Seeing Formic Acid through Experience and Fact
Tracing Formic Acid’s Story
Formic acid takes its name from ants. Scientists discovered it by distilling dead ants in the seventeenth century. Ants produce this substance naturally as a weapon. In nature, this is a chemical meant to defend, to mark a trail, to warn. Chemists later learned to pull it out of other plants too, including stinging nettles. Through the centuries, the applications drifted from curiosity to practical necessity. By the end of the nineteenth century, synthetics led the way. Production ramped up thanks to advancements in catalytic reactions, letting chemical plants supply industry and agriculture at scale. The story follows a line from biology through curiosity to big industry.
Product Overview and Uses with Real-World Impact
You see formic acid everywhere in industrial settings even if you never hear its name. Leather factories rely on it to fix dyes in hides—a key step in making shoes, bags, and jackets that last. Rubber plants turn to it to help coagulate their latex, making products soft enough or solid enough depending on what is needed. Cattle farms douse silage in formic acid to slow spoilage, letting livestock eat safer food longer. Textile dying operations include formic acid to keep colors bright. Even beekeepers use it to control mites threatening their hives. Its grace comes from smallness. One simple molecule, able to nudge reactions forward or hold back spoilage, sometimes just by being acidic in clever ways that other acids cannot match.
Physical and Chemical Properties: Simple Yet Potent
At room temperature, formic acid flows as a colorless liquid with a pungent odor that stings the nose. Touching it leaves a burning sensation, which anyone can feel with a drop even diluted. Chemically, it sits at the bottom of the carboxylic acid family. Its formula, CH2O2, packs punch into a single carbon. The melting point floats just above the freezing point of water and the boiling point sits a few dozen degrees past one hundred Celsius. It mixes with water, ethanol, and ether—straightforward to dilute, effective to use. Many reactions start with this simple liquid, thanks to its reactivity and solubility. This blend of characteristics brings danger and utility in equal measure.
Technical Specifications & Labeling: Reading between the Numbers
Buying formic acid for a plant or a lab demands attention. Labels spell out the purity, sometimes as percentages above 85 percent. Users in agriculture or food preservation care about the level of impurities—metals, water, other acids. The United Nations assigns a number, UN 1779, telling shippers about the dangers. Labels highlight not just chemical Grade but show pictograms for burns and corrosion, since the acid etches skin and attacks metals. These rules grew out of accidents and lessons from history; handling formic acid safely has always meant respecting its bite.
Making Formic Acid: From Past to Present
Old experiments began with ants, but modern plants choose carbon monoxide and methanol, or split oxalic acid. The dominant method flows through a reactor where methanol and carbon monoxide meet in the presence of bases, freeing up formic acid downstream. This process gives better yields and keeps waste low, meeting the needs of scale. Chemists favor these routes not just for efficiency but for environmental footprint, since waste solvents and by-products cost money and invite scrutiny from regulators.
Reactions and Chemical Modifications: Building on Foundation
Chemists know formic acid best for its dual personalities. Sometimes it acts as a reducing agent, sometimes as a feedstock. Add concentrated sulfuric acid and it breaks down to carbon monoxide and water—a laboratory method for gas generation. React it with alcohols, and esters form, each with different fragrances and uses in flavoring. Oxidizing agents can crack it into carbon dioxide. The molecule invites tinkering, putting it at the foundation of more complex syntheses in drugs and materials. Its carboxyl group gives chemists options, with the result that formic acid ends up inside many other finished chemicals without ever being visible.
Other Names on the Shelf
Chemists use the Latin-inspired term, methanoic acid, but most still call it formic acid on the job. Safety data sheets and chemical catalogs sometimes list "aminic acid" from early literature. Some agricultural products call it E236 under food additive regulations. The synonyms remind users about context—biological, industrial, food, or laboratory—and signal what rules apply.
