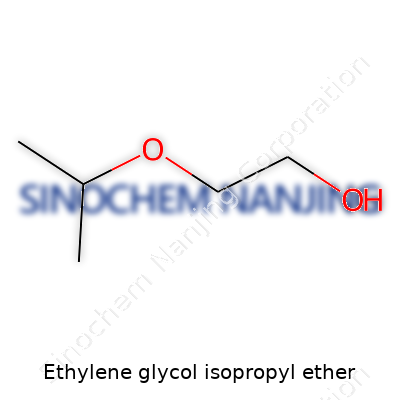
Ethylene Glycol Isopropyl Ether: Today and Tomorrow
How Industrial Chemistry Shapes Everyday Experience
Ethylene Glycol Isopropyl Ether may sound like something most people never touch, but its fingerprints are everywhere. Decades ago, chemists set out to create flexible solvents for the new wave of paints, coatings, and cleaners needed by a rapidly modernizing world. Over time, this ether became prized for how it can dissolve difficult substances, dry at a steady pace, and generally get out of the way after its job is done. Early work built on even older trade in glycols and ethers traced back to 19th-century breakthroughs in organic chemistry. Tinkerers and production chemists learned to value materials like this—not just for technical novelty but for real-world results. Looking back, I remember times in the lab when performance made all the difference: an experiment would stall until the right solvent came along, making the impossible seem simple. That’s the power of practical chemistry.
What Makes Ethylene Glycol Isopropyl Ether Unique?
Unlike some alcohols or straightforward esters, Ethylene Glycol Isopropyl Ether threads the needle with moderate polarity and a boiling point that meets the demands of industrial cleaning and specialized paints. You won’t see it pour as water does; it's got an oily slickness and a faint, almost sweet smell—evidence of its synthetic roots. In solution, it lets stubborn compounds dissolve or disperse, yet it won’t hang around so long that a user waits all day for drying. Technical specifications such as flash point, density, and acid number matter enormously to the folks who actually mix drums or calibrate blending machinery. Nobody making a precision coating or cutting fluid wants surprises from a batch, so trustworthy labeling and solid batch-to-batch consistency end up crucial for trade and for outcomes.
Getting Ethylene Glycol Isopropyl Ether Ready for the World
The story of its production is one of trial and error, patience and pressure. Industrially, it springs from a reaction between ethylene oxide and isopropyl alcohol, commonly carried out under carefully measured heat and pressure with specific catalysts. This reaction needs tight control. Miss the temperature or let water creep in, and yields or product purity can slip. From the perspective of anyone running the process, reliability beats brilliance—no one enjoys scrapping enormous batches because of poorly managed conditions. Chemical engineers, for their part, bring years of learning to tweak throughput and manage the handful of side reactions that can pop up. Once out of the reactor, purification by distillation or filtration removes unwanted leftovers. By the time the ether reaches drums or tanks, the goal is a material ready for jobs as varied as deinking newsprint or prepping an automotive finish.
Names, Synonyms, and Paperwork
Industry keeps track of names because confusion costs money. I’ve seen orders snarl up over a missing synonym or a translation error. You might see this compound called “2-Isopropoxyethanol,” “Isopropyl Cellosolve,” or “IPEG” in different catalogs. It lurks behind strings of numbers on safety documents and trade manifests, but the substance stays the same—an organic solvent bridging old-school and modern needs. Regulatory paperwork reflects the thesaurus of chemical trade: matching up names, CAS numbers, and hazard classes demands care. Those who manage compliance face thick binders and endless revision cycles as the naming conventions shift with each new set of rules from local, national, or international authorities.
Solvent, Synthesizer, and More: Wide-Ranging Uses
Look through the inventory at a coatings plant or a recycler’s workshop, and you’ll spot this ether. Decades of research underline why: it cuts grease, lifts inks, and helps plastics take a shine. As a solvent, it can carry resins, dissolve gums, or thin stubborn polymers. In paint and lacquer manufacturing, it boosts drying while smoothing application. Factories that reclaim used printstock count on it to strip color without damaging substrate. Smaller batches find their way into consumer strippers or degreasers—often carefully labeled and handled because of what we know today about chemical safety. If someone works in surface prep for high-end woodworking, there’s a decent chance they have handled this ether for cleaning or prepping surfaces ahead of staining.
