Ethyl Dichloroacetate: A Closer Look at Its Journey and Impact
Historical Development and Background
Ethyl dichloroacetate sits on a long timeline of chemical research that reflects both curiosity and need. Chemists traced its roots back to early work on haloacetic acids. The dichloroacetic group showed potential early in the 20th century, with researchers digging into modifications that produced a family of compounds. As industry needs shifted during the mid-1900s, makers in the chemical sector used more aggressive halogenation techniques to prepare reagents for synthesis and application. What stands out in the story of ethyl dichloroacetate is the role of persistent, sometimes trial-and-error chemistry that phased out older, less efficient manufacturing routines. Anyone who has sifted through yellowed journals or spoke to an industry veteran can appreciate the incremental improvements that led this compound from laboratory curiosity to a tool with a defined niche.
Product Overview and Characteristics
Ethyl dichloroacetate draws attention among acetic acid derivatives for a simple reason: its balance of volatility and reactivity makes it a handy intermediate. The product’s clear, sometimes faintly yellow appearance, hints at how small changes in process can affect purity and stability. It comes off as a liquid with a sharp, penetrating odor. Those who have worked with it on a bench know it does not behave like a standard ester. Its dichloroacetyl group pushes its behavior toward more aggressive reactivity, inviting use in organic syntheses where selectivity and speed matter. The character of this chemical comes across most clearly the first time you open a container and catch that pungent, characteristic scent—one you never quite forget after the initial exposure.
Physical and Chemical Properties
Dense and volatile, ethyl dichloroacetate packs a molecular punch. It brings a boiling point low enough to require tight control over heating, especially in open settings. The presence of two chlorine atoms drives much of its personality. Unlike aliphatic esters, which can sit quiet on a shelf, this compound responds quickly under the slightest provocation. Those who mix or heat it outside of proper conditions run the risk of sharp releases and even decomposition. Its solubility profile lets it blend with many non-polar solvents but throws up definite limitations in water-based systems, something obvious during separations and extractions in a lab setting. From an energy of reaction perspective, it's both a friend to the creative chemist and a foe to those who ignore the practical side of handling halogenated intermediates.
Technical Specifications & Labeling Practices
Technical data on ethyl dichloroacetate flows from an honest look at purity, stability, and suitability for organic syntheses. Laboratories with rigorous standards rely on chromatography and spectroscopic methods to confirm identity, while industrial batches demand batch-to-batch consistency. Regulatory boundaries set maximum impurity levels, and the best run labs refuse to cut corners. Labels usually flag the presence of halogen atoms and the risks that can arise if storage slips from ideal conditions. Over many years of reading and writing data sheets, I've found that honest labeling improves safety and builds trust, especially where niche chemicals such as this are moving out of strictly industrial domains and into exploratory medicine and research.
Preparation Method and Modern Synthesis
Manufacturers of ethyl dichloroacetate lean heavily on the esterification of dichloroacetic acid with ethanol. The acid chloride route shows up in older literature but has faded, replaced by direct condensation for cleaner yields. This reaction tends to happen under controlled acid catalysis, using reflux conditions. The byproduct water, if not removed, disrupts the process, nudging the reaction backward. In labs where glassware has seen decades of use, this reaction remains a staple, though modern setups often use distillation columns to capture the product without extensive cleanup. The hands-on nature of this synthesis means that minor process tweaks can have outsized effects on purity, something I saw firsthand working alongside veteran synthetic chemists who knew the tricks for coaxing higher yields from even stubborn starting materials.
Chemical Reactions & Modifications
Ethyl dichloroacetate acts as an intermediate with a knack for inviting further chemistry. The dichloroacetyl group, for all its electron-withdrawing power, offers a reactive site that opens doors to chain extension, nucleophilic substitution, and reduction. Alkoxide bases strip away the ester group under transesterification, while nucleophiles set off reactions at the acyl carbon. This compound also sits at a crossroads between stability and lability. Gentle conditions let it slide through reactions unchanged, but push too hard, and you end up with messy mixtures. Small modifications to its core—exchanging the chloro groups or altering the alcohol moiety—turn it into new esters, key for those who want fine-tuned properties in pharmaceuticals or polymers.
Synonyms & Product Names in Circulation
Not everyone calls it ethyl dichloroacetate. In catalogs, one finds names like ethyl 2,2-dichloroacetate or simply the DCA ester. Some publications drop “ethyl” and lean on shorthand, leading to confusion for a newcomer. Clarity in naming remains a struggle, and seasoned scientists often take time to double-check structures. A wrong name, especially in a multi-step synthesis, can mean the loss of weeks of work. The importance of concrete, consistent nomenclature only becomes clearer with experience, and I’ve learned over years of research that tracking synonyms averts costly mistakes.
