Ethyl Lactate: Charting the Journey from Byproduct to Modern Green Solvent
Historical Development
Ethyl lactate didn’t rise to attention overnight. For a long time, many saw it as little more than a niche flavor additive, tucked behind synthetic cleaners and classic esters. Years ago, manufacturers just tossed aside byproducts in corn and beet processing without much thought. Fermentation technology changed the game, as folks realized lactic acid and its derivatives could be coaxed out of renewable crops. The late twentieth century brought mounting pressure to swap petroleum-based chemicals for green alternatives. This stirred up innovation, and ethyl lactate caught industry eyes for its clean pedigree and low toxicity. The environmental movement, which picked up speed in the 1980s and 1990s, kept pushing the solvent market to look past tradition. Ethyl lactate steadily earned more attention, growing beyond food flavors and fragrances, heading straight into labs and cleaning tanks across industries that wanted less hazardous waste.
Product Overview
Anybody that’s poured or sniffed ethyl lactate can spot its lightly sweet aroma—reminds some people of butter, maybe a faint note of fruit. That’s not surprising, since it’s an ester made from lactic acid and ethanol, both of which tie right back to basic fermentation roots. Unlike many industrial chemicals, ethyl lactate gives off fewer hazardous fumes, and it breaks down easily in the environment. You’ll find it as a nearly colorless liquid, not sticky or oily. Folks use it to dissolve inks, strip coatings, and even in specialty pharmaceuticals. Tech labs like it for dissolving polymers, especially where traditional acetates or chlorinated solvents create headaches over air quality or residue.
Physical & Chemical Properties
Ethyl lactate is a relatively small molecule, with the chemical formula C5H10O3. It holds together well in storage and doesn’t evaporate super fast at room temperature—its boiling point sits around 154°C. Water can blend with it, though not in all quantities. One key aspect: it won’t ignite as easily as many other solvents, so handling is less hair-raising. Chemists appreciate that ethyl lactate doesn’t leach much water from the air, and it resists picking up outside contaminants, so once sealed, it stays fairly stable. Its mild odor and low vapor pressure make it a quieter presence on the shop floor than aggressive chemicals like acetone or toluene.
Technical Specifications & Labeling
Lawmakers and regulatory agencies came around slowly. Eventually, standards set clear ground rules on purity, residue levels, and allowable trace elements. Typical industrial batches run above 98 percent purity. Labels on drums and bottles need to carry UN numbers and chemical names, but they look a lot less alarming than the skull-and-crossbones of more caustic solvents. Workers don gloves, goggles, and sometimes a mask, especially during big pouring or mixing jobs. Shipping requires tight sealing, but you won’t see specialized armored containers like those used for more dangerous liquids.
Preparation Method
Producing ethyl lactate takes a mix of fermentation and chemical finesse. Lactic acid first comes to life thanks to bacteria munching on carbohydrates—corn, potatoes, and sugar beets often headline the fermenter. Producers then neutralize the acid, isolate it, and react it with ethanol in the presence of a strong acid catalyst. This esterification pushes out water and builds the final product, which just needs careful distillation to purify. Modern factories love continuous processes, since they can recapture waste heat and recycle leftover ethanol, trimming costs and waste. Batch methods, though, still show up when smaller volumes or special grades land on order.
Chemical Reactions & Modifications
In the lab, ethyl lactate serves as both a reagent and a solvent. Chemists value its readiness to play in transesterification reactions. Heating with alcohols or acids encourages these swaps, opening the door to custom-tailored esters. Reducing agents chop it down into smaller alcohols, while the right oxidizers strip off electrons and morph it into new acids. Heat and strong bases eventually break it back to lactic acid and ethanol, which makes it more recyclable than tougher petroleum solvents. Folks pushing boundaries in polymer science find ethyl lactate helpful for dissolving bioplastics and testing new biodegradable blends—something you won’t easily wring out of traditional oil-based solvents.
Synonyms & Product Names
While most folks in the lab or plant stick to “ethyl lactate,” some materials scientists and food technologists might call it “ethyl 2-hydroxypropionate.” European suppliers sometimes list it as “lactic acid, ethyl ester.” Shorthand like “EL” pops up in trade literature. Branding wavers a bit, but chemical catalogs keep it straightforward, listing IUPAC names and CAS numbers for reference. Walk into a paint shop or printing supply warehouse, and you may see eco-solvent blends with ethyl lactate buried among the ingredients, highlighting its behind-the-scenes role.
