Ferrocene: More Than a Curiosity in Chemistry
Historical Development
When chemists put together iron and cyclopentadiene in the early 1950s, no one expected such a curious structure to shake up chemical thinking. The shock was real: ferrocene brought a sandwich complex to life—a central iron atom tucked between two five-carbon rings. Sometimes discovery tales sound more dramatic than science fiction, and ferrocene’s story fits the bill. Until its discovery, organometallic chemistry felt like an obscure corner, not a main stage. Once those orange crystals hit the scene, Nobel Prizes followed, and organometallic chemistry grew into a powerhouse field. Modern chemists still owe a nod to ferrocene's finders, because those years marked a shift in how molecules get built and understood.
Product Overview and Physical & Chemical Properties
Ferrocene’s first impression starts right with its bright, fiery color—a deep orange that sets it apart from other iron compounds. Its solid, stable nature at room temperature makes it easy to handle and investigate. Unlike iron salts that rust fast or leave residues, ferrocene shows genuine durability, resisting air and moisture. Its molecular structure stands out too: those parallel cyclopentadienyl rings give the whole molecule a symmetry that's as visually satisfying as it is chemically robust. Ferrocene melts at about 172 °C, dissolves easily in organic solvents like benzene or toluene, and releases a faint, oily scent. Under normal lab conditions, it barely flinches when exposed to light or oxygen—a far cry from the stereotypical view of “delicate” lab chemicals. Reliability like this means even inexperienced hands can work with it without losing sleep over safety or decomposition.
Technical Specifications & Labeling
Pure ferrocene appears as orange crystals, with a chemical formula of C10H10Fe and a molecular weight hovering around 186 grams per mole. The packaging usually spells out basic handling guides, alerting users to possible mild irritation risks. On the technical side, standard samples should show good solubility in ether, acetone, and certain hydrocarbons. Products on the market sometimes carry warnings for dust generation—nothing wild, but a good reminder for folks who don’t pay enough attention to basic ventilation. Analytical grades need high purity content, usually hitting above 98 percent, so research and industry professionals can count on consistency. Safety data typically emphasize washing hands after use and not inhaling powdered forms—common sense steps in any chemical setting.
Preparation Method
Classic synthesis relies on the reaction of cyclopentadiene with iron(II) salts in a basic medium. In the early days, sodium cyclopentadienide met ferrous chloride, yielding a bright orange product that amazed even seasoned chemists. Fast forward a few decades, and various tweaks exist—some labs opt for Grignard reactions, others for more eco-friendly aqueous syntheses. No matter the method, the underlying principle stays the same: join iron with two cyclopentadienyl rings under mild or moderate conditions. Unlike some complicated molecules, ferrocene’s synthesis doesn’t call for cutthroat temperatures or exotic equipment, opening the door for students and professionals alike to make their own samples without major obstacles.
Chemical Reactions & Modifications
Ferrocene invites chemists to get creative. Its structure allows straightforward substitutions on the rings, where electrophilic reagents can swap out hydrogen atoms for other groups. Friedel–Crafts reactions, acylations, and sulfonations go off without a hitch, and the resulting derivatives bring an impressive range of electronic and physical properties. Fresh graduates in chemistry often practice these modifications as rite of passage experiments, so ferrocene turns into both teaching tool and research asset. Some researchers push ferrocene chemistry even further, attaching it to biomolecules, constructing polymers with it, or using it as a redox-active building block in sensing devices. Its ease of modification stands out as a big reason why ferrocene keeps sparking new projects across different chemical disciplines.
Synonyms & Product Names
Ferrocene’s journey through textbooks and catalogs delivered it quite a roster of alternative names. Some older literature refers to it as “dicyclopentadienyliron,” while trade and scientific circles stick mostly with ferrocene for clarity. These synonyms rarely trip up seasoned researchers, but for beginners, recognizing the aliases helps when digging through journal archives or product listings. No matter the name, the five-ring “sandwich” image gives it away with a quick glance.
