Glycerol Diacetate: A Deeper Look at a Versatile Chemical
Historical Development
Glycerol diacetate, better recognized in science circles as diacetin, came about during a period when chemists explored the wide net of esterification, aiming to transform plant-derived glycerol into more useful compounds. The last century opened a lot of doors for organic chemistry, and the quest to create new materials from old resources. The roots of glycerol diacetate stretch back to the early 1900s, dovetailing with the growth of the fats and oils industry as food, soaps, and plasticizers became critical to industrial economies. Pioneers in ester chemistry noticed that acetic anhydride and acetic acid could react with glycerol, and the resulting compounds held promise for food, pharmaceutical, and technical uses. The ability to fine-tune properties through partial esterification set the stage for modern applications that go beyond the traditional roles of acetic acid esters.
Product Overview
Glycerol diacetate sits at the crossroads between completely esterified compounds like triacetin and the more familiar monoacetin. It features two acetate groups joined to glycerol, creating a molecule that balances hydrophilic and hydrophobic traits, making it useful both in hydrating solutions and as a solvent. As someone who has worked with food formulation and technical ingredients, I have seen manufacturers favor diacetin because it hits a sweet spot—not too volatile, with a pleasant taste profile, and offering more stability than monoacetin. This niche makes it sought after in both direct food applications and as a starting point in synthesis of other chemicals.
Physical & Chemical Properties
Diacetin brings together two worlds. It has a syrupy, mostly colorless appearance, mixes easily with water and many organic solvents, and resists crystallization at room temperature. A gentle, sweet odor reminiscent of vinegar hints at its origins from glycerol and acetic acid. Density lands at about 1.15 g/cm³, a notch above water, making it easy to handle in liquid applications. Boiling points rest in the mid-to-high 200°C range, high enough to survive routine food processing, but not so high as to complicate distillation in clean-up stages. Chemically, the molecule maintains ester bonds that can withstand moderate acid and base conditions, yet remain open to further modification. As with most esters, it hydrolyzes in strong alkali or acid, splitting back to its parent alcohol and acid, which matters for both safety and waste treatment.
Technical Specifications & Labeling
Diacetin often appears in ingredient lists under E-1517, especially in European markets, where it's approved as a food additive. A glance at chemical supply catalogs lists it with a CAS number of 25395-31-7. In my own experience preparing product labels and safety data sheets, compliance with food and pharmacy regulations means specific information needs to be printed—including purity, residual acetic acid, and moisture content. Each region sets thresholds, but there’s agreement about limiting impurities, especially monoacetin—which can bring off-flavors—and triacetin, to guarantee that the targeted diester makes up the bulk of the product. Workers in quality assurance do not ignore such parameters, especially as trace levels of free acetic acid or glycerol can sway the odor and safety profile of finished goods.
Preparation Method
Commercially, synthesizing glycerol diacetate means reacting glycerol with acetic anhydride or, less commonly, with acetic acid under catalytic conditions. I have handled bench-scale preparations myself, and they usually call for careful dosing of reagents and sustained mixing at raised temperatures to drive the formation of the diester without pushing it all the way to triacetin. Control matters—too much heat or excess acetic anhydride leads the batch toward over-esterified or decomposed by-products. Recovery typically depends on liquid-liquid extraction, followed by distillation under reduced pressure to remove lighter monoesters or volatiles. Operators keep a close eye, since impurities or residual acetic acid can make downstream applications trickier.
Chemical Reactions & Modifications
The reactive handle of diacetin rests in its remaining free hydroxyl group, which opens a door to making more intricate molecules. With additional acylation, it turns into triacetin—useful as a plasticizer and fuel additive. Mild hydrolysis brings it back to monoacetin, or further to plain glycerol, depending on the conditions chosen. This hydrocele of reactions provides flexibility for chemical engineers and product designers looking for ingredients with varying solubility or volatility. Over time, more specialized modifications emerged: functionalizing the free alcohol for new pharmaceutical excipients or modifying esters in search of unique polymers and surfactants. Diacetin, by nature of its bridging properties, provides this versatility, supporting everything from rapid-release drug capsules to non-stick coatings in bakery industries.