Safety & Operational Standards
The track record for worker safety with ethyl lactate stands out among many solvents. Risks unwind mostly for people who breathe in big clouds of vapors or let it soak on unprotected skin. Blisters or short-lived rashes sometimes show up with sloppy handling, but the days of handling it with nothing but bare hands are long gone. Ventilation, gloves, and splash-proof goggles make up basic precautions on factory floors. Spills wash down with water and don’t spark fires—in many plants, that’s a relief compared to handling acetone, MEK, or similar compounds. Regulatory guidelines from the EPA and European Chemical Agency ask manufacturers to cap emissions and keep waste from hitting waterways, but these targets land within reach thanks to rapid biodegradation and low volatility. It’s not a chemical to take lightly, yet the margin of safety runs wide in comparison to petrochemical alternatives.
Application Area
Industries buying barrels of ethyl lactate cover a broad map. Printers turn to it for cleaning ink rolls and thinning out tough, pigment-heavy inks. Recovery labs and electronics manufacturers appreciate its edge for washing residues off circuit boards without tarnishing sensitive metals. Paint and coatings firms use it as a swap-in for tougher solvents, where it smooths surfaces and shortens drying time without filling air with hazardous fumes. Agrochemical labs find ethyl lactate effective for blending and spraying pesticide formulations—its low toxicity matters when people handle crops or soil directly. In pharmaceuticals, the push for “green chemistry” led formulators to use ethyl lactate for dissolving active ingredients during tablet coating or encapsulation steps, where regulatory agencies pay extra attention to solvent residues. Even the food and flavor industries keep a place for it; a dash of ethyl lactate brings subtle creaminess to dairy flavors and blends into bakery items.
Research & Development
Academic and industrial chemistry teams publish papers exploring new ways to stretch ethyl lactate’s usefulness. Catalysis researchers believe this green solvent might edge out harsher esters in fine-tuning reaction selectivity. Polymer researchers found that it softens some biodegradable plastics better than ethanol or acetone, leading to prototypes for new single-use packaging. Biorefinery engineers consider ethyl lactate production as a model for value-added chemicals: turning field crops, even crop waste, into high-purity solvents for cleaning and formulation. Toxicologists still run tests to confirm where ethyl lactate’s limits sit, but few flagged issues pop up at permitted exposure levels. Farmers and consumer product companies press for greener cleaning and degreasing alternatives, betting that ethyl lactate’s low residue and natural degradation win over skeptical end-users.
Toxicity Research
Toxicological data stacks up favorably compared to legacy solvents. Lab animal studies run by university labs and government health agencies mostly show low acute toxicity. Swallowing modest amounts or breathing in brief vapor clouds rarely triggers serious problems. Longer-term effects look manageable under responsible factory conditions, though direct, high-dose exposure can irritate eyes and airways. Once inside the body, ethyl lactate metabolizes into lactic acid and ethanol—compounds the body recognizes and can process. Regulators monitor workplace exposure levels, keeping them well below the threshold for harm. This low-risk profile lets regulatory agencies approve it for multiple uses, from food and feed flavorings to pharmaceutical excipients. Open questions linger about chronic high-level exposures, but the trend leans toward favorability.
Future Prospects
Looking ahead, demand for ethyl lactate keeps rising as industries eye decarbonization and safer workspaces. The push for closed-loop, plant-based chemical cycles fuels research into cheaper, cleaner fermentation and purification routes. Biorefineries weigh the merits of producing ethyl lactate from crop waste, promising lower greenhouse gas emissions than routes tied to corn syrup or beet sugar. In the coming years, advanced catalysts will probably boost yields, while water and energy use shrink further. Policy shifts, especially in the EU and North America, create more incentives for low-toxicity, easily degradable solvents—a slot that fits ethyl lactate well. Additive manufacturers, 3D printing startups, and specialty paint brands all hunt for safer, greener solvents that keep up with demanding specs. Ethyl lactate won’t edge out all competitors, but its flexibility and safety record give it a prime seat as industries shift toward greener chemistry for the long haul.
What is Ethyl Lactate used for?
