Furfuryl Alcohol: From Agricultural Origins to Industrial Powerhouse
A Journey Rooted in Agriculture and Chemistry
Many people would overlook the substance furfuryl alcohol when thinking about important chemicals that shaped today’s industry, but its story is closer to the world of farming, pulp mills, and global supply chains than most realize. Starting in the 19th century, furfuryl alcohol emerged as a byproduct of processing corncobs, oat husks, and similar residue left behind after harvest. Chemists in the early 1920s started paying attention, noticing the potential of this slightly oily liquid with its faintly sweet, burned sugar smell. This compound isn’t rare in nature, coming straight out of furfural, which itself gets pulled from agricultural leftovers using acids. In many ways, furfuryl alcohol reminds us how waste from one process can turn into treasure for another.
Behind the Frothy Surface: What Sets Furfuryl Alcohol Apart
Furfuryl alcohol doesn’t just share its roots with bio-based resources; it shows some quirks in day-to-day handling. Anyone who has worked with it knows it tricks the eyes at first—clear and colorless when fresh, then turning yellow to brown as it reacts with air. At room temperature, this chemical flows easily, thanks to a low viscosity and a boiling point higher than water, which means it stands up to moderate heat. Unlike crude oil derivatives, it mixes well with several organic liquids, including alcohols and ethers, but water doesn’t really dissolve it. I remember early on in my chemical engineering days, folks told stories about what happens if you let furfuryl alcohol heat up or let wild acids or bases near it: it gets unruly—polymerizing, gumming up pipes, and occasionally causing headaches for maintenance crews. These physical and chemical properties, coupled with its tendency to oxidize and polymerize, mean storage tanks must stay cool and out of direct sunlight, and containers need to have liners that can stand up to its character.
Labels and Technical Details Really Matter
Factories and storage facilities can’t just stash furfuryl alcohol anywhere, and regulators have caught onto its quirks. Technical standards usually call for purity of over 98%, water content kept to a minimum, and acid values that don’t surpass low ppm levels. Labels should display its chemical name, common synonyms like “2-furylmethanol” or “2-furanmethanol,” and the standard warning symbols that remind handlers to keep it away from flames and oxidizing agents. For anyone working with labeling requirements, I’ve seen auditors gravitate toward details like batch number, manufacturer, and hazard pictograms straight away. All those careful steps help reduce risks during transport and daily use. Mishandling can result in resin jams or even hazardous vapor emissions.
Turning Biomass Into Building Blocks: Manufacturing and Reactions
The journey from corncobs to finished furfuryl alcohol goes through several steps—starting with the hydrolysis of agricultural residues to pull out furfural, then transforming furfural to furfuryl alcohol using hydrogenation. Traditionally, fixed-bed reactors utilizing copper chromite catalysts have dominated commercial production, but new research highlights improvements through vapor-phase methods and the use of safer, more sustainable catalysts. This production underscores the value of biomass conversion for modern green chemistry. Chemists can modify furfuryl alcohol with all sorts of reagents. Acidic conditions produce furan resins—great for foundry sand binders. Alkaline reactions push it toward different polymer forms, or upgrades like tetrahydrofurfuryl alcohol, which has its own set of applications. Its reactivity with formaldehyde builds stable thermosetting matrices, while mild oxidants turn it into furanones and lactones used in aroma compounds.
Where It Ends Up: Foundries, Plastics, and Beyond
Walking through a foundry, the smell gives away if furfuryl alcohol resins are in play. These resins bond sand for casting steel and iron parts, giving molds better mechanical strength and thermal resistance compared to older binders. The plastic and coatings industries also benefit—in paint formulations, it helps improve chemical resistance. I once visited a wood protection plant in central Europe, where furfuryl alcohol had become integral to hardening and stabilizing softwoods, slowing decay and preventing pests. Smaller, niche applications include corrosion inhibitors, flavor intermediates, and solvent formulations, especially in areas trying to replace petroleum-based chemicals with greener options.
