Hexachloroethane: More Than Just a Chemical Name
Historical Development
Hexachloroethane popped up in chemistry labs early in the 1900s. Back then, a growing hunger for new fuels, propellants, and pyrotechnics took root with war-era industry. Researchers explored chlorinated organics, and hexachloroethane drew attention. Its role during the two world wars marked a turning point: the military found it useful in smoke grenades and fogs. That wasn’t by accident. Combustion of hexachloroethane pumped out thick, white smoke, giving soldiers cover on the battlefield. While that may sound dramatic, the move from battlefield curiosity to industrial workhorse came later, shaped by real-life need and experimentation far from any idealized lab bench.
Broader Product Overview
These days, few outside certain fields recognize hexachloroethane as a household name. For the folks who work with it—pyrotechnics specialists, water treatment operators, metal foundries—it’s a regular tool. What you get is a white crystalline solid, with a distinctive, almost camphor-like odor. It gets packed into smoke formulations and used for degassing aluminum. Pool operators know its connection to making some water sanitization products. The general population brushes up against its effects without knowing, during sporting events with colored smoke or when certain pesticide residues end up in the food chain. Real-world applications matter most, so I’ll focus on those instead of anything purely theoretical.
Physical & Chemical Properties
Hexachloroethane melts just above room temperature and evaporates slowly. It isn't soluble in water—yet dissolves in fat or common organic solvents, making cleanup and environmental fate a headache. The molecules stack tightly thanks to the bulky chlorine atoms, which handle heat but break down under a strong enough flame, producing all sorts of byproducts, many bad for the lungs and environment. The solid persists in the environment, resists breaking down under normal conditions, and this durability is one reason it attracted attention when considering long-term safety and industrial hygiene.
Labeling, Technical Specs & Preparation Method
Anyone who has handled a drum of hexachloroethane has seen the warning labels: skull and crossbones for toxicity, the environmental hazard tree, and sharp guidance around ventilation. In the real world, standards for labelling and purity protect the downstream user. Manufacturing often starts with chlorination of tetrachloroethene, using chlorine gas under high pressure. Commercial suppliers tune parameters to balance yield with purity, keenly aware that trace byproducts carry their own dangers and regulatory baggage. The resulting product finds packaging in tightly sealed containers to block out air and water—and to keep its fumes from spreading.
Chemical Reactions & Modifications
The chemistry isn’t flashy but hits hard. Hexachloroethane reacts readily with pulverized metals. That single step gave rise to its broad use in refining aluminum, where it’s chucked in to scavenge unwanted hydrogen and metallic impurities. Add heat, and it splits off chlorine—in smoke grenades, this combines with zinc to make clouds of zinc chloride, which gather into fine aerosols. Tinkerers and researchers alike chased modifications, hoping to dial down the risk by swapping out some heavy chlorines or baking in molecular tweaks, but the core structure holds steady against much interference.
Synonyms & Product Names
Anyone poking around technical papers or safety data sheets will run across a few alternate names. "Ethane, hexachloro-" or the shorthand "HCE" pop up often. Trade names range depending on supplier, giving the same core molecule, occasionally tweaked for the application. Navigating the minefield of synonyms isn’t just a quirk of chemistry; it matters for safety compliance since different industries file it under varying headings.
Safety & Operational Standards
Direct exposure to hexachloroethane is far from trivial. My experience with chemical storage and handling means that I instinctively double-glove and double-check ventilation. Acute effects land squarely in the respiratory system—cough, dizziness, sometimes hitting the liver and kidneys. For decades, there was a tendency to downplay these risks, especially for military field use. Today, the tide has shifted: OSHA, the EPA, and European authorities insist on minimum exposure standards and strict control of effluents. At the very least, companies now dedicate serious investment to worksite monitors, personal protective equipment, and contingency plans for spills.
