Furfural: More Than a Byproduct—A Story of Transformation and Potential
Historical Development
Furfural’s journey started in the mid-19th century, and sometimes, I find it fascinating how this simple molecule, originally tossed aside as industrial waste, sparked curiosity among chemists who saw value where others saw little. Friedrich Ferdinand Runge, a German chemist, first discovered furfural while distilling grain during his experiments, laying the groundwork for what would become an industrial staple. Demand for furfural ramped up during the World Wars as industries brainstormed ways to wring more value from agricultural residues. Furfural’s rise reflects how necessity and innovation often collide, turning what many called “waste” into a building block for solvent production and more. Today, furfural production plants in many regions operate near farmland to secure a steady stream of crop residues, drawing a straight line from its roots to its role in the modern economy.
Product Overview
Furfural, known by names like furfuraldehyde and 2-furaldehyde, stems mostly from agricultural waste, including oat hulls, corn cobs, and bagasse. People who live near processing plants can’t forget its almond-like odor. In industry, furfural often steps in as both a solvent and a starting material for a broad swath of chemicals. Its versatility touches sectors from oil refining to plastics and beyond. Walking through a processing facility, one might see it stored in drums, awaiting its turn to either clean lubricants, produce specialty plastics, or move along into further chemical transformations.
Physical & Chemical Properties
Furfural’s attributes give it an edge. Under everyday light, it shows up as a colorless, oily liquid, turning amber as it ages. Its sharp, almond smell can catch your attention from across the room, a reminder not to handle it lightly. Its boiling point sits just below 162 degrees Celsius and its water solubility only stretches so far, creating the kind of challenges that chemical engineers often relish: extracting and purifying it from a watery broth of biomass breakdown takes some know-how. As a member of the aldehyde family, furfural reacts with many reagents, and its furan ring structure plays a starring role in most of its uses.
Technical Specifications & Labeling
Pure furfural sold for lab or industrial use meets stringent standards for content and allowed impurities. If you ever work with furfural, you’ll spot chemical purity percentages on labels—usually 98% or higher—to match strict processing requirements. These labels also pull no punches about hazards, warning users about its flammability and potential health effects. Safety data sheets recommend goggles, gloves, and proper ventilation. Chemistry labs and chemical plants keep furfural containers tightly sealed, both to preserve purity and to keep that unmistakable smell from taking over the workspace.
Preparation Method
Traditional furfural production leans heavily on acid-catalyzed hydrolysis of pentosans from lignocellulosic material. In practice, that means steaming or boiling agricultural byproducts in strong acid—often sulfuric acid—and capturing the vapors as the pentose sugars break down into furfural and escape. Modern plants look for any edge in improving yield and lowering energy consumption, which leads to pilot programs exploring alternative catalysts or continuous processing systems. Engineers often tweak cooking times, extraction setups, and raw materials based on price and local crop cycles, all with an eye toward squeezing the most furfural out of each shipment of biomass.
Chemical Reactions & Modifications
If you’ve spent any time at a bench in an organic chemistry lab, that furan ring jumps out as both a promise and a challenge. Nucleophilic and electrophilic groups can attack the ring system, and the aldehyde group stands ready for a host of reductions and condensations. Hydrogenating furfural gives furfuryl alcohol, central to making foundry resins and various polymers. React it further and you’ll pull out furan, which plays a part in pharmaceuticals and fine chemicals. These transformations matter in both commodity chemical synthesis and specialty research. The sheer number of available derivatives opens doors for creative scientists, prompting ongoing research into both new products and methods to produce them.
Synonyms & Product Names
Though chemists know it best as 2-furaldehyde, product labels and literature often mention furfuraldehyde or even furfural alcohol (despite that last one being a distinct derivative). These naming quirks have roots in furfural’s complicated history and the evolution of chemical standards. When talking with engineers and project managers from a range of countries, you hear a handful of names, but the sharp scent and liquid appearance always make people nod in recognition.
Safety & Operational Standards
The chemical’s hazards prompt strict storage and handling practices. Flammability risks and its toxic profile keep it under lock and key in most facilities. In the past, some workers faced exposures that caused headaches and skin irritation, reinforcing a culture of careful handling. Today, national occupational safety agencies and international regulators demand tight controls: gloves, respirators in concentrated settings, and forced-air systems to whisk away vapors. Engineers design processing lines to limit leaks and spills, and regular safety audits try to prevent accidents before they happen. Shortcuts put both workers and the local environment in danger.
