Glycerol Triacetate: More Than Just a Chemical Compound
Historical Development: A Substance With Staying Power
Chemists first prepared Glycerol Triacetate, known to some as triacetin, just as the world was learning how to harness organic synthesis for large-scale applications in the late nineteenth century. This wasn't some accidental concoction—there was intent behind binding together glycerol and acetic acid. Glycerol, a byproduct from soap and candle production, found new life through acetylation. The result became more than an intellectual pursuit. Factories that previously saw buckets of glycerol as excess sludge now could turn it into something with value. Triacetin entered wartime industries, providing smokeless propellants for ammunition, and became a laboratory staple. Its early development pointed to a shift in how chemical feedstocks could shape entire markets, giving industries new tools to expand and innovate.
Product Overview: Meeting Needs Across Sectors
Ask someone working in flavorings, explosives, or even plastics about triacetin, and you hear about its adaptability. Chemists appreciate its use as a solvent and as an efficient plasticizer for cellulose-based materials. Food technologists see it as an agent that holds blends together while not distorting flavor or safety. In the world of cigarette filters, triacetin has become indispensable because of its role in keeping fibers bound tightly, enhancing filter performance. Pharmaceuticals, cosmetics, and coatings also benefit—its portfolio crosses industries in ways few compounds do. In my own lab days, I saw triacetin's flexibility firsthand as we engineered both active drug carriers and subtle flavor enhancers using the same batch, highlighting just how well this compound transitions across fields.
Physical & Chemical Properties: Stability in Action
Triacetin takes the form of a colorless, oily liquid. Unlike many organic compounds, it carries a mild aroma—a sort of soft, fruity scent—rather than the sharp or noxious fumes of other esters. The liquid flows with a certain viscosity, neither watery nor syrupy, which matters when dosing small amounts in formulations. Its boiling point approaches 258°C, so it stands up well to moderate heating. Triacetin dissolves readily in a variety of organic solvents, making laboratory work easier, yet it keeps its cool if exposed briefly to acidic or alkaline solutions. That kind of resilience set it apart for me in projects demanding chemical compatibility and process efficiency, keeping reactions steady even when conditions shifted.
Technical Specifications & Labeling: Navigating Compliance
Regulators keep a sharp eye on chemicals that blend into foods or pharmaceuticals, placing strict boundaries on additives. Triacetin often appears labeled by its E-number, E1518, in the food sector. Industrial versions, expected to meet purity thresholds above 98 percent, must minimize water content and restrict heavy metals like lead. Importers and manufacturers track batch numbers and assign hazard symbols in areas where quantities or concentrations call for it. In my experience complying with labeling laws, getting every detail right—down to the ppm—spares companies later hassle with audits and safety recalls, showing that accuracy at this stage pays back in trust and operational smoothness.
Preparation Method: Turning Glycerol Into a Workhorse
Creating triacetin demands a simple but powerful reaction: one combines glycerol with acetic anhydride or acetic acid, using either acidic or basic catalysts to drive esterification. Most facilities operate under heated, anhydrous conditions, with careful water removal to steer the reaction toward the triester rather than leaving diacetin or monoacetin as leftovers. That control makes a big difference. Years ago, I watched a batch falter simply because the catalyst wasn’t fresh—impurities took hold, and downstream purification cost hours of labor. Experienced plant operators know every tweak in the process—from temperature to vacuum levels—dictates quality, underscoring how chemistry’s basics still dominate high-tech industries.
Chemical Reactions & Modifications: Tweaking for Demand
Triacetin loves its role as both a feedstock and a stabilizer. Through transesterification, chemists swap out acetyl groups for others, shaping new derivatives that fit special niches in cosmetics or pharmaceuticals. Its esters also break down under strong hydrolysis, regenerating acetic acid and glycerol, feeding into closed production loops in environmentally conscious facilities. I’ve seen researchers use triacetin’s backbone to link active molecules, making prodrugs that release slowly, like time-release medications. These advanced tweaks continue to drive research demand, illustrating why the base compound stays relevant everywhere new delivery challenges pop up.
Synonyms & Product Names: Finding an Alias in a Crowded Marketplace
Triacetin rarely shows up by only one name. The scientific community knows it as Glycerol Triacetate, but catalogs and sales sheets might offer it as 1,2,3-Triacetoxypropane or just Triacetin. This flurry of names sometimes confuses buyers and researchers, especially when international shipments rely on precise chemical identifiers. From government lists to company standards, harmonizing nomenclature streamlines both commerce and communication. During one conference discussion on ingredient confusion, it became clear shared terminology eases traceability, a must for consumer products that cross borders.
