Guanidinethiocyanate: A Linchpin in Modern Life Science Research
Historical Development: The Rise of a Biochemical Powerhouse
Back in the twentieth century, the focus on decoding the building blocks of life pushed scientists to find chemicals that could separate cells with surgical precision. Guanidinethiocyanate (also called guanidine thiocyanate) emerged during these efforts as researchers dug for substances that could effectively denature proteins and protect nucleic acids. Its full potential blossomed in the years following the DNA revolution, as laboratories across the world turned to this white crystalline powder to tackle the challenges in gene research. It showed particular value by helping explore viral genomes at the dawn of molecular diagnostics, laying the groundwork for the spread of accurate diagnostic techniques.
Product Overview: Beyond the White Powder
Guanidinethiocyanate usually lands in the lab as a distinctive, compact crystal or powder, boasting a strong affinity for water and an uncanny ability to break apart cell structures. This property draws many molecular biologists and clinical chemists since it lets them extract nucleic acids in a single step. Few compounds can match its knack for smashing cellular boundaries without chewing up the genetic material inside. Decades after its first discovery, it remains present in countless kits used for DNA and RNA prep, testifying to its effectiveness.
Physical and Chemical Properties: No-Nonsense Performance
This substance delivers a lot with a simple makeup: a combination of guanidine and the thiocyanate group gives rise to a salt that is soluble in water, not terribly volatile, and packs a punch in breaking molecular bonds. It rarely clumps unless it draws in moisture, and its solubility means it dissolves quickly, saving precious time during protocols. Its action is driven mainly by the strong disruption of hydrogen bonds, scrambling proteins and inactivating pesky enzymes that could destroy nucleic acids. You end up with safer, more reliable samples to work with downstream.
Technical Specifications and Labeling: What Matters for Practical Work
Labels are clear to prevent dangerous mix-ups: one should spot the chemical formula (CH6N4S), hazard warnings, recommended storage conditions, and purity. Most suppliers push for over 99 percent purity to guarantee results in sensitive applications. Storage at room temperature works for most labs, as long as the cap is sealed and humidity stays low. No-nonsense labeling helps guide safe handling, especially since guanidinethiocyanate does carry some health risks.
Preparation Methods: From Factory to Freezer
Manufacturers create this compound by introducing guanidine into a reaction with ammonium thiocyanate. The controlled environment cuts down on impurities. Careful filtration and drying steps make sure the resulting crystals remain stable and potent. In my own experience working on nucleic acid extraction, using freshly prepared guanidinethiocyanate solution always trumps digging into an old bottle, since you cut down on the degradation products that could mess up results.
Chemical Reactions and Modifications: The Workhorse in Molecular Labs
Guanidinethiocyanate by itself doesn’t sit still. In combination with other reagents, it unlocks more potential. Mixing with phenol and chloroform produces potent extraction solutions for breaking down complex biological mixtures. Its reducing action shields RNA from self-destruction and blocks ribonucleases that often destroy samples before you can even analyze them. The chemical also helps refold or denature proteins, depending on the protocol and concentration, which can be a lifesaver during tricky purifications.
Synonyms and Product Names: Clarity in the Catalog
If you spend enough time in a lab, you’ll spot guanidinethiocyanate under several banners. Guanidine thiocyanate is just the tip – you’ll see GT, GuSCN, or even thiocyanic acid, guanidinium salt. Commercial DNA and RNA extraction kits refer to it as part of "buffer RLT" or "lysis buffer," keeping things streamlined. Clear naming matters since mixing it up with other guanidine-based salts could throw off any sensitive genetic analysis.
Safety and Operational Standards: Keeping Risk in Check
Laboratory safety guidelines rarely treat guanidinethiocyanate lightly. Lab coats, eye protection, and chemical-resistant gloves are standard gear, since the compound can poison on inhalation, skin contact, or if splashed near the eyes. It does not get along well with oxidizing agents, and mixing with bleach can trigger toxic gas release. Training and regular refreshers matter more than ever, especially for young scientists eager to jump into the hands-on part of research. Dedicated fume hoods and clear waste disposal protocols reduce the chance for error. I always remind new lab members: short-term exposure can cause headaches and nausea, while long-term exposure isn’t fully understood, so caution wins the day.
