Methyl Pivalate: More Than Just a Chemical Name
Historical Development
Methyl pivalate, tucked away in the wide world of organic chemistry, has a backstory rooted in the efforts to make esters both simple to handle and reliable in reactions. Its discovery dovetailed with the rise of organic synthesis in the twentieth century when chemists started leaning into pivalic acid derivatives for their stability and straightforward behavior. The chemical structure comes out of a desire for molecules that don’t easily roll over in the face of strong acids or bases. The pivalate group’s steric bulk helped researchers shield more fragile parts of molecules, giving synthetic chemists a handy “bump” to control selectivity in reactions. Over the decades, methyl pivalate became more than a reference in handbooks; it played supporting roles from laboratory benches to industrial plants, especially where robustness and predictability mattered.
Product Overview
Every time I pick up a bottle of methyl pivalate in the lab, what stands out is that subtle, estery scent hinting at its makeup. It’s a colorless liquid, lightweight, and slips easily between glassware, echoing the properties of low molecular weight esters. There’s no fuss about it. Chemists often rely on methyl pivalate as an intermediate rather than a finished product, treating it like a reliable stepping stone. For students, this chemical often serves as a gateway to understanding larger concepts in ester synthesis and stability, emphasizing the importance of those two methyl groups flanking a central carbon — a feature that makes it a quirky cousin in the ester family.
Physical & Chemical Properties
With a boiling point in the range of 100-120°C at reduced pressure and a noticeable, almost ripe-apple fragrance, methyl pivalate flies under the radar when compared to some flashier organic solvents. Its low solubility in water stands out, which makes separation after reactions much easier and lends a simplicity to cleanup that I’ve always appreciated during experiments. The molecular structure, featuring a t-butyl group next to the carbonyl, resists many standard attacks by nucleophiles or bases. This resilience makes it a candidate for running reactions where less robust esters would simply break down, and I’ve seen this firsthand when comparing yields under basic conditions.
Technical Specifications & Labeling
Any laboratory bottle of methyl pivalate speaks the same core language: CAS number, chemical formula (C6H12O2), purity (often above 98%), and safety symbols reminding users to handle with care. Glass bottles tend to win out over plastics due to any risk of permeation, even if that’s mostly a concern for long-term storage. Experience has taught me to trust the sharp smell as an indicator of purity — if there’s an off-note, something’s wrong. Labels never get too busy; regulatory standards ask only for what matters, including hazard pictograms and GHS warnings about flammability and potential irritation. This keeps focus on safe handling and proper context for use.
Preparation Method
Making methyl pivalate in the lab doesn’t require elaborate equipment. Mixing pivalic acid with methanol and introducing an acid catalyst, usually sulfuric acid or sometimes a sulfonic acid resin, triggers esterification. Heating the mixture, then drawing off water as it forms, lets the reaction slide toward completion without much coaxing. In my experience, careful removal of water through distillation or with drying agents directly impacts yield. It’s one of those classic organic reactions where attention to detail pays dividends: stirring speed, temperature stability, and clean separation all count for something. Overarchingly, this synthesis method lines up with standard ester production seen across organic laboratories.
Chemical Reactions & Modifications
Methyl pivalate usually sits out the action when strong nucleophiles or bases are in play, mostly due to bulky groups crowding around the carbonyl. The tert-butyl group shields the reactive site, giving it a reputation for resisting transesterifications or hydrolysis unless harsher conditions step in. Acidic hydrolysis can crack it apart, returning pivalic acid and methanol, but high temperatures or catalysts speed this along. This stubbornness explains why synthetic chemists use methyl pivalate to ferry fragile pieces through tough reaction sequences. Derivatization brings opportunities too, swapping out the methyl group after hydrolysis or extending the carbon chain from the acid end, letting creative hands build new molecules off a proven backbone.
Synonyms & Product Names
Names bounce around — methyl 2,2-dimethylpropanoate, methyl trimethylacetate, methyl neopentanoate — but experienced chemists recognize them as shades of the same molecule. Each synonym helps with database searches or regulatory paperwork, but on the bench, they all lead to that clear, faintly fruity ester. This cross-naming sometimes creates confusion for those new to chemical catalogs; knowing the synonyms saves time and keeps research moving forward, especially during literature reviews or ordering supplies.
