Ethylene Glycol Ethyl Ether: More Than Just a Solvent
The Story So Far
Looking back at how chemical industries grew throughout the twentieth century, ethylene glycol ethyl ether stands out as one of those quiet workhorses seen in paints, inks, cleaners, and electronics. Its roots trace to the days when advances in organic chemistry opened doors to new glycol ethers—compounds that blend water solubility with the grease-cutting power that industry craved. This compound, known in labs and on labels as EGEE, made its way into toolkits around the world, doing jobs that demanded both muscle and finesse. Scientists tuned its preparation with reactions involving ethylene oxide and ethanol, chasing better yields and more reliable purity. As globalization reshaped manufacturing, EGEE moved alongside, weaving itself through processes from Europe to Asia.
A Closer Look at EGEE’s Key Features
EGEE shows up as a clear liquid, not so different in appearance from water, but with a scent that hints at the laboratory. Its boiling point, hovering above 130 degrees Celsius, allows it to handle tasks from high-speed printing presses to cleaning circuit boards in electronics plants. Unlike simple solvents that evaporate at the first sign of heat, EGEE lingers just long enough to dissolve resins and dyes fully, offering reliability in commercial applications. Its moderate polarity means it cuts through grease and grime, but unlike heavier ethers or hydrocarbons, its performance stays stable even on a muggy summer day. Pouring EGEE requires care, since it can absorb water from the air and shift any batch’s composition. Its storage and transport benefit from well-sealed drums, which helps avoid unexpected moisture and potential breakdown.
Staying Safe in Everyday Operations
Anyone using EGEE comes to respect the need for safety and discipline. Regulatory bodies in the US and Europe have published standards for handling glycol ethers. Even small spills count, since vapor inhalation or skin contact can build up in the system over time, triggering headaches, nausea, or more serious harm. That means workers turn to gloves, goggles, and ventilation hoods—not out of some bureaucratic requirement, but because old stories from veteran chemists taught the cost of shortcuts. Waste disposal matters too, as glycol ethers in groundwater invite close scrutiny from regulators and environmental groups. Even large industrial operations face tight supervision when using EGEE, especially near areas where people live and water supplies run close to the surface.
Chemical Reactions and Creative Tweaks
Chemists gravitate to EGEE not only for its solvency, but also its flexibility as a building block. Under controlled conditions, ethylene glycol ethyl ether reacts with acids or bases, expelling the ethyl group or opening up opportunities for further chemical grafting. This adaptability works particularly well for creating specialty inks or coatings that demand just the right drying time and finish. Over years, researchers published method after method for nudging the molecule in new directions—lengthening the chain, introducing halogens, or shifting its reactivity. This kind of laboratory creativity allowed smaller firms to differentiate products in crowded markets, underlining EGEE’s essential role as more than just another solvent.
Names on the Label and Across the Industry
Across the globe, EGEE carries several names, from 2-ethoxyethanol to ethyl cellosolve. Some might call it glycol monoethyl ether, but whoever writes the name, the liquid in the drum stays the same. Regulations in different countries sometimes demand specific label language, but most chemists grow used to recognizing EGEE among a list of similar-sounding glycol ethers. This overlap sometimes breeds confusion, but also keeps experienced hands sharp as they sort out what’s in a truckload before letting it onto a production floor.
Sectors That Rely on This Glycol Ether
Walking through a modern print shop, an electronics assembly line, or a paint factory, traces of EGEE’s usefulness show up everywhere. In printing, it carries inks deep into complex substrates, preventing streaks or clumps. Cleaners thickened with EGEE cut oils from delicate electronic parts, letting manufacturers deliver ever-tinier gadgets. In coatings, it balances drying times, helping crews paint cars or machinery even on humid days. Lab researchers like EGEE for dissolving both polar and nonpolar components, speeding up extractions or chromatography work. As innovations in batteries and solar panels call for ever-stricter process control, the consistent, predictable behavior of EGEE proves invaluable in R&D as well.