Chemical Adaptability: Beyond the Basics
In chemical syntheses, Ethylene Glycol Isopropyl Ether sometimes enters as a starting point, not just as a background solvent. Researchers design modifications to the ether or its derivatives, aiming for new properties: adjusting evaporation, reactivity, or compatibility with next-generation polymers. Teams with access to well-equipped labs and creative minds probe reactions that tweak glycols, hoping to unlock new performance for paints, hydraulic fluids, or specialty cleaners. I've read project reports where a small shift in the structure led to marked differences in water solubility or resistance to yellowing—a reminder that chemists keep refining even the most familiar tools.
Double-Edged Properties: Addressing Toxicity and Operational Hazards
What’s useful in small, controlled doses can be troublesome out of bounds. Years of study show that direct contact or inhalation of Ethylene Glycol Isopropyl Ether, especially for those working day in and day out around it, brings some risks. Toxicity profiles put it among chemicals warranting gloves, goggles, and decent ventilation rather than casual handling. Some studies track reproductive effects and long-term organ toxicity, leading regulators in countries like the US and those in the European Union to restrict exposure or press for safer substitution in certain consumer jobs. Responsible companies invest in education, training, and engineering controls—fume hoods, spill kits, down-to-earth reminders never to skimp on protective equipment. People who have watched a safety drill turn into an actual emergency know that skip in protocol is no joke. Those lessons travel fast across factory floors.
Labeling, Shipping, and Staying Compliant
Getting this ether from the plant to the end user demands sharp attention to detail. Shipments cross borders; rules change as fast as lawmakers can adapt to new toxicological research. Correct labeling—hazard stones, safety phrases, risk codes—matters as much for legal compliance as for worker safety. From personal experience, a mislabeled drum at a loading dock can trigger long investigations or fines. Upstream suppliers and downstream buyers both have skin in the game to avoid lost time and liability.
Ongoing Research and the Question of Tomorrow
The push for greener, safer, and more environmentally friendly solvents creates never-ending demand for improvement. University chemists and industrial labs keep searching for ways to reduce exposure risks, improve breakdown in the environment, or extend performance into new materials or techniques. Recent work explores how to reuse or recycle solvents more efficiently—a concern for both environmentalists and penny-pinching plant managers. Professional groups publish regular updates on hazard profiles, technical data, and recommended best practices. In my own experience, some of the best advances begin at the ground level—new handling tools, tweaks in plant layout, or support for field workers are often as important as dreamy new molecules in a scientific journal.
Looking Ahead: Promise and Responsibility
Demand for tough, versatile solvents isn’t fading. Still, broad awareness of chemical hazards brings new urgency. As governments and consumers lean into the goal of less harmful, more transparent production, the challenge isn’t to retreat from solvents like Ethylene Glycol Isopropyl Ether—it’s to make every stage safer, clearer, and more sustainable. Chemists, managers, and policy makers have to work together so robust standards apply not just on paper but in every warehouse and workshop. If the future of industrial solvents heads toward safer substitutes or clever recycling, this will only come from open eyes to real-world risk and collaboration between people who know both laboratory science and day-to-day logistics. Investment in training, deep dives into environmental impact, and respect for everyone handling these materials in real settings push the field toward real progress.
What is Ethylene Glycol Isopropyl Ether used for?
Understanding Ethylene Glycol Isopropyl Ether
Ethylene glycol isopropyl ether helps industries solve cleaning and blending challenges. I remember coming across it for the first time in an auto body shop, mixed into a solvent blend used to clean paint sprayers. It doesn't stand out in the world of chemicals by name recognition, but its practical role in industrial processes means you find it in lots of unexpected places.
Solvent Power in Paints and Coatings
Painters, manufacturers, and DIYers rely heavily on solvents to create a smooth finish. Many paints, varnishes, and coatings need a chemical that thins the mixture and helps it spread evenly across a surface. Ethylene glycol isopropyl ether fits this job well because it mixes oil-based and water-based substances without leaving streaks or residue. The paint looks better and dries more predictably. For big facilities making coatings in bulk, this chemical gives them what they need to deliver both durability and shine.
