Ethylene Glycol Monopropyl Ether: More Than a Solvent
A Look Back at the Path of Ethylene Glycol Monopropyl Ether
Few people give much thought to the chemistry buzzing through factories and labs, but for anyone who works with paints, coatings, inks, or cleaning fluids, ethylene glycol monopropyl ether stands as a familiar name. This chemical didn't just appear on shelves overnight. Its background reaches into the early development of glycol ethers when scientists sought out compounds blending water solubility with effective solvency and a mild odor. By the latter half of the twentieth century, it had carved out a place among other glycol ethers, thanks to a knack for dissolving both polar and non-polar substances. Its timeline mirrors shifts in manufacturing and environmental awareness, with steady improvements in purity, labeling, and worker safety.
Understanding the Compound: What Sets It Apart
Ethylene glycol monopropyl ether pops up in the chemical world as a clear liquid, with a faint, pleasant scent. It doesn’t just mix with water–it gets along just as well with organic solvents, putting it in a unique niche for blending and cleaning. Boiling at a moderate temperature, it brings flexible use in closed or open systems, showing resilience where volatility can be an issue. It flows easily and won’t leave behind greasy residues, which makes it ideal for surface cleaners and degreasers. The chemical formula, C7H16O2, loses people who don’t follow the chemistry, but what matters is its ability to tackle tough grime without the harshness of some predecessors.
Physical and Chemical Behavior That Matters in Real Work
If you’ve spent much time with paints or cleaning solutions, you'd see how fast-evaporating solvents can make working conditions tough—strong fumes, headaches, and rapid drying that leads to uneven coatings. Ethylene glycol monopropyl ether avoids most of those headaches. Its vapor pressure sits comfortably between volatile solvents (which disappear too fast) and heavy glycols (which can be sticky or slow to dry). In the molecular world, those two oxygen atoms let it bridge the gap between water and oil, carrying dyes and detergents across surfaces with fewer streaks or residues.
How It’s Made and Tweaked in Modern Industry
No magic here—industry brings together ethylene oxide and n-propyl alcohol under conditions that favor selectivity and cleanliness. Older processes often carried unwanted byproducts, but better monitoring and improved catalysts now raise purity and lower waste. Manufacturers don't just stop at basic synthesis. They keep improving reaction conditions, working to recover waste heat, reuse solvents, and cut down on hazardous side-streams. These refinements reflect real pressure from environmental groups and health studies showing what even tiny traces of contaminants might do over time.
Other Names, One Chemical
Depending on the country or supplier, ethylene glycol monopropyl ether may show up as propylene glycol monoethyl ether or indicate its true structure in more technical phrasing like 1-propoxy-2-hydroxyethane. People in the paint and ink industry recognize the common synonyms because they know formulas may shift slightly, and knowing these names helps workers avoid mixing up incompatible chemicals.
Labeling Tells a Story—If You Know How to Read It
In a field plagued by mishaps and chemical burns, labeling isn’t just legal red tape. Regulations keep demanding more clarity—signal words, hazard pictograms, advice for keeping hands safe and lungs clear. You won’t find a bottle on a regulated site that doesn’t flag the flammability risk, the mild irritation danger, and the preventative steps for a safer workspace. Some years back, labels carried only vague warnings, but most sites now lean into the specifics to keep staff informed before accidents happen.
Using Chemical Knowledge to Shape Innovation
The base molecule carries a backbone that chemists tweak for specialty tasks. Sometimes the ether group gets replaced for different evaporation rates, or they tag on additional glycol units, tweaking the solvency for bright colors to stand out in inks or for stronger degreasing in mechanical shops. These changes feed constant research, seeking a balance between cleaning power and safety. Labs track shifts in toxicity and keep searching for modifications that lower worker risk without cutting performance.
