Understanding Ethyl Isobutyrate: A Closer Look at an Underappreciated Chemical
Ethyl Isobutyrate’s Journey Through Time
Ethyl isobutyrate might not command headlines, yet its story stretches back to early developments in synthetic chemistry. Chemists in the nineteenth century began isolating and modifying esters like this one, chasing the secrets behind aroma and flavor. What really drew the early researchers was the ester’s distinct scent—somewhere between fresh fruit and youthful cheer. Over time, industries learned to value not just its fragrance but the reproducible method behind making it. Tools got better, purity went up, and by the mid-twentieth century, global flavor and fragrance brands started selecting molecules like ethyl isobutyrate to give their products more character. It often feels like industrial innovation builds on overlooked foundations. The story of this ester reminds me that small discoveries matter far beyond the lab benches where they began.
What Makes Ethyl Isobutyrate Special?
This compound’s strength lies in its profile: a clear, colorless liquid that delivers the tang and pop of pineapple to the nose. Its chemical formula, C6H12O2, frames a simple ester structure with an isobutyric acid base and an ethyl group tag—hardly complex, but surprisingly effective. With a moderate boiling point and good solubility in organic solvents, ethyl isobutyrate fits neatly into a range of applications. Unlike natural extractives, its consistency doesn’t waver from one barrel to the next. Every time I walk through a flavor blending facility, the practicality behind compounds like this stands out: you know what you’re getting, every time, and there’s something comforting in that reliability. That might sound like a small thing, but in food science, consistency often serves as the difference between safe everyday indulgence and unpredictable risk.
Physical Properties and the Science Behind Them
Ethyl isobutyrate presents itself as a volatile liquid, slightly heavier than water, mostly hydrophobic but with just enough polarity to interact with a range of other organics. Its fruity smell stems from how our noses process the molecule, and its volatility lets it release that recognizable aroma fast and efficiently. The density hovers just under 0.9 g/cm³, while its flashpoint falls comfortably below room temperature, classifying it as a flammable substance. In my time working around industrial solvents and additives, I’ve learned that the real challenge isn’t just how a chemical performs on paper; it’s about how these numbers play out in the wild. Flammability means ventilation matters just as much as labeling, and the persistent scent of esters will linger in memory long after they slip into a finished product.
Labeling and Safety at the Forefront
Factories, labs, and flavor houses all need tight operational standards, so safety labeling isn’t just a checkbox. For ethyl isobutyrate, you see hazard pictograms warning about flammability and potential irritant effects. Safe storage asks for cool, well-ventilated spaces that keep distance from sparks. When blending or transferring, proper PPE and fume hoods help keep exposure in check. Years ago, I spent days reviewing Material Safety Data Sheets, and the importance of clear protocols always came down to one thing: mistakes happen most when familiarity turns into carelessness. With an ester like this, staying focused on best practices provides the surest line of defense against spills, vapors, and unnecessary risk.
Making Ethyl Isobutyrate—The Classic Esterification Tale
Production relies on a classic reaction—Fischer esterification—using isobutyric acid and ethanol in the presence of a strong acid catalyst, usually sulfuric acid. Mixing, heating, and refining convert these simple raw materials into the clean, aromatic ester. Yield matters because industrial producers want little waste and strong purity. I’ve watched operators check pH, monitor temperature, and run samples through gas chromatography to keep each batch true. There’s a satisfying kind of craft at play: the equipment hums along, but human judgment closes the loop. Striking the right balance means not just following a recipe, but reading the subtleties of the process every step of the way.
Where Chemical Changes Lead the Way
Ethyl isobutyrate stands up to some gentle chemical play: slight tweaks to the acid or alcohol portions flip the aroma profile, letting flavorists and scientists chase new territory with each derivative. More dramatic modifications, like transesterification, swap ester groups to shape compatibility with other formulation needs. In organic chemistry labs, students start here, learning the ropes before moving to more complex molecules, because small esters tolerate mistakes and still offer up useful data. It’s easy to overlook the building block nature of these compounds, but time spent experimenting with their boundaries drives home the real-world possibilities hidden in basic reactions.
