberculosis treatments wouldn’t exist without intermediate chemicals—often linked back to hydrazine hydrate. This single ingredient connects to entire branches of treatments that have helped millions lead healthier lives.
Taking Care: Safety and Environmental Concerns
Handling hydrazine hydrate never comes easy. This compound burns skin on contact, and the vapors cause nausea and headaches. There’s no wiggle room for mistakes with hydrazine hydrate, as most chemical engineers will tell you. The need for airtight suits, gloves, and goggles goes beyond simple workplace safety—it becomes a matter of survival.
Environmental concerns also loom large. Hydrazine doesn’t break down in soil or water overnight. Regulators require strict controls to prevent spills near water sources and during transportation. The EPA lists hydrazine as a probable carcinogen, pushing scientists and manufacturers to search for cleaner alternatives, especially for rocket launches. Years working around hazardous materials have taught me that prevention beats cleanup every time, and hydrazine hydrate only reinforces that lesson.
What Lies Ahead?
There are research efforts betting on “green propellants” to replace hydrazine in space missions. Some private companies have won contracts to test new formulas that promise better safety with less environmental harm. It’s not just about replacing what works; it’s about making sure the costs—human and environmental—don’t spiral out of control. Whether in a power station or a launchpad, hydrazine hydrate’s story keeps evolving. Until we land on something safer and just as effective, its legacy will carry on, for better or worse.
Is Hydrazine Hydrate hazardous?
Recognizing What Hydrazine Hydrate Means in Daily Terms
Hydrazine hydrate turns up in settings as ordinary as a college chemistry lab and as specialized as a rocket launch site. My own exposure to it started in a dusty university lab, watching an instructor stress again and again about gloves, goggles, and the strict need to work within a fume hood. The reason became clear pretty quickly: hydrazine hydrate isn’t something people want on their skin or in the air they breathe.
Why Hydrazine Hydrate Raises Flags
Even people who haven’t studied chemistry might remember the word “toxic” attached to hydrazine. That label sticks for good reason. Contact can cause burns and blisters. Vapors irritate eyes and lungs. Long-term or repeated exposure can knock the liver and kidneys for a loop. Cancer risk isn’t just a whisper; both the International Agency for Research on Cancer (IARC) and the U.S. Environmental Protection Agency suggest hydrazine is probably carcinogenic to humans.
Safety data sheets and regulatory bodies lay down heavy precautions for a reason. Fume hoods and respirators aren’t accessories in lab coats—they’re lifelines. Think about the Bhopal disaster’s echo through industrial communities; chemical leaks turn entire neighborhoods upside down. Workers in facilities handling hydrazine hydrate rely on these protections every day. The substance also finds itself on the U.S. Occupational Safety and Health Administration’s roster of regulated hazardous chemicals, showing that this risk isn’t tucked away in academic journals but woven into workplace rules.
Why Do We Keep Using It?
Despite the dangers, hydrazine hydrate keeps showing up in industries like water treatment and fuel production, plus niche areas such as pharmaceuticals and space programs. It works well as an oxygen scavenger in power plant boilers, and space agencies use it as rocket fuel propellant. Alternatives exist, but often fall short in terms of cost, performance, or reliability. There’s an irony in using something so risky to keep water clean or put satellites in orbit, but the practicality drives decision-making.
Managing Risks Without Overlooking the Facts
In my time visiting chemical plants, it was clear: strong rules make a difference. Spill protocols, constant air monitoring, quality gear, and good hygiene practices save both lives and business reputations. Many facilities double up on leak detection and backup plans, knowing that a single mistake carries heavy consequences.
Public transparency also plays a big part. Communities living near plants deserve to know what’s in their air and water. Programs for regular information sharing help build trust. Some states enforce “right-to-know” policies, so anyone can read about chemical storage and emergency plans.
Looking Ahead: Can Anything Replace Hydrazine Hydrate?
Innovation works slowly in chemical manufacturing. Researchers chase safer substitutes, especially for boilers and rocket fuel. Some replacements already improve worker safety or environmental impact, but complete swaps remain rare. Scaling up alternatives takes investment and careful validation—nobody wants shortcuts where safety is at stake.