Meeting Solvent Needs Without Harming the Planet
Ethyl lactate comes from simple sources like corn or sugar and shows up in more places than most people realize. It works as a solvent in industries ranging from electronics to food. My interest in green chemistry first brought this substance to my attention. Standard solvents often come with harsh warnings and environmental baggage, but ethyl lactate gives people a way to clean parts or extract flavors with a lighter footprint. While covering a manufacturing story last year, I saw technicians switch from volatile chemicals to this bio-based option. They said clean-up work became safer, with fewer odors and less skin irritation.
Food and Beverage: Quietly Behind the Scenes
Few people think about what touches their food before it hits the table. Yet, ethyl lactate makes a behind-the-scenes difference as a flavor carrier and extraction tool. It's found in vanilla extract, which many home bakers cherish. Regulatory lists class it as a food-safe solvent, so companies use it to pull flavors from fruits or spices. Knowing that my child's favorite yogurt flavor might rely on such chemistry helps me trust the production process. This isn’t just about taste—in studies, ethyl lactate shows lower toxicity compared to many common food solvents.
Electronics and Personal Care: Cleaning Without Compromise
Electronic circuit makers often face hurdles balancing performance with safety. Solder flux residue removal used to involve tough chemicals. Now, several plants use ethyl lactate for cleaning delicate microchips. Colleagues working in electronics have praised how well it removes residues without damaging sensitive components. Some personal care products also lean on this solvent. Perfume companies and cosmetic labs include it in formulas because it helps dissolve scents and acts as a carrier without triggering major skin reactions for most users. This broader adoption hints at a shift toward ingredients that don’t cause as many problems downstream.
Printing and Paints: Lowering Risk, Raising Standards
The printing trade once relied heavily on petroleum solvents. Talking with an old friend who runs a small press shop, I learned how switching to ethyl lactate cut ventilation system costs and helped employees breathe easier. In paint stripping and coatings, manufacturers appreciate how it dissolves stubborn pigments and oils. Several published studies detail its role in reducing volatile organic compound emissions, which helps urban air quality. I’ve visited machine workshops where they switched for this reason, not just for their own health but for neighbors complaining about odors and pollution.
Challenges and Change in Adoption
No product arrives without caveats. Ethyl lactate may cost more, especially if market demand rises. Some companies hesitate because supply depends on crops, which means price swings and availability shifts. Yet momentum grows as governments tighten rules on air toxins and workplace exposure. Innovation follows regulation, and the chemistry behind ethyl lactate keeps improving. Companies seeking safer workplaces and greener reputations look for alternatives, and this solvent stands out as one that has proven its worth across many corners of industry and daily life.
Is Ethyl Lactate safe for humans?
Understanding Ethyl Lactate
Ethyl lactate comes from the combination of lactic acid and ethanol—two substances already found in the human body and many foods. It smells faintly like fruit, mixes easily with water, and dissolves grease as powerfully as some harsh chemical cleaners. This makes it popular in industries aiming to swap petroleum-based solvents for something friendlier.
Safety in Everyday Use
You might touch or breathe ethyl lactate at work if you use paints, coatings, or cleaners. The chemical also shows up as a food additive and is even present in some flavors. Any talk about safety starts with how much one encounters. Data from the US Food and Drug Administration (FDA) lists ethyl lactate as "generally recognized as safe" (GRAS) for use in foods. At low levels, dangers stay far away.
In my experience working with natural solvents, most people who work with ethyl lactate don’t run into trouble with everyday exposure. Spilling some on skin can produce mild irritation, especially with repeated contact over months or years. It evaporates quickly and the body breaks it down to harmless end-products, so occasional small amounts rarely bother healthy adults.
Lab Results and Real-World Cases
Animal studies back this up. Researchers gave high doses to rodents and noted only minor issues, usually at levels much higher than what people face even in industrial settings. Safety data sheets point out possible eye or skin irritation, but don’t list life-threatening outcomes. Agencies like the Environmental Protection Agency (EPA) suggest the risk in most occupational or consumer settings remains low.
At the same time, not everyone has the same reaction. Workers with sensitive skin or asthma might complain of rash or mild breathing problems if exposed daily in unventilated rooms. Wearing gloves, goggles, and keeping good airflow goes a long way toward making handling safe for those at risk.