Safety & Operational Standards
Laboratories treat ferrocene as a compound with low acute toxicity. Gloves, standard protection, and basic extraction fans keep workspaces clean. Laboratories and production facilities alike focus on routine safety—good housekeeping, avoiding the creation of airborne dust, and handling spills with attention. No corrosive fumes or strong acids lurk in the handling of ferrocene itself, so workers appreciate its easy-going nature compared to nastier chemicals often found in synthesis labs. Eye contact, as with any solid material, deserves caution, but few would consider ferrocene hazardous by nature. Regular disposal rules—avoiding sinks and using solvent collection waste—apply like with most research-grade organics.
Application Area
Ferrocene punches above its weight as more than a textbook marvel. In the world of catalysis, modified ferrocenes help drive industrial reactions to produce plastics, pharmaceuticals, and specialty chemicals. Its stable redox behavior gives it a starring role in electrochemical sensors—devices count on its ability to shuttle electrons in a reliable, predictable manner. Materials scientists use it to create new classes of polymers with interesting electrical and mechanical properties. Over the years, fuel research groups have mixed ferrocene into rocket propellants and special fuels as a burn-rate modifier and anti-knock agent. In medicine and diagnostics, ferrocene finds use in biochemical assays, helping detect markers of disease at trace levels. Its wide-ranging uses speak to a reputation built on flexibility and dependability, rather than flash or hype.
Research & Development
Chemists keep finding new ways to stretch ferrocene’s potential. Over the last decade, laboratories devoted extra attention to ferrocene-inspired drugs and imaging agents. The biocompatibility and redox features set it apart from more traditional organic molecules or simple iron salts. In electrocatalysis and battery development, ferrocene delivers a mix of stability and versatility that makes it a valuable test case for new ideas. Researchers, especially those working at the boundaries between organic materials and metals, benefit from ferrocene’s willingness to undergo multiple transformations with little fuss. Its adaptability and the huge volume of published literature mean both young and experienced scientists can launch projects without starting from scratch. Students often learn their first lessons in organometallic chemistry by exploring ferrocene’s properties and derivatives.
Toxicity Research
The safety record of ferrocene measures up well against many other organometallic molecules. Most studies show only mild irritation from high exposure levels, with almost no reports of chronic harm in typical laboratory use. When tested in animals, large doses produced only temporary effects, and regulatory reviews placed ferrocene in the low hazard bracket. Workers stay clear of long-term or high-exposure experiments without established protocols, but no strong evidence points to ferrocene as a cancer risk or persistent toxin. That said, unwise handling, such as ingesting or inhaling dusts, still poses real health risks—standard laboratory discipline keeps these dangers low. As more researchers push ferrocene into biological and medical testing, toxicity screens continue at a higher level, making sure its reputation for safety remains justified.
Future Prospects
Ferrocene’s future already shapes up to be just as interesting as its past. Research groups aim to fold ferrocene into molecular electronics, using its predictable redox chemistry to design smarter, smaller switching devices. Medicinal chemists nurture hopes that ferrocene derivatives will lead to new anti-cancer drugs or diagnostics—its backbone provides a rare blend of stability and metallic behavior ideal for targeting specific cell processes. Green chemistry initiatives look at ferrocene-based catalysts that minimize waste and energy demands. The constant drumbeat of new applications reflects a molecule with staying power, one that meets the demands of emerging technology just as comfortably as it met the needs of the early pioneers in chemistry. For many in the lab, ferrocene stands as a reminder that elegant chemical design has the power to drive whole fields forward, generation after generation.
What is Ferrocene used for?
A Strange-Looking Molecule Makes a Big Impact
Ferrocene’s structure—two five-sided carbon rings sandwiching an iron atom—caught my attention the first time I saw its orange powder in a lab. Chemists call it a “sandwich compound,” and this oddball shape made it a sensation in the 1950s. The discovery helped launch the era of organometallic chemistry, which now influences research in materials science and pharmaceuticals around the world.
Boosting Fuels and Fighting Knocks
Gasoline engines knocking and pinging do not remind anyone of progress. Fuel additives help solve this, and ferrocene plays a practical role. A dose of ferrocene in fuel acts as an antiknock agent. The iron atom in ferrocene breaks down during combustion and helps gasoline burn smoothly. Studies show that it lowers engine knocking and lessens the buildup of carbon deposits inside engines. This has real value for people running older vehicles, or for those in parts of the world where high-quality fuel is out of reach.
