Fluorosulfuric Acid: The Double-Edged Sword of Chemical Progress
Historical Development
Fluorosulfuric acid occupies a peculiar spot in the history of chemical innovation. Chemists first explored its potential in the late 19th and early 20th centuries, driven by the hunger to unravel stronger acids than those sitting on the lab shelf. As stories in chemistry go, it started with curiosity. Otto Ruff, in 1906, managed to isolate and characterize fluorosulfuric acid, marking the birth of an acid that soon gained both fascination and fear among those who handle it. I remember reading journals from that era and noticing just how quickly the acid's unique abilities grabbed the attention of both academic and industrial chemists. Its strong proton-donating power offered immediate appeal. The acid soon drew comparisons to its chemical cousin, sulfuric acid, but chemists kept returning to its much higher acidity and peculiar reactivity. Ever since those early days, the story of fluorosulfuric acid has been a tightrope walk between opportunity and hazard.
Product Overview
Ask a chemist about fluorosulfuric acid, and you'll often hear it referenced as a superacid—an acid even mineral acids can't compete with. Its formula, HSO3F, might look simple, but it packs an extraordinary punch. From my own experience handling chemicals in a research lab, the acid’s properties stand out right away. Once released from its secure container, its fuming tells a story of volatile strength. It pushes the boundaries of what acids can do: protonating even substances that put up a significant fight against other acids. For industrial producers and bench scientists alike, that sort of strength both excites and terrifies. Fluorosulfuric acid is far from mainstream. It's more like a specialty tool—best suited to those who know what they're doing and respect what it can unleash.
Physical & Chemical Properties
It’s one thing to list the freezing and boiling points of a substance, but what really leaves a mark about fluorosulfuric acid is its volatile fuming and the almost surgical clarity with which it carves up organic and inorganic matter. You pour it and it steams in open air, sending sharp, irritating fumes skyward. The liquid appears colorless but hides a burden of risk. Its density and slippery feel can fool the unwary. Lining up chemical properties, its strength blows past sulfuric acid, and it can dissolve glass while finding kinship with only a few other superacids. What always stands out, from first encounter, remains its aggressive reactivity—capable of pushing tough reactions to completion but punishing any mistakes in handling.
Technical Specifications & Labeling
In practice, fluorosulfuric acid comes tightly bottled, carrying standardized warning labels with skulls, bones, and ample precautionary language. In every lab I’ve worked in, anyone who even thinks of opening a container must triple-read its identifiers. Clear hazard symbols, statements about contact dangers, and guidance for spill or exposure responses fill its documentation. The acid’s technical specifications aren’t just a checklist—they’re warnings handed down by hard-won experience. Regular inspections and tight controls don't come from bureaucracy alone but from necessity. We're not talking about compliance for the sake of paperwork; proper storage temperature, container material, and PPE amount to the line between routine and incident.
Preparation Method
Chemists don’t just stumble into making fluorosulfuric acid. The industrial process builds on direct contact between sulfur trioxide and hydrogen fluoride, releasing heat and fumes that demand fortress-level control. Even seasoned researchers step back and double-check every fitting and gasket before pressing on. I’ll never forget watching strict protocols deployed during a demonstration of its synthesis—the focus sharpened across the room, and the silence spoke for itself. Mistakes here do not just spoil a reaction; they threaten lives. This is a substance that tests the mettle of chemical engineers and lab workers alike, driving home the importance of experience and respect for the tools of the trade.
Chemical Reactions & Modifications
Ask any synthetic chemist and you’ll hear stories about the opportunities fluorosulfuric acid offers in pushing reactions that would otherwise stall or crawl along. It can protonate even the most stubborn bases, opening doors to creativity in organic and organometallic synthesis. This acid doesn’t just play a supporting role; it changes the game. People smarter than me have used it to prepare difficult intermediates and carry out alkylations, isomerizations, and rearrangements that other acids simply can’t touch. On the flip side, its extreme reactivity means it can destroy delicate substrates or react with vessel materials, driving up costs and requiring custom glassware. This double-edged trait has sparked innovations in reaction setups but continues to act as a limiting factor when it comes to wider adoption. Laboratories performing these reactions pour time into protocols for neutralizing and disposing of residues—a reality rarely discussed in basic chemistry coursework but solidly present for anyone familiar with cleanup duty.