Synonyms & Product Names
Consumers and industrial users may spot this molecule under the name diacetin, glycerol diacetate, or diacetyl glycerol. Food manufacturers, under food additive regulations, often label it as E-1517. Other identifiers in chemical supply and regulatory circles include terms like glyceryl diacetate and glycerol acetate. This profusion of names complicates risk assessments and supply chain validation, so due diligence and clear documentation become central in food, pharma, and technical production lines.
Safety & Operational Standards
Glycerol diacetate generally enjoys a reputation for safety, especially when compared with related compounds that turn acrid or hazardous during use. Regulatory agencies like EFSA and FAO/WHO mark it as acceptable up to defined intake levels, and in technical workplaces, those handling galloons of diacetin wear gloves and goggles to avoid irritation from splashes or vapors. From years of reviewing workplace safety protocols, I have seen risk analyses focus on its breakdown products—acetic acid and glycerol are familiar substances, but careless spills or heating above recommended limits can release irritating fumes. Storage in sealed, moisture-free containers keeps the material stable, and avoidance of strong bases or acids in proximity minimizes risk of hazardous reactions. Routine air monitoring and emergency wash stations further improve operational safety.
Application Area
The sweet spot for diacetin shows up in food, pharmaceutical, and technical sectors. Bakers value it for its ability to soften crumb textures and extend shelf-life. Chewing gum manufacturers look to it for moisture retention and a clean flavor impact without overwhelming sweetness. In pharmaceutical formulations, diacetin acts as a solvent or excipient, helping dissolve hydrophobic drugs or maintain pill integrity. Elsewhere, the molecule appears in coatings for tablets, ensuring controlled release in digestive tracts. Beyond these, industrial chemists use it as an intermediate in producing more complex esters, or as a component in non-corrosive lubricants, plasticizers, and anti-fogging agents for packaging. Its moderate polarity and stability unlock a broader palette than mono- or triesters can manage. These applications keep diacetin well positioned among multi-purpose esters, providing both immediate practical benefits and room for inventive use.
Research & Development
Researchers keep looking for greener preparation techniques, cutting waste streams and energy requirements in diacetin synthesis. My own experience collaborating on pilot plant design highlighted the value of switching from petroleum-based acetic anhydride to bio-derived reagents, driving both carbon footprint reductions and regulatory compliance in sensitive markets. Ongoing projects aim to refine downstream purification, using membrane filtration or continuous distillation to boost product yield and purity. There’s growing interest in tailoring molecular structure by selective esterification, opening doors to new excipients, improved food texture agents, and even biodegradable plasticizers. Public interest in renewable chemistry and safer additives encourages universities and companies to sponsor more investigation into the long-term effects and technical benefits of this class of chemicals.
Toxicity Research
Years of toxicological testing confirm that diacetin, within approved limits, poses minimal risk to consumers. Animal studies, embraced by food regulatory bodies, show no evidence of carcinogenicity, teratogenicity, or reproductive toxicity. Short-term exposure triggers only mild irritation, even at concentrations much higher than found in finished foods. Cumulative intake studies remain ongoing, and regulators continue to track any signals of unexpected side effects from chronic exposure. If past experience with related esters offers any lesson, it’s that comprehensive, transparent data builds confidence among users and downplays unfounded fears about chemical additives. Vigilance remains a top priority, especially as new uses and higher throughput manufacturing multiply the number of contact points between diacetin and end-users.
Future Prospects
Looking ahead, glycerol diacetate seems poised for expanding roles, shaped by changing consumer and industry preferences. A push for cleaner, plant-based food additives gives it an edge, especially as multinational brands reformulate for clarity and health. Medical device manufacturers keep testing new uses of diacetin as a plasticizer and solvent in bioabsorbable polymers. In food tech, vegan bakeries and confectioneries look set to boost their uptake, especially for clean-label products where single-molecule additives bring lasting changes in texture and shelf stability without introducing unwanted flavors. Innovators in sustainable packaging eye new esters like diacetin, both for their migrations properties and for the low-toxicity profile that earns regulatory and public trust. As research deepens and technology advances, the full palette of what diacetin can offer is likely to widen, connecting old chemical wisdom to modern sustainability and health concerns.
What is Glycerol Diacetate used for?