Safety & Operational Standards
Any chemical with a volatile nature and strong odor brings a few safety lessons. Inhaling vapors can irritate the respiratory system, and accidental splashes sting the skin. Even though methyl pivalate doesn’t come with a high toxicity profile, the simple rules of glove use, eye protection, and good ventilation still matter. A spill in a warm room can turn into a minor mess, so access to spill kits and absorbent pads is key. From my time teaching undergraduate labs, I’ve found students often underestimate the risks, thinking that familiar esters are harmless. I always point out the need for chemical fume hoods and secure storage away from open flames.
Application Area
Methyl pivalate earns its keep mostly as an intermediate. Pharmaceutical synthesis and the production of specialty chemicals tap into its sturdy nature, using it as a protective group or a stable linker. Where more reactive esters buckle, methyl pivalate stands firm during multi-step reactions, making it useful in synthesizing complex molecules, especially those requiring selective protection. Its fragrance also finds a home in the world of flavor and fragrance manufacturing, lending a crisp top note to prepared scents, though use in consumer goods stays limited due to flammability and regulatory questions. In my own projects, I’ve seen its role as a trusted sidekick, helping shepherd sensitive intermediates through rough synthetic waters.
Research & Development
R&D teams explore how methyl pivalate can work in tougher conditions or how its backbone might inspire new, more resilient esters. Studies dig into tweaking the ester’s structure, changing either end to tune reactivity or physical behavior. Fine chemicals firms aim to improve synthesis efficiency by finding greener catalysts or more renewable sources for pivalic acid and methanol. I’ve noticed researchers probe whether similar bulky esters can further boost selectivity in cutting-edge pharmaceutical applications. These efforts highlight an ongoing drive to raise yields, shrink process waste, and avoid toxic reagents, supporting both economic and environmental goals.
Toxicity Research
Existing toxicity data for methyl pivalate don’t raise alarm bells. Acute exposure generally produces mild symptoms, mostly because of the compound’s volatility rather than inherent toxicity. Chronic studies remain sparse, but regulators watch for cumulative exposure or accidental ingestion, especially in manufacturing settings. Recent research checks metabolic breakdown, which leads to harmless pivalic acid excretion in most mammals. Even so, regulations keep limits clear to avoid overexposure, especially for workers handling drums or reactors. Regular monitoring and proactive ventilation continue to serve as best practices, grounded in a history of chemical safety campaigns rather than an urgent health risk.
Future Prospects
As organic synthesis keeps pushing boundaries, especially in pharmaceuticals and advanced polymers, methyl pivalate’s stability and simplicity give it staying power. There’s growing talk about using renewable feedstocks to make both methanol and pivalic acid, closing the loop for more sustainable ester production. Automation and data-driven lab management look set to drive efficiencies, with compounds like methyl pivalate acting as benchmarks for evaluating new equipment or methods. Looking to the future, I see its role shifting only slightly, serving as a reliable standard while new, more specialized esters emerge for demanding reactions. Well-chosen modifications off its framework could offer even more selectivity or environmental friendliness, rewarding both researchers and industry with every round of iteration. In an age looking for greener, safer, and more adaptable chemistry, compounds with proven track records still have important positions to fill.
What is Methyl Pivalate used for?
What is Methyl Pivalate?
Methyl pivalate pops up in conversations about organic chemistry and manufacturing, though most people don’t give it a second thought. As a chemical compound, it features a fruity smell, which often surprises folks when they discover it’s not made for perfumes or food. This substance plays a part behind the scenes in industries that touch everyday items and advanced materials alike.
Real World Applications
Methyl pivalate finds its way into laboratories as an intermediate — a stepping stone for chemical reactions. In my time spent reviewing safety data sheets and product formulations, I’ve come across this compound most often in the context of organic synthesis. Researchers and chemical engineers use it to unlock new molecules for pharmaceuticals, agrochemicals, and specialty plasticizers. That pleasant odor doesn’t hint at how crucial it becomes in assembling drugs that treat anything from infections to rare diseases.