Application Area
Heavy industry remains the main customer base, with furfural’s selectivity as a solvent prized in oil refineries for finishing lubricants and isolating dyes. Foundries rely on furfuryl alcohol resins to make sand casts for metal parts—think engine blocks and machinery components—while plastics manufacturers tap into furfural chemistry to build high-performance polymers. Recently, interest in plant-based chemicals nudged researchers and entrepreneurs back toward furfural as a feedstock for bio-based materials. This renewed excitement springs from concern over petroleum dependence and a push for more sustainable supply chains.
Research & Development
The research world rarely stands still with furfural. Universities and industry labs keep exploring more selective catalysts and greener production pathways. Some projects turn to ionic liquids or solid acids in an effort to drive down waste and improve yields. Teams have also started screening new agricultural byproducts—switchgrass, straw, and even food processing leftovers—hoping to unlock lower-cost feedstocks. The quest for new furfural derivatives moves chemists further into pharmaceutical and agrochemical applications, chasing higher margins and more sophisticated uses. In my work, collaborating with chemical engineers, I’ve seen startups emerge around biorefinery concepts that anchor their models on furfural valorization.
Toxicity Research
Furfural isn’t a benign compound. Classic studies and recent workplace monitoring both underscore its acute toxicity at high vapor concentrations; repeated exposures can cause both health complaints among workers and subtle effects over time. Animal studies point toward liver and kidney impacts at elevated doses, matching findings in occupational medicine. Regulators use this evidence to set strict exposure limits and define cleanup rules for spills. Ongoing research now looks at long-term, low-dose effects, given the chemical’s expanding footprint—especially for those hoping to green-light furfural in consumer products or as an intermediate for new pharmaceuticals. The challenge: balancing economic opportunity with public safety.
Future Prospects
Furfural sits at a crossroads where history meets innovation. The global push for renewables turns up the spotlight on every chemical that can claim “biobased” status, and furfural fits the bill thanks to its roots in waste biomass. Companies now look at next-generation biorefineries as economic engines, with furfural extraction as a key output. Advanced catalytic pathways offer a way to further lower costs and carbon footprints. I’ve watched leaders in both chemistry and sustainability debate whether furfural might underpin new kinds of biodegradable plastics or even pharmaceuticals with a greener story. For researchers, the unanswered questions about health effects, process sustainability, and untapped applications promise years of work ahead—and more stories like furfural’s turning up as industries keep transforming what was once thrown away into tomorrow’s critical materials.
What is furfural and what is it used for?
The Basics of Furfural
Furfural starts out humble, made from stuff like corncobs, oat hulls, rice husks, and sawdust—things folks usually toss aside. It’s a liquid, yellowish, with a smell that might remind some of almonds but with a sharp edge. Chemists pin it as a platform chemical, but that’s only part of the story. From farm waste, it turns into a building block for industries that reach far past the farms where this story begins.
How It Powers Manufacturing
On shop floors across the world, furfural isn’t just another chemical. Factories use it to pull the good out of crude oil and help shape plastics. Oil refiners trust it because it pulls unwanted stuff out of lubricating oils and diesel. Not only does this make engines run better, but it also helps stretch the lifetime of everything from trucks to tractors. In felt tip pens and some plastics, it helps tie molecules together, so the final product stays strong and predictable. The resin made from furfural and phenol turns up in brake linings, grinding wheels, and components that people need to trust when things heat up or need to stop fast.
Big Role in Farming and Food
Anyone who has watched grain rot knows rot isn’t just a minor setback—it costs money. Furfural comes from that same plant material, though, showing there’s value in what usually gets left behind. Some well-managed ethanol plants in America’s Midwest collect furfural during distillation and sell it to keep their books healthy, while local farmers find a new market for plant parts that once had no buyer.
Safety and Sustainability
People worry about chemicals in their food and water, and for good reason. Handling furfural takes care. Vapors irritate the throat and eyes, so workers suit up with goggles and gloves. In high doses, there’s risk, so companies have rules to keep staff safe. But compared to petroleum derivatives, furfural gives the world one more way to move past fossil fuels. A factory using corncobs rather than crude oil cuts back on fossil imports and puts money in the hands of farmers growing corn instead of oil companies overseas.
The Push for Greener Chemistry
Every big step forward in chemistry needs a raw material that doesn’t run dry or wreck the land. Furfural, with its roots in agricultural leftovers, keeps popping up in research journals looking for ways to build better plastics or store renewable energy. Some researchers are close to swapping petroleum-based solvents with furfural in large-scale production. The European Union even backs projects that pay farmers for field stubble so countries don’t burn it as waste. China, India, and Latin America have followed, building factories that take furfural from local agriculture, making this chemical a possible ticket to more sustainable rural economies.