A Look Into the Everyday Uses of Glycerol Diacetate
Glycerol diacetate, sometimes called diacetin, shows up in many corners of daily life—typically outside the spotlight. One bottle of food-grade glaze could include it; some chewing gum, baked goods, or even industrial applications draw on this simple compound too. Most of us wouldn’t pick it out from a line-up of chemicals, but its reach stretches far, thanks to its unique blend of safety and flexibility.
Digging Into Food: Preserving Freshness and Texture
Walk down the snack aisle and pick up a packaged pastry or a piece of soft candy. That moist, inviting texture? Glycerol diacetate often plays a role. Food manufacturers have favored it for its ability to hold onto moisture and improve shelf life. The U.S. Food and Drug Administration has given it Generally Recognized as Safe (GRAS) status when used as a food additive. That opens the door for bread, pastries, and chewing gum to keep their softness and chewiness longer.
A major bonus of using glycerol diacetate instead of other additives revolves around its breakdown in the body. It quickly splits into acetic acid (like vinegar) and glycerol, both naturally found in our systems. Some artificial preservatives have raised more health questions, so food manufacturers looking for ways to minimize consumer worry tend to pick safer options, and this one checks that box.
Industrial Interests: Solvents and Plasticizers
Beyond the kitchen, glycerol diacetate finds its way into the toolbox of chemists and engineers. In the plastics industry, it softens synthetic resins—think flexible PVC. Other applications include inks, dyes, and even components found in cosmetics. As a solvent, it holds colorants or helps dissolve ingredients that stubbornly resist mixing otherwise.
From personal experience working with specialty products in the coatings industry, I’ve come across situations where strict safety regulations ruled out traditional plasticizers. Regulations in the European Union, for example, push toward low-toxicity substances. This compound provides alternatives that don’t compromise worker safety or environmental health. Some big brands in the paint industry have already made clear moves toward these safer compounds, and glycerol diacetate routinely turns up in their new formulas.
Challenges and Questions That Come Up
Even as use grows, the story isn’t free from challenge. Some producers raise concerns over sourcing—most glycerol comes as a byproduct from biofuel or soap-making, yet quality and purity can vary. That poses a challenge for food and pharma companies who need every batch stable and predictable. On the environmental side, biodegradable additives like this spark less concern than many oil-derived chemicals, but buyers still expect strict documentation about the supply chain.
Different industries sometimes set unique impurity limits. I've seen customers in the pharmaceuticals sector request more rigorous testing protocols, since the health stakes climb in that world. The solution often rests with tight supplier relationships and traceability programs, making sure every delivery lives up to quality claims.
Looking at a Safer Future
Interest in cleaner labels and safer ingredients is not letting up. International agencies, including the European Food Safety Authority, have published positive safety assessments for glycerol diacetate. This inspires more companies to swap out controversial additives. Higher public demand for transparency pressures suppliers, so traceable sourcing and quality certificates will remain important. With these steps, users gain peace of mind while keeping up with shifting regulations and customer expectations.
Is Glycerol Diacetate safe for human consumption?
Understanding What Goes Into Our Food
Glycerol diacetate, also called diacetin, comes up on ingredient lists in salad dressings, baked goods, and even some chewing gums. Food companies use it for its role as an emulsifier. People get uneasy about mystery additives, often for good reason. Over the years, I’ve gotten pickier about what I put on my plate, reading ingredient labels and digging into the meaning behind the chemical names. Glycerol diacetate isn’t a name you’ll hear outside food science circles, so it’s fair to wonder—should we be worried?
What Science Says About Its Safety
Most of what we know about glycerol diacetate’s safety comes from food safety authorities. The U.S. Food and Drug Administration includes it on the GRAS (“Generally Recognized as Safe”) list, based on available research and its use in small quantities. The European Food Safety Authority looked at data from animal studies and estimated daily intake in humans. They concluded it doesn’t pose a risk at the levels found in food. The Joint FAO/WHO Expert Committee on Food Additives offered a similar view, setting an “acceptable daily intake” without red flags.
These decisions aren’t made lightly. They use lab studies on rats and mice, looking for signs of toxicity, DNA damage, and long-term health effects. The amounts used in experiments are much higher than what anyone would ever eat. For glycerol diacetate, no liver, kidney, or reproductive issues showed up. At doses given to people, researchers haven’t linked it with allergic reactions or stomach upset.
