2-Methylfuran: Looking Beyond the Laboratory
Walking Through History
If you ask those in the know about 2-Methylfuran, they’ll probably start from the late 19th or early 20th century, a time when the world was piecing together the puzzle of aromatic compounds. This compound found its place early on, mostly because chemists were enthralled by the furan ring’s chemistry and saw potential in methyl derivatives. As interest in bio-based chemicals started warming up in the twentieth century, 2-Methylfuran gained new relevance. Folks struggled through choked cities craving alternatives to fossil-based chemicals, and suddenly, what once seemed an academic curiosity looked more like one ticket out of an oil-soaked rut. In that sense, the name “2-MF” whispers its age-old backstory even while it wades into fresh debates on green fuels and renewable solvents.
Product Snapshot: Not Your Average Solvent
Those drawn to the nuts and bolts will spot 2-Methylfuran as a clear liquid, faintly smelling of sweet ether. It’s flammable, packing more energy pound-for-pound than much of what’s left over from sugarcane or agricultural waste. Chemists who spend enough years in the lab quickly notice the boiling point lands near 65°C, making it evaporate easily but forcing a reexamination of storage needs. The density lies just below that of water, which can make spills difficult to spot; this small but crucial detail matters in busy workshops and production lines. Getting away from the data sheets, the sharp solvent bite and readiness to take on new structures during reactions gives 2-MF a flavor few can ignore.
Technical Lines and Labels: Cutting to the Chase
There’s no room for vague labels here. Technical specifications demand accuracy: purity often tips above 98%, with most suppliers warning about trace water, acids, or aldehydes. Labels, honest as they are, never gloss over the sharp flammability: users store the stuff in metal drums or specialty plastic, safely tucked away from sparks and heat. The rules in chemical plants and research labs bear down hard—no open flames, proper ventilation, safety goggles for all. Workers pay attention to the SDS not from habit but because 2-MF’s low flash point leaves no second chances if things go wrong.
How It Comes Together: From Biomass to Bottle
Most talk of modern chemistry circles back to sustainability, and here’s where 2-Methylfuran bites into the discussion. Instead of being shackled to oil wells, the compound steps up from plant waste. Researchers lean on the acid-catalyzed dehydration of pentose sugars (think corncobs, rice hulls, or bagasse from sugarcane) to generate furfural, which then gets further transformed with hydrogen. Some processes use a copper catalyst under hydrogen to snip and tuck molecules into 2-MF—no mystery, just years of trial and error in those glass reactors. Production is energy-intensive, doesn’t always promise high yields, and engineers keep sweating over how to scale up without driving costs through the roof.
Chemical Reactions and Modifications: Not Just for the Textbooks
When it comes to chemistry, 2-MF holds its own as both stable and reactive. The methyl group at the second position of the furan ring opens the door for substitution reactions, making it a favorite for folks tinkering with pharmaceutical intermediates or exploring fresh flavors and aromas. Research teams have played around with ring-opening, hydrogenation, and even oxidation, aiming at new alcohols or acids for different markets. I’ve seen chemists go wide-eyed at how it can catch radicals and easily blend into polymer chains, showing that this compound refuses to be pigeonholed into one category or specialty.
Synonyms That Matter: More Than Just 2-MF
Ask around the industry, and you’ll hear names like 2-Methylfuran, methylfuran, or MF. Sometimes older hands call it sylvan, though the romance of that name hardly shows up in modern science. Specialty suppliers stick to CAS numbers or standardized names, but in a busy lab, “2-MF” rolls off the tongue in shorthand. I’ve learned to keep an ear out for all of these, since missing a synonym can derail a search for research papers or regulatory updates—not just a linguistic peculiarity but a practical concern for day-to-day work.
Safety and On-the-Ground Practice
Not all chemicals with interesting properties are friendly. 2-Methylfuran is no exception. The faintly sweet scent masks a dangerous volatility; careless handling can end with skin burns, respiratory irritation, or ringside seats in the emergency room. National rules call for explosion-proof electrical systems and strict fire controls wherever this liquid is transferred. Local teams rarely take shortcuts, since stories float around labs—someone mishandling a bottle, a sudden flash, lessons burned into memory alongside chemical burns. Most experts wear gloves, goggles, and most importantly don’t work alone. Fume hoods hum quietly in the background, doing unseen but vital work to keep air clean. Labeling isn’t a bureaucratic headache but a way to keep lives and livelihoods safe.