Cleaning Up Tough Messes
Spray a little of this chemical on a greasy machine part, and grime breaks up much faster. Industries that depend on clean equipment—factories, garages, print shops—reach for blends containing ethylene glycol isopropyl ether. Its grease-busting strength keeps essential machinery in good shape and reduces the time spent on maintenance. I saw this firsthand while cleaning engine parts with a team—parts that would take hours to scrub clear with old-fashioned soap needed only a few rags and a solvent containing this ether.
Contributions in Inks and Textile Dyes
Printing presses and textile plants look for reliable ways to get strong, even color. This chemical gives ink makers more control. It keeps ink from drying up inside machines, reduces smudging, and allows images to stay crisp. The same goes for fabric dyes—vivid colors, less fading, and fewer production headaches. Companies prioritizing high-quality results keep an eye on ingredient lists for just this reason.
Pushing Forward With Safer Practices
The downside cannot be ignored. Ethylene glycol isopropyl ether, like a lot of industrial solvents, poses health and safety risks. Inhaling fumes or getting the liquid on bare skin has the potential to cause short-term irritation and long-term damage. I’ve worn my share of gloves and masks after watching a coworker develop a rash on his wrist during a busy shift. The chemistry delivers impressive cleaning and blending abilities, but workers and companies must always keep safety rules front and center—protective equipment, ventilation, and handling protocols stay non-negotiable.
Finding Better Solutions
I see industries making careful changes these days. Health concerns and tighter regulations mean chemical companies look for safer substitutes when possible. Some firms are testing less toxic blends or investing in technology that requires fewer harsh solvents. For people who work around these chemicals daily, every improvement in safety standards means a noticeable difference at the end of the workweek. Researchers continue to study solvents for better choices, but until safer options become standard, responsibility falls on everyone in the chain—from engineers to frontline workers—to use products like ethylene glycol isopropyl ether wisely.
What are the safety precautions when handling Ethylene Glycol Isopropyl Ether?
The Hazards Lurking in Everyday Work
Throughout years spent working in laboratories, I’ve developed a sharp sense for the real risks behind chemical names. Ethylene glycol isopropyl ether isn't just a mouthful—it’s a chemical that comes with hazards lurking under the surface. Don’t let the word “glycol” fool you, just because similar chemicals crop up in antifreeze doesn’t mean handling is a breeze. This solvent makes its way into coatings, inks, and cleaners, but it doesn't greet everyone who handles it with open arms. Get careless, and you’ll discover skin irritation, headaches from fumes, even trouble with your liver and kidneys lurking down the road.
Simple Habits That Make the Difference
The most practical shield against ethylene glycol isopropyl ether is to cut off any easy path into your body. The eyes, the skin, and the lungs offer a welcome mat if you stand too close or don’t gear up. I keep my skin guarded—nitrile gloves, lab coat zipped, and safety glasses that block splashes. I learned once in my early days how failing to swap out gloves fast enough after a spill leads to itchy rashes that last all week. It stings, and it reminds you the chemical isn’t gentle.
Good ventilation beats bravado every time. A well-run fume hood moves air fast enough to sweep away fumes. More than once, I’ve watched folks take shortcuts, thinking a cracked window is enough. It's not. Without real airflow, vapors stick around, and headaches don’t take long to follow. OSHA points out that repeated breathing can chip away at health, slow but steady, if engineers ignore proper air flow.
How Carelessness Opens the Door to Accidents
Once, I watched a team skip the step of labeling a beaker of what looked like plain water. That “plain” liquid turned out to be ethylene glycol isopropyl ether, and a careless hand left it on an open bench. Anyone in the shop could have reached for it, thinking it was harmless. This taught us labeling isn’t just formality—it’s a barricade against accidents. Bright labels, clear hazard warnings, and putting chemicals back on shelves fast makes harm less likely.
No lunch breaks in the lab, ever. Food absorbs vapors, even with a lid. I keep my drinks, snacks, and sandwiches away, remembering how ethylene glycol derivatives cause nausea and drowsiness long before an official “exposure limit” sneaks up.