Synonyms & Product Names
In the catalogues and research papers I’ve seen, fluorosulfuric acid usually pops up as “HSO3F” or “fluorosulphonic acid.” Some chemists call it “FSO3H,” replacing a vowel but not the underlying punch. Alternative names don’t change what it does, but you’ll see them in patent filings and international texts. That sort of variation can trip up a reader, or even a purchasing agent. It’s a minor point until it causes the wrong material to show up in the mailroom, and then it matters a great deal. Consistency in nomenclature becomes a quiet safeguard that keeps research and production running smoothly.
Safety & Operational Standards
Working with fluorosulfuric acid leaves a deep impression; the safety rules don’t allow for shortcuts. Full-face shields, acid-resistant gloves, fume hoods with reliable airflow—these become routine, not luxury, whenever the bottle makes its appearance. As someone who has witnessed minor exposure incidents, I can say without hesitation that the discomfort, burns, and respiratory irritation aren’t hypothetical. Regulations in Europe and North America demand airtight procedures not as box-checking exercises but as blueprints for survival. Response drills and clear documentation form an unspoken pact among lab workers: your well-being depends on the other person getting things right. Rarely do you see a chemical that compels this level of group discipline.
Application Area
Outside academia, the main users of fluorosulfuric acid sit in the specialty chemical and pharmaceutical sectors. It steps in where conventional acids fall short, helping to forge tough bonds or catalyze persnickety reactions. Petrochemical fields tap its strength for alkylation, synthesizing additives for fuels using a tool others can’t match. High-end research on complex molecules often leans on this acid, squeezing every ounce of yield out of rare and expensive starting materials. Despite those advantages, the acid’s volatility, cost, and risk keep it from becoming a staple in bulk production. Instead, it belongs to the short list of uncommonly valuable yet undeniably hazardous reagents, up there with the kind that both inspire and humble those who wield them.
Research & Development
Research groups continue to dig into ways of harnessing fluorosulfuric acid with less risk. I’ve seen teams experimenting with microfluidic reactors and new protective materials, hoping these approaches can reduce exposure and tame its volatility. Many researchers aim to shrink the required quantities by using catalytic amounts or confining reactions to closed, remote-controlled systems. The push for alternative solvents and upgraded PPE drives a significant chunk of industrial R&D budgets wherever this acid plays a role. People working closely with it share a common goal: maximize benefit, minimize harm, document every lesson, and teach the next person better habits than the last. This kind of vigilance and innovation draws a line between chemistry’s past and its future, showing that the culture around hazardous materials is not frozen in time.
Toxicity Research
The impact of fluorosulfuric acid on living tissue is unambiguous—contact brings burns, and inhalation means respiratory distress that can rapidly escalate into medical emergencies. Toxicology studies underscore its ability to penetrate skin, sometimes even with brief exposure, and its fumes lodge deep in the lungs. Over the years, published case studies and accident reports keep the lessons vivid. I’ve attended seminars where medical professionals and safety officers review response plans, sharing cautionary tales that stick even longer than any regulation. These lessons extend beyond the lab, shaping the policies of companies and universities all over the world. Fluorosulfuric acid has forced the chemistry community to take both prevention and response far more seriously, and for good reason.
Future Prospects
Fluorosulfuric acid will keep its place as a uniquely powerful tool while innovation shapes how it’s used. Growing attention on environmental impact and worker safety shines more light than ever on the need for smarter handling, safer processes, and greener alternatives. I suspect that as new reaction technologies roll out—especially those built on automation and remote handling—the field will open up new applications or safer ways to leverage the acid’s strengths. While its raw power makes it hard to replace in some corners, there’s unmistakable momentum toward methods that seek the same results with cleaner waste streams and fewer hazards. What I see in the younger generation of chemists is not just caution, but creative ambition to do more with less risk. Fluorosulfuric acid started as a marvel of chemistry, and its future relies just as much on respect as it does on raw capability.
What is Fluorosulfuric Acid used for?
One of Chemistry’s Heavy Hitters
Fluorosulfuric acid grabs attention with more than just a complicated name. This stuff belongs to a small group known as “superacids.” Its claim to fame goes beyond being much stronger than sulfuric acid. You drop a little water on it and the reaction gets violent right away. Add a little organic matter and you get carbonization in seconds. For most folks, this acid seems downright terrifying. Still, inside a chemical plant, fluorosulfuric acid has clear jobs that nothing else can handle quite as well.