A Workhorse in the Lab
In the research world, ferrocene serves as a chemical standard, helping scientists calibrate and compare electrochemical instruments. I’ve prepared more than one batch of solutions where ferrocene acts as a reliable yardstick, especially when working with unfamiliar test chemicals. The way it gives a predictable, repeatable signal is a small comfort when experiments feel unpredictable enough already.
New Polymers and Materials
Ferrocene’s sandwich structure forces chemists to rethink what’s possible in designing molecules. Adding ferrocene to polymers creates plastics with unique properties. These materials hold up better under heat and can conduct electricity, unlocking new options for electronics engineers and aerospace designers. Projects that sound futuristic—like flexible electronics and self-healing coatings—depend on molecules like this to push boundaries.
Medicines and Drug Research
One area that surprised me is drug development. Researchers use ferrocene structures as building blocks for new compounds, especially in cancer research and antiviral agents. Its unusual shape and stable iron core make it different from other drug molecules. Some teams have seen promising results when using ferrocene-linked compounds to disrupt disease-causing cells. We don’t get miracle drugs overnight, but the growing number of published studies shows chemists aren’t done exploring these directions.
A Safer Option with Precautions
Lead and manganese additives gave gasoline a bad name for good reason. Compared to those, ferrocene breaks down into less stubborn pollutants. Still, routine exposure to ferrocene powder at work made me pay more attention to gloves and lab ventilation—iron isn’t the problem, but the dust can irritate lungs over time. Practicing good safety means no shortcuts, even for substances with a “safer” reputation.
Making the Most of an Odd Molecule
Ferrocene keeps showing up in surprising places, from fuel tanks to medical journals. Universities and companies with a long track record in chemistry have kept advancing this field. Funding more research on sustainable uses, improving recycling of ferrocene-based products, and keeping safety education front-and-center helps science keep its promise. Starting as a curiosity, ferrocene carved out a place by solving problems, sometimes in small and quiet ways that matter over time.
Is Ferrocene toxic or hazardous to handle?
Understanding Ferrocene
Ferrocene stands out for its sandwich-like structure, which puts an iron atom snugly between two cyclopentadienyl rings. Chemists and lab workers might spot its telltale orange color, almost like saffron, but with a metallic edge. In labs and research centers, it earns its keep as a catalyst, fuel additive, and even as a component in antiknock agents for gasoline.
What Happens on Skin or Inhalation?
Most safety data sheets describe ferrocene as presenting low acute toxicity. Touching it with bare skin won’t bring instant harm, but that’s no green light to toss aside gloves. Irritation happens, especially with long or repeated exposure. I’ve seen lab workers not bother with masks or goggles just because regulations don’t call for hazmat suits. Anyone who has bumbled through an Organic Chemistry lab learns fast: a little carelessness can lead to headaches or irritation from even the “mild” chemicals.
The dust poses the most risk. Once it gets airborne, it can irritate your nose, throat, or lungs during inhalation. Most facilities recommend respirators and tight-fitting goggles, not to look bureaucratic but because lung irritation isn’t something you shrug off. The powder does not explode like some energetic compounds, but static discharge from dry air or friction can ignite a cloud of dust. Anyone working with bulk powder in a dry room notices the crackle and snap of static, a reminder that chemistry does not bend to wishful thinking.
Long-Term Health Impact and Environmental Risk
Animal studies give insight here. Swallowing large amounts caused weight loss and organ changes in rats, but not cancer. Ferrocene’s iron core means the body handles it somewhat like other iron compounds—excess intake can stress organs. Long-term exposure data in humans is thin, which signals caution. No one should treat ferrocene as safe simply because it doesn’t have a long rap sheet. Over the years, stories floated around of researchers with red stains on their lab coats or persistent coughs, always chalked up to “one of those things.” Years down the road, chronic exposure patterns often reveal risk where regulations lag behind science.
The environmental angle can’t be ignored. Iron compounds themselves break down, but cyclopentadienyl rings hold on in soil and waterways. Chemical runoff from labs or factories can build up, affecting aquatic life and soil chemistry. Local universities and research outfits face fines or public backlash for lax handling, especially when the waste leaves a trail traceable back to ferrocene.