Application Area: The Unsung Hero of Modern Medicine
Walk into any hospital or research campus – guanidinethiocyanate has likely played a role in a test result or published finding. The explosion of PCR-based diagnostics since the late 1980s owes a lot to this chemical, which preserves viral or genetic material even from dodgy, hard-to-handle swabs and body fluids. Routine pathogen surveillance, genetic screening, and even breakthrough cancer diagnostics lean on it for reliable sample extraction. Outside the clinic, researchers dissecting gene expression patterns use its strong denaturing power to create clear, reproducible data. This ingredient makes its way into the workflow of plant genetics, food safety, and even some forensic science, offering consistent separation and stabilization of genetic material.
Research and Development: Always Evolving
The race to perfect nucleic acid extraction methods continues as labs handle more samples and demand quicker, cleaner results. Guanidinethiocyanate stands front and center as protocols evolve. Researchers view it as both friend and challenge; new research focuses on reducing the chemical’s environmental impact and making it safer without compromising performance. There is a steady push for formulations that can work in automation and high-throughput screening, driven by large-scale genetic studies and the explosion of personalized medicine. Some teams investigate alternatives or tweaks to reduce toxicity while retaining those unbeatable extraction results. Every year, published studies highlight new tricks, from integrating it into magnetic bead extractions to pairing it with ultra-stable buffers that help protect fragile viral genomes for longer periods.
Toxicity Research: Balancing Progress with Precaution
Concerns about toxicity haven’t faded as guanidinethiocyanate moves through more labs and clinical facilities. Scientists continue to study its acute and chronic impacts. Test data shows direct contact irritates eyes, skin, and the respiratory tract. Inhalation at higher concentrations produces symptoms that can keep seasoned lab staff off their feet. The compound threatens aquatic life when sent down the drain, putting more pressure on policies about chemical disposal. Regulatory agencies keep a close watch, updating safety calls as new risk data appears. Institutions work to improve best practices, offering more hands-on training and clear, visual guides rather than just written manuals. My own work in facility management has shown that even the best scientists aren’t immune to slipups, reinforcing how ongoing education beats ticking boxes on a safety sheet.
Future Prospects: New Challenges, New Solutions
Demand for ever-faster, more sensitive molecular tests will push innovation in extraction chemistry. Guanidinethiocyanate will stay in the conversation for years, though it faces new scrutiny from environmental and green chemistry advocates. Researchers are probing for less harmful alternatives and exploring closed-loop use that recycles or neutralizes waste. Automated robotic platforms need reagents that combine safety, effectiveness, and the ability to scale – a tall order, but not impossible as smarter formulation chemistry enters the scene. As personalized medicine grows, rapid home diagnostics hinge on simple, safe reagents, so formulations based on guanidinethiocyanate or its successors will need to match both lab quality and household safety standards. That’s a high bar, but experience suggests the research community thrives on big challenges and rarely settles for yesterday’s solutions.
What is Guanidinethiocyanate used for?
Understanding a Powerful Chemical Tool
Walk into any molecular biology lab and you’ll likely see bottles labeled “guanidinethiocyanate” stashed near freezers and PCR machines. It’s not an everyday household name, but this chemical touches countless lives, especially for anyone who’s had a DNA or RNA test. Over decades, guanidinethiocyanate has earned respect as a powerful chaotropic agent, which basically means it can disrupt hydrogen bonds and unravel proteins and nucleic acids. Why does that matter so much? Because in the world of genetics, those tough bonds make working with DNA or RNA tricky. Every accurate viral test, cancer research breakthrough, and forensic sample benefits from this hidden workhorse.
Breaking Down Barriers in Disease Testing
The biggest impact of guanidinethiocyanate probably showed during the COVID-19 pandemic. Labs across the globe shifted into high gear pumping out PCR and rapid molecular tests. Many of those kits depend on this chemical to break open cells so that their genetic material can be studied—no fancy robotics required. Having worked on a COVID-19 test validation team, I saw firsthand how critical guanidinethiocyanate was in isolating viral RNA. Without it, the accuracy of those tests would nosedive, wasting both time and resources.