Foundations of Modern Chemistry
The biggest use for fluorosulfuric acid sits in the creation of specialty chemicals. It gets poured into the mix during alkylation, where chemists bond larger hydrocarbon pieces together. Oil refineries and fuel makers get a boost from this acid since it can turn isobutane and other hydrocarbons into high-octane gasoline compounds. No regular acid does the trick. Through personal experience visiting research labs, I saw how scientists used extreme caution with this material. They wore double gloves and heavy face protection. One slip, and there’s risk of nasty burns and fumes that can hurt the lungs in seconds.
Pharmaceuticals and Custom Molecules
Beyond gasoline, this acid makes life easier for drug manufacturers. Fluorosulfuric acid forms a backbone for making sulfonated organic compounds. These building blocks show up in antiviral medicines and dyes. By activating carbon rings or swapping out hydrogen atoms, chemists can craft molecules with new properties. A synthetic chemist friend told me how the acid let her introduce sulfonic groups at spots other acids would not touch. In the world of pharmaceuticals, every small tweak to a molecule can lead to life-changing drugs.
Electronics and Fluorination
Electronics run better on miniaturized, stable materials. Here’s where the acid helps again, especially in making fluorinated chemicals used for chip manufacturing. The acid starts off reactions to add fluorine atoms to organic compounds. These new chemicals can resist heat, water, and wear much better than the base material. Semiconductor engineers trust fluorosulfuric acid with some of their most sensitive work, since mistakes show up in lost yield and expensive downtime.
Risks and a Push for Safety
The risks of fluorosulfuric acid are not just hype. Emergency manuals label it a top-tier hazard. My own experience speaking to lab managers confirms the heavy training required before anyone even opens the storage containers. There’s no room for shortcuts. Fume hoods, spill kits, and chemical-neutralizing agents stand ready. Over recent years, industry groups have invested in better leak detection and safer handling carts, trying to keep accidents from happening in the first place.
A Call for Better Alternatives
Groups like the American Chemical Society push for safer replacements whenever possible. More sustainable routes for alkylation are under research, with solid acids and ionic liquids getting more attention. These alternatives don’t match fluorosulfuric acid’s power yet, but interest grows as safety and environmental concerns rise. Opening this conversation about alternatives helps keep chemists healthy and reduces the risks of disastrous spills.
Fluorosulfuric Acid in Today’s World
Chemistry depends on a few strong but dangerous players, and fluorosulfuric acid is one of them. Artful handling keeps it useful for making fuels, medicines, and tech that modern life depends on. As scientists chase safer options, the hope is that someday this acid stays sealed on the shelf except for truly necessary cases.
What are the safety precautions when handling Fluorosulfuric Acid?
Why Real Precautions Outweigh the Usual Tools
Fluorosulfuric acid has a reputation that’s earned. It packs one of the most aggressive punches among industrial acids. Staring at a bottle isn’t enough to size up the risks. Odorless, it can burn skin deep long before you notice. A single drop spells trouble. I remember someone in my college lab ignoring a splash on their glove—about two minutes later, they realized a burning sensation had already spread beneath the glove. Emergency protocols turned into chaos. That story stuck with me. So, handling this chemical calls for more than ticking boxes.
Personal Protection: No Shortcuts
Think thick acid-resistant gloves instead of flimsy nitrile ones. Face shields, not just safety goggles. Long lab coats of non-permeable material, sturdy shoes, not sandals or cheap sneakers. Even with good gloves, always double-check for tears or pinholes—acid finds every weakness. One forgotten barrier is a quick route to a hospital. Always suit up before opening that bottle.
Know Your Surroundings
Work in a proper fume hood. The vapors have a mind of their own, capable of drifting and turning moisture—even in your lungs—into more acid. Good air pull, clear sashes, and tidy counters are your real friends. I’ve seen crowded benches where someone lost track of what was being mixed. Before you start, clear out clutter, stow unused glassware, and leave only essentials within arm’s reach.
Handling and Disposal: Respect Every Step
Pouring is never a race. Pour this acid slowly, aiming for the container’s inner walls to cut splash risk to zero. Always use glass, Teflon, or similar containers—metals spell disaster. People forget about pressure. Never cap a bottle too tight after a reaction; gas build-up can blow the cap across the room. Dilution demands its own respect: always add acid to water, not the other way around—otherwise, boiling and spitting ensue.