How to Minimize Hazards in Practice
Keeping ferrocene’s dust out of the air is a basic defensive move. Anyone handling it should use gloves, goggles, and a fume hood, not just because the manual says so, but because the alternative courts trouble. If a spill happens, wet methods control dust far better than sweeping. Storage matters too: airtight containers kept dry send the right message, both to workers and regulators.
Disposal requires a step up from tossing it in the trash. Dedicated chemical waste streams withed labeling protect the city’s water and the workers later down the line. Training new lab staff about the hidden risks in “harmless” compounds benefits everyone and helps keep those red stains from showing up in places they shouldn’t.
What is the chemical structure of Ferrocene?
Understanding Ferrocene at the Molecular Level
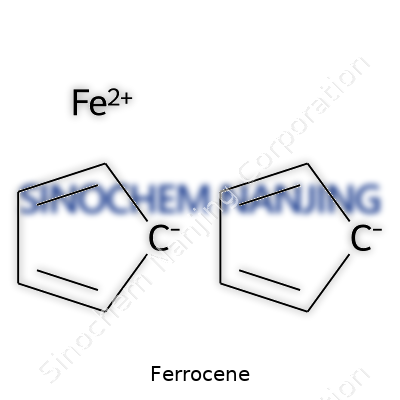
Ferrocene stands out as one of those compounds that reshaped modern chemistry, and not just because it looks cool under the microscope. To grasp its structure, picture two flat, five-membered rings of carbon and hydrogen atoms—cyclopentadienyl rings—stacked one on top of the other. The catch is an iron atom nestled right between these rings. Each ring shares its electrons with the iron center, creating a sandwich that feels as stable as a steel beam supporting two floors. Chemists call this arrangement a "sandwich compound" and credit ferrocene as the first of its kind discovered on a lab bench.
The discovery came in the early 1950s, at a time when organometallic chemistry still felt new and mysterious. Before people understood what was happening in ferrocene, few expected a metal could sit so comfortably between two aromatic hydrocarbon rings. After its structure emerged, research boomed. Folks saw that ferrocene didn't just challenge old ideas—it opened doors to new ways of making stable metal-organic molecules. Curiosity paid off for the teams at Duquesne University and British Oxygen who first uncovered the structure. They didn’t just find a quirky molecule; their work sparked a wave of innovation in medicine, energy, and material science.
Breaking Down the Iron Sandwich
Here's what makes up ferrocene at the atomic scale. Both cyclopentadienyl rings in the molecule consist of five carbon atoms arranged in a pentagon, with each carbon atom bonded to a single hydrogen. The iron atom, in its +2 oxidation state, sits tightly in the gap between these rings. Each ring donates six electrons to the iron, creating a strong bond. That adds up to eighteen electrons around the iron atom—a number that chemists often seek for extra stability in organometallic compounds. The simplicity of this structure almost hides how special it acts chemically.
Ferrocene catches attention because it doesn’t fall apart easily. The bonding between iron and the rings resists heat, acids, and even some strong bases. That resilience explains why ferrocene ends up in so many experiments as a reference molecule or catalyst. Chemists can tweak ferrocene by swapping out hydrogens for other functional groups, creating new versions that serve in medicine, battery technology, and electronic sensors. I remember watching students in the lab marvel at how a small tweak to ferrocene’s rings could give a whole new set of properties—each variant changing something as simple as color or as complex as redox potential.
Lessons Beyond the Textbook
Ferrocene’s discovery teaches the value of keeping an open mind and taking a closer look at unexpected lab results. The bright orange crystals of ferrocene didn’t fit early predictions, yet drove scientists to rethink the interaction between metals and organic molecules. That willingness to learn from surprise plays a key role in developing useful technology today. The molecule’s structure continues to inspire research across chemistry and engineering, from anti-cancer drugs to solar cell components.
Better understanding of ferrocene and similar sandwich compounds could point us toward new energy storage solutions or environmentally friendly catalysts. By unraveling the secrets of such stable and adaptable molecules, chemists can find new ways to tackle persistent issues in sustainable technology. Ferrocene’s chemical structure offers more than a textbook example; it’s proof that looking closer at the building blocks of matter can fuel real progress in the world around us.