Green Chemistry in Action
Ethyl lactate’s strong point lies in how it lets factories switch away from more toxic options. Traditional cleaning and printing chemicals like toluene or acetone bring much greater health risks after repeated exposure, including headaches, nerve problems, and sometimes cancer. Swapping to ethyl lactate helps companies protect both employees and the environment without giving up cleaning power.
For those who want a more sustainable cleaning agent at home, checking for ethyl lactate on product labels is one small way to reduce chemical load in kitchens and garages. Not all “green” products deliver on safety, but ethyl lactate carries a solid track record with regulatory approval to support it.
What Still Needs Watching
No chemical is risk-free. Children, pregnant women, and people with allergies do best with limited exposure to all solvents. Industry also needs to keep monitoring for rare reactions in sensitive groups instead of assuming “natural” always equals harmless. Inhaling high concentrations, even of gentler compounds, may dry out airways or trigger headaches.
Shifting industry toward less toxic options counts for progress, though. Ethyl lactate manages to balance cleaning strength and safety better than many older solvents. Meeting high safety standards doesn’t mean skipping good hand washing or ventilation, but it does highlight how much safer solutions keep showing up as innovation and health science mix.
What are the main applications of Ethyl Lactate?
A Strong Choice for Green Chemistry
Ethyl lactate stands out as a cleaner solvent in an industry packed with harsh chemicals. Sourced from renewable materials like corn and sugarcane, it appeals to those who pay attention to their environmental footprint. For decades, folks working in laboratories have favored ethyl lactate’s less toxic reputation. Regular solvents leave behind residues that need special disposal, but ethyl lactate breaks down easily. This move towards green chemistry makes a difference for both workplace safety and environmental health.
Cleaning Up in Industry
Anyone who has worked with machinery covered in grease knows how hard it is to find something that gets the job done without ruining your hands or the environment. Ethyl lactate steps up as a degreaser for electronics, precision metal parts, and even circuit boards. Many electronics shops, faced with regulations on volatile organic compounds, switched to ethyl lactate and saw workplace air improve. I’ve talked to maintenance staff who appreciate that it cuts through oil without burning their skin or leaving behind a harsh smell. Instead of a clean-up job that brings headaches and rashes, the switch brings peace of mind.
Helping Paints and Coatings Go Green
The paint aisle looks different from what it did years ago. Rules about air pollution forced paint companies to hunt for safer solvents. Ethyl lactate offers the strength to dissolve resins for coatings and inks. Printers, especially those working on food packaging, look for solvents that won’t leave behind toxic traces. Ethyl lactate breaks down quickly and doesn’t add extra risk. I’ve seen printers switch to this solvent and reduce their hazardous waste. Production lines keep moving, but the old chemical headaches fade away.
Food and Flavor: A Gentle Touch
Ethyl lactate pops up in food processing because of its gentle nature. It pulls flavors from fruits during extraction. Food scientists value it since it won’t linger in finished products or warp the taste. As a bonus, its safety record keeps it on the shortlist for many natural flavor recipes. Chefs fiddle with ethyl lactate behind the scenes, knowing that it gives them clean flavors without risk to their customers.
Pharmaceutical and Medical Applications
Hospitals and drug makers work under strict rules for safety. Ethyl lactate gets picked to purify drugs and aid in drug delivery. It dissolves active ingredients smoothly and then leaves no residue. In wound cleaners and other topical products, people trust it not to irritate the skin. Anyone who’s worked in a pharmacy can tell you about the endless hunt for safer additives. With its track record, ethyl lactate proves itself as more than just another solvent—it's a safer path amid a world of complex chemicals.
Clearing the Way Forward
The push for safer, sustainable solutions keeps growing. Ethyl lactate sets an example for how companies can balance effectiveness with a lighter impact on people and the planet. Governments and industry groups can step up by funding more research into bio-based solvents and offering support for companies making the transition. By sharing what works and what doesn’t, the road to a safer workplace and a cleaner environment doesn’t have to be a lonely or expensive one.
How should Ethyl Lactate be stored?
Understanding the Risks
Ethyl lactate, a solvent often chosen for its greener profile and versatility, demands some respect in storage. I came across this chemical in a lab that prioritized eco-friendly products, and even though its reputation seemed harmless compared to harsher alternatives, ignoring safety with any chemical can lead to headaches. While ethyl lactate breaks down to lactic acid and ethanol, both familiar to most people, in the concentrated form it can still irritate skin and eyes and is flammable. OSHA notes its flash point around 46°C (115°F), which means ordinary room temperatures can sometimes approach risky territory, especially in a poorly ventilated storeroom in midsummer.