Multiple Names, One Compound
Walking the flavor corridors, you’ll hear ethyl isobutyrate called several things—Isobutyric acid ethyl ester, 2-methylpropanoic acid ethyl ester, FEMA 2441. Every synonym signals the same core identity to experts, while marketing teams latch onto whichever sounds best for their audience. Recognizing the alternate names brings clarity when reading research, regulations, or product specs, cutting through the confusion that sometimes stalls projects across borders or disciplines. Having the right name maps back to finding the right answer; small detail, big difference in practice.
Where It Ends Up—And Why That Matters
Taste and scent take the lead when it comes to ethyl isobutyrate’s main uses. It brings a burst of pineapple or citrus into candies, beverages, and baked goods. Perfumers lean on its brightness to lift floral and fruity notes above a blend’s base. The reach doesn’t stop there: pharmaceutical development finds aroma critical when masking unpleasant drug flavors, and the animal feed sector uses esters to improve palatability. Stepping into a tasting panel, I’ve watched trained palates spot the ester right away—less as an ingredient, more like a signature hidden in the mix. While consumers rarely notice the name, they absolutely notice the effect. Taste matters in ways numbers can’t quite capture, and every choice about what goes into our food and drink shapes habits and memories.
Ongoing Research and Cleaner Production Methods
Researchers have always looked for ways to make the process safer, greener, and more efficient. In recent years, biocatalytic approaches emerged, using enzyme-driven reactions instead of strong acids—less waste, lower temperature, better selectivity. As sustainability gains steam across production lines, catalysis research attracts chemists hunting for methods that trim emissions and lower hazardous byproducts. Future breakthroughs may hinge on finding balance between cost, safety, and environmental impact. I’ve seen academic posters outlining stepwise improvements, and each incremental change tells a bigger story about chemistry’s evolving conscience—moving beyond scale toward smarter, more responsible production.
Exploring Safety and Toxicity—The Final Litmus Tests
No chemical gets a pass on safety, and ethyl isobutyrate has earned attention from toxicologists and regulatory agencies. Studies suggest it presents low acute toxicity, and historical data shows rare evidence of severe sensitization in food-approved uses. That said, repeated inhalation at high concentrations can provoke irritation, especially in confined workspaces. Chronic studies haven’t uncovered alarming trends among workers, but routine monitoring—and trust in existing occupational guidelines—protects against cumulative harm. As an observer in food and fragrance regulation meetings, I often hear the same core message: the dose, not the substance, is what counts most. Safe operating standards, routine air checks, and solid training anchor real-world safety in everyday work.
Looking Toward the Future—New Frontiers for Ethyl Isobutyrate
Interest in flavor science keeps growing, and demand for reliable, well-characterized ingredients like ethyl isobutyrate won’t let up soon. Green chemistry will keep redefining how manufacturers approach ester production, balancing renewable sources and economic scale. In flavor development, we’ll see even more sophisticated tools—aroma wheels, machine learning-driven profiling—helping tune use rates and discover new masks or enhancers for tough applications. What excites me most is watching new scientific minds draw on the humble building blocks of today for the breakthroughs of tomorrow. Methods once considered standard may look outdated within a decade, as pressure for sustainability and transparency rewire our expectations. In the world of food and fragrance, the real legacy isn’t just better smells or tastes—it’s the smarter, safer, and more responsible future that follows every incremental improvement.
What is Ethyl Isobutyrate used for?
Where You Find Ethyl Isobutyrate
Walk down the grocery aisles, pass by the bakery or the candy shelf, and chances are something you sniff comes from a lab as much as a farm. Ethyl isobutyrate might not ring a bell right away, but this clear liquid works behind the scenes in products you reach for daily. In my own childhood, opening a pack of chewing gum or twisting the lid off a soda, there was a burst of sweet, fruity aroma. That familiar burst owes a lot to compounds like ethyl isobutyrate, which brings a bright, pineapple-like kick to food and drinks.
This chemical appears in the list of “nature-identical” ingredients that flavor companies use to deliver consistent taste. The reason is simple: pineapple and other fruits only yield tiny amounts when pressed and extracted, but flavor houses can blend ethyl isobutyrate to match the real thing at large scale. Regulatory agencies such as the FDA recognize it as safe for use in food, so it finds a home in jams, candies, sodas, ice cream, and bakery goods. Some studies show it’s especially important in soft drinks and fruit-flavored yogurts, where getting that tropical note right can make or break a product line. Its low toxicity means it can add sweetness without health worries—something I pay attention to as a parent choosing snacks for my kids.