Why It Matters—Application and Purpose
The world of chemicals often moves in cycles, and 2-MF is riding high on biofuel research lately. Its boiling point and energy density give hope as a possible gasoline additive or even a primary fuel. Scientists in green tech circles eye it for blending directly into fuel pumps. Those with a background in polymer science or perfume design look for its ability to introduce nutty, caramel notes. Pharmacists and process chemists take notice thanks to the reactivity of the furan ring, which fits neatly into a host of active pharmaceutical ingredients. The push for renewable feedstocks lights up the path for 2-MF’s expansion from lab curiosity to commercial necessity.
Where Research Leads—Digging Into Data
Every year, new studies stack up. Some focus on squeezing more yield from agricultural scraps, others hunt for catalysts that shave pennies off production. The field keeps eyes peeled for scale-up studies that don’t sacrifice safety or purity. Teams worldwide dig into combustion chemistry, mapping what 2-MF leaves in its wake when burned, pushing beyond theory into road and engine trials. On the toxicology front, long-term studies probe the compound’s behavior in air, water, and soil, with data fed straight into regulatory updates. Environmental science crosses paths with chemical engineering more often than outsiders think—the effort to minimize byproducts like furfural is a stubborn and worthy battle.
Toxicity and the Challenges No One Ignores
Anyone looking for a silver bullet in chemistry needs to glance at 2-MF’s safety sheets. Toxicity studies reveal dangers for those exposed to vapors—central nervous system effects, respiratory trouble, and in large enough doses, outright organ damage. The risk increases if the liquid finds its way into groundwater or air. Those in charge of workplace standards set tough rules for exposure, and the best managers enforce them tightly. Disposal has to meet strict environmental codes, given that the compound can persist and won’t quietly break down. This isn’t an untested hazard; decades of animal studies back up the warnings, so abandoning the short-cuts in pursuit of speed makes sense. The truth is, there’s no shortcut to responsible practice, and the hard work protects workers and community alike.
Thinking About the Future
The hunger for greener chemicals keeps companies and academic labs returning to 2-Methylfuran. Every new advance in biomass upgrading, process intensification, or catalyst design brings it closer to market. Some groups hope to see modified forms in medicine or specialty materials, others watch for shifts in car fuels as lawmakers press for better emissions. The cost barrier looms large, at least for now, but as history shows, today’s specialty solvent can become tomorrow’s commodity. The trend toward tighter safety and environmental controls means only the most robust processes will make the cut. For every challenge, there’s a team out there logging the hard hours, and their success could define how widely this compound appears in tomorrow’s factories, fields, and fuel stations.
What are the common applications of 2-Methylfuran?
Keeping Engines Running—Biofuels and Blending
Growing up in a family surrounded by old pickups and endless talk of fuel prices, alternative fuels always sounded more exciting than just saving a few bucks at the pump. 2-Methylfuran shows up in these conversations a lot for good reason. It comes from renewable sources, like agricultural waste, and carries an energy punch similar to gasoline, so engines don’t need special tweaking to run on blends with 2-Methylfuran. Compared to regular ethanol-gas blends, you get better mileage and lower tailpipe emissions. Studies by the U.S. Department of Energy back this up, showing how its use can cut carbon monoxide and hydrocarbons without sacrificing the driving experience.
Aroma with a Punch—Flavor and Fragrance Applications
Back in college, monitoring air quality during the fall harvest, I learned that tiny differences in molecules can change how we experience flavors and smells. 2-Methylfuran slips into that world, too. It shows up naturally in coffee, roasted nuts, and certain fruits, adding that slight caramel or burnt sugar edge. Professional flavorists rely on it to round out the aroma in processed foods or to bring punch to beverages. The European Food Safety Authority points out its low toxicity at the levels used in foods, supporting consumer safety in the use of this compound.