Safety Considerations and Operational Standards
Ethyl dichloroacetate reminds handlers what “respect for the bottle” means. Fumes irritate the eyes and throat, and even a small spill calls for keen attention and immediate cleanup. Guidelines focus on direct ventilation and resistant gloves, but more importantly, they tell people to anticipate problems before uncapping a bottle. Many accidents stem from overconfidence and poor planning. Training everyone who works with it—regardless of experience—prevents most incidents. The strict routines adopted by chemical manufacturers are not suggestions but sharp lessons written in hard-won experience. Adherence to local and international safety standards grounds the daily practice of chemical handling.
Application Area and Uses
Interest in ethyl dichloroacetate waxes and wanes across the pharmaceutical, agricultural, and research sectors. Medicinal chemistry teams turn to it for its part in metabolic modulators, including research into mitochondrial disorders. Lower-level applications include its role as a building block for other esters, especially those that need the weight and polarity given by the dichloroacetyl unit. Some pesticide chemistries have leaned on its structure for effect, though shifts toward greener solutions undercut this role in recent years. The research crowd values it in proof-of-principle syntheses and as a reagent for tuning reactivity in sensitive reaction sequences. Real-world experience shows that, even if commercial products carry its name less frequently on shelves today, the variety of papers and patents built on its chemistry grows steadily.
Research & Development Efforts
Chemists and researchers sharpen their focus on new uses for ethyl dichloroacetate. Biomedical investigations seek routes to treat metabolic disease. Recent studies hint at roles in modulating lactic acid pathways, with small pilot results drawing cautious interest. While some of these efforts move painstakingly, lab-scale adaptation of synthesis and handling unlocks creative uses. New synthetic methodologies arise—such as greener esterification techniques—motivated by environmental pressure and the high costs of downstream purification. Research communities now connect faster, share protocols, and build ever more sophisticated studies on this backbone. My own time in collaborative groups emphasizes the value of open discussion between synthetic, analytical, and application-focused teams. The true power of such a niche molecule shows itself in how quickly new ideas can move from paper to bench when people work across boundaries.
Toxicity Research and Health Effects
Safety studies on ethyl dichloroacetate point to the need for careful risk assessment. Animal models show both acute and chronic effects, ranging from liver stress to carcinogenic concerns at high or repeated exposures. Medical researchers sometimes explore it as a therapeutic agent, but push and pull between risk and benefit means rigorous trials and close monitoring. No room exists for shortcuts or assumptions. People handling or administering it in research protocols wear the consequence of incomplete data—so protocols often default to heightened precaution. A growing movement pushes for alternatives that limit body burden and environmental impacts. Regulatory bodies like the EPA and EU agencies periodically review available evidence and fine-tune exposure limits. Researchers seeking to use it in advanced applications keep a close eye on toxicological updates and err on the side of caution. From an ethical perspective, full disclosure and respect for potential risks have moved from best practices to essential duties.
Future Prospects and Potential Directions
The outlook for ethyl dichloroacetate will hinge on innovation and responsibility. Chemical users will ask where it fits best, especially as public health and environmental consciousness sharpen regulatory lines. Makers of advanced pharmaceuticals might find niche benefits by exploiting its reactivity, but only if the safety profile stands up under continued scrutiny. Interest from the biotech and synthetic biology realms may drive new approaches, such as precisely engineered analogs that trade hazards for high value. Those of us in the industry now watch for breakthroughs—not just in end uses, but in process technology and green chemistry. Safer, more efficient manufacturing offers the promise of lowering the risk profile while keeping the benefits alive for another decade or more. At the end of the day, the pathway this molecule takes will depend not on simple economic forces, but on relentless curiosity and a commitment to doing right by those who work with and around it.
What is Ethyl Dichloroacetate used for?
Not Just Another Chemical
Ethyl dichloroacetate might sound like something out of a chemistry textbook, though its presence stretches far beyond the lab bench. This compound grabs attention because of its traces found both in industrial processes and biomedical circles. People often spot it under the microscope for its role in several curious experiments linked to energy metabolism and potential medical treatments.
Its Place in Industry
Factories dealing with specialty chemicals sometimes reach for ethyl dichloroacetate during the synthesis of other products. Folks in organic synthesis use it as an intermediate, thanks to its reactivity. By serving as a building block for more complex molecules, it feeds into the production chain of dyes, agricultural compounds, or even active pharmaceutical ingredients. Workers handling it spend time checking for leaks—this chemical’s volatility demands respect, and safety rules get followed to the letter.
Digging into Energy Disorders
On the medical side, researchers test ethyl dichloroacetate for its unique sway over how cells use energy. It acts as a modulator of the enzyme pyruvate dehydrogenase kinase. In plainer terms: This means it can coax cells to burn sugar more efficiently. Some doctors and scientists looked at it as a possible option for treating lactic acidosis, a condition when the body’s cells drown in lactic acid because they can’t shuttle it out quickly enough. Staring at rare inherited energy disorders—things like mitochondrial diseases—medical pioneers tried using this compound as a possible therapy. Evidence remains preliminary and side effects remind people to tread carefully.