Cleanup is no place for improvising. Keep piles of sodium bicarbonate and copious water at hand for neutralizing spills. Once a spill happens, tackle it immediately with proper neutralizer, never just water, or the situation will escalate. Tools and glassware need separate cleaning—nothing that touched the acid should mix with your normal wash-up pile.
Emergency Habits: Practice Means Survival
Have a wash station within sprinting distance and know exactly how to use it—fifteen minutes rinsing under running water, no shortcuts. Phones for emergency calls should work. Don’t keep the accident protocol in a folder across the building; post it on the wall. Drill those steps on a quieter day, not during a crisis. Quick, confident moves save skin and sometimes lives.
Sharing Wisdom and Reducing Risk
Every workplace that lets employees handle fluorosulfuric acid should run safety workshops, not just pass out pamphlets. Encourage people to retell mishap stories, learn from near-misses, and demand accountability from everyone at the bench. Supervisors should observe, correct, and never assume someone already knows the right way. No fear-mongering—just a steady push to make safety habits second nature.
Serious chemicals demand serious respect—from the moment they hit the bench, to every scrap of cleanup. Experience has shown me that shortcuts and overconfidence never end well. Real safety comes from treating every drop as if it’s the one that could change your life. That’s a lesson you carry long beyond the lab.
What is the chemical formula of Fluorosulfuric Acid?
Getting Straight to the Chemistry
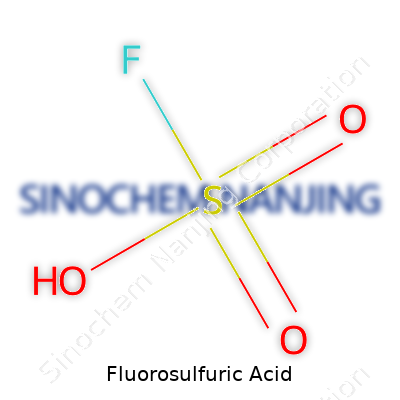
Fluorosulfuric acid is a name that doesn’t grab headlines, but its formula—HSO3F—packs some serious power. Add a single fluorine atom to sulfuric acid and the world of molecules changes. HSO3F, sometimes called “magic acid” when mixed with antimony pentafluoride, redefines what chemists can accomplish in the lab.
Why HSO3F Draws Attention
Groups working in research or specialty manufacturing count on certain superacids to get jobs done where classic acids stumble. Looking at fluorosulfuric acid, its lone hydrogen can jump off much faster than in sulfuric acid. That makes it a champion proton donor. Labs use it to drive reactions that shape complex organic compounds, tweak pharmaceutical molecules, and clean up stubborn surfaces in microelectronics and chemical synthesis. The industrial world wants materials that resist almost anything—HSO3F steps in for etching, making fluorinated chemicals, and creating catalysts. In this chemical’s world, strength brings out possibility, but also responsibility.
Balancing Use and Care
Getting hands on fluorosulfuric acid means serious training, proper gear, and respect for safety. Direct contact with HSO3F burns skin and eyes in seconds. Mixed with water, its reaction blasts out heat and harmful fumes. I’ve seen small acid spills handled by experts, and even then, the air gets tense. No matter how familiar it becomes to chemists, shortcuts never pay off. Laboratories build redundancies—fume hoods, neutralizers, acid-proof gear—because safety lapses turn one mistake into a medical emergency.
Globally, only facilities with seasoned staff and designed setups get clearance to use or ship this compound. Regulations stand between skill and disaster because the chemical doesn’t forgive. Producers keep tight records on quantities and movement. This keeps the material out of amateur hands and stops improper disposal from ruining waterways or drinking supplies.
Backing Up the Facts
Each year, agencies like OSHA and the European Chemicals Agency revise handling guidelines based on new case studies and lab accidents. The strong acidity of HSO3F boosts its role in pushing boundaries in industrial chemistry. Its pKa sits around -10, a number that doesn’t just confirm its class, but signals the level of preparation required. In the wrong conditions, a simple spill can produce toxic hydrogen fluoride vapors. That’s a real risk to anyone nearby, so real-time air monitoring often runs in parallel with routine lab work.
Working Toward Solutions
There has been a steady push for safer, greener alternatives in chemistry. If a process can swap out superacids for weaker acids or avoid acid use altogether, research and industry jump on board. Green chemistry programs at universities dig into finding reactions that cut back the need for hazardous acids like HSO3F. Some startups invest in new tech for residue neutralization or safer reaction vessels to blunt accident risks. Plenty of industries keep using the old standbys, but every year technology pulls more tasks away from superacid territory.