What the Future Looks Like
Furfural turns waste into economic fuel. More economies built on farm waste and less on oil means rural regions could get stronger while cities breathe cleaner air. Over the years, I’ve met mill managers who keep a close watch on furfural’s market price just as much as they watch corn futures. As science finds new uses, the people growing, processing, and handling crops have more chances to profit. The more furfural gets woven into daily manufacturing, the clearer it seems that simple changes in what we value—waste turned resource, local over imported—can push chemistry, industry, and agriculture to work better together.
Is furfural hazardous to health or the environment?
Knowing What Furfural Is
Furfural comes from agricultural leftovers like corn cobs, oat hulls, and even sawdust. Industry folks use it to make resins, solvents, and certain kinds of plastics. I’ve seen furfural pop up in discussions among chemical workers and even among people looking for safer, renewable materials. But every so often, someone asks if it’s really safe for people or the environment. This question deserves honest, practical answers.
Looking at Health Risks
People who handle furfural in factories have reported headaches, skin rashes, and eye irritation. Inhalation can bring coughing or sore throats. These symptoms happen even with short-term exposure, so personal protective gear matters. The US National Institute for Occupational Safety and Health (NIOSH) labels furfural as a potential occupational hazard. At high levels, furfural exposure has caused liver and kidney issues in lab animals. The National Toxicology Program describes it as “reasonably anticipated to be a human carcinogen,” based largely on animal studies. No solid proof so far links furfural exposure to cancer in people, but the possibility can’t be ignored.
As someone who has visited sites where chemical vapors linger in the air, proper ventilation and regular checks go a long way. If ignored, small problems—dry skin, red eyes—can turn big quickly. Decades ago, workplace safety rules were more relaxed, and chemical odors went unnoticed. Now, with stricter rules, complaints have dropped, but the chemical still has a bite if workers get careless.
Environmental Effects
Furfural does not just stop at workplace health. It mixes with air, seeps into water, and can impact plants and animals nearby. Direct spills into rivers or soil affect aquatic life. Furfural breaks down in the open air over days, but it can be toxic to fish and tiny freshwater creatures at low levels. The US Environmental Protection Agency keeps tabs on furfural in water, mainly near big processing plants. In rare cases, furfural in local water sources has led communities to close off wells or limit fishing seasons.
The Bigger Picture
Most furfural today comes from renewable plant waste, which helps reduce our reliance on petroleum. Switching to sources like these makes sense, though no chemical comes without risks. Some manufacturers use strict containment, water treatment, and scrubber systems to capture or break down furfural before release. These methods often cost extra, but in my view, avoiding lawsuits and bad press always beats cutting corners.
Better education for people who work around furfural is one path forward. Handwashing stations, leak detectors, and training sessions aren’t just for paperwork—they keep people safe. Technology opens new doors for monitoring and early warning systems. Remote sensors in work areas now alert managers before levels get risky, helping prevent health scares.
Realistic Solutions
Furfural brings real benefits to industry, especially if handled with care. I support stronger safety policies on the shop floor and accountability for leaks or spills. Engineers can design more efficient, closed systems that limit air or water contamination. Investing in greener chemistry reduces the need for toxic solvents in the first place. Long-term, partnering with health officials and researchers helps sharpen guidelines and keep communities in the loop.
Anyone working near furfural should pay attention to visible, simple warnings—itchy skin, watery eyes—and not brush them off. The community gains when companies act fast on safety, and the environment benefits from steady improvements in technology. Furfural serves a purpose, but smart, everyday precautions matter most in keeping people and places safe.
How is furfural produced or manufactured?
Understanding Furfural's Real Value
Furfural comes up more and more in talks about eco-friendly industry. Digging into how it’s made shows why companies care about both quality and where it comes from. Furfural is pulled from agricultural leftovers, like corncobs, oat hulls, and even wood chips—things you’d find at the end of a harvest that people often considered waste. Bringing this material to life as something useful proves how much value we can pull from what many folks tend to overlook.
Step-By-Step: From Biomass to Furfural
Furfural manufacturing boils down to a few main stages. It starts by collecting those raw leftover bits from crops. They’re shredded and wetted down, then mixed with acid (typically sulfuric acid). This acidic mix gets heated with steam inside tall reactors. The process creates hydrolysis—a fancy word for breaking down the tough plant fibers. The real goal is to crack open the hemicellulose in those plant leftovers, releasing pentose sugars.