Adding Scent to Everyday Products
Step out of the kitchen and ethyl isobutyrate still sticks around. Fragrance brands mix it into perfumes and air fresheners to create a fresh, uplifting character. Its volatility means it jumps from liquid to vapor fast, delivering scent quickly. I remember testing different cleaning sprays at a supermarket, pausing for a moment over one with a tropical scent. A glance at the label—tiny print, but there it was—ethyl isobutyrate among the ingredients. Brands like it because it lasts through the life of the product and doesn’t overpower milder base notes. Sometimes, even cosmetics like shampoos and lotions use it to get that subtle fruity twist.
Why It Matters in Food Safety and Regulation
Not all chemicals make it into consumer goods. Companies depend on strict guidelines from bodies like the Joint FAO/WHO Expert Committee on Food Additives and the FDA. Safety data and decades of toxicology research give consumers confidence that trace amounts pose no threat. If regulators withdrew the status of ethyl isobutyrate, the food and fragrance industries would scramble to reformulate or pull products. Reliable ingredients build public trust—a key point if, like me, you’ve watched food recalls spiral out of control due to overlooked details.
Facing Sustainability and Natural Demand
More shoppers now want natural ingredients, so the pressure grows to source compounds like ethyl isobutyrate from sustainable, bio-based processes. Labs keep finding ways to produce it without fossil fuels. Some companies turn to fermentation, coaxing yeast or bacteria into making flavor chemicals just as nature does. This shift brings the chance to lower carbon footprints and cut chemical waste, a step I care about as the impacts of climate change become more urgent.
Looking Forward
Ethyl isobutyrate demonstrates the push and pull between tradition and modern technology in the flavor world. It keeps snacks, sodas, and body sprays bright and fruity, but industry leaders can keep looking for renewable sources and transparent labeling. As consumers learn more, the companies with clear sourcing and proven safety records will keep earning trust.
Is Ethyl Isobutyrate safe for use in food and beverages?
What Ethyl Isobutyrate Really Is
Open a bottle of fruit juice or flavored soda and you’ll probably find a cocktail of food flavorings behind that refreshing taste. Ethyl isobutyrate brings a sharp, pineapple-like aroma that gives candy, drinks, and even baked snacks their fruity punch. Most folks don’t recognize this name on a label. Still, it’s been a go-to in the industry for decades. It’s a common choice because a little goes a long way—and because it manages to deliver that “real fruit” impression without draining budgets or spoiling on the shelf.
What the Experts Say About Its Safety
I remember the first time I checked an ingredient label and ran into a list of chemicals that sounded intimidating. It’s natural to worry. Ethyl isobutyrate has been looked at by groups like the Flavor and Extract Manufacturers Association (FEMA), as well as by the U.S. Food and Drug Administration. The FDA places it on the GRAS (Generally Recognized as Safe) list for use in food. That’s not a free-for-all label; it follows plenty of safety testing by toxicologists and food scientists. These evaluations track daily intake, the way bodies break down this compound, and any potential for known allergic reactions.
Addressing Real Concerns
Safety isn’t just about whether something is “technically” harmless. People want to know if consuming this molecule over years could mean trouble. Scientists have run tests on animals at much higher doses than people would ever get from food or drinks. The data show that the body quickly processes ethyl isobutyrate and clears it out without leaving harmful byproducts. No strong evidence links it to cancer or other chronic illness at levels used in food manufacturing. Allergic reactions tied to this additive show up rarely in reports, and symptoms usually result from much bigger exposures than you’d get sipping soda.
Why Food Science Keeps Watch
Taste trends change, and companies always look for new flavor shortcuts. Whenever a synthetic compound becomes popular, watchdogs keep updating research. Staying sharp on food safety means reviewing studies, adjusting intake guidelines, and sharing knowledge across countries. For example, European regulators take a tougher stance on some additives and restrict levels to a fraction of the U.S. limits. That cross-checking actually strengthens consumer protection. For those who choose to limit processed foods or avoid “artificial” flavors, alternatives like real fruit juice come with their own shelf-life and cost trade-offs.