Application Area
Factories wedge hexachloroethane into obscure but vital corners of production—metal degassing, smoke munition fillers, and even specialty rubber compounding. Pool maintenance crews encounter it indirectly through its role in making disinfectants. In the past, agriculture toyed with it as a pesticide, but mounting safety worries stopped that line cold in many regions. Foundries, pyrotechnicians, and defense contractors make up the core repeat customers. General consumers experience its effects only at a distance, maybe cheering on a football team while colored smoke drifts from the stadium.
Research & Development
Work on hexachloroethane keeps shifting alongside new regulations and environmental findings. Researchers dig into ways to replace it with less persistent alternatives, especially in aluminum refining and military-grade smoke. Attempts to capture or neutralize its breakdown products keep labs busy, since accidental releases mean environmental oversight or costly clean-up. Academics tracking chemical fate often start with this molecule, tracing where the chlorine goes and how it sticks around in soil or water. For regulation veterans, it's a game of chasing the real risk down every rabbit hole: exposure monitoring, safer substitutes, or new formulations for smoke grenades that don’t punch holes in the ozone.
Toxicity Research
The science around hexachloroethane’s toxicity leaves few doubts. Animal studies link it to organ damage and possible carcinogenic effects at high doses. Workers in old-school metal shops sometimes paid the price, showing up with liver function abnormalities or lasting lung problems. Environmental researchers cite troubling persistence and bioaccumulation in wildlife, urging an exit plan for its continued use. Public health campaigns and scientific advocacy groups press for stricter standards, reflecting a consensus that risks outweigh convenience in many existing uses. My own engagement with occupational health teams taught me that “safe enough” never means safe for everyone; vigilance continues to trump complacency.
Future Prospects
Outlook for hexachloroethane stays cloudy. Metal casting and pyrotechnics still count on its unique chemistry, but pressure mounts to ditch it in favor of safer, greener compounds. Politicians, regulators, and advocacy groups won’t let up, demanding transparent reporting, tighter limits, and more robust alternatives. R&D teams inside chemical giants know change isn’t just coming—it’s necessary, with dollars shifting from status quo products toward the next generation. In another generation, I expect most of its military and civilian uses will look old-fashioned, replaced by sharper, less harmful molecules. Pushing this transition means keeping open lines between industry, academia, and the people living next door to the factory fence. Stubbornness serves nobody when safer paths can be found—and shared—by everyone.
What is hexachloroethane used for?
Hexachloroethane’s Place in Industrial Life
People tend to ask what hexachloroethane is used for because it seems to float on the edge of industrial relevance—old-fashioned, yet still present. This grey-white solid showed up often in metallurgical work during the twentieth century. Factories, especially those dealing with metals like aluminum and magnesium, used hexachloroethane to degas molten metals. The chemical helped workers strip away unwanted hydrogen gas by turning it into bubbles that could be scraped off or let rise up and out. The goal was to produce cleaner billets and castings since hydrogen pockets will weaken finished metals.
Welders, particularly in naval yards, grew up working with hexachloroethane in smoke or signaling grenades. You’d toss a disk or chunk into a device, set it off, and the heavy smoke would cover troop movements or help mark rescue zones. For decades, militaries sourced tons of it for those purposes. By the time I worked part-time in a hardware store during university, hexachloroethane was something we’d see listed on old safety sheets whenever guys bought foundry kits.
Looking at Health and Safety Concerns
We can’t talk about chemicals like this without bringing up safety. Over time, it became clearer that this stuff is no friend to the lungs, kidneys, or liver. Inhaling the smoke or dust, even during tests, left workers with headaches, nausea, and much worse if exposed for long stretches. There are case reports: Navy crew on amphibious craft had their lungs damaged after using hexachloroethane grenades in confined spaces. Researchers found links between exposure and liver enzyme changes in animal studies. The U.S. Environmental Protection Agency points to risks for people breathing air near demolition training—or neighbors downwind from unregulated scrap metal recycling.
Regulators in Europe and North America have pushed hexachloroethane off the main stage. You simply can’t import or sell it for most non-industrial uses in the EU and Canada. Factories in the U.S. replaced it with other degassing agents, or retooled manufacturing to need less of it. Workers today have access to better respirators, hazard controls, and—overall—less direct contact.