Weighing Risks and Keeping Workplaces Healthy
Health questions cast a long shadow over glycol ethers, especially those with a track record like EGEE. Medical literature points to a link between exposure and effects on blood, reproductive health, and organ systems, with evidence growing out of animal studies or past workplace accident reports. Regulators have lowered permissible exposure limits with each decade, pushing firms to upgrade gloves, improve ventilation, and test the air more regularly. End users face a real challenge: capturing the benefits of an effective solvent without sliding into practices that risk long-term illness for workers handling the drums and mixers. Industry groups, watchdog organizations, and community activists continue to debate where the line falls between safety and productivity. This is a fight unlikely to end soon.
Current Research: Seeking Safer Alternatives
Innovation in solvents focuses today on reducing health and environmental hazard while holding on to performance. Research groups in academic and industry labs screen alternatives for both toxicity and technical capability. Some promising candidates in the alkyl ether category show less risk for chronic harm, but struggle to match EGEE’s clean solvency. Collaborative projects look at green chemistry approaches, including biodegradable or plant-derived alternatives, though few have cracked the balance needed for industrial scale. The conversation among chemists recognizes that perfect solutions come slow, but the stakes—human health, environmental quality, and economic competitiveness—push work forward despite the hurdles.
Looking Down the Road: What Comes Next
As industry trends shift toward stricter regulation, demand for sustainable chemicals increases, and the global workforce gets more informed, EGEE finds itself at a crossroads. Many legacy facilities adapted processes over years to rely on this compound; switching out comes with real cost and loss of institutional expertise. Yet, pressure mounts each time a new medical study highlights another potential health concern. The future likely holds a mix of evolution and revolution: improved handling technologies, better training for workers, and—eventually—innovative substitutes that can match EGEE’s performance without its drawbacks. Firms that succeed will be the ones willing to recognize both the track record of this old staple and the need for change, showing respect for history without letting nostalgia get in the way of progress.
What are the primary uses of Ethylene Glycol Ethyl Ether?
What Makes It Stand Out
Ethylene glycol ethyl ether lands in more places than most people think. Most have never heard of it, and that’s not surprising. This clear, flammable liquid moves quietly through the world of manufacturing and consumer goods, often as a silent helper in daily life. I worked in a paint supply store for a few years, and it surprised me how many labels read like chemistry homework. Ethers like this one often handle the tough jobs behind the scenes.
Main Uses in Industry
The number one job for ethylene glycol ethyl ether shows up in paints and coatings. This chemical takes on the role of solvent, thinning the paint so it goes on evenly and dries without leaving brush or roller marks. Anyone who’s watched a wall stay sticky for days probably didn’t have enough of the right solvent in the mix. It also keeps paints stable inside the can, stopping clumps or separate layers. Chemists pick it out because it works with both water and oil-based formulas, something you won’t find in every solvent.
Cleaning products rely on it for much the same reason. It gives glass cleaners, degreasers, and ink removers the muscle to lift off dirt, oil, and marker without harming most surfaces. In the janitorial supply world, managers pay close attention to what goes in these mixtures, since the wrong solvent leaves streaks or dulls the shine on glass and counters.
Printing inks would struggle without it. Commercial printers use this chemical for inks that need to dry fast but resist smudging after drying. With so much package labeling and book production running at top speed, slow-drying ink causes jams and lost revenue.
Other Critical Uses
Manufacturers of textiles and leather use ethylene glycol ethyl ether, often under brand names, to soften materials and make dyes take hold without fading. A run of plain white t-shirts or weatherproof jackets looks a lot better after the fabric passes through a bath laced with this solvent. The same goes for shoe or furniture leather—color and texture both improve, which means less waste and higher profits.
The electronics industry gets more attention as gadgets fill up homes and offices. Here, ethylene glycol ethyl ether shows its value in cleaning and prepping circuit boards before assembly. Tiny, delicate parts need solvents that aren’t too rough, since leftover residue can ruin connections. I once talked with a plant engineer who swore by a special blend containing this ether because he saw fewer short circuits in finished products.