This compound serves in the formation of pivalic acid derivatives. For instance, certain antibiotics come from processes involving methyl pivalate. Finding reliable intermediate compounds holds the key to effective, safe, and scalable drug manufacturing. The story doesn’t stop with medicines. Pesticides and herbicides also need building blocks. A robust supply of quality intermediates like methyl pivalate helps agrochemical producers respond to the global demand for food production while managing costs and regulatory hurdles.
Why Quality Control Matters
Safety always trumps convenience, especially with chemicals. Through the years, supply chains have tightened controls to prevent contamination and mistaken identity between similar-looking substances. Methyl pivalate, like many organic esters, poses real risks if handled carelessly. Exposure may irritate the skin or eyes, so workers need proper training and protective gear. Responsible companies maintain traceable documentation and standardized handling procedures. This approach keeps operations compliant with health and environment standards, reducing workplace accidents and product recalls.
I’ve spoken to chemists who recall near-misses—mixing up esters or failing to label containers—causing delays and added costs. Manufacturers who invest in reliable supply networks ensure smooth production runs and fewer interruptions. Quality assurance programs have a huge impact on everything downstream, from the price consumers pay for medications to the economic health of the chemical sector.
Looking Forward: Safer and Greener Solutions
Sustainability concerns shadow every step in the chemical supply chain. Traditional methods for making methyl pivalate rely on fossil raw materials and energy-intensive processes. By investing in greener chemistry—using renewable feedstocks and lowering overall emissions—companies can stay ahead of regulations and public demand for cleaner products. I’ve read case studies showing incremental improvements: recycling solvents, recovering heat, switching to biobased alcohol inputs. Small tweaks add up over years, leading to lower production costs and a smaller environmental footprint.
In summary, methyl pivalate plays a supporting role in crafting many products vital to modern life. Paying attention to how it’s produced and used ensures safer workplaces, more accessible medicines, and greater environmental protection for communities down the line.
What is the chemical formula of Methyl Pivalate?
Understanding the Backbone: The Formula and Structure
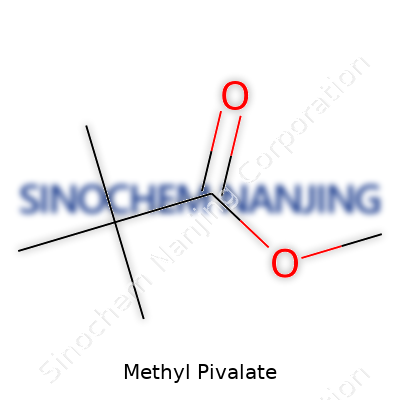
Methyl pivalate comes down to a pretty straightforward chemical formula: C6H12O2. That simple arrangement brings together carbon, hydrogen, and oxygen in a way that points to its role as an ester. Esters like this stand as the result of combining an acid and an alcohol—in this case, pivalic acid and methanol. The methyl group gets attached to pivalate via oxygen, locking in its identity within organic chemistry. Its structure (with the full name methyl 2,2-dimethylpropanoate) packs significant bulk, thanks to the three methyl groups clustering on one side.
Why Methyl Pivalate Matters
As a writer with a background in chemical engineering, I've come across methyl pivalate in both academic labs and manufacturing floors. It holds its own in fragrances, solvents, and even in synthetic flavors. Its fruity, apples-and-pears scent brings value for perfumers looking for something that lasts, since the bulky tert-butyl group won’t easily let go of the molecule, keeping the aroma steadier over time.
Looking at the facts, esters like methyl pivalate commonly show up where predictable volatility and relative safety are needed. It brings lower toxicity than many other organic solvents, so labs prefer it when trying to minimize unnecessary risks. Handling it requires the usual chemical safety savvy: using gloves, goggles, working in good ventilation. It’s not the kind of substance to drink or pour down the drain, being mildly irritating to skin and eyes.
Production Realities and Challenges
I’ve seen methyl pivalate made through straightforward Fischer esterification. Toss methanol and pivalic acid in a flask, add some acid catalyst, and coax them together at higher temperatures. From a production standpoint, waste management matters. Acidic residues, leftover methanol, and water byproducts can gum up a process if facilities lack the right storage or treatment.