Real Safety Lessons Learned on the Job
Anyone who’s worked in industrial settings understands the routine of donning gloves and checking ventilation before pouring out a solvent like ethylene glycol monopropyl ether. The chemical tracks through lungs and skin, and although milder than older glycol ethers like ethylene glycol monomethyl ether, it still holds hazards—especially with chronic exposure. Over time, workplace studies linked it to headaches, mild skin irritation, and in rare cases, effects on red blood cells at high exposure levels. Over the last decade, stricter time-weighted average exposure limits and rapid detection methods have built a safer environment for workers. Safety data gets updated often. Sites test air quality, check PPE compliance, and offer practical tips, not just boilerplate warnings.
Getting to the Heart of Product Use
You don’t see ethylene glycol monopropyl ether lining grocery store shelves, but it runs through the veins of commercial products everywhere. In paints, it carries pigments onto walls and metal without quick evaporation stalling the job. In inks, it allows for sharp lines that don’t run, especially important for high-speed commercial printers. At hospitals, cleaning crews use products that blend this ether to cut grime without strong odors lingering. Think of it as the “quiet hand” that lets cleaning happen fast and leaves behind little residue or scent. The cleaning business values its strength without harshness, and auto shops prize its ability to cut oily residues off tools and engine parts.
Trends and Developments Shaping the Future
Science never sits still, especially with chemicals under scrutiny. Consumer demand for greener products keeps pressure on researchers to uncover substitutes nontoxic to humans but just as tough on stains. Some companies experiment by reformulating with bio-based glycols or by using enzyme additives in cleaners. Legislation in Europe and North America turns up the heat, and manufacturers adapt by cutting solvent content and limiting hazardous air pollutants. Research into acute and chronic toxicology provides steady feedback for regulation, product reformulation, and worker safety improvements.
Exploring the Edge: Toxicity and Human Health
No chemical gets a free pass in today’s world. Toxicologists have taken hard looks at ethylene glycol monopropyl ether. Studies in lab animals and reports from real factory work show relatively low acute toxicity, but lingering effects appear when exposure gets high or lasts for years. Concern about reproductive health effects pushed reviews of older glycol ethers, but fewer red flags emerge here. Even so, regulators press for tighter exposure controls. The balance always comes down to real-world risk—exposure level, protective equipment, and handling habits. Facts matter more than vague warnings, and data from health monitoring keeps shaping best practices.
Where Do We Go from Here? Future Directions
Life in a world that values high performance yet fears hidden dangers means chemicals like ethylene glycol monopropyl ether face scrutiny from all sides. Green chemistry seeks replacements but has yet to find a single molecule that does as many jobs so quietly. The next wave of research will focus on alternatives and process improvements as much as on the compound itself. New standards push the industry to lower exposure, cut emissions, and share clear information not just with facilities but also with consumers. In years ahead, expect continued investigation of the subtle health effects, sharper labeling standards, and cleaner production—driven as much by workers who expect safety as by scientists writing new laws.
What is Ethylene Glycol Monopropyl Ether used for?
An Everyday Chemical Behind the Scenes
Walk down any grocery store aisle filled with cleaning sprays and you’ll find products that get grime off countertops or glass without much effort. Ethylene glycol monopropyl ether stands behind these powerful cleaners. Most people don’t notice it because companies list it among many ingredients, but it pulls a lot of weight by helping other chemicals dissolve. Many cleaning jobs call for a mix that lifts grease, removes sticky fingerprints, and doesn’t leave streaks. This solvent cuts through those problems, leaving surfaces looking sharp and clean.
In Factories, One Ingredient Makes Many Products Work
In manufacturing, production lines run smoother with a reliable solvent. Factories that make paints, varnishes, and inks add ethylene glycol monopropyl ether for a few reasons. It keeps mixtures stable so they coat surfaces evenly. Paint won’t dry too fast or too slow, which means fewer defects. As a paint worker for several years, I remember how a shift in solvent blend could throw off the whole process — making paint too sticky to roll or quick to clump in cans. This chemical brought products back to the right feel and flow.
Helping Care for Machines and Workplaces
For workers in metal shops or auto garages, cleaning tough machine parts comes with daily tasks. Grease, metal shavings, and caked-on residue build up fast. Strong solvents like ethylene glycol monopropyl ether break those stains apart, so mechanics spend less time scrubbing. When used correctly, it can extend the lifespan of tools and engines by making it easier to keep surfaces clean and reduce wear from grit.