How is Ferrocene synthesized?
Getting Down to the Nuts and Bolts
Ferrocene’s nickname—the “sandwich compound”—comes straight from the way iron sits between two organic rings. Building this molecule reflects the kind of creativity chemists thrive on. I remember the first lab demo I saw in college where orange ferrocene crystals appeared like magic from a dark blue solution. It's eye-opening, proof that even simple ingredients like powdered iron and a common organic molecule spark surprising results.
Why Chemists Care About Ferrocene
Ferrocene’s unique structure makes it both sturdy and versatile. Before its discovery in the early 1950s, nobody imagined metals could sneak into rings of carbons so smoothly. This opened new doors for chemistry as a whole—from teaching molecular bonding to designing fancy catalysts.
I’ve seen ferrocene help explain stability in organometallics. Its crystal structure lands in textbooks. Its chemical behavior has influenced the design of drugs, sensors, batteries, and fuel additives. When I tutored organic chemistry, students always connected better with real-world examples like ferrocene than with abstract reaction arrows. The compound’s bright color and odd shape make it memorable.
The Classic Synthesis—Step by Step
Most college labs still use the same time-honored process researchers first published decades ago. It starts with cyclopentadiene—a five-carbon ring, as volatile as it sounds. This gets deprotonated using a strong base like sodium metal in a non-polar solvent such as diethyl ether. The mix becomes sodium cyclopentadienide, a salt that’s both reactive and ready to combine with iron.
Iron(II) chloride shows up as a green powder or a crystalline solid. Toss it into the sodium cyclopentadienide solution. Iron slides itself between two of those five-carbon rings. You know the reaction’s kicked off when you spot an orange hue—the hallmark of ferrocene.
Risks and How To Work Smart
Synthesizing ferrocene requires prep. Cyclopentadiene dimerizes at room temperature, so splitting it up before use is a step you can’t skip. Sodium metal reacts fiercely with water, throwing off hydrogen gas and heat—eye protection and dry glassware cut down on injuries. Diethyl ether isn’t just flammable, it can form dangerous peroxides. I always respect the fire risk and store it far from any open flame. If you’ve worked in a teaching lab, you know these hazards can wake up anyone who drifts into autopilot.
Beyond the Basics—Keys to Success
Fresh reagents matter. Impurities in solvents or moisture ruin yields. Keeping everything washed, dry, and free from oxygen solves more problems than fancy equipment ever could.
The popularity of ferrocene also comes from how easy it is to purify. Recrystallization or chromatography cleans up crude samples quickly. Even students with shaky hands can manage a good batch, which makes ferrocene a staple for learning and a reliable building block in professional labs.
Finding Progress in Reliable Chemistry
Building ferrocene stands as a great introduction to the possibilities found at the intersection of metal and organic chemistry—a junction chemists visit often when designing materials for the future. Labs fine-tune the steps, try greener solvents, and push for safer bases, but the essential curiosity stays the same. Seeing that orange color form is chemistry at its most accessible and inspiring.
What are the physical properties of Ferrocene?
Understanding Ferrocene in the Lab
Ferrocene looks like nothing you’d expect from a typical iron compound. Forget the rust-red or metallic vibe that usually shows up with iron. What you see is a bright orange, fine crystalline solid that comes off the spatula almost like a powder. The color alone catches the eye long before any measuring or weighing starts, and that is a real gift if you’re used to drabber organometallics. Handling ferrocene, you notice it can stain gloves and paper quite easily at room temperature, but it doesn’t melt in your palm—this substance stays solid right up until about 173 degrees Celsius, where it melts to a deep orange liquid. Beyond that, it boils above 249 degrees Celsius. The high melting and boiling points match well with its toughness in the lab, standing up to air and even moisture for quite some time without much fuss.
No Bad Smells, No Real Hazards—but Not Quite Harmless
Ferrocene doesn’t leave the nose wrinkling. It has little to no smell. This non-volatile nature means you aren’t clouding up your workspace just by uncapping a bottle. That makes it a lot easier and safer to work with than many other organometallics, which often release strong, unpredictable odors or toxic vapors. At the same time, orange stains can last if ferrocene gets into cloth or plastic, so wiping up spills quickly is a must.