Building Blocks—The Chemical Industry
Working at a chemical plant during one hot Texas summer taught me that every end product has a backstory. 2-Methylfuran sits near the start of many. Chemists love it as an intermediate because its furan ring opens up options in synthesizing pharmaceuticals, pesticides, and specialty polymers. It reacts smoothly with other compounds, providing pathways to anti-inflammatory drugs or creating insecticides that keep crops productive. The World Health Organization and several academic journals document its use as a cost-effective step in new drug discoveries and green chemistry projects.
The Science Angle—Solvent and Lab Applications
Anyone who’s spent hours at a workbench knows that solvents are the unspoken backbone of chemical labs. 2-Methylfuran makes life easier here, too. Its moderate polarity lets it dissolve substances that water or non-polar solvents leave behind. Research groups focusing on renewable chemistry or organic synthesis often reach for it when isolating sensitive molecules, and it keeps the loss of product to a minimum.
Thinking Ahead—Environmental and Economic Potential
Every time oil prices spike or climate talks heat up, renewable chemicals step back into the spotlight. 2-Methylfuran comes from plant materials, slashing dependence on fossil reserves. Its production from biomass goes hand in hand with waste reduction. Government reports in the U.S. and Europe highlight how sustainable sourcing keeps rural economies working even as industries pivot to bio-based production. Small towns with access to agricultural byproducts benefit, bringing growth where new jobs are needed.
Sustainable Choices—What’s Next?
From filling fuel tanks to flavoring snacks, 2-Methylfuran quietly shapes daily life. Its ability to improve air quality, support farmers, and build new jobs makes it stand out. Clear regulation, investment in new technology, and honest communication with the public can help this molecule deliver on its promise without leaving old problems behind.
Is 2-Methylfuran hazardous or toxic?
What Researchers Know About 2-Methylfuran
2-Methylfuran pops up in places most folks might not expect. Found naturally in roasted coffee, cigarette smoke, and even some processed foods, it also shows up in labs and industrial settings. Chemists pay attention to it because it can act as a solvent and has potential in fuels. Most of the time, the average person will only bump into this compound in small amounts. Things shift when workers, researchers, and manufacturers handle it on a larger scale.
Health Risks Connected to 2-Methylfuran
This stuff has a strong odor, which already clues people in: it’s not something to take lightly. Animal studies give us more concrete evidence about its toxic side. Researchers report that rats and mice forced to inhale or ingest 2-methylfuran show signs of liver and kidney damage. Those health effects line up with what toxicologists often find with other volatile organic compounds (VOCs).
The National Institute for Occupational Safety and Health (NIOSH) and the Environmental Protection Agency (EPA) have flagged the need for more study, especially because animal studies point to central nervous system depression and blood changes – headaches, dizziness, even confusion turn up pretty quickly after exposure. Most folks don’t hang out in labs, but for professionals who regularly work with chemicals, these studies matter a lot.
Hazards in the Air and Workplace
Handling 2-methylfuran in closed or poorly ventilated spaces ramps up the risks. This chemical evaporates quickly at room temperature, so it fills the air before most people even notice. I learned that a whiff while working with solvents can leave you with a pounding head in minutes. Some folks have shared stories of nausea or nose and throat irritation after cleaning up spills or running tests. Over time, that kind of low-level exposure adds up.
Fire risks don’t take a back seat. This chemical flares up with a simple spark, giving safety officers more to worry about than just health exposure. I’ve seen work areas where one missed label or sloppy cap brings everybody closer to danger – not the kind of lesson anyone wants taught the hard way.
Protecting People and Places from Harm
Fact is, common sense and safety gear form the backbone when working with any volatile compound. Proper ventilation makes a big difference. Some labs and factories have switched up their equipment, adding local exhausts and improving room air exchanges. That cuts down how much 2-methylfuran drifts through a workspace.
Labels and regular training don’t get enough credit. Some workplaces set up routine hazard assessments, which brings in outside consultants or safety managers. The biggest improvements often come once everyone understands the risks. Clear instructions, marked storage, and correct gloves and masks stop health problems before they take root.