Staying Safe: Health, Handling, and Industry Standards
Personal experience and data from occupational health research both underscore that furfuryl alcohol demands real respect in the workplace. Acute exposure—whether from spilled liquid or vapor leaks—can irritate skin, eyes, and the respiratory tract. Chronic contact raises more serious questions, as animal studies have pointed to possible cancer risks. Regulatory agencies in the United States, European Union, and parts of Asia place limits on acceptable workplace exposure, requiring exhaust ventilation, chemical-resistant gloves, and regular air monitoring. Fire marshals insist on keeping furfuryl alcohol away from open flames and oxidizers. Industry safety committees often push for substitution or enclosure of processes wherever emission risks stay high. These steps slow down workplace injuries and chronic illness, grounding best practice in lives, not just compliance.
Pushing Boundaries: Research and a Greener Future
Environmental advocates and researchers keep hunting for cleaner synthesis methods and safer applications for furfuryl alcohol. Today, efforts focus on swapping out heavy-metal catalysts used in hydrogenation and cutting down residual impurities that can trigger unwanted side reactions. Academic labs investigate new pathways for bio-based resin production, hoping to lower environmental footprints and improve product longevity. On the toxicology front, wider epidemiological studies still need to fill in the blanks about long-term exposure. So far, regulatory consensus points toward careful handling, monitoring airborne concentrations, and favoring closed-system operations wherever possible. These steps create a safer work environment and also encourage the design of new, less hazardous chemical pathways.
Looking Ahead: Opportunity in a Circular Economy
Furfuryl alcohol today sits at the crossroads of sustainable industry, reduced waste, and modern manufacturing needs. As more industries embrace the idea of a circular economy, demand for chemicals derived from agricultural byproducts stands to grow. There’s clear opportunity for those willing to invest in cleaner catalysts, greener processes, and advanced waste treatment. Additional research into health effects and exposure will help shape new standards, moving us toward safer, more responsible chemical industries. Companies able to tap into local agricultural streams to source raw furfural, and who use updated technology to minimize hazards, will likely secure a steady place in a greener industrial landscape.
What is furfuryl alcohol used for?
What People Actually Do With Furfuryl Alcohol
Furfuryl alcohol sounds like something that belongs in a lab, glassware lined up on a shelf, but for anyone who works with cast metal, its smell is oddly familiar. Most people don’t see it in daily life, yet furfuryl alcohol connects to industries that make the raw backbone of trains, tractors, and sewer grates all over the world: metal foundries. Workers mix it with sand and other resins to create molds for casting heavy iron pieces. Once the mixture cures, it holds the shape under intense heat and then breaks away clean after the metal cools.
More Than Just Foundries
You’ll hear about furfuryl alcohol most from foundry workers, but paper chemists and agricultural engineers use it too, just for different reasons. Paper mills use furfuryl alcohol sometimes because it helps improve the water resistance of wood-based boards and certain specialty papers. Farmers looking for smarter ways to protect crops may encounter it in the world of pesticides, since some chemical makers use furfuryl alcohol as an ingredient for making active compounds that fight off fungus.
Safety Doesn’t Take a Back Seat
In my time around manufacturing sites, safety training comes up as often as coffee breaks. Furfuryl alcohol, like many chemicals, requires serious respect. Too much inhalation can cause headaches, and skin exposure isn’t a good idea either. Regulators in the U.S., Europe, and Asia keep a close watch on workplace exposure and insist on training, proper ventilation, and gloves. The requirements didn’t come out of nowhere; folks got sick before stricter rules set in.
Environmental Responsibility
Here’s what matters to me: how furfuryl alcohol gets managed when it leaves the factory. If it spills, it sticks around in water and soil longer than most folks would expect. In the ’90s, researchers started paying more attention to water monitoring around older chemical plants, and the lesson stuck: small leaks add up fast. Companies today track their handling, storage, and waste treatment much more closely than before—some even run dedicated wastewater treatment just for this chemical.