Why Safety and Knowledge Matter
People sometimes bring chemicals home without knowing much about them. Label reading goes a long way, especially since ethylene glycol monopropyl ether can irritate skin or eyes if used carelessly. I’ve seen folks get rashes or headaches because they didn’t open a window or put on gloves. Training workers to handle even everyday chemicals safely can prevent health problems. Employers need to stress real stories, not just recite guidelines.
Environmental health agencies study these types of solvents because overuse or accidents can affect indoor air. Regular ventilation and disposal rules keep workplaces healthier. If you work around these chemicals, pay attention to guidelines and look for safer alternatives as technology improves. Some companies now use water-based systems with less reliance on harsh solvents—these protect both workers and the environment without lowering product performance.
Looking for Better Solutions
People ask about greener options. The market shifts toward formulas with fewer risks, but price and performance keep ethylene glycol monopropyl ether in use. Researchers continue to test new blends and educate the industry about safe handling. Next time you use a glass cleaner or paint your walls, remember that chemistry shapes daily routines in ways most folks never see. Products feel easy to use only because someone studied the science and made careful choices. Keeping eyes open for better solutions and sharing that knowledge can help families and workers stay safer, while industries find ways to improve.
Is Ethylene Glycol Monopropyl Ether hazardous to health?
What It Is and Where You Might Find It
Ethylene glycol monopropyl ether pops up in a surprising number of places. Folks who have spent time cleaning or working in auto shops probably have brushed past it without knowing. It shows up as a solvent in cleaners, paints, inks, and sometimes in coating products. A quick glance at a safety data sheet reveals that it's there to help things mix well or spread smoothly. Not everyone gives it a second thought.
Breathing It In and Touching It
Vapors from this chemical don’t linger like a stubborn paint smell, but inhaling them can still cause headaches, eye or throat irritation, and a feeling of dizziness. It's pretty common for someone to wipe down a counter or spray a surface, think nothing of it, and then realize minutes later their eyes sting or their hands itch. Skin contact can trigger redness, and a few folks notice it dries out their hands faster than dish soap ever could. Workers in manufacturing can get exposed over longer days, not just in passing.
What's Backed by Science?
Research on ethylene glycol monopropyl ether shows it can affect the blood if large amounts build up in a person. A study from the late 90s highlighted blood changes after repeated exposure in animals. The European Chemicals Agency points out risks grow for folks who get exposed daily at work. That kind of info gets around in industrial safety circles, and most plant managers take it seriously. There’s no need to whip up panic, but facts help put things in focus.
Small Exposures, Big Questions
Short bursts — like mopping a floor once or using a paintbrush for about an hour — rarely lead to obvious problems. The real challenge comes for workers clocking forty hours a week with this stuff in the air. The problem isn’t always the product itself, but using it without gloves or in rooms with windows closed tight. Industrial hygienists measure air levels, urging companies to keep them well below legal exposure limits. Those rules matter. They can mean the difference between a mild headache and much bigger issues after years on the job.
What Can People Do About It?
Gloves and goggles go a long way. If the product label suggests ventilation, crack open a window or run a fan. Manufacturers know folks trend toward shortcuts, especially in busy shops or warehouses, but skipping safety habits adds up. Some cleaning team veterans talk about the difference they feel after switching to products with fewer chemicals or using wet wipes instead of sprays. For parents or pet owners, storing chemicals out of reach is more than a tip; it prevents unplanned trips to the ER.
Where Companies and Regulators Step In
Most reputable producers don’t hide what goes into their bottles. Laws force them to show ingredients and warn about side effects if you use a lot or for a long time. European and US regulators don’t always agree on exact limits, but both update rules as new research rolls in. Labels grow clearer by the year, but it still falls to workers and consumers to read them, share concerns with supervisors, and push for better air flow or training in busy spaces.
Final Thoughts on Staying Safe
Nobody expects to inhale or absorb chemicals while scrubbing out the sink, but knowing the small print on a bottle matters. If an instruction label mentions gloves and fresh air, take it as a simple step toward better health. A little caution helps everyone—from the seasonal painter to the person stocking shelves at midnight—get through the job without the nagging headache or itch that many quietly blame on “just the job.”