Safety & Operational Standards: No Room for Complacency
Using triacetin does not mean tossing aside safety protocols. Its relatively low toxicity often lulls operators into a false sense of security, but spills still call for cleanup and even minor exposures should get logged. Standard regulations require proper impact analysis and waste management. Despite its food-grade pedigree, uncontrolled emissions or accidental mixing with incompatible substances lead to safety reviews or worse. I’ve seen factories tighten protocols after minor incidents, requiring ongoing training for staff working with triacetin-based processes. Safe operations come from repeated practice and treating every chemical—even the mild ones—with respect.
Application Area: Real-World Utility Defines Value
Open a discussion about triacetin and someone in the crowd always mentions its function in flavor and fragrance formulations. Its ability to carry and stabilize volatile compounds saves costs and boosts performance in baked goods, candies, or chewing gum. In pharmaceuticals, it serves as both a solvent for active ingredients and a film-coating agent for tablets. Even the tobacco industry leans on triacetin to keep cigarette filters effective. These examples show that a single molecule can ride through multiple supply chains, blending seamlessly into products that people encounter every day. I’ve handled projects crossing from lab synthesis to mass-market rollouts, showing how functionalities discovered in research settings end up driving profits on store shelves.
Research & Development: Pushing for Innovation
Laboratories continue testing triacetin’s range to see where limits might lie. Scientists have delved into its use for controlled drug delivery, using it to wrap and release therapeutic molecules in time-dependent profiles. Others experiment with using it as a renewable fuel additive, improving cold stability in biodiesel blends. In food science, ongoing trials look at new uses as an emulsifier or moisture-retention agent for healthier and longer-lasting products. The cross-pollination of ideas between disparate fields keeps triacetin in focus at technical conferences and in specialty publications, encouraging a culture where no property or avenue of use stays unexplored for long. My own participation in such forums has made clear that this molecule’s utility is far from exhausted—researchers see possibilities everywhere.
Toxicity Research: Trust, But Verify
Despite its widespread acceptance and historical use, researchers keep scrutinizing triacetin’s toxicological profile. Peer-reviewed animal studies, tests for carcinogenicity, and acute exposure reports all point to a comparatively safe profile, with oral and dermal exposures tolerated at levels far above normal consumer use. The compound breaks down quickly in the body, converting to metabolic byproducts already familiar from natural food digestion. Scientists debate its long-term safety in applications with chronic exposure, and regulatory bodies such as the U.S. Food and Drug Administration continue to review new data. Keeping up with fresh research helps companies adopt better practices, and it ensures consumer confidence as products circulate. Transparency in these findings underpins public understanding, defending the place of triacetin in everyday products.
Future Prospects: Still Evolving
As industries push for greener processes, triacetin faces both challenges and chances. Companies look for more sustainable acetic acid and glycerol sources, hoping to build biobased supply chains that lower the lifecycle footprint for everything from pharmaceuticals to food flavorings. Researchers engineer better catalysts and cleaner processes to cut down energy consumption during manufacturing. There’s growing interest in new applications—especially where triacetin stabilizes sensitive mixtures or helps create biodegradable materials. Watching these trends from both the lab bench and business meetings, I see a clear signal: demand for adaptable, safer, and more environmentally friendly chemicals has never been higher. Triacetin, with its versatility and proven track record, stands well positioned to ride that wave of change.
What is Glycerol Triacetate used for?
Everyday Product, Hidden Impact
Glycerol triacetate holds a place in far more corners of modern life than most folks notice. Often called triacetin, this substance popped up on my radar years ago when I started looking at the ingredient lists in foods and personal care items. At first, the name sounded intimidating. But it’s just a colorless liquid, chemically a simple ester made from glycerol and acetic acid. Its uses run the gamut from food production to plastics, all thanks to its ability to mix with both water and oils and its low toxicity.