Why Getting it Right Matters
Our roads, machines, and even plumbing need cast metal, and furfuryl alcohol helps keep the process efficient. Skipping it would mean slower production, higher defects, or more expensive alternatives. Still, just because the stuff works doesn’t mean we get sloppy. Balancing efficiency with safety keeps the industry honest and workers safe.
Looking for Better Options
A few startups and research labs are trying to create fresher, safer alternatives. Bio-based resins from plants like soy or corn have made progress in lab settings, but matching the performance of furfuryl alcohol hasn’t been easy so far. The best next step: keep investing in safer chemistry and never let old habits guide new decisions. Being responsible with chemicals like furfuryl alcohol makes industry safer for everyone, from the folks on the floor to families living nearby.
Is furfuryl alcohol hazardous to health?
Understanding Furfuryl Alcohol
Furfuryl alcohol turns up in plenty of industrial settings. Used mostly in foundry work, resin production, and as a flavoring agent, its reach stretches into food contact materials and even some lab work. Some folks working with it probably remember that pungent odor. Anyone with experience in manufacturing or laboratories quickly learns that dealing with this stuff calls for some healthy respect.
Toxicity and Everyday Exposure
Years back, my first real brush with furfuryl alcohol came during a college chemistry lab. Gloves on, fume hood active, warnings about not letting anything touch bare skin pretty much burned into my memory. Occupational scientists have kept a close eye on how furfuryl alcohol behaves. At room temperature, it’s a colorless liquid, but it readily evaporates, letting vapors fill the air. That’s where most risks start—breathing the fumes or getting skin contact.
The International Agency for Research on Cancer (IARC) labels furfuryl alcohol as possibly carcinogenic to humans. Doesn’t mean everyone who smells it gets cancer, but animal studies raised enough red flags. Rats exposed to high levels developed tumors, though concrete links in people haven’t been nailed down. Short-term symptoms pop up quicker—headaches, nausea, throat and nose irritation. Eye and skin irritation follows quickly. Asthma-like symptoms have shown up among foundry workers in factories with poor ventilation.
Long-Term Concerns
Working around furfuryl alcohol day in, day out, without solid protection, puts lungs and skin at risk. Chronic exposure, especially in jobs with little air movement, can cause respiratory issues. The U.S. Environmental Protection Agency lists it as a hazardous air pollutant. Safety data sheets urge consistent protective equipment and ventilation. This isn’t just bureaucratic noise—it’s based on real risks documented in workplace health studies.
There’s also the bigger picture. Furfuryl alcohol breaks down to furan, which researchers link to potential liver toxicity and cancer in lab animals. Most offices or households don’t run into the stuff, but workers in casting, resin manufacturing, or chemical processing face these risks unless controls get enforced and updated.
Putting Safety First
What actually makes a difference? Speaking from experience, nothing beats fresh air and proper gear. Respirators, gloves, goggles, and hood fans are worth their price, especially after seeing co-workers with nasty rashes or shortness of breath. Environmental agencies keep exposure limits strict for a reason. The American Conference of Governmental Industrial Hygienists sets an airborne limit of just 2 parts per million over an 8-hour shift to protect workers.
Accidents still happen, especially in small shops where safety programs lag behind. Education and transparency lower those odds. Giving workers real health information, not just a binder on a shelf, saves skin—literally. Management should put money into safety training and real ventilation upgrades rather than waiting for enforcement visits.
Better Choices Going Forward
Some companies have swapped in less risky chemicals where possible. Furan-based resins, for instance, have substitutes on the market. Big changes can take time, and not every process can change overnight, but sharing knowledge on hazards and control methods moves the needle. As more research fills in the blanks on long-term health, industries and workers can make smarter choices.
What is the chemical formula of furfuryl alcohol?