Balancing Performance and Safety
People tend to overlook what makes products work until a recall or a health scare hits the news. Ethylene glycol ethyl ether comes with some risks—workers exposed to high amounts, especially in older factories, can run into headaches, dizziness, or worse. That’s why regulations now require strong ventilation and protective gear for those handling it. In my time around chemical warehouses, I always spotted big signs pointing out the extra steps taken to keep air clean.
Safer alternatives exist for some applications, but few match this solvent’s blend of power and flexibility. Environmental groups keep the pressure on companies to find greener replacements. Some new water-based paint systems promise similar results, and more cleaning product companies push for plant-based ingredients.
In the end, ethylene glycol ethyl ether stays in the mix because it works—fast, reliably, and across so many fields and products trusted every day. The push for innovation grows each year, but this chemical holds its ground on factory floors and store shelves, always working behind the scenes to make the rest of life a bit smoother.
What are the safety precautions when handling Ethylene Glycol Ethyl Ether?
What Makes Ethylene Glycol Ethyl Ether a Concern?
Ethylene glycol ethyl ether, often found in cleaning agents, paints, and inks, works as a strong solvent. Its sweet smell and clear appearance don’t hint at the risks beneath. It can move quickly into the body through skin, inhalation, or swallowing, and its impact doesn’t stop at a simple rash or cough. Upset stomach, headaches, drowsiness, and even kidney or liver damage show up in people who overlook the safety basics. No one forgets the first time they spill a little on bare skin—the redness and discomfort are pretty quick lessons.
Smart Handling Starts with Protective Gear
The safest crews gear up before uncapping a container. Nitrile gloves block the solvent far better than latex. Cotton and regular work gloves soak up spills and trap the chemical against skin. Splash-proof goggles protect from the drifting fumes or accidental sprays. I’ve watched new workers ignore this step until their eyes burn or the skin between their fingers goes tender and red. After just one exposure, they reach for gear every time.
A good cleanup workspace uses an apron or lab coat that resists solvents. Street clothes soak up spills and carry them long after a shift ends. A respirator certified for organic vapors clears out fumes that linger near open containers. Even on days that seem calm, a slow build-up in the air can lead to headaches within an hour. It doesn’t take long to see which companies care enough to supply the right masks and which ones gamble with medical costs.
Essential Steps Before and After Use
Ventilation means more than opening a window. Local exhaust hoods, fume extractors, and running air systems drag away low-level vapor and keeps exposure in check. Pouring without a vent system can make the difference between feeling fine and getting the shakes halfway through a shift. I’ve spent time in a shop where opening a door simply didn’t cut it—even mild symptoms forced management to install fans and extraction units fast.
People sometimes skip basic hygiene. Washing hands and arms thoroughly after handling solvents is not old-fashioned advice—skin absorbs more than you’d think. Eating or smoking without cleaning up leads to swallowing dangerous residues. In my own experience, workers would skip this in a rush, but the stomach problems and sick days that followed changed habits quick.
Storage and Emergency Planning
Steel drums and tightly sealed plastic containers keep this chemical from leaking into the workspace. Labels, bold and clear, ensure no one mistakes one drum for another. Accidents still happen—spills spread fast, and without spill kits nearby, a small problem becomes a costly one. Absorbent pads and neutralizing agents stored within reach cut down chaos. Fire extinguishers must be close, since vapors catch a spark quicker than many realize.
Training all staff, not just the safety manager, makes the biggest difference. New regulations from OSHA and other agencies push for annual training sessions, but the best shops refresh this info every few months. Workers learn what dizziness or red skin signals, and managers respond with speed instead of confusion. Medical checkups focus not just on obvious symptoms but also on early markers for nerve or organ stress.
Building a Culture of Safety
Working with solvents always carries a risk. Respecting that risk, wearing the right gear, setting up real airflow, and having emergency plans ready keeps everyone healthy. Personal experience and plenty of workplace stories show that small steps—like washing up, storing products right, or using spill kits—really save trouble down the line. This isn’t just about checking a safety box. It’s about making sure everyone finishes the workday without hidden harm or regrets.