Switching to Safer Alternatives
If a foundry still uses hexachloroethane, you can bet cost drives the decision. In my experience, old-school shop managers sometimes don’t want to swap out what has “always worked.” Yet, plenty of companies now favor argon or nitrogen to do the same job—no toxic cleanup required, no health scares for the neighborhood. Welding shops can buy signaling pyrotechnics using safer, less persistent chemicals—ones that don’t stick around in soil and runoff.
The path forward: modernize where possible, invest in equipment upgrades, and listen to workers who know firsthand what chemical exposure feels like. Hazard training can’t be an afterthought. Companies should revisit their Material Safety Data Sheets and look beyond just legal compliance—public health matters, and communities read news about chemical exposure more closely than ever.
The Role of Accountability
Research and dialogue are key. Industries should team up with public health scientists, not just government inspectors. Sharing data on contamination, reporting near-misses, and inviting worker feedback do more for safety than secretive management ever could. If a factory still relies on hexachloroethane, be transparent about why, and plan the switch out loud. Every workplace owes that to its team and to the public.
Is hexachloroethane hazardous to health?
What People Should Know About Hexachloroethane
Hexachloroethane always sounded to me like one of those chemicals you’d find in an old science class, tucked behind glass, never mentioned again. Yet, this chemical finds its way into metal manufacturing, smoke grenades, and even some pyrotechnic devices. People tend to forget these substances don’t disappear after use. They often stick around in air, soil, or even make their way into water. I’ve worked around industrial sites and saw firsthand how little most workers talk about long-term health risks despite direct exposure.
Health Hazards: What Research Says
Breathing even moderate amounts can cause throat irritation and coughing. If you handle or inhale large amounts, things get worse: headaches, dizziness, and sometimes more severe problems with heart or liver function. The U.S. Environmental Protection Agency lists hexachloroethane as a possible human carcinogen. That doesn’t mean it’ll always cause cancer, but it’s a strong warning sign. I remember reading a study where repeated exposure in animals led to kidney and liver tumors. My first thought was always: If animals showed clear damage, humans probably aren’t immune.
Looking at real-world cases, military and industrial workers exposed in the past have shown liver and kidney issues. Risks don’t just come from breathing fumes at work. Sometimes contaminated groundwater in communities near plants turns safety upside down for families living nowhere near a factory floor. Kids and elderly folks can’t handle these chemicals running through their bodies. Every chemical that builds up in tissues carries hidden costs.
Regulation and Real-World Oversight
Government agencies recognize the dangers. The Occupational Safety and Health Administration (OSHA) set strict limits for workplace exposure. An average person can’t see or smell this stuff, so rules and safety training become the first line of defense. Still, from what I’ve seen on site, safety gear gets neglected, especially in high-heat settings, or when deadlines push people to cut corners.
Plenty of companies now look for safer alternatives, especially in pyrotechnics and aluminum casting. Military suppliers have switched out old smoke compositions for those without hexachloroethane after health complaints from soldiers. Responsible industry should take a cue from that shift. If safer options exist, use them. It shouldn’t take lawsuits or employee illness to spark change.
Personal and Community Responsibility
Safe handling and storage matter at every stage—factory, shipping, and disposal. I’ve talked with local parents panicked after reading water quality reports. Communities deserve transparency about what’s in their air and water. Regular testing, public disclosure, and real enforcement—these steps build trust. Homeowners living near industrial sites shouldn’t have to hire their own testers to understand health risks. I’d like to see community clinics given resources to test for these chemicals when someone shows symptoms out of the blue.
Moving Forward
Hexachloroethane’s risks show up in facts, not just chemical reports: higher odds of cancer, organ damage, and threats to new generations. Governments, businesses, and everyday people share the job of minimizing those risks. Putting health before convenience or profit usually saves money and lives down the road. Safer workspaces and cleaner neighborhoods are impossible if people turn away from uncomfortable facts.
Anyone can ask questions—workers, neighbors, parents. The plain truth never harmed anyone as much as ignorance or secrecy about what chemicals linger in the background.
How should hexachloroethane be stored safely?