From Food to Flavors
I saw triacetin on the back of a chewing gum pack and later in a bottle of vanilla essence on my kitchen shelf. In food, it works as a solvent for flavors and colors. Manufacturers reach for it because it carries flavor compounds while keeping them fresh and stable. In cakes and candies it acts as a humectant, keeping treats moist longer—a win for anyone who can’t finish cookies in a day. Triacetin has approval from both the FDA and the European Food Safety Authority, which sets it apart from many additives—regulators have studied it and found it safe in reasonable amounts.
Cigarettes and Pharmaceuticals
Triacetin’s link to cigarettes troubled me the most. It shows up as a plasticizer in cigarette filters, which means it makes the cellulose acetate fibers softer and helps hold them together. While it doesn’t add to the nicotine, its use in cigarettes puts more strain on discussions about ingredient transparency in tobacco products. By softening plastics, it also turns up in capsules for pills, helping those hard shells dissolve more predictably.
Tech, Textiles, and Beyond
Outside the kitchen and pharmacy, triacetin pops up again in nail polish and lipstick. It does double duty: softening polymers and slowing down evaporation so makeup feels better and lasts longer. Tech companies grab it as a plasticizer in making films, flexible plastics, and even special-purpose coatings. It doesn’t have the environmental baggage of some older plasticizers, which matters to anyone watching chemical footprints. In textiles, it can help certain fibers hold dyes or stay softer.
Environmental and Health Questions
No chemical gets a free pass. Triacetin plays nice with the body in tiny doses, because enzymes break it down to safe materials—acetic acid and glycerol. Yet large-scale manufacturing demands attention. Every year, thousands of tons end up in factories, so proper controls for waste and worker exposure matter. Researchers have found little evidence that it builds up in the environment or causes chronic health issues at common exposure levels, but strict monitoring keeps folks safe.
Finding Better Choices
Many companies keep looking for safer, greener plasticizers and food additives. Some renewable-based alternatives show early promise but face hurdles at scale. Open disclosure and more research into long-term safety could nudge manufacturers toward greener chemistry and help shoppers make better decisions. Triacetin isn’t a villain, but its story covers everything from supermarket cookies to what lands in rivers out back. Paying closer attention to these hidden helpers in daily products, and urging transparency from companies using them, helps everyone make smarter choices about health and the planet.
Is Glycerol Triacetate safe for human consumption?
A Closer Look at Glycerol Triacetate
Glycerol triacetate, often called triacetin, ends up in more foods than you might realize. This liquid has a clean taste, no real smell, and blends easily with water or fats. Food companies like it for stabilizing flavors, softening chewing gum, and keeping baked goods moist. Triacetin also appears in pharmaceuticals and cosmetics, which means people ingest or apply it in small amounts daily. My own curiosity about food ingredients came from reading labels and noticing words that didn’t sound like food. Triacetin was one of them.
What Regulators and Experts Say
The U.S. Food and Drug Administration (FDA) includes glycerol triacetate on its Generally Recognized as Safe (GRAS) list. The European Food Safety Authority (EFSA) also reviewed it and gave the go-ahead for restricted use. Studies tested triacetin in rodents and provided a wide margin of safety. At standard food concentrations, no evidence links triacetin to cancer, birth defects, or toxicity. In my view, these conclusions matter because regulatory reviews try to catch long-term risks before they become a threat. Regulators do not simply trust a chemical’s name or history in older recipes—their process demands real scientific proof.
How Much Is Too Much?
Most people eat or drink far less triacetin than any limit regulators recommend. Soft drinks, dairy desserts, and even gel capsules deliver small doses—usually less than 30 milligrams per serving. Animal studies fed much larger amounts before any minor side effects showed up, and those effects faded after triacetin was removed from their diets. From a practical standpoint, eating several kilograms of yogurt or gum each day for a year would be closer to an unrealistic experiment than a real-world risk.
Why Worry?
Skepticism makes sense because chemical names can spook anyone. Food allergies, personal sensitivities, and past scandals (like the melamine crisis in China or red dye scares) all make shoppers pay closer attention. I don’t buy the argument that every approved additive gets a free pass. Companies and regulators have made costly mistakes before. Watching for updates or reports on specific food ingredients remains important, especially for people with digestive issues, allergies, or auto-immune problems. But for triacetin itself, long-term review across continents shows no evidence of trouble at normal food levels.
Potential Solutions for Ongoing Safety
Food manufacturers need to keep testing ingredients in the real world, not just laboratories. Third-party lab testing and transparent sourcing of raw materials give people more trust in what lands on their table. Labels should ditch scientific jargon when possible, so shoppers understand what they eat. Digital tools can now scan barcodes or ingredient lists, translating chemical names into plain language and linking to studies or safety data. More honest communication and simple explanations about why additives show up in foods would go a long way toward building confidence.