Clean-Up and Emergency Know-How
Spills happen. I always keep an absorbent spill kit nearby. Mop it up right away, dispose of rags in proper containers, and never hose chemicals down the sink. Training steps in here. Everyone who handles this stuff should know their shop's emergency phone numbers by heart. Splashes in the eyes need a fifteen-minute eyewash rinse, and inhaling a heavy odor means getting outdoors fast. I’ve watched people freeze, unsure what next step comes. Drills and clear job-site instruction keep panic at bay.
A Culture of Safety Builds Trust
People rarely talk about how the best protection doesn’t just come from gloves and goggles. It comes from workplace trust. Colleagues check each other’s gear. Supervisors enforce safety stops. Tidy record-keeping makes sure no container gets left behind. Health and safety agencies like NIOSH lay out the exposure limits, but in real life, it’s daily habits that keep that invisible line from getting crossed.
The lessons I take from years surrounded by chemicals aren’t dramatic, just steady. Every routine—gloves, airflow, labels, cleanup—locks shut the door that ethylene glycol isopropyl ether is ready to open. Avoiding shortcuts gives everyone a better shot at leaving shift with their health intact.
What is the chemical formula of Ethylene Glycol Isopropyl Ether?
The Formula at a Glance
Ethylene glycol isopropyl ether has the chemical formula C7H16O2. In the lab, this name sometimes gets lost among rows of similar-sounding compounds, but every part of that formula tells a story. C7—seven carbon atoms—signals its modest size; H16 gives a nod to its hydrocarbon base; O2 signals its key oxygen links. The structure spells out practicality: a glycol ether backbone with an isopropyl group tacked on the end, making it both a strong solvent and safe enough for use in multiple applications.
Why Chemical Names Matter in Daily Life
Most folks never see this compound outside of a label on cleaning products or industrial safety sheets. But for anyone working in paints, coatings, or specialty cleaners, knowing what C7H16O2 actually means brings its own set of responsibilities. Chemical formulas aren’t just for textbooks—they’re roadmaps for how a compound acts in the real world. The presence of the ether link gives it a balance, keeping it less aggressive than pure alcohol-based solvents but still strong enough to dissolve tough greases and inks.
Safety: More Than Just Lab Coats and Gloves
From my experience around industrial settings, I’ve learned fast that glycol ethers walk a fine line between usefulness and risk. The same property that makes ethylene glycol isopropyl ether an effective degreaser—its ability to cut through oily compounds—means it can also cut past the skin barrier. Chronic exposure has sometimes led to headaches or skin irritation among plant workers, especially when folks get lax about personal protective equipment. Safety data sheets spell out the need for good ventilation, gloves, and eye protection, but real-world habits drive home that point even harder.
Regulators pay close attention to glycol ethers, since years of research have shown that long-term exposure to some kinds can cause health issues. Ethylene glycol isopropyl ether has managed to avoid some tighter restrictions because of its moderate toxicity compared to others in its family, but workplace safety always calls for respect and prepared teams.
Push for Greener Alternatives
Industry has started looking for alternative compounds that do the same job with less environmental impact. Many users—especially in Europe—demand lower volatility products with lower risks for air quality and workplace safety. Some companies have made progress with bio-based solvents, blending plant-derived molecules to replace petrochemical options. These eco-friendly options often cost more, but more buyers have shown they’re willing to pay, given strong proof that safety goes up and long-term environmental costs go down.
Training Still Makes the Biggest Difference
Easy access to chemicals brings advantages and responsibilities. In every shop floor training session I’ve attended, the message rings clear: formulas like C7H16O2 aren’t just numbers. They’re blueprints for handling, storage, and disposal. Someone once tossed a leftover pail in regular trash, not realizing a spill could send volatile fumes into a crowded storage area. Incidents like that drive home why chemical literacy matters, not just for scientists but everyone in the supply chain. Safer habits and honest communication about risks ultimately make science work better for everyone.
How should Ethylene Glycol Isopropyl Ether be stored?