Safety and Handling Concerns
Strong chemicals demand respect, and guanidinethiocyanate definitely fits the bill. Exposure can cause burns or breathing trouble, so it makes sense that labs have clear safety rules. People new to the field quickly learn the precautions: eye protection, gloves, and fume hoods aren’t optional. The principle is simple—science can’t move forward if the people powering it aren’t protected.
Expanding Uses Beyond Diagnostics
While its claim to fame sticks closely to RNA and DNA extraction, guanidinethiocyanate also finds fans in protein research. In protein purification, it breaks apart complexes that resist milder chemicals. Hospitals, universities, and food scientists use these tools to push medical and agricultural research, drive safer food production, and spot contamination earlier in the cycle. The chemical doesn’t usually make headlines, which seems unfair given its wide reach. Looking at the rise in genetic therapies, personalized medicine, and food safety concerns, we’ll probably see even more creative uses in the coming years.
Problems with Pollution and Disposal
Large-scale lab operations create another challenge: chemical waste. Guanidinethiocyanate can’t just go down the drain. I’ve seen labs scramble for affordable, safe disposal options, only for costs to pile up or local facilities to fall behind on pickups. In cities where scientific research thrives, this problem gets worse. Since this chemical can harm water systems and wildlife, managing waste responsibly counts as both a moral duty and a regulatory headache. Research groups keep pushing for greener alternatives and better recycling, but progress feels slow at times.
Finding a Balance: Innovation with Responsibility
The value guanidinethiocyanate brings is real. It drives breakthroughs in healthcare and basic science every day. That said, as its use grows, so do the challenges— protecting lab workers from harm and communities from polluted waste. Scientists, companies, and policymakers need to share responsibility, pushing for safer procedures and less toxic alternatives whenever possible. My hope is that as new testing demands emerge, research keeps both innovation and public safety top of mind, remembering the power— and limits— of the invisible tools behind the world’s most important discoveries.
How should Guanidinethiocyanate be stored?
Understanding What We’re Dealing With
Anyone who’s spent real time in a laboratory has handled chemicals that encourage respect. Guanidinethiocyanate sits among those. Lab folks know it for its role in breaking open cells, stripping proteins, and prepping nucleic acids for all kinds of analysis. In the wrong conditions, though, this white powder can present big risks – both for safety and reliability of results. Mishandling it introduces a cascade of headaches, from ruined experiments to dangerous exposures.
Getting Storage Right
Over the years, I've seen too many labs treat storage as an afterthought. Guanidinethiocyanate is hygroscopic, which means it absorbs moisture from air. Leave a jar loosely capped on the bench and you’ll soon spot caking, clumping, and sometimes a crust that pays no heed to scientific schedules. Moisture triggers decomposition, generating poisonous fumes like hydrogen cyanide. The pungent smell alone demands attention, but the real danger comes quietly – chronic exposure steals health over time.
Proper Practices Count
Experience shows that storing this chemical in a cool, dry, and well-ventilated area makes all the difference. Use airtight containers. Polyethylene bottles work, and glass jars with PTFE-lined caps add an extra layer of insurance against leaks. Avoid containers that have seen acidic substances, since even tiny traces accelerate breakdown and hazard potential.
Too often, folks stash bottles on open shelves close to the workbench for convenience. Don’t take that shortcut. Place them away from direct sunlight, far from acids and oxidizing agents. Many labs build dedicated “toxic chemical” cabinets– it’s not overkill, it’s common sense. In shared spaces, clear labeling stops confusion and prevents the kind of mistakes that lead to emergency drills becoming real emergencies.
Health and Safety Demand Respect
You learn quickly that lab safety isn’t just box-checking. NIOSH and OSHA recommend strategies not because they want more paperwork, but because inhaling or touching guanidinethiocyanate leads to sore throats, burns, and worse. In my years working with undergraduate researchers, glove slips and inattention happened more often than anyone admits. Nitrile gloves, lab coats, and splash goggles help. So does a fume hood, if handling involves weighing or mixing.