Everyday Impact
Understanding a substance like HSO3F’s formula isn’t just trivia for chemists. It plugs into bigger questions about risk, responsibility, and science’s role in shaping safer products. Modern manufacturing and research always walk a line between possibility and care, and the story of fluorosulfuric acid shows that respect for the details builds the backbone of progress.
How should Fluorosulfuric Acid be stored?
Real Risks in Every Drop
Fluorosulfuric acid sounds like something straight out of a villain’s lab, and honestly, that isn’t too far off. This acid eats through skin, metal, and almost everything else. Even folks with years of chemistry under their belt feel uneasy around it. I remember my first training session with this acid; a box of reinforced glass bottles sat in a fume hood, wrapped up tight like museum relics. My supervisor leaned in and said, “Respect this stuff or you’ll regret it.” That warning stuck with me.
Don’t Trust Weak Containers
A regular plastic or glass bottle loses a fight against fluorosulfuric acid before the bell rings. One tiny leak, and you get fumes strong enough to bring down a laboratory crew. Storage calls for special containers—only PTFE or pure Teflon stands a chance, and even then, I check the seals before every use. Some companies use steel drums lined with thick fluoropolymer, but nobody in their right mind stores this acid outside secondary containment. Any spill turns into a full-scale emergency; I’ve seen floors pitted and walls streaked from a single dropped vial.
Control the Surroundings, Not Just the Acid
Keeping fluorosulfuric acid locked away isn’t just about the bottle. Temperature swings make containers brittle or even explode under pressure. I always store the acid in a cool, well-ventilated chemical cabinet, never above other reactive agents and never near anything organic. Mixing mistakes kill. Some labs stick to special acid cabinets with extractor fans running day and night; the smell alone—sharp and metallic—acts as a constant reminder to stay cautious.
Emergency Gear Sits Front and Center
Rules say you won’t need a spill kit or an eyewash station until you really need one. I keep full-face shields, chemical gloves, and acid aprons in arm’s reach. Anything less feels like tempting fate. Colleagues tell stories about burns that went straight through regular nitrile gloves within seconds. Having the right training matters just as much as the gloves: quick action can mean the difference between a scary close call and a hospital stay.
No One Handles It Alone
I never work with this acid solo, no matter how tight the deadline looks. Someone else always watches, ready to call for help if the worst happens. Calling out container sizes, logging each transfer, and double-checking labeling prevents disaster. Every job gets another set of eyes—no shortcuts, no lone-wolf chemistry.
Safe Practices Built on Hard Lessons
Plenty of regulations reinforce these ideas for a reason. Evil-sounding acid or not, mistakes don’t wait for a second chance. Training and attention make or break storage and handling. Even small spills mean alarms and cleanup teams. The best safety record I’ve seen came from a group obsessed with redundancy and preparation. Their setup included floor pans, vapor alarms, and rigid checklists. They earned that confidence, drop by dangerous drop.
Invest in Storage, Not Clean-up
It’s tempting to cut corners, but repairs cost more than proper cabinets and containers ever will. I’ve seen bosses try to skimp, only to face fines or worse. Investing in the right stuff up front keeps accidents rare. My advice: respect the acid, listen to those who’ve seen what it can do, and put real effort into safe storage. Any different approach invites disaster that nobody wants to clean up.
What are the hazards associated with Fluorosulfuric Acid exposure?
Facing the Risks Up Close
Fluorosulfuric acid rarely grabs headlines, but it deserves close attention in any space where safety matters. This colorless, fuming liquid promises some of the harshest chemical burns and long-term damage if it sneaks past the usual layers of gloves and goggles. My work in laboratories, watching how even experts tread carefully around this acid, taught me respect for its power—it eats through protective clothing and even basic glassware, so handling it without the right gear practically asks for trouble.
Personal Exposure Stories Matter
Few experiences match the fear of accidental splash or spill in a lab. Colleagues who had near misses with fluorosulfuric acid felt burning pain within seconds, despite emergency drench showers and fast responses. Unlike more forgiving acids, this one pushes past the skin, searing through tissue, and unleashing toxic fumes that choke the lungs. Hospitalization becomes a real possibility, since the toxic route continues deep into the respiratory tract—and burns can take months to heal. The clear lesson: you don’t cut corners or improvise with this stuff.