Picking the Right Spot
Many storage disasters start with a rushed decision. A bottle left in sunlight on a windowsill, for instance, can degrade and build up pressure. For ethyl lactate, a cool, dry, and well-ventilated place without any direct sunlight works. Keeping chemicals at a temperature below 25°C lowers risk, especially if the local climate has a habit of spiking. Shelves in a chemical storeroom should offer stability—nobody wants broken glass and spilled solvent kicking off an evacuation.
The Importance of Proper Containers
People sometimes think saving on containers is harmless, but cheaper plastic can break down, especially after repeated use or exposure to chemical fumes. Ethyl lactate comes in glass or stable HDPE plastic bottles for a reason. Containers need tight-fitting lids; air exposure not only invites contamination but can kick off evaporation, costing money and safety.
Labeling every container
I’ve seen confusion lead to mistakes that bring labs grinding to a halt. A missing or fading label, especially after a bottle transfer, means guessing games nobody enjoys. Date received, date opened, and full product name help anyone stepping into the store room. Clear hazard symbols make risks obvious—fire pictograms, irritant warnings—so routine checks never become rushed glances.
Keeping Separate from Incompatibles
Mixing unknowns is an invitation for dangerous results. Ethyl lactate should not share shelf space with acids, oxidizers, or strong bases. The idea isn’t paranoia; it’s past experience. Spillages or leaks can lead to toxic fumes or fires, even if the volumes seem trivial. Segregating solvents, acids, and water-reactives has saved more than one person from a close call.
Planning for Spills
Even tidy people have spills, especially in busy workspaces. Absorbent spill kits, neutralizers, and gloves ought to sit close by. If someone splashes a small amount onto a surface, fast cleanup keeps irritation and damage to a minimum. Good ventilation pulls any vapor away before it reaches nose level, which helps avoid headaches and dizziness.
Training Matters
People often overlook the value of regular safety training. Having worked through more than one discipline—analytical labs, workshops, even art studios—I’ve noticed the difference between spaces that treat safety as a checklist and those that weave it into routine. Workers should know why these rules exist, not just follow them blindly. Short, hands-on sessions cut through overconfidence and build good habits. Proper storage pays off not just for safety, but for long-term product reliability and peace of mind.
What are the chemical properties of Ethyl Lactate?
Understanding Ethyl Lactate on a Chemical Level
Ethyl lactate focuses on both simplicity and usefulness in the chemistry world. Made through the reaction of lactic acid and ethanol, it forms a colorless liquid with a pleasant, fruity smell. It feels right at home in everyday products, whether in the form of a cleaner or a flavoring agent. With the chemical formula C5H10O3, this compound includes an ester bond, which comes from the marriage between lactic acid’s carboxyl group and ethanol’s alcohol group.
Solubility and Stability: Ethyl Lactate in Action
Ethyl lactate blends easily with water and most organic solvents, so it handles many mixes, both in labs and in commercial settings. Its ability to dissolve a wide range of substances makes it a solid choice for use as a solvent. That solubility comes handy for industries trying to move away from traditional petroleum-based solvents, and it gives way to more sustainable options.
This compound shows solid stability under typical storage conditions, yet strong acids, bases, or high heat will break it down, returning it to lactic acid and ethanol. This trait works in its favor from an environmental perspective, since natural processes or even simple composting can help break it down without leaving stubborn residues.
Boiling Point, Flash Point, and Safety
Ethyl lactate boils at around 154°C, which sits higher than many other common esters. The flash point settles near 46°C, meaning the liquid can catch fire with enough heat. So, from the perspective of safe storage and use, it should not hang around open flames or excessive heat sources. Like many organic solvents, good airflow and careful handling matter, especially in larger quantities.
Biodegradability: Moving Toward Better Solutions
Nature plays a role in the chemical makeup of ethyl lactate—not just in how it’s made, but in how it breaks down. With its origins in substances found in food and drinks, ethyl lactate leaves less of a footprint than the harsher solvents often used in industrial settings. Research supports its quick breakdown in water and soil, thanks to the work of bacteria and natural enzymes. Because of this, companies wanting to move to greener chemicals look seriously at ethyl lactate as a realistic option. The U.S. Environmental Protection Agency gave it the thumbs-up for use in “Safer Choice” products.