Practical Steps for Transparency
Food trust doesn’t grow from secrecy. Brands gain goodwill by making ingredient information easy to find and explaining why each one gets used. In day-to-day life, the most dependable approach looks simple: read labels, look out for any unusual personal reactions, and stay tuned to new information coming from recognized government agencies or reputable science groups. I’ve seen food recalls take place fast when new evidence pops up, which shows the system can respond if problems emerge. Keeping an open channel between consumers, scientists, and industry pushes everyone toward safer products and more choices on the shelf.
What is the chemical formula of Ethyl Isobutyrate?
Ethyl Isobutyrate in Everyday Life
Ethyl isobutyrate gets a fair bit of attention from chemists and food scientists, but hardly anyone outside those circles talks about it, except maybe winemakers or folks in the fragrance business. Cooks or bartenders might not know the molecule by name, but chances are good they’ve worked with flavors or essences that owe their punchy fruit notes to compounds like this one. It’s found its way into artificial flavors, perfumes, and probably more than a few candies we grew up eating. It’s easy to write off the study of chemical compounds as a detail for scientists only, but understanding the makeup and role of these substances connects everyday products to basic science.
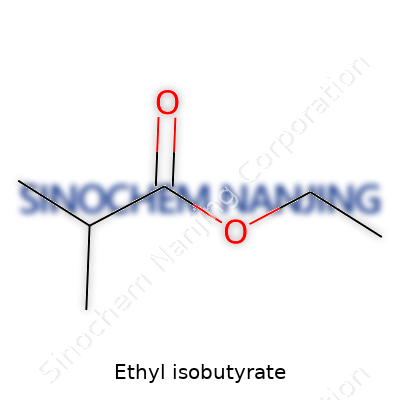
The Formula: C6H12O2
Ethyl isobutyrate carries the chemical formula C6H12O2. What does this shorthand mean in practical terms? Each molecule contains six carbon atoms, twelve hydrogens, and two oxygens. The atoms link up to form a structure called an ester, specifically the result of combining isobutyric acid with ethanol. This merger shapes not only the formula on paper but also the way ethyl isobutyrate smells and tastes. The compound smells like juicy pineapples, which explains its popularity in pineapple-flavored treats.
Why Formula Matters Beyond the Classroom
Knowing that ethyl isobutyrate’s formula is C6H12O2 helps manufacturers avoid unwanted by-products. Wrong proportions can trigger off-flavors, and in the food industry, even a slight chemical misstep can mean the difference between a sweet aroma and a strange aftertaste. The United States Food and Drug Administration monitors the use of compounds like these, and safety data sheets hinge on the exact chemical structure. College chemistry students memorize formulas, but that rote learning turns into crucial troubleshooting in laboratories and quality assurance setups.
Health and Safety Concerns
Ethyl isobutyrate isn’t just a flavor powerhouse; it also carries the need for clear safety guidelines. The formula gives toxicologists and emergency responders the information needed to understand how the compound might interact with the body or the environment in case of a spill. Science journals publish toxicological reviews pinning the formula to specific metabolic pathways, so researchers can predict potential side effects or allergic reactions.
Responsible Production and Environmental Footprint
Large-scale manufacturers can’t ignore the environmental impact of any chemical, even one approved for foods and cosmetics. Ethyl isobutyrate’s structure allows for efficient, high-yield synthesis from relatively benign starting materials. Factories keep emissions and waste low by using precise chemical engineering, guided by the clear-cut formula C6H12O2. That’s not just good business; it makes a difference in how sustainably the world meets growing demand for processed foods and household goods.
Solutions and Better Practices
Chemists continue to streamline synthesis, removing impurities before flavorings hit production lines. Quality control scientists use analytical techniques to confirm the structural match to the target molecule, always referencing the molecular formula. Industry watchdogs, armed with accurate formulas and a solid grasp of chemistry, help spot mislabeling or contamination. These steps, rooted in a strong foundational knowledge of ethyl isobutyrate’s formula, protect consumers and reinforce trust in product labels.
What does Ethyl Isobutyrate smell like?