There’s an environmental layer worth talking about here. As with all esters, small spills evaporate fast, so air quality and proper ventilation come into play. The chemical breaks down quickly in sunlight, limiting large-scale persistence, but still calls for responsible disposal practices. Workers and managers need clear safety sheets, spill protocols, and backup containment.
What Could Make Handling and Use More Sustainable?
In my experience, training employees beyond the basics makes a difference. Not every worker comes in with a chemistry background; straightforward guidance on methyl pivalate’s hazards versus its uses can mean fewer mistakes on the job. Partnerships with local hazardous waste processors turn what could be landfill-bound material into reusable chemical feedstocks or cleaner incineration. Streamlining risk assessments and container labeling systems has sped up audits and improved compliance where I’ve worked.
On the research side, keeping an eye on green chemistry methods pays off. Swapping out traditional acid catalysts for solid acids or enzymes has been piloted in a few facilities, which slashes mixed waste. Labs experimenting with continuous flow systems create less byproduct than batch processes. While the cost remains a factor for many smaller operations, the growing toolkit of cleaner synthesis offers hope for better balance between cost and sustainability.
Supporting Safe and Smart Use
Methyl pivalate highlights the importance of understanding what’s in use, where it ends up, and who handles it. With C6H12O2, simple structure supports a surprising range of flavors, fragrances, and chemical products—and with thoughtful management, its benefits far outweigh its risks.
Is Methyl Pivalate hazardous or toxic?
Understanding the Risks
When a chemical like methyl pivalate comes up in research or manufacturing, most people outside chemistry circles probably wouldn’t blink. It’s not a household name. Yet, many folks working in industries—whether in laboratories or plants—run across it as a reagent or solvent. If you’re not a chemist, the main questions are pretty straightforward: does it harm you, and what does the science say?
What Science Tells Us About Toxicity
Like many organic esters, methyl pivalate slips under the radar in popular discussion. Some folks might look at the faintly fruity odor and clear appearance and assume it’s basically harmless. That instinct gets plenty of people in trouble. Studies show this compound can cause irritation if it contacts skin or eyes. Vapors may pose risk to the respiratory tract in poorly ventilated areas. The U.S. National Library of Medicine lists it as a mild skin and eye irritant, and Safety Data Sheets back that up.
Animal testing hasn’t found methyl pivalate to be acutely toxic at low doses. That doesn’t mean it’s risk-free. Inhalation, especially at higher concentrations, will hurt the throat and possibly lungs. Anyone with allergies or asthma might feel symptoms sooner than others. Chronic exposure data remains sketchy, but we know enough to say it doesn’t belong in drinking water or food. I’ve seen gloves left off and goggles skipped, and I’ve also seen the rashes and red eyes that follow. Even if a compound isn't listed as carcinogenic or deadly, complacency never works in the lab.
The Hidden Trouble—Ease of Ignition
No conversation about chemical hazards feels complete without talking about flammability. Methyl pivalate takes a spark quickly. Its flash point sits below room temperature, so even a summer day can make vapors build up. Improper storage or handling creates recipe for disaster, something experienced chemists have learned through close calls. Left near heat, this liquid forms vapors that can ignite and escalate a small accident into a major one. Fire departments worry less about the chemical’s toxicity and more about explosions and burns.
Handling and Real-World Safety Choices
Most laboratories have rules: gloves, goggles, fume hoods, good ventilation. They aren’t just for show. Neglect leaves workers exposed to chemical burns or lung irritation. At home, folks sometimes order chemicals online for hobbies without really understanding the risks. Without industry-grade ventilation, the risks climb. I’ve walked into garage labs—best intentions, poor airflow, methyl pivalate open on the bench. The room smells sweet, but the headache comes quick.
For industry, the right approach means treating methyl pivalate like any flammable, irritant liquid. Store it away from flames and oxidizers. Use gloves and goggles, not only for accidents but also for everyday work. Industry studies never find short-cuts to be worth the risk; a moment of protection avoids days of missed work and medical bills. Even at home, small quantities should get locked up and opened only with windows open or outdoors.