Why Chemical Properties Matter Day-to-Day
As a chemist and a citizen, I see firsthand the headaches caused by harsh, lingering chemicals. Ethyl lactate helps bridge the gap between performance and responsibility. Used in cleaning agents, coatings, and even food processing, its chemical properties support real, practical change. If every industrial solvent could tick off boxes for effectiveness, safety, and minimal environmental impact, the supply chain would look very different.
Real solutions start with better choices at the chemical level. Selecting ingredients like ethyl lactate turns vague sustainability promises into actual, working alternatives. By understanding how properties such as solubility, boiling point, and breakdown pathways tie into the bigger picture, industries and consumers both take steps toward safer and more responsible chemical use.
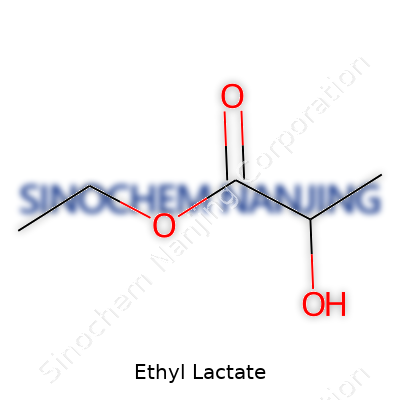
| Names | |
| Preferred IUPAC name | ethyl 2-hydroxypropanoate |
| Other names |
Lactic acid ethyl ester Ethyl 2-hydroxypropanoate Ethyl 2-hydroxypropionate Ethyl lactate 2-Hydroxypropanoic acid ethyl ester Dowfrost |
| Pronunciation | /ˈiːθɪl ˈlæk.teɪt/ |
| Identifiers | |
| CAS Number | 97-64-3 |
| Beilstein Reference | **1365055** |
| ChEBI | CHEBI:27750 |
| ChEMBL | CHEMBL1356 |
| ChemSpider | 8010 |
| DrugBank | DB04325 |
| ECHA InfoCard | 03c36c40-1b8e-4777-983e-9e2536f1b4a7 |
| EC Number | 97-64-3 |
| Gmelin Reference | Gmelin Reference: **102148** |
| KEGG | C02302 |
| MeSH | Ethyl Lactate [D02.241.223.211.570][D01.268.150.370.570] |
| PubChem CID | 7348 |
| RTECS number | KO4375000 |
| UNII | K72NS3N40G |
| UN number | UN1192 |
| CompTox Dashboard (EPA) | DTXSID1022742 |
| Properties | |
| Chemical formula | C5H10O3 |
| Molar mass | 118.13 g/mol |
| Appearance | Clear, colorless liquid |
| Odor | Mild, fruity odor |
| Density | 1.03 g/cm³ |
| Solubility in water | miscible |
| log P | -0.24 |
| Vapor pressure | 0.46 mmHg (at 25 °C) |
| Acidity (pKa) | pKa ≈ 15.3 |
| Basicity (pKb) | pKb ≈ 15.3 |
| Magnetic susceptibility (χ) | -7.87 × 10⁻⁶ |
| Refractive index (nD) | 1.414 |
| Viscosity | Viscosity: 2.5 mPa·s (at 25 °C) |
| Dipole moment | 4.74 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 362.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -635.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2224 kJ/mol |
| Pharmacology | |
| ATC code | A07EC02 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | **"GHS02,GHS07"** |
| Signal word | Warning |
| Hazard statements | Hazard statements: "H315: Causes skin irritation. H318: Causes serious eye damage. H335: May cause respiratory irritation. |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 46 °C |
| Autoignition temperature | 225 °C |
| Explosive limits | 4 - 16% |
| Lethal dose or concentration | LD50 (oral, rat): 5,939 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 5,000 mg/kg |
| NIOSH | WI9275000 |
| PEL (Permissible) | 100 ppm |
| REL (Recommended) | 25 mg/m³ |
| IDLH (Immediate danger) | 1500 ppm |
| Related compounds | |
| Related compounds |
Ethyl acetate Lactic acid Methyl lactate Propylene glycol Butyl lactate |