Staying Safe: Rules and Habits that Save Lives
Working with formic acid means staying alert. Skin contact brings immediate pain and possible burns. Inhaling vapor irritates lungs and can trigger coughing fits or worse if the concentration climbs. Accidents in plants have led to burns and even fatalities. Good safety habits matter—eye protection, gloves resistant to acids, keeping ventilation high, storing containers away from metals and flammable liquids. Familiarity breeds carelessness, but the acid always punishes mistakes.
Where Formic Acid Makes a Difference: Application Areas
Cattle farmers lean on formic acid to store silage through humid summers. Tanners integrate it into their schedules as they treat hides. Automotive and electronics makers sometimes use it to adjust acidity in specialty applications. Textile mills put it into dyes to lock in complex colors for fabrics. Pest control specialists and amateur beekeepers alike deploy it against varroa mites in apiaries. Water treatment plants might turn to it when specific pH levels must be maintained for hours at a time. It quietly links agriculture, chemistry, manufacturing, and even apiaries in a way few basic chemicals manage.
Exploring Research and Development
Researchers explore catalytic processes to make formic acid more sustainably. Electrocatalysis receives attention, especially using renewable electricity. Some labs see formic acid as a candidate for hydrogen storage. Load the hydrogen onto its structure; release it later using a catalyst and liberate clean gas on demand. Other scientists put it under microscopes, curious about new esters or novel solvents. The field persists because the molecule’s simplicity hides room for creative chemical transformations.
Understanding Toxicity: Knowledge That Protects
Doctors and toxicologists record cases of accidental poisoning, mostly from accidental ingestion or workplace accidents. The acid can prove fatal in large doses. Even short, sharp exposures leave marks—burns, ulcers, lung irritation. Regulatory boards set strict exposure limits for workers, with time-weighted averages and mandates for airing out workspaces. Vigilance lowers the risk, but training and design matter too. Equipment must resist corrosion, emergency protocols must be second nature, and training must never get stale.
Future Prospects: Where Formic Acid Heads Next
Attention turns to green chemistry and sustainability. Production methods look for routes that avoid fossil inputs and lower emissions. The push toward hydrogen as a future fuel brings new research in storing and releasing the gas safely, with formic acid offering real-world advantages thanks to its liquid nature and chemical stability. Industrial producers search for ways to reduce by-products and boost overall efficiency for chemical footprints that can meet new regulatory standards. From farm to factory, from the lab bench to possible hydrogen economies, the coming years look busy for formic acid’s story.
What are the main uses of formic acid?
Beyond the Chemistry Lab
Formic acid shows up in some surprising places. Farmers use it almost every day, especially those working with hay or silage. When storing winter feed, nobody wants it to rot. Formic acid acts as a preservative, stopping the growth of bacteria and mold in silage and haylage. This keeps fodder fresh and livestock healthy. About a third of global formic acid goes to animal feed and silage preservation, according to data from the European Chemical Industry Council.
Workhorse in Leather and Textiles
Tanneries need acids to handle hides. Leather processing uses formic acid to help remove hairs and open up fibers, prepping them for more treatment. The textile trade relies on it too. Dyeing cotton or wool calls for the right pH balance, and formic acid helps tweak the acidity, making colors come out deeper and longer-lasting on fabric. It also works to clean wool, washing away grease and lanolin.
Role in Industrial Chemicals
Formic acid forms part of many industrial reactions. It steps in as a reducing agent—basically giving up electrons to help other chemicals react. Some rubber factories use it to "coagulate" latex, turning sap into bouncy sheets that will become tires, gloves, or shoes. The rubber industry wouldn’t run as smoothly without it.
Food Additive and Safety
Not everyone realizes that formic acid gets into food production as well. It's approved as an additive for animal feed in the EU and even in some foods. The acid drops the pH, keeping bacteria like salmonella in check. Grain storage, fish meal, and silage all benefit from this. The U.S. Food and Drug Administration rates it as "Generally Recognized As Safe" for use at certain levels. Even so, most food on grocery shelves has only trace amounts, far away from the strong stuff handled in factory tanks.