Is Ethylene Glycol Ethyl Ether hazardous to human health?
Understanding the Risks
Ethylene glycol ethyl ether, sometimes known in technical circles as EGEE, shows up in industrial work. The colorless liquid doesn’t have much of a smell, but it slips through the air easily. I’ve come across it in paints, cleaning fluids, inks, and even degreasers. Most folks outside specialty trades don’t bump into it every day, but those who do should not underestimate it.
Direct exposure, especially over long periods, leads to serious health trouble. Workers in auto shops or factories often breathe in vapors or get droplets on bare skin. Skin starts feeling dry or irritated, eyes sting, and after longer exposures, problems go deeper. Studies have found that ethylene glycol ethyl ether can damage blood, kidneys, and even reproductive organs after enough time. Researchers from the American Conference of Governmental Industrial Hygienists have set exposure limits based on tests showing developmental toxicity. Lab animals exposed over pregnancy showed birth defects. The U.S. Environmental Protection Agency and other groups flagged this chemical for those exact reasons.
Dangers of Routine Exposure
Folks working in manufacturing know how misleading some chemicals can be. No scent, nothing visible—still does harm. Short-term symptoms don’t always stand out, and people brush them off as fatigue or a simple headache. Mixing paint or cleaning a printing press, I’ve felt a headache set in later, only learning about chemical exposure much afterward. Prolonged inhalation of EGEE vapors can slow reaction times, cloud thinking, or worsen asthma. Chemicals like these don’t usually cause dramatic symptoms overnight, but they quietly build up harm. Some evidence suggests long-term exposure may contribute to anemia, and health agencies have tracked kidney damage after repeated incidents in industrial settings.
A friend who worked in boat maintenance shared stories of coworkers ignoring gloves or fans during the rush to finish a job. Within months, a few started seeing skin rashes or reporting forgetfulness. Occupational safety records back up these stories. The World Health Organization and CDC warn that these symptoms often go unreported, yet long-term harm sneaks up all the same.
Reducing the Harm
There’s real room for improvement. A few simple steps can cut the risk sharply: local exhaust ventilation, proper gloves, and keeping containers tightly sealed. I worked at a shop where the boss brought in better vent fans and made gloves standard issue. You could feel the difference in the air, and complaints about aches and rashes dropped. Training workers to recognize the subtle symptoms makes a difference, too. Manufacturers who swap this chemical for something safer lower the risk right away, and several companies have already started doing that.
Regulatory watchdogs play a role, but proactive shop owners and workers protect health every day. No sense waiting for disaster. I keep up with the latest safety guidelines from trusted sources like the National Institute for Occupational Safety and Health because it pays off. Health matters more than cutting corners.
How should Ethylene Glycol Ethyl Ether be stored and disposed of?
Why Storing This Chemical Takes Real Effort
Ethylene Glycol Ethyl Ether shows up in a lot of places, from paints and coatings to electronics manufacturing. Anyone who has spent time around this liquid knows a few facts: its sweet smell doesn’t make it any less risky, and a splash on bare skin can turn into a headache quickly. The stuff evaporates easily, so it doesn’t play nice with open air or loosely sealed containers.
A steel or high-density polyethylene drum with a tight-fitting lid has proven reliable over the years. I remember an old job site where a forgotten can leaked, and the result was no joke—peeling labels, headaches that lingered, and a frantic cleanup. The lesson hit hard: store the drum in a cool, well-ventilated spot, away from sunlight or heat sources. Temperature swings speed up vapor leaks. Leaving it near an open flame or a running motor stacks up risk for fire and even explosions. Storing this solvent near oxidizers or acids also ramps up danger.
A clear storage schedule and regular container checks save time and trouble. One tip from seasoned safety supervisors: label everything with clear chemical names and hazard warnings. Don’t rely on memory; taped-on scribbles fade fast under splashes or sunlight, and a missing warning can put someone in a world of trouble.