Scent That Works Behind the Scenes
Ethyl isobutyrate pops up more than most people realize, especially in places that want to smell just a bit better. Its scent hits the nose with a lively fruitiness that feels familiar if you’ve ever bitten into a ripe pineapple or sipped on a mixed fruit punch. Even before learning its name, most recall catching a hint of it in candies, chewing gum, or those nostalgic fruit-flavored drinks from childhood. Unlike some chemicals that try to stay out of the spotlight, ethyl isobutyrate shapes a sweet, tropical background note—almost like a supporting actor who manages to steal a scene.
Experience in Everyday Products
Spending years working in food processing, I learned to identify certain artificial flavors just by their scent, and ethyl isobutyrate never failed to give away its fruity vibe. Every time a new jelly or beverage rolled out onto the test table, this ester would leave its mark with a punchy, pineapple aroma. Sometimes, I wondered whether the recipe was too heavy-handed, as the sweet, slightly creamy note would remind me immediately of those gummy candies stacked along convenience store counters.
This connection between scent and memory runs deep. This volatile compound quickly escapes liquids or gels, filling the air with sweetness and making the mouth water just by association. People aren’t always aware of the impact such molecules have on their cravings, but the right aromatic balance can boost a product’s appeal instantly. Food science leans on this knowledge—and so do perfume-makers who use similar scents in tropical or summer-themed offerings.
Tracing the Source
Found naturally in apples, bananas, and pineapples, ethyl isobutyrate doesn’t feel out of place in a basket of mixed fruit. Once extracted or synthesized, it manages to cross into the laboratory world, turning up on ingredient lists with reassuring regularity. Safety studies from organizations like the Flavor and Extract Manufacturers Association and regulatory reviews from the U.S. Food and Drug Administration point to its acceptance for use in foods and fragrances as long as it stays within certain concentrations.
Many flavor chemists will mention how ethyl isobutyrate blends smoothly with other fruity notes: mango, passionfruit, even coconut. Those involved in recipe development often mix it with acids or sugars to create a sensation that punches through blandness, giving flat drinks or candies a brighter, more inviting identity. Its strength, and sometimes its downfall, lies in its intensity—a little goes a long way. Too much, and the flavor starts to feel artificial, masking the subtlety of natural ingredients.
Challenges and Smarter Use
Growing concern exists over artificial flavors and additives, not just from a health perspective but about transparency and truth in labeling. Some people feel uneasy when ingredient lists look unfamiliar. Brands trying to build trust with consumers find themselves pulled in two directions: deliver the vivid flavors people crave while sticking to simple, clear ingredient lists. Science suggests ethyl isobutyrate isn’t harmful at normal concentrations, but it still carries the baggage of being “synthetic” in many minds.
Encouraging wider use of natural sources or improved extraction techniques may help, but food safety and flavor stability remain crucial. Research must continue into how such additives interact with the body and with each other, especially as new alternatives enter the market. Meanwhile, anyone curious about the chemistry of flavor should try smelling ethyl isobutyrate—just a careful sniff from a properly labeled sample shows why it matters so much in the modern food world.
What are the storage conditions for Ethyl Isobutyrate?
The Nature of Ethyl Isobutyrate and Everyday Storage Risks
Ethyl isobutyrate brings a fruity note that finds its way into flavors and fragrances. This liquid looks clear and carries a strong odor. Its flammability demands attention, especially in workspaces that juggle different chemicals. Picture a warehouse in summer: bottles of the stuff stacked near a window, heat radiating in, and not enough ventilation. Over time, that scenario creates hazards most people do not anticipate until a whiff of fumes or a misplaced spark turns into real trouble.
Conditions That Keep Things Safe and Stable
Cool and dry places do more than keep things comfortable—they prevent loss and damage. Heat pumps up vapor pressure, and with a low flash point around 18°C (64°F), ethyl isobutyrate climbs quickly toward a risk zone. Metal containers with tight-fitting lids reduce the chance of leaks and keep vapor inside. In practice, companies tap into flame-proof storage cabinets, setting aside a room with steady temperatures below 25°C (77°F). Moisture triggers hydrolysis, so leaks around pipes or cracked seals can ruin the entire batch and corrode metal parts.
Some people picture storage as just a shelf in the corner. Experience shows that physical barriers—sealed drums or IBC totes—act as checkpoints against accidental mixing. Ethyl isobutyrate reacts with strong oxidizers, and careless storage next to bleach or peroxides has caused incidents in smaller facilities. A clear labeling system, plus segregation from incompatible chemicals, keeps the pressure off emergency responders if something ever goes wrong.