Green Cleaning and Renewable Energy
Most household cleaners don’t list formic acid on the label, but it can dissolve limescale and rust, making it handy for heavy-duty descaling in industry. Its popularity is growing among cleaner-makers looking for an alternative to more corrosive chemicals. Some researchers are also looking at formic acid for storing hydrogen. If hydrogen cars ever catch on, formic acid could help solve big problems in storage and transport since it holds hydrogen in a stable, liquid form.
Human Health and Safety
People learn early in chemistry lab that formic acid can sting. The stuff in ant bites and stinging nettles is naturally occurring formic acid. Factory-strength acid can burn skin or damage eyes, so strict safety controls protect workers. Firms using formic acid must plan for air handling and spill control. Studies show that long-term low-level exposure doesn't build up in the body, but careful handling stays non-negotiable on the job site.
Room for Safer and Greener Choices
While formic acid is far less toxic than many acids, recycling and disposal pose challenges. More companies now look into biobased production, using renewable feedstocks and greener processes. As greener chemistry advances, costs drop and safety improves. This change helps people, animals, and the environment at once—and drives innovation through the supply chain.
Is formic acid hazardous to health?
Understanding Formic Acid’s Place in Daily Life
Formic acid pops up in surprising places. Ants use it as a defense mechanism, and it shows up in certain foods and industrial settings. Growing up on a farm, I remember the sharp, stinging smell escaping from the barn whenever concentrated cleaning agents were used. Turns out, that sting often came from formic acid lurking among the chemicals that kept hay from going moldy and kept pests away.
Industrial workers know the risks all too well. Anyone who’s worked with concentrated formic acid will tell you that even a quick splash on the skin leaves a red, angry welt. It acts fast and burns deep. Eyes and lungs feel the impact, too. Inhaling even moderate levels of vapor can leave someone coughing and short of breath. Skin contact can lead to blistering. If it gets inside the eyes, that becomes a medical emergency.
Everyday Exposure and Long-Term Risks
Most people run into formic acid in weak concentrations—think of traces found in some foods. In those small doses, it doesn’t bother most people. The concentration makes a huge difference. In controlled scientific studies, researchers discovered that solutions under 1% caused little reaction. Higher concentrations ramp up the risk fast. The U.S. National Institute for Occupational Safety and Health (NIOSH) calls for strict exposure limits: workers shouldn’t breathe in more than 5 parts per million over an eight-hour shift.
Prolonged or repeated exposure isn’t something to dismiss. Chronic contact with formic acid can lead to skin rashes known as dermatitis. There are even cases of asthma-like symptoms, particularly in folks handling chemicals all day. Eyes and respiratory tract line up as the most sensitive areas. Burns in these tissues take a long time to heal, and sometimes leave behind scars or long-lasting sensitivity.
What Science Tells Us
Studies have tracked formic acid’s effects on both animals and humans. In rodents, high doses did significant organ damage. Human case studies reveal that swallowing concentrated formic acid leads to painful burns in the throat, mouth, and stomach. This rarely happens by accident in households, but it does occur in workplaces with lax safety measures. The International Agency for Research on Cancer hasn’t labeled this acid as a cancer-causer, yet the real health burden comes from irritation, chemical burns, and, in rare cases, organ damage.
Handling Risks with Simple Precautions
Personal experience and health data both highlight the need for proper handling. Gloves, safety goggles, and good ventilation offer the best line of defense. On the farm, we used cheap cotton gloves by default—not a good call for acids. Nitrile or rubber gloves stop formic acid from seeping through. Well-sealed goggles protect eyes and take away a major source of worry.
Regulators echo the need for training and clear labeling. Every workplace using chemicals should run safety drills and keep emergency eyewash stations ready. At home, products containing formic acid deserve respect, too: keep them out of reach of kids, and never transfer them into unmarked containers.
Better Awareness Equals Better Health
The hazards tied to formic acid are real, but so are the solutions. Knowledge, the right safety gear, and proper storage make all the difference. This isn’t about scaring people away from an important industrial tool. It’s about respecting formic acid’s potential and putting health first in any setting where people might encounter it.