Understanding the Basics
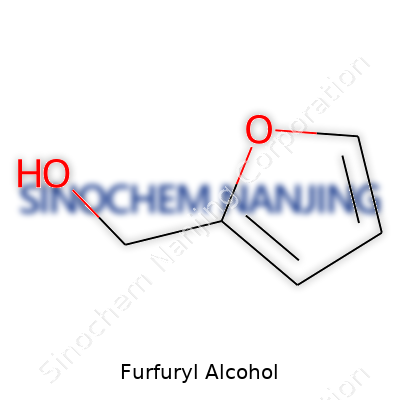
Furfuryl alcohol carries the chemical formula C5H6O2. Breaking this down, the structure holds five carbon atoms, six hydrogen atoms, and two oxygen atoms. Simple enough, yet behind these letters and numbers sits a compound that’s become important for many industries, especially in resin production, agriculture, and even with certain flavors and fragrances. I remember my first encounter with furfuryl alcohol during a chemistry lab session in college, surprised by the faint, somewhat sweet odor that hinted at its presence outside the beaker.
Where It’s Used
Resins call for stable, effective binders. Foundry workers rely on furfuryl alcohol to form molds for metal casting, taking advantage of its ability to form strong bonds after polymerization. The heat-resistant quality stands out, helping to shape intricate metal parts. I once visited an automotive parts plant, and staff there showed me the difference between poorly bonded and properly bonded casting molds. Without furfuryl alcohol, they said, quality would drop and waste would soar. The efficiency in the production line often traces back to reliable core bonds, and furfuryl alcohol consistently steps up to the plate.
Why Chemical Knowledge Counts
Knowing the chemical makeup isn’t just for chemists. C5H6O2 isn’t just a random set of atoms. Recognizing its arrangement helps predict how it reacts. For example, the alcohol group gives furfuryl alcohol its water solubility and its potential to form connections with other chemicals. This matters in safety, too. Its exposure limits and reactivity depend on this arrangement. Workers who deal with it daily must understand storage risks. Spills can lead to fires. This is more than theory—one misplaced drum or carelessly placed rag, and you risk an accident.
Environmental Impact
Furfuryl alcohol doesn't break down instantly in the environment. Some studies from regulatory bodies in the United States and Europe point to moderate persistence and the possibility of aquatic toxicity if released untreated. Years ago, after reviewing pollution data for a local river, I learned how chemical leaks from nearby plants added furfuryl alcohol to the water. A simple formula on paper impacts fish and ecosystems downstream. Knowing its structure allows regulators to set limits and devise cleanup strategies that actually address the real risks.
How We Can Address the Risks
Minimizing harm starts with education. Anyone mixing, transferring, or applying furfuryl alcohol needs clear guidelines on personal protective equipment and environmental controls. Facilities often choose closed systems to reduce airborne emissions. Investing in capture technologies like activated carbon filters cuts down on fumes leaving the building. Alternatives get considered, but efficiency and effectiveness keep furfuryl alcohol in the mix for now. I worked on a project advising a foundry on safer use, which showed that process tweaks and better spill response trimmed down incidents, without major cost increases.
Looking Ahead
Chemical formulas can seem abstract, but C5H6O2 leads to real-world choices—better safety, smarter production, and environmental care. That awareness makes a difference far beyond the laboratory or factory floor. Every person from plant worker to environmental advocate benefits by understanding what these molecules mean, and why it’s worth paying attention.
How is furfuryl alcohol produced?
What It Takes to Make Furfuryl Alcohol
Furfuryl alcohol has a strong link to agriculture. Factories rely on materials like corn cobs, oat hulls, or sugarcane bagasse because these contain pentosans—a specific group of carbohydrates. Here’s the interesting part: the journey from plants to furfuryl alcohol starts with furfural. To get furfural from plant bits, processors use acid to break the pentosan chains down and release furfural through steam distillation. The process is quite old, first pioneered in the 1920s, and still drives much of what goes on in many factories, especially in China and India.