The Bigger Picture With Food Additives
Even though glycerol diacetate passes safety reviews, that doesn’t mean people won’t have concerns. Food regulators work on averages. They can’t always account for folks who eat more processed food than others or who might be sensitive to certain chemicals. Everyone’s body is different. A healthy diet limits processed foods, not only because of the additives but also due to the salt, sugar, and unhealthy fats they sneak in.
Long-term safety also comes up for debate. Additives that seem fine now might get new scrutiny if fresh research comes along. We’ve seen ingredients pulled off shelves decades later when health risks surfaced. Trust grows when industry improves transparency—by sharing research, origin, and the reason an additive lands in your food.
How We Can Stay Informed
People want more control over what they eat. It helps to read up on ingredients, question strange names, and reach out to health professionals if something raises a red flag. The science on glycerol diacetate points to low risk at the levels used. Folks with specific allergies or food sensitivities should still talk with their doctor, since reactions to certain substances do pop up, even in items rated as safe.
Public health works best when scientists, companies, and ordinary eaters share information. We vote with our wallets and our words, steering companies to consider what matters most to us—not just flavor, but the safety and honesty behind every meal.
What is the chemical composition of Glycerol Diacetate?
The Simple Structure Behind a Common Ingredient
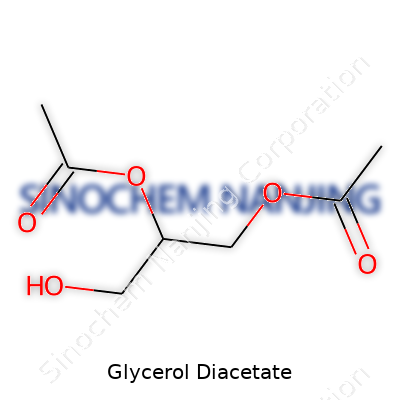
Glycerol diacetate doesn’t grab many headlines, but plenty of food scientists, pharmacists, and industrial chemists handle it every day. This mouthful of a name refers to a chemical made by combining glycerol and acetic acid. At its core, the molecule features a three-carbon backbone (from glycerol), with two of those – usually the first and second positions – capped off with acetyl groups from acetic acid. The last carbon sticks with its original hydroxyl group. To folks with a chemistry background, the formula is C7H12O5, or more plainly, it’s a colorless, oily liquid with a mild odor, soluble in water and a baker’s dozen of organic solvents.
Out in the real world, it usually shows up as a mixture of 1,2- and 1,3-diacetin isomers, thanks to the nature of the esterification process. Some technical-grade supplies even have a squirt of monoacetin hanging around. Seeing a mix doesn’t bother industry, as the differences between these isomers barely shift performance for most needs; the molecular backbone does the heavy lifting.
Why Knowing Ingredients Isn’t Just for Scientists
My own path as a former food safety auditor brought me face-to-face with dozens of ingredient statements, labels, and certificates of analysis. Plenty of people glaze over chemical terms, but knowing what’s in your food or products matters more than ever—especially when regulations tighten. Glycerol diacetate falls in the “generally recognized as safe” category according to the U.S. Food and Drug Administration. That came after years of toxicology data and practical use – it earned its stripes in the E number system as E1517, clearing hurdles most additives never cross.
Glycerol diacetate’s acetyl groups tug at water and oil, letting it slip between both with ease. In foods, I’ve watched it act as a humectant, keeping baked goods moist far longer than their shelf could usually bear. It stops things from drying out, keeps flavors locked in, and can even help dyes and flavors spread evenly across a dough or syrup. As a solvent in the pharmaceutical world, it gives active ingredients an even ride, turning unpredictable mixtures into predictable outcomes. I once spoke with a tablet production manager who swore by this ester for consistent coatings. The consistency is no accident—it’s chemistry at work, but also experience and a dose of industry regulation keeping watch.
Safety and Transparency: Room for Improvement
Transparency around chemicals in common goods came a long way since I started in quality control. Still, the general public rarely knows more than the jargon stamped on ingredient lists. The best solution comes from sharing straight facts in plain language. Consumers do not need to become chemists themselves, but they deserve accurate information to make informed choices. I noticed that the more people learn how simple structures like glycerol diacetate work and what roles they fill, the less fear and confusion clouds the conversation around food and personal care safety.