What is the concentration or purity of formic acid sold?
Why Purity Levels Matter in Everyday Use
Buying formic acid isn't like picking detergent off a supermarket shelf. The bottle doesn't just list "formic acid," and that's that. Purity levels can make or break whatever project or product you're working on. In my years handling everything from industrial cleaning to gardening, purity often stands between results and disappointment. The wrong concentration can ruin leather, eat through equipment, or mess up a beehive treatment.
Typical Purity Ranges You Actually See
If you browse online marketplaces or chemical suppliers, most bottles list a purity of 85%. This figure didn't spring from nowhere. Suppliers settled on this because it balances safety and performance pretty well. Anything above 90% brings risk. Even experienced hands get nervous dealing with near-pure formic acid since it's not just corrosive—it fumes, and a splash can cause real harm.
Some industries want stronger stuff. Labs sometimes source 95% or even higher, but it’s uncommon to see that for everyday industrial tasks or retail shelves. These ultra-high grades cost more, require special storage, and demand careful handling protocols. There’s only a handful of everyday uses for such concentration, like certain lab syntheses or niche manufacturing.
Meanwhile, diluted solutions fill another market need. Anything below 50% doesn’t pack much reactive punch. Beekeepers—folks I’ve known for years—often work with solutions hovering around 65%. It gets the job done without scaring the pants off anyone in the hive. Even here, purity matters because residues and impurities can threaten bees. Gardeners might use diluted forms as an herbicide, but they’re not lining up for 99% bottles.
What Ends up in the Bottle?
Industrial companies know that pure chemicals attract contamination fast. Most formic acid drums ship sealed tight, but the nature of the stuff means even a small air leak changes the game. Besides water, trace metals and organic residues creep in, especially if storage tanks or pipes aren’t flawless. Over the years, I’ve seen rusty connections spoil batches before they ever hit the package line.
Retailers usually list only one number—that headline purity. Below the surface, there’s another story. Safety Data Sheets show water content, and responsible sellers share extra info like iron, sulfate, or chloride levels measured in parts per million. It’s worth checking the paperwork if your process can’t handle surprises.
What Can Buyers Really Do?
Check the label. Beyond headlines, ask for test results. Reputable suppliers are open about the lot analysis for every shipment. If a supplier dodges simple questions about impurities or can’t send documentation, consider that a warning sign. Over the years, I’ve called out sellers whose bottles didn’t match their promises, and problems rarely got better the longer you waited.
Smaller users avoid risk by buying only what they can use quickly, reducing time for contaminants to sneak in. Anyone handling high-concentration formic acid should invest in gloves, goggles, and solid ventilation. It’s not paranoia—it’s learned from close calls and scars.
Looking for Solutions and Safer Access
Bigger outfits lobby for clearer regulations and better labeling. More transparency helps everyone avoid accidents and saves money by reducing waste. As a buyer, never cut corners or believe you’re immune to risk. If purity and safety sound boring, try cleaning up a chemical spill with a flimsy mop. Everyone from chemists to farmers deserves the truth on that label. That’s how you make formic acid useful, not dangerous.
How should formic acid be stored and handled?
Formic Acid Isn’t Ordinary—It’s Strong Stuff
Folks who work with formic acid won’t forget its pungent, eye-watering smell. As a simple carboxylic acid, it finds its way into farms, tanneries, and labs. Most people remember its sting from ant bites, but a diluted form skips from chemical drums to countless industrial uses. The risk runs deeper, though. Spills and splashes create real trouble. After a few years in a biotech facility, the lessons stick: respect the acid, and it won’t bite you.
Protecting People Comes First
Skin burns, eye damage, even breathing trouble—a quick mistake working with formic acid can send folks to urgent care. Safety goggles, chemical-resistant gloves, and an apron go on before anyone starts pouring or mixing. Goggles never feel like overkill when you see how fast acid vapor can irritate eyes or burn skin. Disposable nitrile or neoprene gloves, not latex, bring better protection. In tight spaces, full-face shields help. For larger quantities, workers step behind splash guards. Respirators with acid cartridges become mandatory if fumes get thick. The right personal protection stops accidents from turning into stories shared in hospital waiting rooms.