In the meantime, thorough training, updated equipment, regular audits, and emergency response drills offer the best defense against accidents. Hydrazine hydrate requires real respect, not just from chemists and engineers, but from anyone sharing the space with this potent chemical.
How should Hydrazine Hydrate be stored?
Real-World Hazards Demand Smart Choices
Every chemist or engineer who has handled hydrazine hydrate knows the stuff isn’t forgiving. It’s a colorless liquid, strong-smelling, highly reactive, and volatile. Even with decent chemical knowledge, it’s easy to underestimate the risks it brings – slippery hands, a fume here or a drop there, and the stakes rise quickly. Stories have circulated about severe burns and violent fumes just from a moment’s inattention. Add in its toxic, potentially carcinogenic nature, and the need for good storage isn’t just technical talk—it’s basic workplace safety.
Why Hydrazine Hydrate Turns Dangerous
This compound reacts with air, metals, and ordinary chemical containers. Light kicks off decomposition, while heat boosts vaporization, increasing the likelihood of leaks. Contaminants—dirt, metal ions, even moisture—set off rapid, unpredictable chemistry, sometimes with explosive results. Once, a colleague in a poorly ventilated environment popped open a container for a routine check. The sharp, ammonia-like tang filled the room and his mask didn’t stand a chance. That day stuck with the entire team: safeguards aren’t for show.
Proven Storage Approaches
Industry handles hydrazine hydrate by turning to reliability. Storage happens in tightly-sealed, corrosion-resistant containers. Stainless steel or certain plastics like HDPE do the job much better than ordinary metals, since common containers can trigger unwanted reactions or pitting. The container’s seal means more than just leak prevention—it blocks out air and moisture. Fumes escape easily, contaminating air and creating inhalation dangers. Good sealing keeps these problems out of mind and out of the workspace.
Those containers don’t sit on just any shelf. They belong in cool, shaded storage areas, separated from direct sunlight, steam pipes, or any spot where heat might build up over time. Exposure to warmth makes decomposition more likely and increases the odds of vapor leaks. In my own lab experience, someone once left a small drum near a window. Sunlight crept over, the drum heated up, and by lunchtime, vapor built so much pressure that it compromised the cap, resulting in a costly cleanup.
Safety Protocols Backed by Evidence
Emergency gear—spill containment, eyewash stations, gloves, goggles—forms the first line of defense. No shortcut makes sense here. Material Safety Data Sheets from global suppliers say hydrazine exposure can cause severe skin and eye damage, respiratory distress, and long-term health effects. These aren’t just theoretical. Documented workplace accidents lead to hospital visits and expensive regulatory trouble.
Fire hazards sit at the top of the risk ladder. Hydrazine hydrate burns with a nearly invisible flame, so smoke detectors and automatic suppression systems play a key role. Routine checks for leaks, corrosion, and temperature changes in storage areas give a heads-up before small problems grow. Mixing with oxidizers, acids, or even rusty tools creates a chain reaction—one accident at Texas City involved improper separation and resulted in a fire lasting days.
The Solution: Rely on Rigor, Not Luck
Simple fixes save lives. Label containers clearly, keep incompatible chemicals away, and educate every handler—seasoned chemists and newcomers alike. Invest in storage areas with proper ventilation, sturdy secondary containment to catch spills, and regular inspections. Hydrazine hydrate punishes carelessness. Rigorous storage and handling practices respect its risks and give everyone a better chance at safe, incident-free work.
What is the chemical formula of Hydrazine Hydrate?
Why Chemical Formulas Matter in Chemistry
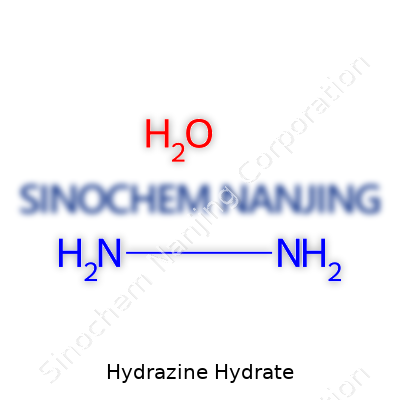
Every chemist remembers the first time they cracked open a textbook, staring down at a jumble of letters and numbers that looked more like a secret code than a solution to real-life problems. Chemical formulas look simple, but they pack information that shapes how compounds are handled in labs and factories. Hydrazine hydrate shows this perfectly. Its formula—N2H4·H2O—looks like a dry string of elements, but so much about workplace safety, transportation, and research rest on getting these formulas right.