Why Storage Matters
Hexachloroethane isn’t some run-of-the-mill substance you can throw anywhere and forget about. Stories about chemical accidents serve as blunt reminders that chemistry class lessons don’t end at the lab bench. I once worked near an old warehouse where drums of chemicals shared space with farm supplies—one spill taught us quickly how unforgiving mistakes get with industrial compounds like this.
This powder or crystalline compound, often used in the metals industry or for smoke grenades, turns hazardous when stored without care. Hexachloroethane can break down and release toxic fumes or react with other chemicals during fires. I remember a news clip showing authorities in moon suits, all because a storage room lacked decent ventilation. People in the community panicked, health officials scrambled, and local businesses closed for days. It’s clear: mishandled storage doesn’t just threaten workers; it puts entire neighborhoods at risk.
Getting Storage Right
Solid, leak-proof containers make a real difference. Hexachloroethane belongs in drums or sturdy containers built for chemical resistance. People sometimes overlook material compatibility and assume every plastic barrel is equal. Polyethylene or high-quality metal containers stand up well, while thin plastic can crack or dissolve.
Fire isn’t just a worry for woodpiles; hexachloroethane becomes especially dangerous around heat. Keeping it in cool, ventilated rooms away from sparks or open flame goes a long way. I’ve seen old warehouses where chemicals sat near space heaters, waiting for disaster. Fire codes in the U.S. set strict rules for a reason: smoke from hexachloroethane carries carcinogens and other bad actors. Think about sprinklers, smoke detectors, and fireproof storage cabinets—these investments pay for themselves the first time they prevent an emergency.
Labels and Training Can’t Get Ignored
Sometimes the biggest trouble comes from simple confusion. Clear, permanent labels showing both the name and hazard symbols help workers know what’s inside a drum. Every skilled safety manager I’ve worked with insists on routine checks and signs—no improvising with hand-written tags that fade after a month.
Experience tells me most accidents begin with someone thinking, “I didn’t know that was in there.” Regular staff training makes a difference. Team members need to recognize not just the label, but what those warning symbols mean. Telling someone “don’t touch this” isn’t enough; understanding why encourages caution in real-world situations. In places with frequent staff turnover, this kind of training turns into an ongoing responsibility, not a box checked during orientation week.
Managing Risks Long-Term
One stubborn issue is old or surplus chemical stock. Leaving barrels to gather dust creates buildup—and unknown risks. Inventory audits show up in government safety guidelines for a reason. Trained staff should keep track of what’s on-site, move unused hexachloroethane for proper disposal, and document changes. I’ve seen companies dodge fines and major headaches just by keeping clear logs of every chemical shipped in or out.
Leaks and spills need more than just a mop. Fast cleanup tools, with absorbent materials and neutralizing agents, stand by in most responsible workshops. Anyone who’s felt the shortness of breath or headache from chemical vapors knows these basics aren’t optional; they keep people out of hospitals.
Room for Improvement
I’ve watched regulations catch up as more incidents make headlines. Community knowledge about what's stored nearby could get better, too. Opening up safety data to the public and setting up clear plans for emergencies means fewer nasty surprises for first responders or neighbors. Hexachloroethane may not get the front-page treatment like oil or gasoline, but mishandled storage attracts the wrong attention quickly.
What are the physical properties of hexachloroethane?
The Substance at a Glance
Hexachloroethane shows up as a white, crystalline solid with a faint, musty odor. Some folks compare its smell to camphor, although it’s less noticeable unless you really stick your nose near a container. Sitting on a lab bench, this stuff looks completely innocent—like rock salt or sugar—but it tells a very different story thanks to its chemistry.
Melting Point and Boiling Behavior
The melting point marks one of its standout features. Hexachloroethane melts at around 230 degrees Celsius. That’s way higher than the temperatures most people encounter in a kitchen or even in routine lab work. It skips boiling in a traditional sense under normal pressure, breaking down before it can vaporize neatly. This changes how you have to handle and process it. In my time working with chemicals, you quickly realize such thermal stability makes the compound appealing in some industrial settings but can also make cleanup slower if there’s a spill.