Containment: No Shortcuts Here
Formic acid likes to eat through ordinary containers—the stuff chews metal, soft plastics, and even concrete in high enough concentration. Polyethylene or stainless steel (316 grade and above) works well for storage. Only containers with strong, chemical-resistant linings hold up over time. Caps stay tight, vents prevent pressure build-up, and labels list concentration and hazard class loud and clear.
Drums land in cool, well-ventilated rooms away from sunlight and heat sources. No one stores drums near anything reactive, especially bases—mixing with caustic soda leads straight to a hazardous reaction. Acid isn’t stored on rickety shelves or alongside flammable solvents. One misstep leaves folks cleaning corrosive leaks for days. Bear in mind, keeping it below 30°C helps reduce vapor and makes storage safer.
Spill Response: Fast Action Matters
Spills happen, even if everyone tries their best. Acid finds gaps, runs under pallets, slips between tiles. Having a ready-to-go spill kit changes everything. Neutralizing powders—like sodium carbonate—soak up and neutralize spilled acid before mopping. Water alone doesn’t help; diluting just spreads the danger. Proper training teaches staff to clear the area, air out fumes, and notify their supervisor immediately. Quick team response spares serious injury and lost work hours.
Why Documentation Helps
Chemical safety sheets—SDSs—don’t just fill binders. They give workers the facts: exposure limits, emergency first aid, and storage do’s and don’ts. Strict record-keeping also means every drum gets tracked, shelf lives don’t creep by unnoticed, and any near-misses get logged for future review. This attitude not only satisfies inspectors, but protects everyone down the line.
Better Training, Fewer Incidents
Not everyone grows up knowing what “corrosive” truly means. Regular training opens eyes. Hands-on demos, simple instructions posted by storage rooms, and mechanical ventilation keep new and experienced staff on the same page. Managers who invest in ongoing safety meetings notice fewer injuries and tighter teamwork.
Safer Options and Smarter Choices
Switching to less concentrated forms, using closed delivery systems, or automating processes reduces the number of people exposed to risk. Consulting with certified chemical safety pros can uncover small changes that bring big safety gains. No shortcut matches the peace of mind earned from safe storage and sound habits.
Can formic acid be shipped internationally?
The Challenge of Moving Chemicals Globally
A lot of folks don’t think twice about what it takes to send a chemical like formic acid from one country to another. If you’ve ever dealt with hazardous materials, you know the process requires constant vigilance. Formic acid stings if you get it on your skin, but it brings big benefits for farms, textile mills, rubber makers, and even folks keeping beehives healthy. The trick comes in moving it safely, protecting workers, and meeting the rules that each country writes into law.
Hazards on the Shipping Dock
Formic acid belongs to a class of chemicals the United Nations calls “Dangerous Goods.” Why the label? It eats through metal, irritates skin and lungs, and reacts fiercely if the wrong stuff gets mixed in during transit. In my experience working with shipment restrictions, even a drum that leaks a few drops can lead to big headaches; fire departments don’t take kindly to spur-of-the-moment chemical spills on their watch.
International shipping starts with international rules. The UN’s International Maritime Dangerous Goods (IMDG) Code, the IATA guidelines for air, and a string of national regulations layer up. If you’re sending formic acid to India, Brazil, or Germany, each stop means fresh paperwork. Anyone skipping steps in packaging, labeling, or emergency plans will find their delivery stuck in customs—or facing fines that can wipe out several quarters’ profit.
Protecting People, Safeguarding the Planet
The reason for strict rules shows up every day in chemical mishaps. Poor labeling, or cutting corners with packaging, invites spills in port warehouses, triggering evacuations and environmental damage. Take the 2019 incident in Houston, where a shipping container of chemicals, including acids, triggered fires and air warnings for miles. No business wants their product to be the cause of the next headline.