Practical Advice for Concerned Shoppers
I always suggest reading ingredient lists and sticking with brands that disclose more than the law requires. If triacetin or any unfamiliar name shows up often in your diet, search for brands that use fewer flavor or texture enhancers. Most people using medication in capsule form or eating processed snacks won’t notice triacetin one way or another. But checking for new science and trusting your own experience—especially if you have allergies—keeps you firmly in charge of your health.
What are the chemical properties of Glycerol Triacetate?
Understanding the Structure
Glycerol triacetate, also known as triacetin, stands out for its straightforward yet fascinating structure. Three acetate groups attach to a single molecule of glycerol, swapping out each hydroxyl group for an acetic acid ester. This tweak makes the molecule far more hydrophobic than plain old glycerol. If anyone has tried mixing it into both water and oil, it tends to blend better with the oily side of things, but shows enough versatility to dissolve partially in water. Its molecular formula clocks in at C9H14O6 and you can smell its faint fruity, sweet scent. Having handled laboratory solvents, that odor comes as no surprise since esters often carry gentle aromas.
Physical Stability and Reactivity
Triacetin flows like a light, clear oil and doesn’t harden up under normal conditions. It stays stable in both air and regular lighting. Pouring a little in a beaker, you'll notice it resists breaking down in most environmental settings. Reports show it can handle a wide range of temperatures before giving up any of its structure—boiling only after reaching about 258°C (496°F). That point matters for folks involved with food and pharmaceuticals because the compound holds together when heated in industrial processes.
Acids and bases put it to the test. Strong bases, especially caustic soda, can crack open those ester bonds through hydrolysis, taking triacetin back to its original components—glycerol and acetic acid. The same happens, though a bit slower, with strong acids. If someone worries about chemical buildup over time in storage tanks or pipelines, it's this risk of slow hydrolysis that calls for materials compatibility checks.
Solubility and Compatibility
This compound dissolves well in organic liquids like alcohol, ether, and chloroform, but go heavy on water and it barely mixes. Its limited water solubility allows both ease of handling and flexibility in formulation. Any chemist working on formulating flavors or plasticizers keeps this property in mind, knowing triacetin will deliver active components without watering down mixtures.
Function and Health Considerations
Triacetin steps into food as a flavor carrier or moisture deterrent. It finds a second home in cigarette filters and pharmaceuticals as a plasticizer or solvent. Regulatory bodies like the FDA approve it as safe, but studies recommend sticking to these established use levels—the compound earns its Generally Recognized as Safe (GRAS) status for a reason. Tinkering with excessive doses in food or medicine can put consumers at unnecessary risk, and watching for signs of hydrolysis matters over time, since released acetic acid can tweak the taste or pH of a product.
Years of manufacturing have shown that reliable testing and storage protocols keep triacetin’s profile clean. These measures help guard against impurities that can slip in through sloppy handling. In my own work, cross-checking supplier certificates helped us flag high water content, which can speed up decomposition and limit shelf life. Staying on top of such routine checks can make or break a production cycle’s quality level.
Toward Responsible Chemical Use
As industries keep finding new uses for compounds like triacetin, transparent research processes and clear safety labeling anchor responsible development. Publicly accessible chemical safety databases and shared industry data let users make sound decisions about handling and storage. My direct experience? Involving hands-on teams in safety discussions gets better up-take of protocols. Talk through those chemical properties with folks on the line, and you boost both workplace confidence and end-user safety—two things no lab should overlook.
How should Glycerol Triacetate be stored?
Why Storage Matters
Glycerol triacetate, sometimes called triacetin, pops up a lot in labs and factories, especially if you work with plastics, food additives, or pharmaceuticals. Many folks see it as no big deal—clear liquid, faint smell, nothing flashy. But treating it like just another bottle on the shelf can backfire. I’ve seen colleagues learn this the hard way, dealing with ruined batches, leaky containers, or the stress of a surprise safety review. Controlling how this stuff is stored isn’t nitpicking—it keeps products pure and workspaces safe.