Training and Culture Matter
A safe storage policy means little if people ignore it. Clear protocols work best when echoed in daily routines. I’ve watched good habits spread from veteran techs to newcomers simply by setting up storage areas with colored labels and posted instructions. Nobody benefits from secrets in the lab: when one person shares a scare story about a cracked bottle or vapor leak, others listen. Sharing these lessons as a team keeps everyone sharper.
Solutions and Improvements
Labs can always get better at handling chemicals. Invest in reliable containers, train every new set of hands, and walk storage areas for checks before end-of-day lockup. Use digital inventory systems so chemicals don’t get forgotten and go past their prime. When mistakes happen – and they do – honest reporting beats covering them up. This builds trust, and that trust forms the backbone for safety with guanidinethiocyanate and everything else that asks for respect on the bench.
Is Guanidinethiocyanate hazardous or toxic?
Understanding Guanidinethiocyanate
Guanidinethiocyanate shows up in a lot of molecular biology labs. People use it because it breaks open cells fast and helps scientists get to the RNA they’re after. The thing about this white, crystalline powder—a chemical that science folks sometimes call GITC—is that it doesn’t belong in anybody’s kitchen or regular workplace. It has bite. Touch, inhale, or even splash it, and you start to see why manufacturers slap hazard labels on every bottle.
Why Guanidinethiocyanate Can’t Be Taken Lightly
What stands out with GITC is the way it messes with the body’s building blocks. At the concentrations used in labs, GITC can irritate the skin and eyes in no time. If someone breathes in the dust or the vapors, the risk goes up—cough, sore throat, and more severe lung trouble can follow. Exposure isn’t just a minor inconvenience. In large enough amounts, this chemical’s toxicity can bring nausea, headaches, or difficulty breathing.
Accidents happen, even to the careful. One colleague of mine brushed a powder spill off a bench, forgot her gloves, and by the afternoon, she had a rash running up her forearm that stuck around for days. Safety data sheets back these real events—serious damage is possible if someone gets careless.
What Science Says About The Risks
Studies show that GITC acts as a protein denaturant. That explains why it’s so good at breaking open cells, and also why it’s rough on living tissue. Not a surprise, since denaturing proteins in a test tube isn’t all that different from doing so on your skin or in your eyes. Research indicates that swallowing the chemical or prolonged exposure can hurt kidneys and liver in animal models. Long-term effects on people aren’t well-studied, but early reports warn about potential chronic health risks.
Some safety evaluations say GITC isn’t a classic “carcinogen” or “mutagen," but just because it’s not labeled as such doesn’t signal an all-clear. Lab chemicals with cell-killing power deserve respect, no matter what the regulations lag behind.
Making Lab Work Safer
Gloves, lab coats, and eye shields earn their keep here. Beyond personal gear, anyone using GITC should measure and mix under a fume hood to dodge breathing in airborne particles. All spills want swift cleaning with plenty of water and, if it ends up splashing on the skin, a good rinse. Basic hygiene—no food or drink near the bench, wash hands before breaks—keeps accidents down.
Most big suppliers provide detailed guidelines. The U.S. National Institutes of Health, for example, lays out clear best practices. Disposal shouldn’t involve the sink or trash can—special chemical waste routes matter every time. When training new people in the lab, GITC safety always comes up near the top of the list, right along with bleach and phenol. There's no shortcut around safety lessons for this stuff.
Keeping Risks in Perspective
GITC unlocks a lot of modern medical and genetics work. But its benefits don’t erase the real-world risks for people working with it. Respecting safety guidance and calling out careless moments as soon as they show up keeps both science and scientists healthy.
What is the recommended concentration for RNA/DNA extraction protocols?
Why Concentration Matters in Extraction Protocols
Getting pure nucleic acids from any sample starts with concentration. Anyone who runs a lab or has spent hours pipetting at a bench knows how much this step makes or breaks experiment results. Getting concentrations wrong means the next part of the workflow, from PCR to sequencing, faces trouble—too much inhibits reactions, not enough and nothing amplifies at all.