Why Attention to Air and Light Pays Off
Direct sunlight degrades the ester. Bottles start to change color, and the contents lose value fast. Keeping this chemical in dark containers or shadowed spots inside storage areas extends shelf life. I once saw a lab lose three months’ inventory simply because the blinds hadn't been drawn. Even overhead lights can spark enough UV to slowly break down sensitive substances over weeks. Routine checks—simply running a hand along seals and caps, reading labels for fading, noticing unusual smells—make a big difference in stopping problems before they escalate.
Hazard Management Beyond the Storage Room
Ethyl isobutyrate smolders unseen if a vapor builds up. Ventilation prevents accumulation. Sash hoods or local exhaust systems stand between a bad day and a regular one. Training is not just paper-pushing—it teaches folks how to handle spills, extinguish small fires, and read warning signs. At one plant, routine drills meant the staff caught a leak at the gasket before it turned into a blown cap and vapor cloud. Read enough accident reports and you see simple steps make the difference.
Quick cleanup kits—sorbent pads, non-sparking tools, and chemical waste bins—offer peace of mind. Fire extinguishers rated for flammable liquids should sit an arm’s length away. Clear emergency aisles prevent panic if someone scrambles for help.
Practical Solutions for Real-Life Scenarios
Locks on cabinets stop wandering hands and curious visitors. Inventory records help track old stock nearing expiration. Outdated material builds pressure and increases risk, and inventory checks let you cycle out what doesn’t belong. Regular inspections—not just during audits—reinforce a culture of safety.
Good habits around labeling, using compatible storage materials, and steady supervision keep ethyl isobutyrate doing its job and out of the news. It’s not just about following a checklist, but taking ownership, staying curious, and remembering what’s at stake beyond the lab or warehouse floor.
| Names | |
| Preferred IUPAC name | Ethyl 2-methylpropanoate |
| Other names |
Ethyl 2-methylpropanoate Isobutyric acid ethyl ester Ethyl isobutanoate |
| Pronunciation | /ˈiːθɪl aɪsəˈbjuːtɪreɪt/ |
| Identifiers | |
| CAS Number | 97-62-1 |
| Beilstein Reference | 635582 |
| ChEBI | CHEBI:88573 |
| ChEMBL | CHEMBL3185227 |
| ChemSpider | 63919 |
| DrugBank | DB14141 |
| ECHA InfoCard | 03ef3b19-2d6e-4ed2-bb18-fc292732b6cb |
| EC Number | 203-293-5 |
| Gmelin Reference | 6037 |
| KEGG | C12560 |
| MeSH | D005002 |
| PubChem CID | 7764 |
| RTECS number | EL5425000 |
| UNII | LI2X64QM44 |
| UN number | UN1179 |
| CompTox Dashboard (EPA) | DTXSID4020977 |
| Properties | |
| Chemical formula | C6H12O2 |
| Molar mass | 116.16 g/mol |
| Appearance | Clear colorless liquid |
| Odor | Fruity; pineapple |
| Density | 0.868 g/mL at 25 °C (lit.) |
| Solubility in water | 0.09 g/100 mL |
| log P | 1.85 |
| Vapor pressure | 2.9 mmHg (20°C) |
| Acidity (pKa) | Estimated pKa ≈ 25 |
| Basicity (pKb) | 17.3 |
| Magnetic susceptibility (χ) | -7.33 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.3890 |
| Viscosity | 1.101 mPa·s (25 °C) |
| Dipole moment | 1.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 267.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -464.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3130.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 20 °C |
| Autoignition temperature | 459 °C |
| Explosive limits | 1.2% - 7.0% |
| Lethal dose or concentration | LD50 (oral, rat): 7,850 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 7,340 mg/kg |
| NIOSH | NIOSH: NH2450000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Ethyl Isobutyrate: "PEL: 25 ppm (TWA) |
| REL (Recommended) | FEMA 1.1 mg/kg |
| IDLH (Immediate danger) | IDLH: 1700 ppm |
| Related compounds | |
| Related compounds |
Ethyl butyrate Isobutyric acid Isobutanol Butyric acid Ethyl acetate |