Food and drink companies keep a close watch on how much 2-methylfuran ends up in what we eat or drink. Regulatory limits come from animal testing data, so these agencies keep tabs as new studies give a fuller picture. Home cooks won’t ever reach those levels, but industry has more at stake.
Where Things Go from Here
Future studies are likely to close more gaps. Better testing with human volunteers is rare for these kinds of chemicals, but improved technology, like air monitoring and personal exposure badges, mean that those at risk can see real-time results. I think the road ahead leads to smarter workplace rules, stronger limits, and more careful handling – none of which happen by accident.
What is the chemical formula and molecular weight of 2-Methylfuran?
Chemical Formula of 2-Methylfuran
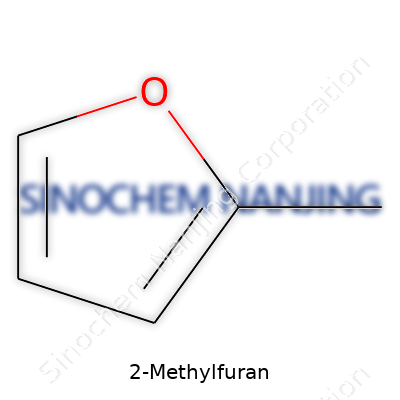
2-Methylfuran stands out as a simple organic compound, commonly labeled with the chemical formula C5H6O. Certain enthusiasts in the world of organic chemistry might picture this structure as a furan ring sprouting a single methyl group at the number two position. This slight tweak gives the molecule some distinct characteristics compared to its parent furan.
Molecular Weight Matters
2-Methylfuran records a molecular weight of about 82.10 g/mol. This value isn’t just an academic footnote. It plays a part in lab work, from dosing reactions to calculating yields. Recognizing a compound’s molecular weight lets researchers estimate how much material they’re handling and points to how that chemical could behave or move through a reaction setup.
Why Chemists Care About Small Molecules
Even compounds as modest as 2-Methylfuran can chain together a web of insights in chemistry research and production. Years working alongside analytical chemists, I’ve seen how even minor structural changes, like adding a methyl group onto a furan ring, can tweak physical and chemical properties in meaningful ways. Lab techs care about volatility, solubility, flammability, and odor. A shift in molecular formula often kicks off plenty of questions about handling and safety.
Where 2-Methylfuran Pops Up
This molecule isn’t just a curiosity in university labs. Researchers investigate it for potential roles in biofuels, pharmaceuticals, and fragrance manufacturing. Its relatively low boiling point and aromatic features lead many to consider it as a component in gasoline alternatives. That’s not just wishful thinking. Reports suggest 2-Methylfuran can punch up octane ratings and lends itself to more eco-friendly fuel blends, reducing reliance on petroleum sources.
Medicinal chemists sometimes investigate molecules like these for their building block potential. Because 2-Methylfuran comes with both a reactive ring and a methyl group, it sometimes steps in as a synthetic precursor or an intermediate in certain pharmaceutical preparations. Its versatility ties back to the understanding of molecular structure—anyone in drug discovery will tell you how a single extra carbon can turn a puzzle piece into a perfect fit.
Safety and Handling Insights
Speaking from experience, safety in the lab always tracks closely with the characteristics of the chemicals being handled. 2-Methylfuran’s volatility means extra ventilation isn’t just a nice touch, it becomes non-negotiable. Its flammability brings up memories of fire drills that felt over-the-top at the time, but those protocols shape habits that pay off over a long career. Proper labeling, sturdy spill kits, and up-to-date Safety Data Sheets help prevent mistakes that never show up in journal articles but make all the difference in daily lab work.
Building a Responsible Chemical Future
As industries lean harder into sustainable solutions, molecules like 2-Methylfuran get a new look. Bio-based starting materials and greener synthesis routes can shrink the environmental footprint of traditional chemical processes. Continued evaluation of toxicity and long-term impacts remains a cornerstone of responsible development. People want cleaner fuels and safer products without giving up performance, and 2-Methylfuran demonstrates how basic chemistry knowledge feeds into larger-scale changes.