Those sugars aren’t furfural themselves, but, under heat and acid, they transform into vaporized furfural. The hot vapor rises and gets sent into a condenser, where it cools back into a liquid. There’s a water-furfural mix at first, so the next challenge is separating the two. Furfural is heavier and settles out, letting producers skim off the pure stuff. Later, distillation helps clean out any impurities, delivering a product that chemical and refining industries actively seek for solvents, resins, and even as a diesel fuel additive.
Why Sourcing Matters
Most furfural on the market comes out of countries with rich supplies of agricultural leftovers. China and a handful of African nations, for example, lead the way, largely because their farming systems kick out huge piles of raw material. In places where corn isn’t so plentiful, making furfural can get expensive or even unfeasible. That highlights a key point: access to waste biomass turns into a real economic advantage.
Hurdles on the Production Line
Workers and communities near furfural plants deal with a mix of challenges. The acid involved introduces health and environmental risks. Old-school furfural factories sometimes dumped waste acids or vented noxious fumes, putting local air and water at risk. Newer plants try to capture and re-use those acids, or neutralize them before release, but big gaps remain between top-tier sites and low-budget operations. Tackling these issues needs investment—there’s no trick around that.
Another point: these processes draw plenty of heat energy, which often comes from burning coal, wood, or other fuels. This means furfural carries a carbon footprint, though some new methods turn to biomass-derived steam or waste heat from other plant operations.
Looking Forward: Smarter and Cleaner Furfural
As people look for greener chemicals, furfural stands out as an option—especially where it’s made wisely. New research highlights milder catalysts, less corrosive acids, and reactors that squeeze out higher yields with fewer emissions. Switchgrass and other energy crops, bred for land efficiency, could join the list of raw materials in the next few years. Companies focused on sustainability can speed up the change, proving that industrial chemicals don’t have to come with heavy pollution or deplete the earth’s resources.
From personal experience watching rural communities take control over their waste and channel it into new income streams, honest production of furfural helps everyone along the chain. From producers to workers to the folks living nearby, it comes down to demanding cleaner processes and holding companies accountable for every stage—from field to finished product.
What industries commonly use furfural?
A Hidden Workhorse in Manufacturing
Furfural doesn’t show up in flashy ads on TV or command headlines in business news, but this simple organic compound has powered more sectors than most folks realize. It comes from agricultural waste like corn cobs and oat hulls. I remember touring a chemical plant where stacks of brown husks, discarded from food processing, found their second life in giant reactors—turning what would be trash into something surprisingly valuable.
Refining and Chemicals
Refineries depend on furfural to clean up base oils. Engineers use it to strip out unwanted aromatics, making lubricants for cars and trucks run smoother. This makes a real difference for both vehicles and the environment, cutting down pollution and cutting costs for everyone involved. Manufacturers also use it as a building block to make other chemicals, including pesticides, plastics, and flavors used in food. Years ago, I interviewed a chemist who explained that without furfural, producing nylon and some foams would get a lot tougher, if not impossible.
Pharmaceuticals
Drug-makers extract active ingredients and synthesize new medicines with furfural-based solvents. These solvents let chemists fine-tune reactions to get exactly the product they want, in the amount the world actually needs. My friend in pharmaceutical R&D says their team cannot skip furfural when developing specific painkillers and antibiotics, especially with modern regulations getting stricter each year.
Agrochemicals and Crop Protection
Furfural-based products protect fields from pests and weeds. American growers in the Midwest rely on these formulations because they keep crops alive in tough seasons without breaking the bank. The efficiency here isn’t just in spraying fields, but also in cutting down production time for herbicides and fungicides. Talking with a farmer at a county fair, I learned just how much their yields owe to these chemical protectors, even if city folks rarely notice their role.
Food and Beverage
Even though furfural can be toxic in high doses, trace amounts help produce some flavorings and preservatives. The almond, caramel, and coffee notes in multiple processed foods often owe something to this compound. Food scientists like to experiment with it to get exactly the right scent or flavor, especially for products where cost matters just as much as taste. Once, during a visit to a flavor lab, I tasted several vanilla extracts and learned how chemical tweaks with furfural can shift a product from synthetic to almost gourmet.