Why Storage Demands Attention
Ethylene Glycol Isopropyl Ether doesn’t draw much attention unless you work with chemicals, but stories from project sites show a sloppy storage setup can bring big risks. Spend a little time around chemical plants or talk to folks in the auto industry—everyone has a tale about unexpected leaks or vapors. Small mistakes during storage spark expensive clean-up jobs, health scares, or, at worst, dangerous fire. Think of this not as a regulatory hoop but the kind of thing that ensures everyone goes home at the end of the day.
Storing Safely Means Knowing the Hazards
This solvent evaporates easily and burns without much effort. If it drips onto a surface, fumes come off quickly, and those vapors can irritate your lungs. Eyes, skin—they’re both at risk if you get careless. The solution starts with limiting evaporation and keeping open flame well away from storage spots. A friend once ignored an old rag near a container, and before long, the stench filled the whole workshop. Vapors from this ether aren’t just an annoyance—they carry health consequences, especially in a cramped or stuffy storeroom.
The Right Container Makes Life Easier
Always reach for a container made from high-grade, compatible plastic or steel. Flimsy or rust-prone containers lead to leaks and big headaches down the line. I’ve watched a few folks learn the hard way: one punctured drum and half the storage room needed scrubbing. Seals matter just as much. A properly closed lid keeps vapors in and air out, slowing any reaction or spoilage—not to mention avoiding the pungent smell wafting through the building.
Ventilation: Fresh Air for Safety
Good ventilation isn’t just a detail—fresh airflow removes any vapor that escapes when containers open. I remember a facility that skimped on fans in the warehouse. The spot nearest the door felt fine, but fumes lingered farther in, making every trip to that aisle risky. A simple fix with duct fans and open vents made a world of difference. Clean, circulating air drops the risk of accidental intake and prevents headaches.
Keep It Cool and Dry
Warm, sunny spots spell trouble for this solvent. Elevated temperatures speed up evaporation and increase pressure inside sealed drums. That extra pressure threatens to pop lids or fittings free. Store drums away from heaters, boiler rooms, or anywhere temperatures spike. Set them in a shady corner, preferably on a raised pallet, since a water leak in the building or a wet floor can corrode bases and ruin labels. Labeling gets overlooked, but nothing good happens when a drum turns up unmarked after a few weeks. Fading markers cost people time and can even lead to mishandling.
Small Steps and Big Returns
Sticking to simple practices—strong containers, dry storage, clean ventilation, and solid labeling—protects everyone in the area. Training staff helps too. Handling chemicals safely becomes a habit when the right setup and clear procedures back it up. With accidents and fines looming over every workplace, cutting corners never pays off. A modest investment in proper storage can repay itself many times over by preventing unexpected emergencies. Experience shows that these habits form the backbone of safe, efficient workplaces.
What are the health hazards of Ethylene Glycol Isopropyl Ether exposure?
Understanding What We’re Dealing With
Ethylene Glycol Isopropyl Ether sounds like lab jargon, but it’s actually a chemical that shows up in products such as cleaners, paints, and industrial solvents. Some people might walk past it every day on the job or handle it through DIY projects without knowing the dangers lurking in that clear liquid.
How the Body Reacts to This Chemical
Anytime I have handled solvents in the past, the smell alone gave me a hint of what’s at stake. This one carries even more risk beneath the surface. Skin contact proves risky, causing irritation or rashes that can get much worse with repeated exposure. My neighbor, who worked in janitorial services, wound up with red, painful hands after weeks of handling chemicals without gloves. What nobody warned him about, he learned the hard way: symptoms may creep up slowly, and by the time they become obvious, the damage could run deep.
Inhaling vapors spells real trouble. The lungs don’t like these fumes, which can end up causing coughing, headaches, dizziness, and even confusion. In closed spaces, breathing gets harder, and the chance of feeling faint rises. I remember repainting my own garage with inadequate airflow; halfway through the job I needed to step outside and felt woozy for hours. These aren’t just annoyances—the chemical targets the nervous system, hinting at wider effects inside the body.