The Building Blocks of Hydrazine Hydrate
Hydrazine hydrate comes from combining hydrazine, a well-known reducing agent, with water. This forms a colorless, nearly clear liquid that usually comes in sealed drums marked dangerous. The chemistry behind the formula N2H4·H2O tells the story: one molecule of hydrazine joins with one molecule of water. See those subscripts? They count the atoms—two nitrogens, four hydrogens for hydrazine, two hydrogens and one oxygen for water. This simple math shapes everything from how much heat the compound releases to its tendency to react violently with oxidizers.
Why Industry Depends on Hydrazine Hydrate
I spent years in laboratory settings and remember hydrazine hydrate for its sharp, ammonia-like smell and the nervous care technicians showed each time they unscrewed the bottle. Hydrazine hydrate plays an essential part in chemical manufacturing—blowing up in the news sometimes for its role in rocket fuel, though most of it powers much less glamorous work. Chemical engineers rely on it for making foaming agents in plastics, cleaning boiler water in power plants, and prepping pharmaceuticals or agricultural chemicals. It’s not the sort of compound you’d find in a high school lab drawer, but scale up to an industrial plant, and the importance of getting the formula correct becomes crystal clear.
Why Safety Hinges on a Simple Formula
The dangers of hydrazine hydrate make headlines for good reason. Toxicity ranks high. Inhaling even small amounts can cause headaches, nausea, and in large doses, damage to vital organs. Its corrosiveness burns skin and eyes. In factories, the drum labels shout warnings, and safety data sheets spell out protocols in tedious detail. Emergency response teams learn these formulas, so they know exactly what to do if a spill hits the floor. I have seen teams rehearse this: gloves, goggles, masks on, working from real chemical spills. Those high-stakes drills focus on compounds like hydrazine hydrate precisely because its formula tells them how it will behave, what neutralizes it, and why water alone sometimes won’t help.
Supporting Safe Use Through Knowledge and Practice
Most of the time, protecting people and the environment depends on accurate, up-to-date information. In my years working in research and regulation, a recurring lesson was to double-check everything, especially the formulas behind high-risk chemicals. Anyone studying, handling, or shipping hydrazine hydrate needs to understand what N2H4·H2O means beyond paper—the heat it might release, the vapor it gives off, the way it can react with common materials. Good training, clear protocols, and trustworthy suppliers all tie directly back to this knowledge. It’s a reminder that chemistry starts with what’s written on the bottle, and grows from there to touch safety, the environment, and countless lives worldwide.
What precautions are required when handling Hydrazine Hydrate?
Understanding the Danger
Hydrazine Hydrate isn’t something you spot on a grocery shelf or handle like a regular cleaning agent. This chemical carries real risks. Most people don’t know much about it unless they’ve worked in a lab or one of those specialized industries—think rocket fuels, pharmaceuticals, water treatment, or some plastics manufacturing. In my time working with engineering teams, stories about mishandling chemicals have always rung alarm bells. Hydrazine Hydrate gets a bad reputation for a reason.
Health Risks on the Line
Even short contact can irritate skin or eyes, and the vapor can hit lungs hard. Exposure can feel like a headache or cause nausea first, but the danger doesn’t stop there. Prolonged or high-level contact raises cancer risk, damages liver and kidneys, and leaves lasting respiratory problems. These facts come straight from studies published by organizations like the Centers for Disease Control and the International Agency for Research on Cancer.
Protective Gear: Not Optional
Over the years, I learned to respect what a good pair of chemical-splash goggles and a decent respirator can do. For tasks that toss fumes around, a full-face shield beats just safety glasses. Gloves made from a strong material—like butyl rubber—keep the stuff off your skin. I’ve seen co-workers reach for the wrong gloves and end up regretting it. Chemical-resistant aprons, lab coats, or full body suits make sense if spills seem even remotely possible.