Density and Solubility
It packs a pretty heavy punch by weight—a density of roughly 2.1 grams per cubic centimeter puts it well above the density of water. That means if you drop a chunk into a glass of water, it sinks to the bottom without a second thought. Also, its solubility tells a straightforward story. Water hardly touches it—less than a milligram dissolves in a whole liter. Yet many organic solvents, including chloroform and benzene, dissolve it just fine. This habit to separate from water influences everything from wastewater management to emergency cleanup procedures.
Stability and Reactivity
Hexachloroethane doesn’t decompose easily in the open air at room temperature, and you can store it safely if you keep it dry and out of strong sunlight. Heat shifts this balance. Over around 250 degrees Celsius, it can break down, giving off toxic fumes—hydrogen chloride and phosgene land at the top of the list. I always read the fine print before working with it, and anyone else should too. Phosgene, after all, has a brutal history in chemical warfare, so you can’t treat this step lightly.
Crystal Structure and Handling
Looking at its crystals under a microscope, you’ll see hexachloroethane forms flat, plate-like shapes. These crystals glide between your fingers, not unlike soap flakes, which can feel harmless until you remember the risks. Gloves become non-negotiable. I remember seeing a coworker brush away some spilled powder bare-handed once—training fixes that mistake, but the lesson sticks.
Environmental and Health Impact
Dust control ranks up there as a vital point. Hexachloroethane particles can float into the air easily if you’re not careful. Breathing in the dust isn’t exactly something you should shrug off—it can irritate the lungs and bother the liver and kidneys if you get enough exposure. Long-term contact might build up in fatty tissues, and sometimes it pushes out toxins you weren’t expecting. That puts an extra burden on safe disposal and rigorous lab protocols.
Tackling the Challenges
I’ve worked with teams that put up local exhausts and use sealed containers just for this reason. Modern practice leans on these guardrails because anyone who’s tried cleaning up a spill with only shop towels soon learns how important proper planning becomes. Investing in personal protective equipment, ventilation, and strict training helps cut down the hazards.
Looking Forward
Understanding these properties—the melting point, density, solubility, and risks—cuts right to the heart of responsible handling. Anyone planning to use or dispose of hexachloroethane owes it to themselves and others to respect its quirks. Every fact here traces back to a real consequence, and that’s what shapes the daily routines in labs and factories that deal with this compound.
How is hexachloroethane disposed of or treated as waste?
Why Hexachloroethane Matters
Hexachloroethane, often used in metal working and military applications, brings real headaches when it’s time for cleanup. I’ve seen old chemical stockrooms filled with dusty cans labeled with hazardous materials, hexachloroethane among them. Tossing them in the regular trash isn't an option—these compounds pose major health and environmental threats if they break free into soil or water. The long-lasting effects on ecosystems are no joke; even low levels can damage animal organs and long-term exposure affects people, too.
Disposal Methods: The Dirty Details
Chemical waste managers face a tough job deciding what to do with old or spilled hexachloroethane. Unlike household cleaners, this isn't a matter of diluted soap down the drain. Instead, highly regulated processes handle the toxin. High-temperature incineration is one of the primary routes. You need intense heat—often above 1,000 degrees Celsius—to break down its chemical bonds. Only specialized facilities can reach such temperatures and properly capture the unwanted byproducts, especially hydrochloric acid and nasty organic residues, keeping them from creeping into the air.
Landfills equipped for hazardous waste sometimes receive stabilized hexachloroethane, but that involves mixing it with materials like lime or cement. This keeps the chemical locked away from water or roots that might spread the problem. Encapsulation works but it's more of a bandage; above all, nobody wants leaks in future generations.
Health Impacts That Demand Respect
I remember talking to a friend who worked on remediation sites. Their protective gear told you everything: one splash, one inhaled breath in the wrong place, and they faced real risks. Hexachloroethane vapors hammer the liver and kidneys and can cause cancer with enough exposure. The U.S. Environmental Protection Agency classifies it as a hazardous waste under RCRA, which means strict oversight each step along the chain. Lax handling means real consequences. Waterways face contamination, and schools and homes nearby pay the price for short-term convenience.