Steps Toward Safer Practice
No chemical achieves a perfect safety record, but companies, labs, and individuals can cut risks. Investing in good storage, training, and ventilation makes handling methyl pivalate much safer. Manufacturers and suppliers have a duty to share clear labels and data sheets. As people become more aware of chemical safety, the chance of injuries sink. At the end of the day, a few careful choices make sure science and industry keep moving forward, without sacrificing worker health or safety.
What are the storage requirements for Methyl Pivalate?
Understanding the Realities of Safe Storage
Methyl pivalate turns up in labs and factories as a colorless liquid, and a lot of folks might not know it’s got a low flash point—about 25°C—which signals some fire risk. That’s something you can’t just gloss over. Direct sunlight or warmth from a nearby heat source takes this compound from “inert” to “hazard” quicker than people expect. In my own time spent around similar esters, I’ve seen how an overlooked shelf near a sunny window turns into a problem at the worst moment. Companies with strong safety cultures tend to lock down their flammable stocks far from steam lines and HVAC vents for a reason.
Why Packaging Matters
A big worry with methyl pivalate isn’t only about heat. It evaporates into a sharp-smelling vapor. That vapor can hit eyes and airways, especially in a stuffy room. My stint in manufacturing taught me that the cheapest fix—sturdy, tightly sealed, chemical-resistant jugs—pays off in the long run. Polyethylene or glass containers hold up well against leaks and don’t react with the ester. Skipping this step might save a few bucks at first but often causes expensive headaches if the liquid seeps out and taints other supplies. Regulatory bodies, like OSHA, make a point of flagging improper storage for chemicals like this.
Labeling and Ventilation
I’ve walked into storage rooms where identical bottles sat shoulder-to-shoulder, one marked and one not. That lack of labeling blurs the line between safe and sorry. Every container needs a clear, permanent label showing what’s inside, the date it arrived, and hazard warnings. That way, even kids straight out of school can check and get it right. As for the air, strong ventilation doesn’t just help people breathe easier—it keeps vapor levels low so nobody gets a surprise headache or worse.
Fire Protection and Spill Control
Some people just stick chemicals on a regular shelf and hope for the best. That approach fails once a fire breaks out nearby—or someone knocks over a jug. I’ve seen companies use flame-resistant cabinets to cut risks way down. This isn’t paranoia; it’s smart management. Keeping a spill kit close by, stocked with absorbent pads and neutralizing agents, helps stop accidents from becoming disasters. Workers need training, not just written rules they never read.
Legal and Environmental Pressure
Handling chemicals these days comes under plenty of scrutiny. Enforcement from local authorities on chemical storage has only grown sharper, especially after the headlines from warehouses gone wrong. I remember a time when an audit inspector showed up unannounced and walked straight to the chemical cabinet. Safe handling was the main focus. Even the community around an industrial site watches for any hint of leaks or “chemical smells” that drift their way. Mishandling methyl pivalate, or any similar solvent, only grows risk for everyone, not just the ones at the bench.
Looking Ahead: Simple Steps for Safer Storage
Turning storage rules into everyday habits lowers risk. Cool, dry, well-ventilated spaces, away from flames or sparks, keep methyl pivalate safer than any high-tech alarm. Tight seals, good labeling, and a basic foam extinguisher nearby make all the difference. Workers who know what’s in the room and how to act in a pinch help companies avoid fines, accidents, and nervous neighbors. Chemistry’s never just about reactions in a beaker—the real trick comes in handling what you can’t always see.
Where can I purchase Methyl Pivalate?
Looking for Methyl Pivalate?
Methyl pivalate shows up in plenty of labs, from organic chemistry classes to fragrance makers. People outside those groups might never hear its name. For researchers, finding a trusted place to buy this chemical can be a headache. Years back, working as a graduate student, I recall standing in front of a stockroom with a purchase order, facing a shelf full of unfamiliar bottles and slightly intimidating warning labels. Safety comes first: not every shop or online store checks if buyers know the rules around proper handling and legal purchases.
Who Sells It?