Disposal Isn’t a DIY Job
Pouring Ethylene Glycol Ethyl Ether down a drain or into regular trash shouldn’t even cross your mind. Tried-and-true rules from environmental agencies are strict for a reason. This solvent can wreck aquatic habitats and stick around in the environment, long after it leaves the shop.
Used rags, filters, and containers that held the solvent count as hazardous waste, too. In my years in manufacturing, waste collection day always meant double-checking every storage closet. All containers should feed into a locked, labeled, fire-resistant cabinet before they go into a licensed hazardous waste program.
Working with a certified waste disposal contractor keeps everyone above board. These folks handle the transport, tracking, and correct treatment—usually involving incineration at high temperatures. It’s pricey, but the cost of skipping steps runs much higher if you factor in potential fines and cleanup after a spill.
Education and Training Make All the Difference
Every new worker I’ve trained on a shop floor has needed to see, step-by-step, how serious proper storage and disposal get. It’s never enough to hand someone a sheet of official rules. Real learning happens through clear demonstrations and regular drills. Hands-on practice with spill kits, fire extinguishers, and emergency ventilation wins out over a hundred PowerPoint slides.
Rarely does a year pass without a new story about solvent mishandling and workplace injuries. Those stories often start with someone cutting corners on storage or treating a small spill as “no big deal.” Company leaders shape safe behavior by making it easy to do things right with the right equipment and clear procedures.
You won’t find a shortcut to safe handling of Ethylene Glycol Ethyl Ether. Setting higher standards for labeling, storage conditions, and disposal keeps people and the environment safe. It’s the kind of investment that pays off every day, one safe drum at a time.
What is the chemical formula and physical properties of Ethylene Glycol Ethyl Ether?
Chemical Formula and Structure
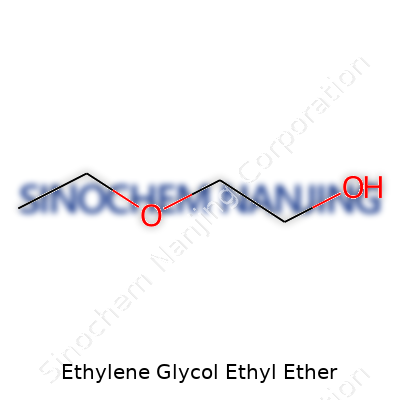
Ethylene glycol ethyl ether, known in the trade as EGEE, carries the chemical formula C4H10O2. Structurally, it stands as a simple glycol ether, with ethylene glycol providing the backbone and an ethyl group joined to one of the oxygen atoms. The systematic name, 2-ethoxyethanol, highlights the arrangement—a two-carbon chain with a hydroxyl group on one end and an ethoxy group on the other.
The molecular weight clocks in at 90.12 g/mol. In factories, labs, or workshops, it’s the chemical’s unique balance of water solubility and ability to dissolve greases and oils that gives it real-world value.
Physical Properties
Pour a bit of EGEE into a glass beaker and you’ll see a clear, colorless liquid. Many recognize the slightly sweet, sometimes bitter odor even at low concentrations. At room temperature, it moves like light syrup—its viscosity sits low enough to handle easily, but it clings more than water. This liquid boils at about 135°C (275°F), which lands well above water's boiling point, making it stable in plenty of industrial processes that generate heat.
EGEE’s freezing point doesn’t come into play unless a room gets extremely cold, dropping below –70°C (–94°F). It evaporates slowly, releasing vapor into the air without immediate notice. Yet, its flash point, at roughly 49°C (120°F), means handling and storage always need care in workrooms to avoid accidental ignition.
Water and oil both succumb to EGEE’s dissolving power. Painters, ink makers, and cleaners value this solvent for its ability to carry pigments, clean up sticky residues, or thin out stubborn grease—all without the toxicity that comes from older, harsher chemicals.