The chemical makeup of glycerol diacetate might seem simple, but it delivers on functionality and safety, thanks to its unique blend of atoms. Keeping this ingredient on the label—and in discussions about safe, effective food and drug additives—matters for anyone interested in what’s really going into the things they use and eat every day.
Are there any side effects associated with Glycerol Diacetate?
Understanding Glycerol Diacetate
Glycerol diacetate often shows up on food labels as an additive. It’s part of the group of acetylated mono- and diglycerides, and manufacturers lean on it to keep foods fresh longer and improve texture. Bakers count on it for softer bread, and it keeps salad dressings from separating. This compound has been through various safety assessments. The FDA and the European Food Safety Authority have allowed its use, though with set limits. Yet, consumers and anyone who cares about what goes into their meals deserve to know about any possible risks tied to it.
Everyday Exposure and Possible Effects
Most people eat tiny amounts of additives like glycerol diacetate. People rarely see big health shifts from such low levels. Consuming much larger doses, though, could result in digestive upset—things like gas, loose stools, or stomach cramping. These effects happen when the digestive system meets more emulsifiers than it's ready to handle. People with sensitive guts or chronic stomach problems might notice these side effects sooner. For instance, someone with irritable bowel syndrome might experience more pronounced symptoms after meals with added emulsifiers. Animal studies note mild irritation at high doses, but these levels go far beyond what most would eat in a regular diet.
Looking at Allergic Reactions and Rare Cases
Allergy to glycerol diacetate in food remains rare. Still, our bodies react differently, and even foods considered safe can cause trouble for some. If a person feels unwell after eating foods that list glycerol diacetate, logging symptoms and discussing them with a healthcare provider makes sense. In my own experience reading ingredient lists and talking to dietitians, folks who watch out for chemical sensitivities often have broader dietary challenges. For them, tracking how they feel after eating processed foods helps spot problem additives early.
Assessing Long-Term Intake
There’s plenty of discussion about the long-term effects of food additives. Decades of studies exist, but most follow short-term exposure. Scientists call for better research examining what happens after years of low-level intake. Chronic inflammation worries some experts, especially for those eating loads of processed foods. Over the years, lifestyle changes, higher consumption of prepackaged goods, and less access to whole foods have made these questions more urgent. If there’s one thing nutrition research confirms, it’s that a balanced, varied diet supports good health more than any single ingredient's risk.
Practical Steps and Safe Practices
A person aiming to minimize exposure can keep meals centered around unprocessed foods. Learning how to read food labels goes a long way. If emulsifiers or additives regularly pop up in your grocery haul, consider swapping in fresh veggies, fruits, and simple, homemade meals a few times a week. Parents, caregivers, and health-conscious shoppers can talk to doctors or registered dietitians for guidance on additives. The food world features thousands of chemicals, with most proving harmless in the tiny amounts eaten. Still, everyone deserves honest information and safe choices. Food makers, in turn, should keep up with new research and clearly share ingredient information on labels.
How should Glycerol Diacetate be stored?
Getting Practical with Chemical Storage
Anyone who’s ever spent time around a warehouse, lab, or even a modest stockroom knows how easy it is for basic safety to slip when things get busy. Glycerol diacetate doesn’t jump out at people like bleach or ammonia, but it still calls for respect. Chemical handling isn’t just about ticking a box for compliance; it’s about protecting everyone who sets foot near that shelf or drum. Over the years, I’ve witnessed plenty of preventable blunders—slippery floors, mislabeled containers, fumes nobody can identify—often caused by someone stashing a bottle in the wrong place because “it’ll only sit there overnight.” Occasionally, that’s all it takes for a minor mistake to snowball into a big mess.
Sensible Steps Beat Tech Jargon
Think about the space. Dry, cool, low-light conditions always work better for chemical stability. Glycerol diacetate, being a liquid ester, holds up best where sunlight and drastic temperature changes can’t reach it. Storing it a short walk away from heat sources—think radiators, boilers, direct sunlight through a window—cuts down the risk of slow chemical transformations that no one notices until the next inspection rolls around. Temperature swings don’t always scream danger at first, but they shorten shelf life and, given time, mess with purity.