Common Recommendations: Numbers from Experience
Good extraction kits offer advice: for RNA, most research sticks to a concentration between 20 nanograms per microliter (ng/μL) and 200 ng/μL. For DNA, protocols often target at least 50 ng/μL. Clinical labs look for even cleaner samples but stay within these ranges. I’ve worked with plant leaf tissue and blood—across all the projects, aiming for these concentrations produced more repeatable and trustworthy data.
The Science Behind the Numbers
Enzymes like Taq polymerase or reverse transcriptase can get picky. High levels of salt, leftover phenol, or proteins throw off their function. Low concentration exposes samples to more contaminant effects. A UV spectrophotometer throws up a warning when nucleic acid ratios stray; a 260/280 ratio below 1.8 for DNA or below 2.0 for RNA suggests unwanted material is hanging around. Kits from companies like Qiagen or Thermo Fisher set benchmarks for a reason: years of troubleshooting make their recommended concentrations trustworthy.
Extraction is Just the Start
After extraction, quantifying the nucleic acids with a Qubit or NanoDrop can feel like the moment of truth. In undergraduate research, we once ran an overnight extraction on tissue from an invasive plant, chasing an elusive band on a gel. Concentrations came out far too low, and PCR failed three times. Eventually, going back to the basics and sticking to the recommended input improved everything—the difference wasn’t subtle.
Common Issues and How to Fix Them
Samples with odd ratios or concentrations outside the norm show up all the time. Sometimes, too much tissue or cell debris overwhelms the lysate; other times, old reagents play tricks. Recent research claims adjusting lysis time, increasing ethanol in wash steps, or using fresh reagents helps. For RNA, keeping things cold from start to finish preserves yield. DNA lagging behind? Readjust the ratio of binding buffer or double-check for any stubborn proteins hanging around.
Improving Reproducibility in the Lab
Small adjustments matter. Standardizing how much starting material goes into every tube removes guesswork. Using clean tips, calibrating pipettes, and recording each step catches issues before they pile up. One postdoc I know shared how repeated failures haunted her project—once she tightened up her sample handling and stuck with the proven concentration range, her data stopped fluctuating.
Better Protocols for Better Science
Working with nucleic acids sometimes feels like a test of patience, but following recommended concentration guidelines makes science more solid. Troubleshooting and optimization never stops, yet clean extractions set the foundation for reliable results. For students and seasoned researchers alike, it always helps to return to the fundamentals and respect the numbers that come from years of collective effort. The more attention paid to this simple metric, the less time wasted chasing unreliable results down the road.
How should spills or exposure to Guanidinethiocyanate be handled?
Understanding the Stuff We’re Dealing With
Every lab worker gets introduced to guanidinethiocyanate at some point. This chemical shows up when isolating RNA, handling cell lysis, and busting open samples for DNA work. It’s strong, breaks down proteins, and gets right into the guts of things. It’s also hazardous if not treated right. I’ve seen new team members freeze up once they realize what a spill means, so talking about direct steps we all need to take could save plenty of trouble.
The Risks Are Real
Guanidinethiocyanate harms on contact: skin, eyes, lungs, all at risk. The powder creates a dust cloud quickly, sneaking through lab airways or onto exposed arms. Touching your face after a spill can mean burns or an itchy rash, and inhaling it means coughing fits or worse. Data sheets back this up, not just my experience. Poisoning symptoms may include headaches, nausea, and respiratory distress if a cloud spreads out. Old-timers in the lab remember stories of folks ignoring this stuff and ending up in an emergency room.
Spill Response: Straightforward Steps, No Drama
Avoid panic, act with focus. At my last job, our spill kits sat in plain view, not hidden beneath cabinets. The process begins by getting colleagues away from the spill site. I’ve learned clear communication works best—call out the danger right away.