Some firms train their staff once, then assume everything will go smoothly the next dozen times. Reality treats complacency harshly. Regulators run inspections, rejecting shipments that don’t match paperwork or that miss the required spill kits. Emergency phone numbers need to work anywhere the cargo travels, or a single accident can escalate fast without backup.
Making Sense of a Tangled Web
Shipping formic acid safely takes more than meeting the bare minimum. Freight forwarders with deep experience in chemical logistics help smooth the process by keeping up with the rules that change every few years. Clear communication between shipper and carrier matters as much as following the law. I’ve watched small exporters lean on advice from industry groups or collaborate with larger, established partners to keep on top of the latest updates.
A fix worth the effort means investing in better training for handlers, using leak-proof drums, and double-checking compliance with both the outgoing country and the destination. Technology helps, too; digital track-and-trace can flag route changes, or delays that could cause cargo to sit in hot, corrosive conditions. If one country’s customs office blocks a delivery, the real value comes from a team problem-solving in real time, not pointing fingers.
Learning from the Experience
From a practical perspective, any company thinking about global markets should budget for regular compliance audits and keep an open line to regulators. Small errors scale up fast at the international level. People, not just protocols, drive the safest possible outcome for each drum and container that leaves one shore and arrives on another.
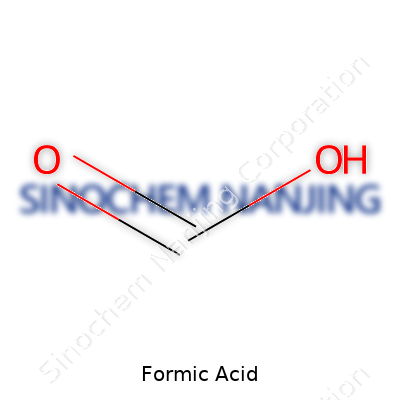
| Names | |
| Preferred IUPAC name | methanoic acid |
| Other names |
Methanoic acid Aminic acid Hydrogen carboxylic acid Formylic acid |
| Pronunciation | /ˈfɔːrmɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 64-18-6 |
| Beilstein Reference | 505924 |
| ChEBI | CHEBI:30751 |
| ChEMBL | CHEMBL715 |
| ChemSpider | 967 |
| DrugBank | DB01942 |
| ECHA InfoCard | 03d43d43-d708-4e7a-9279-32e4bbf9649e |
| EC Number | 200-579-1 |
| Gmelin Reference | Gmelin Reference: 604 |
| KEGG | C00047 |
| MeSH | D005547 |
| PubChem CID | 284 |
| RTECS number | LQ4900000 |
| UNII | 9YRO3M6FFE |
| UN number | UN1779 |
| Properties | |
| Chemical formula | HCOOH |
| Molar mass | 46.03 g/mol |
| Appearance | Colorless, fuming liquid with a pungent, penetrating odor. |
| Odor | Pungent, penetrating, characteristic |
| Density | 1.22 g/cm³ |
| Solubility in water | miscible |
| log P | -0.54 |
| Vapor pressure | 43 mmHg (20°C) |
| Acidity (pKa) | 3.75 |
| Basicity (pKb) | 10.3 |
| Magnetic susceptibility (χ) | χ = -19.6 × 10⁻⁶ |
| Refractive index (nD) | 1.370 |
| Viscosity | 1.57 mPa·s (at 25°C) |
| Dipole moment | 1.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 129.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −425.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −254.0 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | J01XX01 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H226, H314, H331 |
| Precautionary statements | P261, P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | ~68°C |
| Autoignition temperature | 601 °C |
| Explosive limits | 3.7–33% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 730 mg/kg |
| LD50 (median dose) | LD50 (median dose) of formic acid: **730 mg/kg (oral, rat)** |
| NIOSH | FF6300000 |
| PEL (Permissible) | PEL: 5 ppm |
| REL (Recommended) | 30 mg/m³ |
| IDLH (Immediate danger) | 30 ppm |
| Related compounds | |
| Related compounds |
Formate Methanol Acetic acid |