Ventilation Changes Everything
A fume hood or chemical exhaust system goes from “nice to have” to non-negotiable in rooms using Hydrazine Hydrate. Vapors float into the air quicker than expected, spreading through a whole room in no time. Just opening a window isn’t enough. Industrial fans and closed ventilation systems keep the work area safer for longer periods and for everyone, not just the person doing the handling.
Storage: Tight, Locked, Labeled
Leaving Hydrazine Hydrate on a bench or next to acids counts as asking for trouble. This chemical catches fire easily and reacts badly with lots of regular lab items. I always check that containers have tight-fitting lids and get stored in a spot away from sunlight and sources of heat. Good storage means locked acids cabinet, clear hazard labels, and only keeping as much as you need. It seems like overkill—until something leaks or a label gets smudged.
Training and Emergency Response
In every place where I worked, proper training wasn’t just a formality—it was common sense. People who regularly deal with Hydrazine Hydrate need drills for spills and clear steps for medical emergencies. I remember drills where someone had to shout out for the eyewash station blindfolded to practice in case of chemical splashes. Emergency kits don’t fix every mistake, but they make the difference when the clock is ticking. Regular checks of safety showers and first aid stations should be part of the routine.
Better Habits Build Up Over Time
No one gets chemical safety perfect on the first round. Habits stick by practicing, staying curious, and keeping safety talks short and real. The more you know about why protocols matter, the less likely you are to cut corners—the stakes with Hydrazine Hydrate leave no room for error. It isn’t about being paranoid; it’s knowing each step means protecting your health, your team, and your workplace.
| Names | |
| Preferred IUPAC name | Hydrazine monohydrate |
| Other names |
Hydrazine Monohydrate Hydroazanium hydroxide Diamidogen oxide N2H4·H2O |
| Pronunciation | /haɪˈdræziːn ˈhaɪdreɪt/ |
| Identifiers | |
| CAS Number | 7803-57-8 |
| Beilstein Reference | 4036807 |
| ChEBI | CHEBI:38757 |
| ChEMBL | CHEMBL1235727 |
| ChemSpider | 7556 |
| DrugBank | DB01382 |
| ECHA InfoCard | EU0000190774 |
| EC Number | 206-114-9 |
| Gmelin Reference | Gmelin 973 |
| KEGG | C00228 |
| MeSH | D006730 |
| PubChem CID | 783 |
| RTECS number | MV8050000 |
| UNII | K50XQU1029 |
| UN number | UN2030 |
| Properties | |
| Chemical formula | N2H4·H2O |
| Molar mass | 50.06 g/mol |
| Appearance | Colorless, fuming, oily liquid with an ammonia-like odor |
| Odor | Ammonia-like |
| Density | 1.03 g/cm³ |
| Solubility in water | Miscible |
| log P | -1.48 |
| Vapor pressure | 13.3 mmHg (20 °C) |
| Acidity (pKa) | 7.98 |
| Basicity (pKb) | pKb: 5.89 |
| Magnetic susceptibility (χ) | +0.7e-6 |
| Refractive index (nD) | 1.418 |
| Viscosity | Viscosity: 0.99 mPa·s (at 25 °C) |
| Dipole moment | 1.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 121.65 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −133.7 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -417.5 kJ/mol |
| Pharmacology | |
| ATC code | V60X |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes severe skin burns and eye damage; may cause an allergic skin reaction; may cause cancer; suspected of causing genetic defects; very toxic to aquatic life. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H225, H260, H301, H311, H331, H314, H317, H341, H350 |
| Precautionary statements | P210, P260, P261, P280, P301+P310, P303+P361+P353, P304+P340, P311, P320, P330, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | 52 °C |
| Autoignition temperature | 270°C (518°F) |
| Explosive limits | 4.7–100% |
| Lethal dose or concentration | LD50 oral rat 60 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat: 60 mg/kg |
| NIOSH | K681 |
| PEL (Permissible) | PEL = 1 ppm (as Hydrazine) |
| REL (Recommended) | 30 mg/m³ |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Hydrazine Hydrazinium sulfate Monomethylhydrazine Unsymmetrical dimethylhydrazine |