Solubility and Its Curious Ways
This compound loves organic solvents but not water. Shake ferrocene into a test tube with something like ethanol, diethyl ether, or benzene, and you’ll get a clear, orange solution with very little left behind. Drop it in water, and you’ve got a mix that won’t blend. Organic chemists lean hard on this habit in both synthesis and purification—if you want to separate, purify, or analyze ferrocene, the choice of solvent makes a big difference.
Crystal Structure: Stacked Like a Sandwich
Beneath the microscope, ferrocene reveals another surprise. Its structure earned the name "sandwich compound" for good reason. Two flat rings of cyclopentadienyl face each other with an iron atom tucked perfectly in the middle. Every molecule stacks with others to make a stable crystal lattice. The symmetry helps explain its physical resilience. The structure held up in X-ray crystallography studies, long before organometallic chemistry exploded as a field.
Stability and Longevity in Real Applications
Leaving iron exposed to the air or moisture tends to end badly. Not here. Ferrocene stands tough thanks to its "aromatic" stability—electrons in those rings spread out, meaning less tendency to react with oxygen or water in the environment. You can leave it open on the bench for a bit without seeing rapid breakdown or loss, so it lives longer in the bottle. That helps explain its shelf life and why so many researchers trust it for calibration or as a standard in experiments.
What We Can Learn From Working With Ferrocene
From undergraduate chemistry to advanced research, I’ve always found ferrocene rewarding to handle. The visual markers like color and solid nature, the safety from lack of fumes, and its eager solubility in non-water solvents—these lessons hit home quickly. If you ever needed to judge moisture, handle a low-hazard compound, or simply appreciate the quirks of organometallic structure, ferrocene draws the line between curiosity and practical knowledge. Its physical properties teach us more than just facts; they help build habits that carry over to more challenging compounds.
| Names | |
| Preferred IUPAC name | bis(η⁵-cyclopentadienyl)iron(II) |
| Other names |
Bis(η5-cyclopentadienyl)iron Dicyclopentadienyliron Sandwich compound |
| Pronunciation | /fəˈrɒsiːn/ |
| Identifiers | |
| CAS Number | 102-54-5 |
| Beilstein Reference | 136369 |
| ChEBI | CHEBI:30212 |
| ChEMBL | CHEMBL14009 |
| ChemSpider | 6618 |
| DrugBank | DB01538 |
| ECHA InfoCard | 100.036.325 |
| EC Number | 205-489-7 |
| Gmelin Reference | 63632 |
| KEGG | C06725 |
| MeSH | D005247 |
| PubChem CID | 7489 |
| RTECS number | NL0175000 |
| UNII | LB4S318YO6 |
| UN number | UN1325 |
| Properties | |
| Chemical formula | C10H10Fe |
| Molar mass | 186.04 g/mol |
| Appearance | Orange crystalline solid |
| Odor | Odorless |
| Density | 1.490 g/mL |
| Solubility in water | Insoluble |
| log P | 1.69 |
| Vapor pressure | 10 mmHg (20 °C) |
| Acidity (pKa) | pKa = 13.6 |
| Basicity (pKb) | 4.6 |
| Magnetic susceptibility (χ) | +70.7×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.503 |
| Viscosity | 3.2 mPa·s (at 25 °C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 186.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -23.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1583 kJ/mol |
| Pharmacology | |
| ATC code | B03AA02 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H228, H302 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P370+P378 |
| NFPA 704 (fire diamond) | 1-1-0-⨁ |
| Flash point | > 102 °C |
| Autoignition temperature | 400 °C (752 °F; 673 K) |
| Lethal dose or concentration | LD50 (oral, rat): 1320 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 1320 mg/kg |
| NIOSH | NL1400000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Ferrocene: "No specific OSHA PEL established |
| REL (Recommended) | 100 mg |
| Related compounds | |
| Related compounds |
Ferric acetylacetonate Ferrocene derivatives Cobaltocene Nickelocene Chromocene Manganocene |