Science and Immediate Health Effects
The main challenge is the acid’s aggressive reaction with water, which releases toxic fumes of hydrogen fluoride and sulfur trioxide. Exposure to just a breathful can trigger violent coughing, choking, and lung damage. Eye contact often blinds, even with rapid rinsing. Prolonged or repeated contact reaches bones, causing necrosis and tissue loss. Statistics show even experienced industrial workers remain at risk if ventilation or equipment fails—accidents have forced shutdowns and led to permanent injuries.
Workplace and Environmental Dangers
Factories and research centers handling fluorosulfuric acid run drills for spills and exposure for good reason. A single accident can leave a lab or warehouse uninhabitable for weeks, with hazardous waste cleanup and hazmat crews on site. Water reacts explosively, so standard fire suppression can worsen the incident. I’ve witnessed firsthand how panic sets in when alarms sound, knowing that a few lost seconds could seal someone’s fate. No shortcuts exist—scrupulous storage, ventilation, spill kits, and training remain the only safeguards that stand up to this acid.
Building Safer Systems
Prevention means respecting engineering controls: acid-resistant coatings, sealed containers, and airtight transfer systems. I’ve worked in groups where peer checks before handling materials added an extra layer of security. Regulations should require high visibility signage, real-time air monitoring, and strict policies about entering areas with fresh or lingering fumes. Medical facilities need training in chemical burn treatment and exposure protocols, since delayed care leads to lifetime scars or even fatalities.
Why It’s Worth Our Attention
Fluorosulfuric acid teaches humility. Every year, new generations of chemists and factory workers meet this compound for the first time. Only by sharing real stories, hammering home the dreadful side effects, and doubling down on practical safety steps can we reduce the toll. Clear communication, strong safety culture, and better emergency planning help keep people safe, whether in a laboratory, industrial plant, or during transport. No other acid delivers such a harsh lesson about the rules—and why they exist.
| Names | |
| Preferred IUPAC name | Fluorosulfuric acid |
| Other names |
Sulfuryl fluoride HSO3F Fluorosulphuric acid Sulfuric fluoride Hydrogen fluorosulfate |
| Pronunciation | /ˌflʊə.roʊˌsʌlˈfjʊr.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 7789-21-1 |
| 3D model (JSmol) | _Fluorosulfuric Acid_ JSmol 3D model string: ``` F[S](=O)(O)=O ``` |
| Beilstein Reference | 1711318 |
| ChEBI | CHEBI:29375 |
| ChEMBL | CHEMBL1230426 |
| ChemSpider | 29311 |
| DrugBank | DB14680 |
| ECHA InfoCard | 100.029.130 |
| EC Number | 231-975-3 |
| Gmelin Reference | 69540 |
| KEGG | C01739 |
| MeSH | D005563 |
| PubChem CID | 66624 |
| RTECS number | WS4900000 |
| UNII | S1Y1GX0423 |
| UN number | UN1774 |
| Properties | |
| Chemical formula | HSO3F |
| Molar mass | HSO3F: 102.07 g/mol |
| Appearance | Colorless, fuming oily liquid |
| Odor | Pungent, choking |
| Density | 1.77 g/cm³ |
| Solubility in water | Miscible |
| log P | -2.0 |
| Vapor pressure | 40 mmHg (20 °C) |
| Acidity (pKa) | −10 |
| Basicity (pKb) | -15.1 |
| Magnetic susceptibility (χ) | -43.5e-6 cm³/mol |
| Refractive index (nD) | 1.335 |
| Viscosity | 2.1 mPa·s (25 °C) |
| Dipole moment | 1.88 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 157.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –780.0 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | –970 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Corrosive, causes severe burns to skin and eyes, toxic if inhaled or ingested, reacts violently with water and organic materials, releases toxic fumes. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H290, H314, H335 |
| Precautionary statements | P260, P264, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 4-0-2-W |
| Autoignition temperature | 310 °C (590 °F; 583 K) |
| Lethal dose or concentration | LD₅₀ (oral, rat): 214 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2140 µL/kg (rat, oral) |
| NIOSH | TT6300000 |
| PEL (Permissible) | 1 mg/m3 |
| REL (Recommended) | REL (Recommended Exposure Limit) for Fluorosulfuric Acid: "0.1 mg/m3 (as fluoride), 8-hr TWA |
| IDLH (Immediate danger) | 30 ppm |
| Related compounds | |
| Related compounds |
Sulfuric acid Chlorosulfuric acid Triflic acid |