What are the storage requirements for Ethylene Glycol Monopropyl Ether?
Why Proper Storage Matters
Ethylene glycol monopropyl ether isn’t just another clear, colorless liquid sitting on a warehouse shelf. It plays a big role in cleaning products, paints, coatings, and even inks. Yet it comes with headaches if treated without respect. Leaky drums, vapors building up in closets, and careless stacking can put both workers and operations in a tough spot. I’ve seen a maintenance team scramble after a spill because a container sat too close to a steam line and failed. So putting extra thought into storage makes real-world sense.
Temperature and Ventilation
Keep this liquid far from heat sources or direct sunlight. Overheating shortens shelf life and speeds up the breakdown of product quality. Ethylene glycol monopropyl ether gives off vapors even at room temperature; warm conditions bump up those vapors, making strong ventilation crucial. I recall a shop where workers got headaches until a well-placed exhaust fan cut down on vapor buildup. Tightly sealed containers don’t fix the problem by themselves—a combination of temperature control and airflow has a bigger effect.
Compatible Containers and Stacking
Containers should be made from materials that don’t react with the chemical. Steel drums with special linings or high-grade plastics do the job. A supply manager I know once tried switching to a lower-cost plastic drum, and a week later found the container showing early wear and slight bulging. Saving a few bucks up front didn’t pay off, since a leaking drum costs more in cleanup and product loss.
Stacking shelves or pallets never goes higher than manufacturer recommendations. Heavy drums on shaky shelves risk tumbles, and banged-up containers can spill or rupture. Every warehouse worker dreads the moment a punctured drum leaks.
Avoiding Incompatible Materials
Ethylene glycol monopropyl ether doesn’t belong near oxidizers, acids, or strong bases. Even strong warehouse folks have found out that mixing up storage zones can mean serious chemical reactions, released gases, or a fire scare. Set clear labels, train staff, and keep good documentation for all materials on site. In my experience, a simple checklist near the chemical storage zone cuts out confusion.
Spill Preparedness and Weekly Checks
No amount of planning replaces regular checks. Weekly inspections catch cracked seals, rusting drum rings, or shifting containers before they become emergencies. On-hand spill kits—including absorbents, PPE, and clear instructions—turn a stressful spill into a manageable cleanup. Practical habits always beat best intentions.
Safe Handling Culture
The difference between safe and risky chemical storage often comes down to small habits and shared responsibility. Even a new warehouse team learns fast by seeing old hands label containers clearly, check expiration dates, and log routine inspections. Management support for training pays off by cutting both workplace accidents and wasted product, and staff should always have direct ways to flag concerns.
Supporting Safe Storage
The relevant OSHA and NFPA guidelines give clear ground rules for chemical storage. Anyone working with ethylene glycol monopropyl ether should be familiar with the safety data sheet and follow local fire codes and environmental regulations. Keeping chemicals safe begins and ends with teams working together, checking off routines, and keeping an eye out for the details nobody thinks about—until it becomes the only thing that matters.
How should Ethylene Glycol Monopropyl Ether be handled safely?
What Makes This Chemical Different
Ethylene Glycol Monopropyl Ether tends to show up in everything from paint strippers to cleaning fluids. Working with it, you notice the sweet smell—almost like it’s not a threat. That’s when the risk sneaks up. The body absorbs this chemical through the skin as easily as inhaling it through the air. It targets the central nervous system, affects kidneys and liver, and can hurt red blood cells. Eye and skin irritation come first, but long-term exposure draws out the more serious health trouble.
How Exposure Happens—Knowledge from the Field
Walking through industrial floors, you often see workers spraying cleaners or degreasers without gloves. Even a splash can sting the skin or eyes. Lab workers sometimes forget how easily vapors spill over the edge of a loosely-capped container. Breathing in the fumes brings headaches and sometimes nausea, all signs the body’s under attack.
Routes to Safer Handling
Shutting the door on risk starts with rethinking how this solvent gets handled. Facilities laying out procedures rarely base them on paperwork alone—older workers share stories about close calls, or mistakes that led to an emergency room trip. That earned wisdom matters more than a poster on the wall.