Turning furfural into furfuryl alcohol only needs one key step: hydrogenation. Factories feed furfural and hydrogen into a reactor fitted with a catalyst, usually copper chromite. With the right conditions—careful heat, specific pressure, reliable catalyst—they convert furfural into furfuryl alcohol with good efficiency. This step doesn’t come easy. The chemistry wants to produce all kinds of byproducts, so skilled operators pay close attention, tweaking variables to get yields above 85%. Clean catalysts, reliable feedstock, and steady hands keep it all humming.
Why Furfuryl Alcohol Still Matters
The global foundry business runs on this alcohol. It acts as a resin ingredient for sand molding, making strong, reusable molds for casting metals. Without it, the process stumbles—costs climb, parts grow less precise, and defects sneak in. Furniture, paints, and even the agricultural sector depend on furfuryl alcohol for resins, adhesives, and protective coatings. Many folks outside chemical fields rarely notice, but most urban lives depend on products that pass through a furfuryl alcohol stage.
Production brings its share of issues. Pentosan-rich farming waste goes up in value as regions find more reasons to convert it. Plants often cluster close to where feedstock grows. This makes furfuryl alcohol a good story about waste turning into value, but logistics create hurdles. Monopolies or price gouges sometimes pop up as a few players control regional supply. I’ve seen local farmers get excited as industrial buyers show up only to feel squeezed once middlemen take over.
Facts and Pressure Points
China and India produce over two-thirds of all furfural and its alcohol cousin, leaving smaller players on unstable ground. The hydrogenation step, while mature, generates emissions. For every ton of furfuryl alcohol, over 200 kilograms of CO2 can enter the air if no effort’s made to scrub waste gases. Spills and improper waste disposal turn this useful chemical into a real headache, impacting streams and soil nearby. Strong safety standards, regular site audits, and experienced technicians go a long way toward keeping communities safe.
Finding a Better Way
Cleaner options exist but make headlines less often. Electrochemical reduction uses less energy, but labs don’t scale up well. Some newer biocatalysts bring down emissions, but high costs keep them from mainstream adoption. Sharing best practices—like better feedstock contracts or supporting research that strips out heavy metals from catalysts—can close the gap between massive producers and local operations. In my view, investment in plants close to where agricultural waste gathers could reduce both shipping costs and emissions. Real improvement comes through those little tweaks, not grand reinventions, and a steady commitment to protecting both wallet and waterway.
What are the storage and handling precautions for furfuryl alcohol?
Understanding Furfuryl Alcohol Risks
Furfuryl alcohol plays an important role in producing resins and foundry sand binders. It comes with a bitter almond smell and carries its own set of challenges, especially during storage and handling. Its liquid form doesn't make it less risky—it can irritate skin, eyes, and respiratory tracts. Anyone who spends time around industrial chemicals knows it’s easy to underestimate just how quickly a spill or exposure can happen, especially if corners get cut on safety.
Safe Storage Heats Up
Many chemicals prefer consistent, cool conditions, furfuryl alcohol included. The liquid begins to break down when exposed to heat or even just warm sunlight streaming through a warehouse window. Keep it in a well-ventilated, shaded spot, away from direct sources of heat like boilers or radiators. Once, during a summer heatwave, I saw drums start to sweat condensation inside a poorly ventilated storage room. The labels peeled off, turning retrieval into guesswork—no one wants to play that game with hazardous materials.
Sealed drums stand up better to temperature swings and keep out moisture, which can trigger unwanted reactions. Stainless steel and carbon steel containers stand up well, but avoid storing furfuryl alcohol in anything made with copper, brass, or bronze, since those metals can react and speed up decomposition. Factories that label their storage containers clearly and keep detailed logs find far fewer surprises later on.
Watch the Air and Flame
Despite not being as flammable as some industrial solvents, furfuryl alcohol catch fires with the right spark and enough vapor in the air. Smoking anywhere near storage or transfer points shouldn’t even be up for debate. Proper electrical wiring, explosion-proof lighting, and readily available fire extinguishers save both product and lives. Modern safety data sheets recommend keeping foam, carbon dioxide, or dry powder extinguishers nearby.