Potential Solutions and Looking Forward
Concerns crop up with sustainability. Heavy reliance on agricultural waste as feedstock gets tricky as land use shifts. Long-term supply of raw material can run dry if farming practices change or weather takes an odd turn. Making these supply chains resilient means blending in new sources, like wood waste or even municipal biosolids, and not just sitting back waiting for next year’s harvest. Companies also need to tighten safety protocols to avoid air and water pollution during production and processing. By staying aware, listening to both workers and neighboring communities, and pushing research into cleaner furfural synthesis, industry leaders can keep both the product and the planet in balance.
What are the storage and handling requirements for furfural?
Furfural’s Role in Industry
Furfural often shows up behind the scenes in chemical and agricultural industries. Factories take advantage of its usefulness making resins, solvents, and certain plastics. Some might even remember its sharp almond-like smell from a high school lab or from stories of workers in processing plants. Furfural offers benefits, but falling short on its storage or handling can lead to big problems—both for workers and for the environment.
Hazards Few Talk About
Furfural isn’t as forgiving as some other chemicals you find in the warehouse. It catches fire at lower temperatures, spreading flames quickly, and its vapor can form explosive mixtures with air. In my years walking plant floors, managers who ignored good storage faced real scares. Even a brief spill smelled up entire buildings and left people coughing and red-eyed—documented in numerous workplace safety incidents. Long-term exposure can lead to skin problems and internal organ stress, according to facts on file with the CDC.
Storage: Real-World Advice
The best warehouses treat furfural the same way they treat other flammable liquids. Drums or containers should always stay in cool, shaded spots, far from anything hot or prone to spark. Rather than relying on fancy automated systems, well-run places check container seals, lines, and valves each shift—by hand. Leaky drums have no place in a safe warehouse.
One overlooked lesson from older hands: never store furfural near strong acids, bases, or oxidizers. Combinations like these have sparked more than a few warehouse evacuations over the last decade. Steel tanks work, but only the right kind—those lined to resist corrosion. That’s not just to follow a rulebook, but because unlined tanks have led to disastrous leaks, as reported by OSHA case studies. Workers there learned quickly that it’s less costly to invest in suitable storage than to patch up chemical damage and clean spills.
Ventilation and Fire Control
Ventilation might sound simple, but too many facilities get lazy, letting vapors collect around storage areas. In practice, using fans and exhaust systems keeps air moving and removes buildup that could ignite from a spark or static. Sprinklers and foam, along with ready-to-use fire extinguishers (rated for chemical fires), make a difference in emergencies—those who keep equipment dusty or blocked with boxes are taking needless risks with lives and property. A few moments spent clearing aisles and staging extinguishers has saved more than one warehouse from burning to the ground.
Training and PPE: No Shortcuts
No fancy rulebook replaces experience. Workers who respect furfural wear goggles, gloves, and flame-resistant coveralls, plus simple respirators for vapor-heavy areas. PPE falls short if employers don’t train their teams, though. It’s clear: teams familiar with emergency shutdowns, spill control, and first aid react quicker and prevent injuries. Written instructions posted in changing rooms—real-life examples from my own years supervising a crew—help keep skills sharp and encourage everyone to watch out for neighbors.
What Good Practice Looks Like
So much of safe furfural storage comes down to habits. Well-lit labels, clear signs, and scheduled checks beat good intentions. Industries that treat chemical safety as a daily task—not just training paperwork—avoid costly mistakes. From my own work and from stories passed down through the trades, nothing compares to a warehouse where every worker owns the responsibility for safe handling. That’s the real protection against accidents and environmental headaches—personal vigilance backed up by practical routines.