Environmental Conditions: Keep It Cool, Dry, and Dark
Heat does glycerol triacetate no favors. Direct sunlight or high temperatures will speed up its breakdown. I remember a summer several years ago, when a shipment spent a bit too long in a poorly ventilated storeroom. By the time I got my hands on it, the material looked a little cloudy—never a good sign. Recommended temperature caps out at around 30°C (86°F), with less swing the better. I stick to low shelves in shaded rooms, avoiding radiators and sunny windows.
Humidity seeps into everything in old buildings, and glycerol triacetate is no exception. Moisture can mess with its chemical stability. A dry place works well; think of rooms with dehumidifiers or those classic moisture-absorbing packets thrown in with supplies. Low humidity saves you on mess and keeps your material true to label.
Light breaks down lots of chemicals slowly. Amber bottles or opaque containers limit contact with sunlight and bright lights. If you’ve got clear containers, wrap them with aluminum foil or store them in closed cabinets. I keep mine out of sight as insurance against slow, silent damage.
Choosing Containers: Material and Seals Matter
I’ve seen friends use whatever container is handy. That cuts corners. Polyethylene or glass bottles with tight, chemical-resistant lids work best. I check containers every few months for cracks or bulging—old plastics get brittle, and glass marred with scratches cracks more easily. Factory seals keep out unwelcome air and water. Don’t ever store this stuff in containers that had strong-smelling chemicals before; even tiny traces will shift glycerol triacetate’s purity.
Labeling and Inventory Controls
Label everything, every time. Name, batch, and arrival date stay in plain sight. In my early days, I skipped this after a rush order—I ended up throwing out a half-used jug since I couldn’t prove when it arrived. Regulations in many countries expect clear labeling. Tracking what comes in and out of storage not only meets standards but stops waste and confusion.
Safe Storage Locations
Keep glycerol triacetate far from strong acids, bases, and oxidizers. Chemical reactions do not belong in storage cabinets. I use color-coded shelves or segregated cabinets; my last workplace stuck bright tape along shelves that held incompatible chemicals.
Ventilation matters for bigger tanks or lots of bottles—small leaks in closed rooms cause headaches or worse. Storage rooms with fans, vents, or air exchanges reduce risks.
Emergency Preparedness: Complete the Picture
It pays to keep spill kits nearby. Absorbent mats, gloves, and goggles won’t stop an accident, but help you clean up fast if you tip over a container. Training everyone who handles glycerol triacetate about hazards and procedures helps avoid panic, keeps everyone safe, and stops spills from turning into real emergencies.
Is Glycerol Triacetate environmentally friendly?
Looking Closer at Glycerol Triacetate
Glycerol triacetate pops up in a lot of places: food products, pharmaceuticals, cigarette filters, even industrial manufacturing. People sometimes call it triacetin. This colorless, oily liquid usually comes from the reaction of glycerol—a byproduct from making soap or biodiesel—with acetic acid. Since glycerol often comes from natural fats and oils, some folks think of triacetin as a “green” ingredient.
Raw Materials and Sourcing
Any real conversation about triacetin and the environment should start with the source. Biodiesel and the soap industries generate large volumes of glycerol. Without secondary uses, unwanted glycerol could pile up or get dumped. Processing it into triacetin reclaims material that would have gone to waste. In that respect, the story starts off on the right foot. But there’s a catch: acetic acid doesn't always come from renewable sources. The industry still pulls petroleum-based acetic acid into the mix, particularly where cost matters more than carbon footprints. Synthetic sources drag down the environmental grade, even if only some batches use renewable methods.
What Happens After Production?
Triacetin has a reputation for stability. It doesn’t break down right away if dumped in the environment. High persistence means residues could hang around, depending on conditions. That raises questions about long-term impacts in soil or waterways. Most studies suggest triacetin is not especially toxic and can break down under the action of microbes in water or soil. Reports from the European Chemicals Agency show no big red flags about acute toxicity, mutagenicity, or danger to aquatic life when used in moderate volumes. In places handling tons of this stuff, though, spills or misuse could change the picture.
Production Footprint: Water and Energy
Factories producing triacetin do use energy, and they generate some waste. Converting glycerol to triacetin means heating, mixing, and sometimes running distillation. That’s true with any chemical, but energy sources and how they’re handled make a difference in environmental impact. Some companies are starting to use renewable power and tweak manufacturing to cut emissions. In my experience walking through a midwestern biodiesel plant, the difference between a modern, well-managed facility and one running off old fossil-fuel plants hits you in the nose, literally and figuratively. Good ventilation and energy recovery change a smelly, hot room into something much more tolerable for people and the planet.