Everyday Applications and Concerns
Look at a fresh coat of paint, a well-printed magazine, or polished industrial equipment, and odds are that EGEE played some part somewhere along the line. Decades ago, workplaces turned to glycol ethers for their efficiency—but overlooked health impacts. Today, government regulations subscribe to safety reviews, as EGEE, in large doses or with chronic exposure, can punch harder than expected against livers or kidneys.
Lots of people in manufacturing—myself included—learned early on to respect ventilation rules, gloves, and basic chemical hygiene. The ease with which EGEE crosses skin demands care, since it doesn’t just sit on the surface. Workers report headaches or dizziness if air monitoring gets ignored. OSHA and Europe’s REACH regulations encourage strict monitoring, safer alternatives, and training for handlers.
Improving Chemical Safety
Plenty of companies now swap in glycol ether blends with lower toxicity or improved biodegradability for certain uses. Substitutes only work if they handle the same tasks—dissolving, cleaning, thinning—without bumping up health risks or environmental persistence. Industry partnerships with universities keep pushing for safer solvents, often drawing on green chemistry innovation.
At home, the best advice means reading product labels and following manufacturer recommendations. Both users and regulators now expect full disclosure of ingredients, spill cleanup plans, and safe disposal routes to prevent groundwater and air contamination.
Staying informed, supporting research into new alternatives, and keeping a healthy respect for established safety protocols make all the difference—protecting workers, end users, and the larger world that sits beyond factory walls.
| Names | |
| Preferred IUPAC name | 2-ethoxyethan-1-ol |
| Other names |
2-Ethoxyethanol Ethyl cellosolve Glycol monoethyl ether Cellosolve EGE Ethylene glycol monoethyl ether |
| Pronunciation | /ˈɛθɪliːn ɡlaɪˈkɒl ˈiːθɪl ˈiːθər/ |
| Identifiers | |
| CAS Number | 110-80-5 |
| 3D model (JSmol) | `load =C(CO)OCC` |
| Beilstein Reference | 0623407 |
| ChEBI | CHEBI:45035 |
| ChEMBL | CHEMBL1338847 |
| ChemSpider | 7687 |
| DrugBank | DB14019 |
| ECHA InfoCard | 100.003.004 |
| EC Number | 203-804-1 |
| Gmelin Reference | 5768 |
| KEGG | C06508 |
| MeSH | D005005 |
| PubChem CID | 8008 |
| RTECS number | KK9275000 |
| UNII | FB6DM43UQT |
| UN number | UN1171 |
| CompTox Dashboard (EPA) | DTXSID7020375 |
| Properties | |
| Chemical formula | C4H10O2 |
| Molar mass | 134.18 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Mild, pleasant odor |
| Density | 0.930 g/cm3 |
| Solubility in water | Miscible |
| log P | -0.32 |
| Vapor pressure | 0.67 mmHg (20°C) |
| Acidity (pKa) | 14.6 |
| Basicity (pKb) | 7.64 |
| Magnetic susceptibility (χ) | -9.87×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.404-1.408 |
| Viscosity | Viscosity: 2.36 mPa·s (at 25 °C) |
| Dipole moment | 2.45 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 356.2 J/mol·K |
| Std enthalpy of formation (ΔfH⦵298) | -471.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3099.7 kJ/mol |
| Pharmacology | |
| ATC code | D07AX01 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes serious eye irritation. |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 50°C (122°F) |
| Autoignition temperature | 235 °C (455 °F; 508 K) |
| Explosive limits | 3% - 20% |
| Lethal dose or concentration | LD50 oral rat 2380 mg/kg |
| LD50 (median dose) | 2,000 mg/kg (rat, oral) |
| NIOSH | K160 |
| PEL (Permissible) | PEL: 100 ppm (parts per million) |
| REL (Recommended) | 18 mg/m³ |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Ethylene glycol Diethylene glycol Ethylene glycol methyl ether Diethylene glycol monomethyl ether Ethylene glycol butyl ether Propylene glycol Diethylene glycol ethyl ether |