Protective clothing stands between you and real harm. Gloves rated for organic solvents give fingers a fighting chance. Face shields and goggles protect from splashes. Lightweight aprons and sleeves keep the solvent off skin. The more comfortable the protection, the less likely it ends up tucked in a locker.
Ventilation systems keep the invisible vapors on a short leash. Open windows and a few fans don’t cut it—local exhaust hoods right where the chemical gets poured or used pull fumes away from your breathing zone.
Training rooted in real situations helps people remember the stakes. Running drills on what to do after a spill—step by step—makes all the difference. Supervisors who walk around, spot what’s missing, and hand out new gloves or masks, send a clear message: safety is part of the job, not a checkmark at orientation.
Storage and Emergency Response
I’ve seen barrels stored in rooms too hot, with lids only half secured. This stuff burns. Storing it in tightly sealed, clearly labeled containers, away from sparks or open flames, drops the chance of fire. Marking chemical storage zones and keeping spill kits within reach means no running around if something spills. Kits stocked with absorbent pads, neutralizers, and backup gloves let people clean up without guessing how to stay safe.
Pushing for easy access to safety showers and eyewash stations cuts recovery time from minor accidents. Every second counts after a splash. Workers who know where the nearest station stands—and have practiced getting there with eyes closed—don’t freeze up under pressure.
Why It All Matters
I’ve heard the phrase, “It’s just part of the job.” That attitude opens the door to shortcuts and accidents. Ethylene Glycol Monopropyl Ether does its work quietly, but so do its dangers. Real safety involves gloves that fit, training that sticks, and the courage to speak up before pouring another jug. Setting higher standards keeps dangerous mistakes out of daily life—one careful step at a time.
What is the chemical formula and structure of Ethylene Glycol Monopropyl Ether?
Chemical Identity and Structure
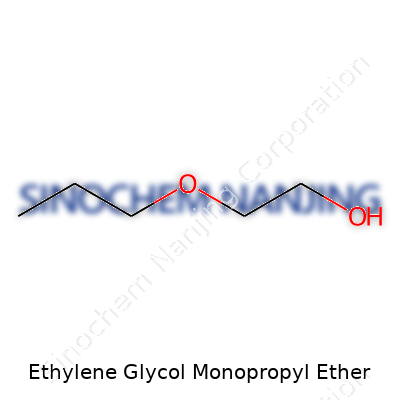
Ethylene Glycol Monopropyl Ether—often found in cleaning fluids, paints, and inks—stands out with a simple and clear chemical formula: C5H12O2. It carries the IUPAC name 2-propoxyethanol. Its structure paints a story of chemistry and industry coming together. Picture ethylene glycol’s two-carbon backbone, then swap one of its endings with a propyl group. What’s left: a molecule holding a hydroxy group (–OH) connected via an ethyl linker to a propoxy group. Mapping it out, the structure runs CH3CH2CH2OCH2CH2OH, a backbone that gives it unique traits in both solubility and reactivity. In labs and factories, this molecule gets called by its trade names as often as by its chemistry.
Importance in Real Life
I’ve handled products that rely on solvents like this one for decades. Ethylene Glycol Monopropyl Ether behaves like a workhorse in paints and cleaning agents, thanks to that mix of an alcohol and ether within the same molecule. It brings just the right balance of dissolving power. You’ll find it speeding up cleaning jobs where oily grime won’t budge for plain water or simple alcohols.
For those working in the coatings industry, the choice of solvent sets the tone for everything from finish quality to drying speed. C5H12O2 slips easily between water and oil phases. This lets it stabilize and mix ingredients that usually keep to themselves. In my own experience, swapping out older, harsh-odor solvents for monopropyl ether versions brought a better working environment—less stink, fewer complaints of headaches during long jobs.