Digging into the Science
Studies have shown that ingested Glycol Ethers can damage red blood cells and the liver. The National Institute for Occupational Safety and Health (NIOSH) warns workers about potential links to kidney failure, reproductive issues, and even birth defects in the unborn child. Blood work on exposed animals revealed harmful breaks in blood chemistry and organ function. That tells me the risk’s not just on the surface—internal health can get sideswiped, too.
Chronic low-level exposure is just as concerning. A research article in the Journal of Occupational and Environmental Medicine flagged long-term cognitive and memory problems in workers with persistent solvent exposure. People might blame aging or stress, not realizing the harm could stem from jobs they’ve held for years.
Steps for Protecting Ourselves
Some folks still roll up their sleeves without gloves or skip the mask in the paint booth, but the facts make clear protection has to come first. Gloves made to resist solvents, goggles, and fitted masks cut risk dramatically. Good airflow helps vapors escape before they reach the lungs. In my experience, people who take shortcuts learn quickly—one incident can lead to weeks of misery, all for saving a few minutes.
Workplaces ought to take up regular air quality checks and keep material safety data sheets handy in plain language. Nobody gains from hiding the facts or hoping for the best. Even at home, reading the fine print on product labels helps folks steer clear of unnecessary trouble. Parents, in particular, need to store chemicals out of kids’ reach.
Pushing for Safer Alternatives
Demand for gentler cleaning products and low-tox paints keeps growing. I’ve made the switch where I can, and noticed far fewer headaches or skin issues. Supporting companies that phase out harsh chemicals nudges the whole industry in a healthier direction.
Real knowledge puts power back where it belongs: with people who want to keep themselves, their families, and their coworkers healthy. The best defense will always be staying informed, keeping protection gear close, and not taking the risks lightly.
| Names | |
| Preferred IUPAC name | 2-(Propan-2-yloxy)ethan-1-ol |
| Other names |
2-Isopropoxyethanol Isopropyl cellosolve Propylene glycol isopropyl ether IPGE Glycol isopropyl ether Ethylene glycol monopropyl ether Isopropoxyethanol |
| Pronunciation | /ˈɛθɪliːn ɡlaɪˈkɒl aɪsəˈprəʊpɪl ˈiːθər/ |
| Identifiers | |
| CAS Number | 109-86-4 |
| 3D model (JSmol) | `3d:CCCCOC(CO)CO` |
| Beilstein Reference | 1340748 |
| ChEBI | CHEBI:81937 |
| ChEMBL | CHEMBL59130 |
| ChemSpider | 18523 |
| DrugBank | DB14189 |
| ECHA InfoCard | 03bba2e6-d03a-44e7-9640-d922b729cb66 |
| EC Number | 203-689-7 |
| Gmelin Reference | 8096 |
| KEGG | C19597 |
| MeSH | D017568 |
| PubChem CID | 8126 |
| RTECS number | KK5075000 |
| UNII | 48E51C00S4 |
| UN number | UN2379 |
| CompTox Dashboard (EPA) | DTXSID7054726 |
| Properties | |
| Chemical formula | C5H12O2 |
| Molar mass | 134.18 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Mild ether-like |
| Density | 0.92 g/cm3 |
| Solubility in water | miscible |
| log P | 0.14 |
| Vapor pressure | 0.3 mmHg (20°C) |
| Acidity (pKa) | 14.7 |
| Basicity (pKb) | 15.03 |
| Magnetic susceptibility (χ) | -47.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4100 |
| Viscosity | 1.89 mPa.s (25°C) |
| Dipole moment | 2.16 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 310.52 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -537.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4357 kJ/mol |
| Pharmacology | |
| ATC code | D07AX05 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H302 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P301+P310, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P362+P364, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | Flash point: 49°C (120°F) |
| Autoignition temperature | 215 °C |
| Explosive limits | 1.0% - 12.5% |
| Lethal dose or concentration | LD50 oral rat 3089 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 3089 mg/kg |
| NIOSH | NIOSH K160 |
| PEL (Permissible) | PEL: 25 ppm (140 mg/m3) |
| REL (Recommended) | 25 ppm |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Ethylene glycol butyl ether Ethylene glycol methyl ether Ethylene glycol ethyl ether Diethylene glycol monomethyl ether Diethylene glycol monoethyl ether |