Like most chemicals, the importance of 2-Methylfuran rests not just in its name or formula, but in understanding what that formula makes possible—better fuels, creative syntheses, and safe handling. Scientists, engineers, and regulators all benefit from staying fluent in the basics. That’s where world-changing ideas take root.
How should 2-Methylfuran be stored and handled?
Keeping It Safe in the Workplace
2-Methylfuran, a colorless liquid with a strong scent, pops up in labs and industrial settings as a solvent and a building block for other chemicals. I’ve worked with enough flammable liquids to know one thing: giving safety short shrift leads to ugly surprises. You pick up the bottle, catch the odor, and the label practically says: “Treat with respect.” This stuff doesn’t ask for much, but a little extra care prevents accidents.
What Makes 2-Methylfuran Tricky
Vapors from 2-Methylfuran ignite fast. Left uncapped, the fumes hang in the air, waiting for a spark. The flash point sits well below room temperature. It evaporates like ether. So, the storage space matters just as much as the container itself. Forgetting to seal the lid, or failing to vent the space, can turn a regular day into a fire drill. I saw one shelf of solvents go up because someone left a cap loose; things like this remind everyone to double-check each time.
The Storage Game Plan
Regular metal shelving won’t cut it. Flammable cabinets built for chemical storage keep bottles cool and out of direct sunlight. Storing 2-Methylfuran away from oxidizers is essential—this rule isn’t just on some test you pass in college. These chemicals play badly together and spark violence without warning. Putting incompatible substances together is like storing matches with fireworks: people do it until they don’t.
Glass bottles or metal cans with sturdy seals work better than plastic. Some plastics soften or break down if the solvent sits long enough. Secondary containment trays help catch leaks and spills, and labels must stay legible. People rush, they grab the wrong thing, and mistakes happen—clear labeling with permanent markers keeps confusion at bay.
The Everyday Handling Routine
Laboratories and workshops using 2-Methylfuran see the same hazards: splashes and fumes. Lab coats, gloves rated for solvents, and chemical splash goggles protect your skin and eyes. You start your day by cracking a window or flipping on a fume hood. Every bottle gets opened on top of a spill mat, not over notebooks, phones, or lunch. I’ve noticed that once PPE becomes a habit, fewer accidents show up in the incident log; muscle memory doesn’t leave much room for shortcuts.
Spills demand action, not panic. In practice, granulated absorbents or spill pillows go down before paper towels or water. Open flames, hot plates, and cigarettes don’t belong in rooms with 2-Methylfuran—lines on the floor and big red stickers do the talking long before someone reaches for a Bunsen burner.
Thinking Past the Basics
Training sits at the core of chemical safety. Someone new on the job learns how fast vapors build up and how easy it is to get careless. Regular safety drills and reminders in break rooms keep procedures from gathering dust. In my experience, honest conversations about near-misses teach valuable lessons. People respect clear rules around storage and waste disposal, especially when they’re part of the discussion.
So if the goal is to keep 2-Methylfuran from turning routine work into emergency calls, a bit of discipline goes a long way. Ventilated storage, good labels, sturdy containers, and a steady dose of attention help everyone on the team get home with ten fingers and two eyebrows—every single shift.
What precautions are required for the safe disposal of 2-Methylfuran?
Understanding the Risks and Realities
2-Methylfuran shows up in labs and manufacturing settings as a clear liquid with a strong, ether-like smell. It serves as an intermediate in chemical synthesis and can end up as a solvent or a fuel additive. Its straightforward appearance masks some nasty dangers. Vapors can catch fire quickly, and breathing in the fumes or splashing it on skin irritates and damages just about anything it touches. After working around chemicals for years, it is clear to me that you can’t take shortcuts with hazardous liquids like this. Taking a careless route does not just put a person at risk—it affects both co-workers and the surrounding environment.
Safe Handling Before Disposal
No one should think of tossing 2-Methylfuran down the drain or in the regular trash. That simple action could release toxic fumes into the air or pollute water. Working around this stuff, I’ve seen that the right gear saves a lot of trouble. Sturdy nitrile gloves, goggles, and a lab coat keep it off the skin and away from the eyes. Good ventilation is not just a luxury; an open window or reliable fume hood keeps fumes from building up. Spills need attention fast with absorbent, non-sparking materials—no sweeping under the rug, no makeshift fixes.