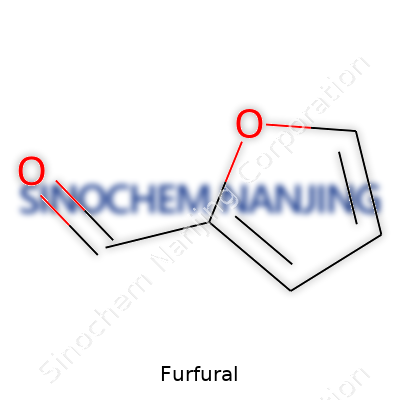
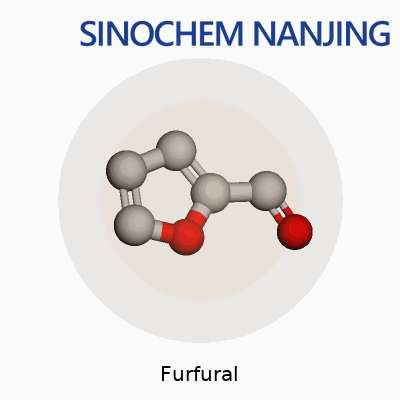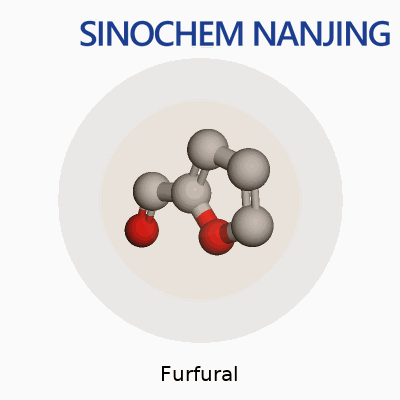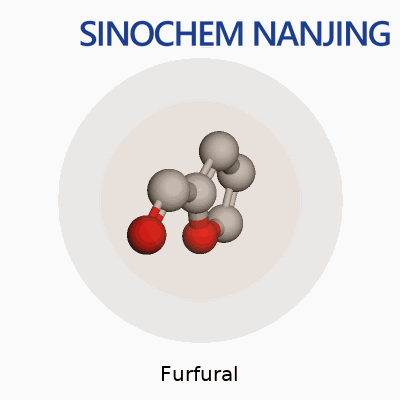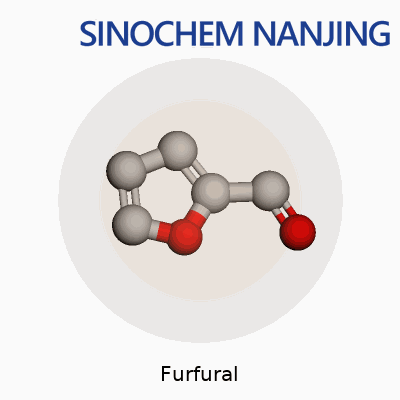
| Names | |
| Preferred IUPAC name | Furan-2-carbaldehyde |
| Other names |
2-Furaldehyde Furancarboxaldehyde 2-Furylmethanal Furfuraldehyde Furfurol |
| Pronunciation | /ˈfɜːrfjʊˌræl/ |
| Identifiers | |
| CAS Number | 98-01-1 |
| 3D model (JSmol) | `3D model (JSmol)` string for **Furfural**: ``` C1=CC=C(C=C1)C=O ``` |
| Beilstein Reference | 636068 |
| ChEBI | CHEBI:17146 |
| ChEMBL | CHEBI:17147 |
| ChemSpider | 7138 |
| DrugBank | DB02348 |
| ECHA InfoCard | echa.europa.eu/information-on-chemicals/infocards/100.003.080 |
| EC Number | 200-837-2 |
| Gmelin Reference | 87835 |
| KEGG | C00783 |
| MeSH | D005673 |
| PubChem CID | 7362 |
| RTECS number | LU8400000 |
| UNII | W7P3KQJ7ED |
| UN number | UN1199 |
| CompTox Dashboard (EPA) | urn:lsid:epa.gov:compToxDashboard:DTXSID0027092 |
| Properties | |
| Chemical formula | C5H4O2 |
| Molar mass | 96.08 g/mol |
| Appearance | Colorless to amber-colored oily liquid |
| Odor | Aromatic, almond-like |
| Density | 1.159 g/cm³ |
| Solubility in water | 9.8 g/100 mL (20 °C) |
| log P | −0.41 |
| Vapor pressure | 1 mmHg (20°C) |
| Acidity (pKa) | 7.98 |
| Basicity (pKb) | The pKb of furfural is 13.60 |
| Magnetic susceptibility (χ) | -7.44·10⁻⁶ |
| Refractive index (nD) | 1.524 |
| Viscosity | 1.23 mPa·s (25 °C) |
| Dipole moment | 2.52 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 160.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -189 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1367 kJ/mol |
| Pharmacology | |
| ATC code | D03AX17 |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H226, H301, H311, H331, H319, H351, H373 |
| Precautionary statements | P210, P261, P280, P301+P310, P305+P351+P338, P308+P311 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 60°C |
| Autoignition temperature | 437 °C |
| Explosive limits | 2.1% (LEL) - 19.3% (UEL) |
| Lethal dose or concentration | LD50 (oral, rat): 65 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Furfural: 65 mg/kg (oral, rat) |
| NIOSH | K0221 |
| PEL (Permissible) | 50 ppm |
| REL (Recommended) | 2 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Furan 2-Furoic acid Furfuryl alcohol Tetrahydrofurfuryl alcohol Tetrahydrofuran |