What About Safer or Greener Alternatives?
Triacetin works in places where you want plasticity or a stable carrier for flavors and drugs. Safer alternatives exist, but sometimes they just don’t hold up. Natural esters, like those from plant oils, might serve in a few applications, but they break down, react with other ingredients, or have supply chain troubles. So, as much as I'd love to see a totally plant-based, biodegradable replacement everywhere, the chemistry says triacetin isn't going away soon.
Steps Toward Progress
To improve the environmental story, policy-makers and the industry should nudge producers toward renewable acetic acid. Incentives for biodiesel coproducts already encouraged glycerol recycling. In some regions, green labels or buyer pressure push companies to certify their sourcing and emissions data. Tighter regulation of waste handling, along with public tracking of accidental releases, could close the gap further. From what I’ve seen, real change comes when customers—especially big consumer brands—demand cleaner inputs and back up those goals with long-term contracts. Scientists continue looking for more biodegradable, lower-impact plasticizers. Until one of those takes over, improving triacetin’s supply chain stays important for anyone who cares about making everyday products cleaner.
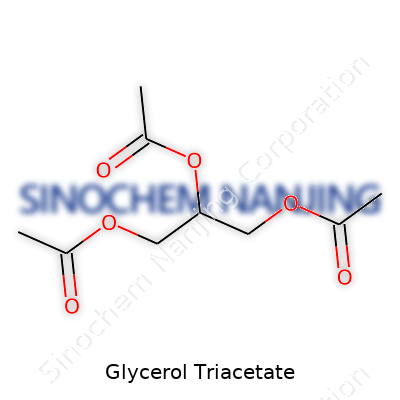
| Names | |
| Preferred IUPAC name | 2,3-Diacetoxypropyl acetate |
| Other names |
Triacetin 1,2,3-Triacetoxypropane Glycerol triacetate Glyceryl triacetate |
| Pronunciation | /ˈɡlɪs.ə.rɒl traɪ.əˈsiː.teɪt/ |
| Identifiers | |
| CAS Number | [“102-76-1”] |
| Beilstein Reference | 1630785 |
| ChEBI | CHEBI:53609 |
| ChEMBL | CHEMBL1209510 |
| ChemSpider | 5916 |
| DrugBank | DB02028 |
| ECHA InfoCard | 100.025.273 |
| EC Number | 204-822-2 |
| Gmelin Reference | 597 |
| KEGG | C06561 |
| MeSH | D005957 |
| PubChem CID | 5499 |
| RTECS number | AKK02847OQ |
| UNII | 8KQ31790D6 |
| UN number | UN1993 |
| CompTox Dashboard (EPA) | DTXSID2020186 |
| Properties | |
| Chemical formula | C9H14O6 |
| Molar mass | 218.21 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Odorless |
| Density | 1.15 g/cm³ |
| Solubility in water | Miscible |
| log P | 0.25 |
| Vapor pressure | 0.1 mmHg (20 °C) |
| Acidity (pKa) | 12.5 |
| Basicity (pKb) | 9.43 |
| Magnetic susceptibility (χ) | -8.46e-6 |
| Refractive index (nD) | 1.430 - 1.433 |
| Viscosity | 165.9 mPa·s (25 °C) |
| Dipole moment | Dipole moment of Glycerol Triacetate is 2.05 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 531.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1611.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3708.1 kJ/mol |
| Pharmacology | |
| ATC code | A07EC02 |
| Hazards | |
| Main hazards | May cause respiratory irritation. |
| GHS labelling | Not a hazardous substance or mixture according to the Globally Harmonized System (GHS) |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P370+P378 |
| Flash point | Flash point: 177 °C |
| Autoignition temperature | 401 °C (754 °F; 674 K) |
| Explosive limits | Explosive limits: 2.7–19% |
| Lethal dose or concentration | LD50 Oral Rat 7,500 mg/kg |
| LD50 (median dose) | 7,200 mg/kg (rat, oral) |
| NIOSH | WA2625000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 50 mg/L |
| IDLH (Immediate danger) | No IDLH established |
| Related compounds | |
| Related compounds |
Glycerol diacetate Glycerol monoacetate Glycerol Acetic acid |