Respirators and nitrile gloves matter. The first pair might rip, so keep extras in your pocket. Don’t use cloth or bare hands—this chemical chews through latex fast. Make sure eye shields fit snug. If granules or solution lands on skin, rinse with water for fifteen minutes. Anything spilled on clothing gets ripped off immediately, then flooded with water.
Contain liquid or solid spills with absorbent pads or dedicated spill granules. Scoop up the mess with disposable tools—never anything heading back to the communal wash basin. The waste should go straight into labeled chemical disposal buckets, locked down so no curious hands come into contact later.
Cleaning Up Well
Soap and water won’t handle this alone. The bench, floor, or any involved gear needs a detergent that breaks down hazardous residues. Some teams rely on simple bleach solutions; I always recommend double-checking compatibility for ongoing safety. Once cleaned, double-bag the gear, then fill out chemical incident forms. This stuff isn’t bureaucracy—it reminds you and the safety officer what went wrong so future spills shrink in impact.
Training and Culture Matter Most
Every place I’ve worked, the teams thriving with fewer accidents simply cared more about training. Veterans showed the rookies each kit, did mock drills, and pushed a “speak up” culture. No one mocked new folks for triple-gloving, which built confidence. Management replaced used-up safety supplies immediately—they never said “use soap instead.” Written protocols were posted big and bold at every bench. Training gives people clarity, not just rules.
Labs can get frantic, but the basics stay the same: Respect your chemicals, keep your gear sharp, and look out for each other. Shortcuts may save a minute. In reality, that risk lingers for weeks in the form of uncovered wounds or hospital forms.
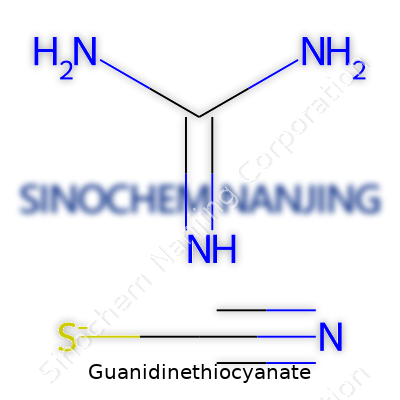
| Names | |
| Preferred IUPAC name | Carbamidine thiocyanate |
| Other names |
guanidine thiocyanate guanidinium isothiocyanate guanidinium rhodanide |
| Pronunciation | /ɡwəˌnɪd.iˌnoʊˌθaɪ.oʊˈsaɪ.ə.neɪt/ |
| Identifiers | |
| CAS Number | 593-84-0 |
| Beilstein Reference | 1098720 |
| ChEBI | CHEBI:63043 |
| ChEMBL | CHEMBL1234891 |
| ChemSpider | 16010 |
| DrugBank | DB11105 |
| ECHA InfoCard | 07bb0b0c-24d2-4de6-8d77-5a86934a16e0 |
| EC Number | 209-812-1 |
| Gmelin Reference | 83444 |
| KEGG | C01416 |
| MeSH | D016229 |
| PubChem CID | 3520 |
| RTECS number | MF1400000 |
| UNII | F82E255159 |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID2020092 |
| Properties | |
| Chemical formula | CH5N3·SCN |
| Molar mass | 118.16 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.306 g/cm3 |
| Solubility in water | Very soluble |
| log P | -1.4 |
| Vapor pressure | 0.01 hPa (20 °C) |
| Acidity (pKa) | 13.6 |
| Basicity (pKb) | pKb 0.4 |
| Magnetic susceptibility (χ) | -47×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.542 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 220.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | V03AB24 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled. Causes severe skin burns and eye damage. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H312 + H331, H302 + H312 + H332, H314, H410 |
| Precautionary statements | P261, P280, P301+P312, P304+P340, P305+P351+P338, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 2-1-1 |
| Flash point | 176 °C |
| Autoignition temperature | 400 °C |
| Lethal dose or concentration | LD50 oral rat 593 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 593 mg/kg |
| NIOSH | BT5000000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 50g |
| IDLH (Immediate danger) | 500 mg/m3 |
| Related compounds | |
| Related compounds |
Amidine Thiocyanate Dicyandiamide Thiourea |