Room for Smarter Approaches
Right now, too much chemical waste hangs around storage closets, forgotten until a spill or inspection. Safer, greener chemistry tries to replace compounds like hexachloroethane, but in many industries, they still rely on it. Source reduction stands as a big hope: using less, opting for cleaner alternatives, and designing processes that steer away from persistent chemicals.
Government regulations should never be seen as red tape for its own sake. Oversight keeps communities, workers, and kids safe. Public agencies and private sectors both need to stay updated on disposal laws and best practices. Training workers who handle such waste matters as much as scientific know-how. Proper labeling, documentation, and secure containers form the most basic line of defense.
Tangible Solutions—Moving Beyond Wishful Thinking
Research keeps trying to bust the traditional mold of disposal. Emerging methods, like advanced oxidation or bioremediation, promise cleaner outcomes, but scaling them to real-world volumes lags behind. Communities should insist on better funding for hazardous waste management, stronger penalties for illegal dumping, and robust collection events to clear out forgotten stock.
Anyone with leftover hexachloroethane—whether a school lab or a welding shop—should contact local hazardous waste services. It’s not just about following the rules; it’s about making sure our water, soil, and neighbors stay healthy. Disposing of dangerous chemicals with care brings real peace of mind, not just box-checking for compliance.
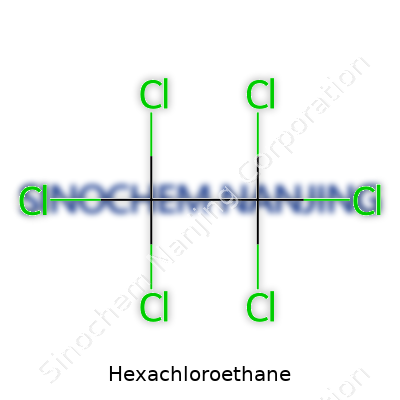
| Names | |
| Preferred IUPAC name | 1,1,1,2,2,2-Hexachloroethane |
| Other names |
Carbon hexachloride Ethane hexachloride Hexachloroethene Hexachloroethylene |
| Pronunciation | /ˌhɛk.səˌklɔːr.oʊˈɛθ.eɪn/ |
| Identifiers | |
| CAS Number | 67-72-1 |
| Beilstein Reference | 1204112 |
| ChEBI | CHEBI:39251 |
| ChEMBL | CHEMBL1331 |
| ChemSpider | 10429 |
| DrugBank | DB06733 |
| ECHA InfoCard | 100.001.150 |
| EC Number | 201-733-2 |
| Gmelin Reference | 724 |
| KEGG | C07453 |
| MeSH | D006600 |
| PubChem CID | 8221 |
| RTECS number | KI4175000 |
| UNII | K4E21C5H7W |
| UN number | UN1181 |
| Properties | |
| Chemical formula | C2Cl6 |
| Molar mass | 236.744 g/mol |
| Appearance | White crystalline solid |
| Odor | Mild, camphor-like |
| Density | 1.68 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.94 |
| Vapor pressure | 0.0025 mmHg (25 °C) |
| Acidity (pKa) | -0.3 |
| Magnetic susceptibility (χ) | -87.7·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.527 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 340.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -8.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -107.3 kJ/mol |
| Pharmacology | |
| ATC code | V03AB19 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335, H410 |
| Precautionary statements | P260, P261, P264, P270, P271, P273, P301+P310, P304+P340, P305+P351+P338, P312, P330, P403+P233, P405, P501 |
| Autoignition temperature | 215°C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 2,840 mg/kg |
| LD50 (median dose) | 3,000 mg/kg (rat, oral) |
| NIOSH | NIOSH: MN0875000 |
| PEL (Permissible) | 1 ppm (10 mg/m3) |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | 300 ppm |
| Related compounds | |
| Related compounds |
Tetrachloromethane Tetrachloroethene Trichloroethene |