Ventilation seems boring, but it matters. Over time, even low-odor chemicals like glycerol diacetate can build up in unventilated storage areas. I remember a cramped storeroom in a college lab, where opening the door sent an odd, vinegar-like wave into the hallway. Staff just shrugged, but old textbooks in the room went brittle, and the glassware picked up a weird taint. Something as basic as a working exhaust fan could have avoided that whole situation—and kept air quality inspectors happy.
Containers and Labels: Small Details, Big Results
Chemists and warehouse crew love to debate the “right” container. For glycerol diacetate, stainless steel, HDPE, or glass work well—so long as lids screw on tight and nothing eats through the material. I once saw a plastic bottle with the wrong chemistry start to soften after a month, forcing a scramble before anything leaked onto a concrete floor. There’s no substitute for double-checking compatibility charts, even if they seem boring. And as someone who’s peeled off sticky, half-legible labels in a hurry, clear labeling in bold print saves far more headaches than it causes. Full name, concentration, hazard symbols—no exceptions. Sloppy handwriting or missing hazard tags have real-world consequences.
Plan for Everything, Not Just the Best-Case
No storage plan holds up if nobody can find the emergency gear. Spill kits sit in the corner for a reason. Staff needs to know where to grab absorbent pads and goggles, without digging through boxes or unlocking cabinets. Training isn’t a checkbox—it’s an investment in people coming home in one piece at the end of their shift. Each year, chemical incidents spike because someone assumes “just a splash” won’t matter or that a quick wipe with a rag solves everything. Experience says otherwise.
Bring Regulation to Real Life
Rules aren’t about scaring people; they make sense, especially when tailored to daily frustrations. Following OSHA and local fire authority guidance tightens up protocols, but what locks it all in is culture. If people buy in—if checks happen out of habit, labels stay sharp, and stories about “close calls” get shared at every staff meeting—storage practices stick. That’s where safety moves off the checklist and into daily routine. Nobody needs to learn the hard way.
| Names | |
| Preferred IUPAC name | 1,2-Diacetoxypropan-3-ol |
| Other names |
Diacetin 1,2,3-Propanetriol diacetate Glycerin diacetate |
| Pronunciation | /ˈɡlɪs.ə.rɒl daɪˈæs.ɪ.teɪt/ |
| Identifiers | |
| CAS Number | [25395-31-7] |
| Beilstein Reference | 1630783 |
| ChEBI | CHEBI:8630 |
| ChEMBL | CHEMBL1504265 |
| ChemSpider | 16213 |
| DrugBank | DB14096 |
| ECHA InfoCard | ECHA InfoCard: 03-2119951587-27-0000 |
| EC Number | EC 204-772-1 |
| Gmelin Reference | 5987 |
| KEGG | C02455 |
| MeSH | D005957 |
| PubChem CID | 7793 |
| RTECS number | AJ1925000 |
| UNII | WL7H6P18PS |
| UN number | UN3082 |
| CompTox Dashboard (EPA) | DTXSID6022812 |
| Properties | |
| Chemical formula | C7H12O5 |
| Molar mass | 160.17 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 1.18 g/cm3 |
| Solubility in water | miscible |
| log P | -0.48 |
| Vapor pressure | 0.03 mmHg (20°C) |
| Acidity (pKa) | 13.48 |
| Basicity (pKb) | 8.5 |
| Magnetic susceptibility (χ) | -6.1e-6 |
| Refractive index (nD) | 1.417 |
| Viscosity | 10.7 mPa·s (20 °C) |
| Dipole moment | 4.30 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 176.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1016.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3071.7 kJ/mol |
| Pharmacology | |
| ATC code | A16AX11 |
| Hazards | |
| GHS labelling | GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 145°C |
| Autoignition temperature | Autoignition temperature: 400 °C |
| Lethal dose or concentration | LD50 Oral Rat 6,200 mg/kg |
| LD50 (median dose) | 5,200 mg/kg (rat, oral) |
| NIOSH | WA7350000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 200 mg/kg |
| IDLH (Immediate danger) | No IDLH established |
| Related compounds | |
| Related compounds |
Glycerol Triacetate Glycerol Monoacetate Glycerol Acetic Acid |