Suppliers like Sigma-Aldrich, TCI Chemicals, and Alfa Aesar stock methyl pivalate. They ship to universities, companies, and labs across the globe. It is easy to find product pages with purity grades, documentation, and shipping options. They stand out with detailed Certificates of Analysis and safety data. Not every seller offers that.
Most chemical suppliers expect buyers to prove some sort of professional or institutional connection before sale. A registered business, a university lab, or an authorized institution often satisfies this requirement. Some even go so far as to ask for a statement of use or a copy of a project proposal. That keeps chemicals out of unsafe hands, but can frustrate hobbyists with legitimate projects. Hobbyists sometimes turn to general marketplaces, believing it streamlines the process. Yet here’s the catch: quality and safety drop. I have seen containers with faded labels or no documentation from gray-market listings. That feels like gambling on purity, and no experiment—let alone personal safety—deserves that risk.
Rules Around Buying
Plenty of people want to know why chemicals don’t just ship like any other product. Laws, liability, and safety concerns shape those rules. Methyl pivalate isn’t considered the most dangerous substance you can buy, but even “low-to-moderate hazard” solvents can burn skin, pollute water, and release fumes. In some countries, customs agents block shipments without the right paperwork. In the United States, chemical suppliers and shippers follow rules from the EPA and DOT. Europe sticks to REACH registration. Australia and Canada have their own frameworks.
Supporting Good Use and Safe Access
Buying through established vendors remains the safest path. Their sales teams walk you through any compliance paperwork, answer questions about storage, and supply guidance if disposal becomes necessary. Recently, a friend’s small business contacted a supplier about a bulk order for fragrance research. The process proved slower than a simple online cart checkout, but brought peace of mind with real-time safety advice and assurance of quality.
More transparency and guidance would help. Vendors could lay out clearer rules on their websites—who qualifies, what documents work, which countries face restrictions. Educational steps, detailed safety sheets, and up-front support cut down on confusion and wasted time. I learned quickly that patient, honest questions with reputable suppliers get better results than chasing fast delivery from unreliable sources.
Getting It Right
The world of chemistry depends on trust, safety, and knowledge. Following those leads buyers to sellers who treat methyl pivalate and other specialty chemicals with respect. That protects people, the planet, and honest progress in research and industry.
| Names | |
| Preferred IUPAC name | methyl 2,2-dimethylpropanoate |
| Other names |
Pivalic acid methyl ester Methyl trimethylacetate Methyl neopentanoate 2,2-Dimethylpropanoic acid methyl ester |
| Pronunciation | /ˈmɛθɪl pɪˈveɪleɪt/ |
| Identifiers | |
| CAS Number | 554-12-1 |
| 3D model (JSmol) | `COC(=O)C(C)(C)C` |
| Beilstein Reference | 1719477 |
| ChEBI | CHEBI:17841 |
| ChEMBL | CHEMBL424324 |
| ChemSpider | 54686 |
| DrugBank | DB14102 |
| ECHA InfoCard | ECHA InfoCard: 100.008.463 |
| EC Number | 203-310-6 |
| Gmelin Reference | 108605 |
| KEGG | C19157 |
| MeSH | D014959 |
| PubChem CID | 8056 |
| RTECS number | AY7850000 |
| UNII | 863P5V17CO |
| UN number | UN1993 |
| Properties | |
| Chemical formula | C6H12O2 |
| Molar mass | 116.16 g/mol |
| Appearance | Colorless liquid |
| Odor | Fruity |
| Density | 0.895 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 1.82 |
| Vapor pressure | 3.1 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 25 |
| Magnetic susceptibility (χ) | -8.59 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.387 |
| Viscosity | 0.754 mPa·s (25 °C) |
| Dipole moment | 1.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 223.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -426.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3017.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P301+P310, P303+P361+P353, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 72°C |
| Autoignition temperature | 420 °C |
| Lethal dose or concentration | LD50 oral rat 4,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 7,010 mg/kg |
| NIOSH | SKH07250 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Methyl Pivalate: Not established |
| REL (Recommended) | 100 ppm |
| Related compounds | |
| Related compounds |
Pivalic acid Neopentyl alcohol Methyl acetate Methyl esters Ethyl pivalate |