Choosing the Right Disposal Route
2-Methylfuran qualifies as hazardous waste in most places. That label means strict rules. It cannot mix with regular trash, recycling, or pour down the drain. From years of coordinating lab clean-outs, the number one thing: never mix leftover products unless you’ve done your homework on chemical compatibility. Some labs keep it in a labeled, sturdy, sealed container made of material that resists organic solvents—for many, high-density polyethylene or glass does the trick. Keeping the waste away from heat sources and sunlight avoids accidental ignition.
Many local governments set up hazardous waste collection programs. Calling ahead and explaining exactly what you have keeps surprises to a minimum—for both the holder and the waste handlers. Spilling, leaking, or worse, a fire during transport usually happens when people get cavalier. Padding and proper sealing make a difference. Paperwork—waste manifests—should always list all contents and quantities, so handlers know what to expect. Each step matters: honesty keeps accidents out of the news.
Environmental and Legal Responsibility
Poorly discarded 2-Methylfuran seeps into groundwater, destroys aquatic life, and can hang around in soil where it doesn't belong. Local, state, and federal rules all draw clear lines about improper disposal. Penalties and fines reach into the thousands. Laws aside, being careless with chemicals just fuels the public’s suspicion toward scientists and businesses handling these substances. Communities deserve informed, responsible neighbors, not more headaches and contamination.
Building a Safer Chemical Culture
Lack of training shows up in statistics reporting chemical accidents. Regular workshops, plain-language checklists by sinks and doors, and easily available safety data sheets all make a real difference. Sharing stories about close calls and lessons learned keeps new staff more alert. I’ve watched new lab members pick up good habits by seeing senior colleagues double-check waste labels or gently correct sloppy handling. Swapping out old policies for practical, experience-based approaches doesn’t just keep a site compliant—it keeps everyone safer, healthier, and avoids disruption when disposal time comes.
| Names | |
| Preferred IUPAC name | 2-Methylfuran |
| Other names |
Silvan 2-Furylmethane Methylfurane Methyl-2-furan |
| Pronunciation | /ˈtuːˌmɛθɪlˈfjʊəræn/ |
| Identifiers | |
| CAS Number | “534-22-5” |
| Beilstein Reference | 1206405 |
| ChEBI | CHEBI:34485 |
| ChEMBL | CHEMBL14294 |
| ChemSpider | 68295 |
| DrugBank | DB04114 |
| ECHA InfoCard | 100.011.382 |
| EC Number | 01-2119967775-19-0000 |
| Gmelin Reference | 1361 |
| KEGG | C01434 |
| MeSH | D013693 |
| PubChem CID | 7019 |
| RTECS number | LQ8925000 |
| UNII | F9F9G063EJ |
| UN number | UN2264 |
| Properties | |
| Chemical formula | C5H6O |
| Molar mass | 96.13 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | aromatic |
| Density | 0.917 g/mL at 25 °C |
| Solubility in water | slightly soluble |
| log P | 1.84 |
| Vapor pressure | 11.6 kPa (20 °C) |
| Acidity (pKa) | 5.75 |
| Basicity (pKb) | pKb = 7.55 |
| Magnetic susceptibility (χ) | -8.11×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.431 |
| Viscosity | 0.545 mPa·s (25 °C) |
| Dipole moment | 1.590 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 122.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −35.2 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2426 kJ/mol |
| Pharmacology | |
| ATC code | R05DB24 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H301, H319, H335 |
| Precautionary statements | P210, P261, P280, P301+P312, P304+P340, P312, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-2 |
| Flash point | 11°C |
| Autoignition temperature | 255 °C |
| Explosive limits | 1.8% - 14.8% |
| Lethal dose or concentration | LD50 oral rat 410 mg/kg |
| LD50 (median dose) | LD50 (median dose): 400 mg/kg (oral, rat) |
| NIOSH | RN2909 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-Methylfuran: "75 ppm (skin) |
| REL (Recommended) | 1,000 |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Furan 2,5-Dimethylfuran Furan-2-carboxylic acid |