Health and Safety Facts
Any honest discussion about solvents needs to face up to health considerations. Monopropyl ether versions outshine some older glycol ethers for lower toxicity but still require smart handling. Inhaling high concentrations can irritate eyes and lungs. Longer exposure, especially without proper ventilation, may turn into bigger health risks—just ask anyone who’s ignored the guidance on using gloves or masks. I learned the hard way, picking up headaches and skin irritation on a long repainting job before I paid more attention to labels and protective gear.
Public safety groups and regulatory bodies keep an eye on this ether. Their fact sheets and exposure limits grew out of real cases, not desk theory. Most manufacturers swapped in monopropyl ether for nastier solvents, taking safer chemistry to heart. This doesn’t mean the coast is clear, though. The CDC and OSHA call for careful storage, well-ventilated spaces, and regular breaks for folks in contact with the vapor or liquid for long periods.
Practical Solutions and Alternatives
Industry keeps searching for safer yet effective solvents. Blends have trended toward lower toxicity, with more focus on worker health, especially as green chemistry takes hold. Engineers test for solvents with similar dissolving strength, but with a lower health burden. Plant-based or biodegradable options are under the microscope, but few match the efficiency and price point found in ethylene glycol monopropyl ether. Some applications still reach for traditional chemistry because replacements come with cost or performance trade-offs.
So, it pays to know the facts: the formula C5H12O2, the common uses, and the clear side effects tied to poor handling. Upgrading ventilation, investing in better personal protective equipment, and choosing greener alternatives where they work can push risk down for those who rely on this ether for a living.
| Names | |
| Preferred IUPAC name | 2-propoxyethan-1-ol |
| Other names |
Propylene glycol ethyl ether 2-Propanol, 1-ethoxy- Propoxyethanol Ektasolve EP Dowanol EP Propylene glycol monoethyl ether |
| Pronunciation | /ˌɛθ.ɪˌliːn ɡlaɪˈkɒl ˌmɒn.oʊˈproʊ.pɪl ˈiː.θər/ |
| Identifiers | |
| CAS Number | 2807-30-9 |
| Beilstein Reference | 1209282 |
| ChEBI | CHEBI:50956 |
| ChEMBL | CHEMBL45698 |
| ChemSpider | 83449 |
| DrugBank | DB06710 |
| ECHA InfoCard | ECHA InfoCard: 03e1d7cd-6654-45e7-aafb-f55e42fe02f1 |
| EC Number | 203-951-1 |
| Gmelin Reference | 1147 |
| KEGG | C06508 |
| MeSH | D005006 |
| PubChem CID | 8124 |
| RTECS number | KC8575000 |
| UNII | HPY9X25F66 |
| UN number | UN1992 |
| CompTox Dashboard (EPA) | DTXSID5020609 |
| Properties | |
| Chemical formula | C5H12O2 |
| Molar mass | 118.17 g/mol |
| Appearance | Clear, colorless liquid |
| Odor | Sweet |
| Density | 0.913 g/cm3 |
| Solubility in water | miscible |
| log P | 0.075 |
| Vapor pressure | 0.34 mmHg @ 25°C |
| Acidity (pKa) | 14.8 |
| Basicity (pKb) | pKb: 5.7 |
| Magnetic susceptibility (χ) | -46.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.405 |
| Viscosity | 1.7 mPa·s (at 25 °C) |
| Dipole moment | 2.48 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 249.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -482.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4057.2 kJ/mol |
| Pharmacology | |
| ATC code | D07AX |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319 |
| Precautionary statements | P210, P280, P301+P312, P305+P351+P338 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 60°C |
| Autoignition temperature | 215 °C (419 °F) |
| Explosive limits | 1.1% (LEL) – 10.1% (UEL) |
| Lethal dose or concentration | LD50 oral rat 3089 mg/kg |
| LD50 (median dose) | 1,900 mg/kg (rat, oral) |
| NIOSH | KN1575000 |
| PEL (Permissible) | PEL: 25 ppm (skin) |
| REL (Recommended) | 25 ppm |
| IDLH (Immediate danger) | 700 ppm |
| Related compounds | |
| Related compounds |
Ethylene glycol monoethyl ether Ethylene glycol monobutyl ether Diethylene glycol monomethyl ether Diethylene glycol monoethyl ether Diethylene glycol monobutyl ether |