Ventilation systems deserve attention, otherwise vapor can pool in low-lying spots. I remember visiting a plant that swapped out simple fans for a proper extraction system after a small vapor cloud went unnoticed. Afterward, the workers said headaches and watery eyes became rare, which speaks louder than any corporate policy memo.
Gear Up for Handling
Gloves, eye protection, and chemical-resistant clothing aren't “nice to haves” around furfuryl alcohol—they're non-negotiable. Even brief skin contact can cause burns or rashes, and breathing in even modest amounts can trigger coughing and sore throats. Reliable procedures for cleaning up spills make a real difference: a spill kit with absorbents and neutralizers should stay close to the action, not buried in a back office.
Training stands at the core of any good practice. Rushed or only “on paper” instructions increase the odds of an accident. Regular safety drills and clear signage inside warehouses or work areas reinforce good habits. The best results come from a culture that puts worker health first, not from a binder of policies left to gather dust.
Preparing for the Unexpected
Furfuryl alcohol, like other reactive chemicals, demands respect. That means more than just posting a few warning signs. It means storing it in the right place and the right type of drum, double-checking valves and seals, and teaching every worker what to do if something goes wrong. Looking at chemical accidents—often rooted in routine shortcuts or lazy oversight—shows how essential it is to stay vigilant. Reducing unnecessary risks doesn’t just save inventory; it protects everyone on the job.
| Names | |
| Preferred IUPAC name | (furan-2-yl)methanol |
| Other names |
2-Furylmethanol Furan-2-ylmethanol 2-Furancarbinol Furfuryl alcohol α-Furylcarbinol |
| Pronunciation | /ˈfɜːrfjɪl ˈæl.kə.hɒl/ |
| Identifiers | |
| CAS Number | 98-00-0 |
| Beilstein Reference | 1360640 |
| ChEBI | CHEBI:42786 |
| ChEMBL | CHEMBL15722 |
| ChemSpider | 6809 |
| DrugBank | DB02185 |
| ECHA InfoCard | 04f1346c-ef42-4856-9360-ed9b53970fd4 |
| EC Number | 200-945-0 |
| Gmelin Reference | 80916 |
| KEGG | C00573 |
| MeSH | D005693 |
| PubChem CID | 7308 |
| RTECS number | LU5950000 |
| UNII | 38DTA8DV2A |
| UN number | UN2874 |
| CompTox Dashboard (EPA) | DTXSID0022275 |
| Properties | |
| Chemical formula | C5H6O2 |
| Molar mass | 98.10 g/mol |
| Appearance | Clear, colorless to pale yellow liquid |
| Odor | Musty burnt odor |
| Density | 1.129 g/cm³ |
| Solubility in water | Miscible |
| log P | 0.28 |
| Vapor pressure | 0.6 mmHg (20°C) |
| Acidity (pKa) | 14.0 |
| Basicity (pKb) | 15.36 |
| Magnetic susceptibility (χ) | -8.55·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.485 |
| Viscosity | 7.0 mPa·s (at 25°C) |
| Dipole moment | 1.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 116.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -333.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2114 kJ/mol |
| Pharmacology | |
| ATC code | D01AE22 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS02,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H301, H311, H314, H331, H341, H351 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P260, P264, P270, P271, P273, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P308+P311, P312, P314, P330, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0-W |
| Flash point | 60 °C |
| Autoignition temperature | 🔥 372°C (702°F) |
| Explosive limits | 1.5% - 16.7% |
| Lethal dose or concentration | Lethal dose or concentration (LD50/LC50) for Furfuryl Alcohol: "LD50 oral (rat): 132 mg/kg |
| LD50 (median dose) | LD50 (median dose): 300 mg/kg (oral, rat) |
| NIOSH | NIOSH: L516 |
| PEL (Permissible) | 50 ppm |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Furan Tetrahydrofurfuryl alcohol 2-Methylfuran Furfurylamine Furan-2-carboxylic acid |