Challenges and Careful Handling
Safety takes priority. Ethyl dichloroacetate doesn’t line up alongside everyday household cleaners. Glove-wearing, respirators, and good ventilation become part of the daily routine for those who deal with it. Health authorities classify it as an irritant; even a quick splash on skin or a single inhaled whiff tells muscles and lungs something’s up. Cautious storage and thorough training for anyone handling or transporting it help keep mishaps low. The Centers for Disease Control and Prevention and other regulatory groups lay out detailed safety sheets and reinforce accident procedures.
Environmental Impact: A Need for Balance
My experience working on chemical safety audits showed me how legislation guides the disposal of volatile solvents. Ethyl dichloroacetate must not end up down the drain or in open landfills. Special waste management teams handle its destruction using high-temperature incineration. Runoff or leaks can turn into a headache for water treatment workers. Constant monitoring and strict containment become more than buzzwords—they keep real people and ecosystems out of trouble.
The Ongoing Search for Solutions
Scientists recognize the opportunities in compounds like ethyl dichloroacetate, but nobody turns a blind eye to the risks. Long-term studies keep rolling out, targeting its medical potential while hunting for cleaner, safer analogues or substitutes in industry. Advocating for stronger workplace training, transparent reporting of incidents, and investment in engineering controls, professional health inspectors and chemists push for a safer environment.
A Tool—Handled with Respect
Ethyl dichloroacetate isn’t a household name, yet it sits quietly in research and manufacturing corners, shaping important projects and sparking new ideas. Respect for its risks, backed by solid science and thorough guidelines, helps safeguard those who rely on it. Care, curiosity, and a commitment to safety anchor every conversation about this potent, sometimes promising, sometimes perilous, chemical.
What are the safety precautions when handling Ethyl Dichloroacetate?
Getting Real about Hazards
I’ve spent time with lab chemicals that look harmless at first glance and end up causing more problems than you’d expect. Ethyl dichloroacetate lands squarely in that unpredictable zone. It’s a clear liquid—easy to mistake for something less toxic. Give it an open container for more than a few minutes, and sharp fumes fill the room. Your lungs and eyes feel the sting. Folks who work with chemicals pick up on this quickly, but inexperience or impatience opens the door to accidents.
Gear That Protects
When handling ethyl dichloroacetate, the best gear choice means goggles with side shields and nitrile gloves. Standard lab coats aren’t just for show—they block splashes most days. I once skipped gloves for a “quick transfer” and ended up washing my hands for ages, hoping the tingling would fade. Don’t trust this compound to play nice with bare skin. Chemical fume hoods aren’t optional either. One whiff outside a hood taught me the hard way: nausea and watery eyes can knock your focus clean off.
Good Habits Prevent Trouble
A little bit of spilled ethyl dichloroacetate creates a slippery mess, making the floor a hazard fast. Clean-up goes better with absorbent pads, not paper towels, since paper falls apart and leaves residue that spreads the compound. I always keep eye wash stations and safety showers close. No one plans on an accident, but the folks who know where safety equipment sits never lose precious seconds searching during a crisis.
Storage Can’t Wait
Leaving ethyl dichloroacetate out on a bench even for “just a minute” is an invitation for trouble. This liquid needs locked cabinets away from sunlight and away from acids or bases that cause it to break down or react. Vapors build up pressure if the cap isn’t tight or if the storage area heats up, and then even a gentle shake pops the seal.
Disposal Eats Up Time
Dumping leftover ethyl dichloroacetate down the drain sounds convenient, but this shortcut pollutes local waterways and lands folks in legal trouble. Every place I’ve worked has a hazardous waste protocol, and it chews up minutes most don’t want to spare. Still, following that process saves longer headaches with health inspectors or environmental regulators.
Training and Watchful Eyes
Newcomers to lab work learn quickly from experienced coworkers who don’t let safety steps slide. I remember a time a trainee forgot to close a waste bottle; the next person downwind felt the effects. Sharing these stories does more than a rulebook ever could. Written policies matter, but regular drills and open communication keep everyone sharp.
Professional and Community Responsibility
Mistakes with chemicals reach beyond the lab. Volatile substances like ethyl dichloroacetate easily seep into shared airspace. Take a short walk near some industrial labs and you’ll notice the air gets sharp and chemical. People who live and work nearby depend on us to stick with safe practices every time – not just when the boss is watching.
Real Solutions Make a Difference
Strict supervision, modern safety gear, and a culture of speaking out when something feels off stop problems before they start. Skipping steps never beats the peace of mind you get from going home healthy at the end of the day.
What is the chemical formula and structure of Ethyl Dichloroacetate?
The Makeup of Ethyl Dichloroacetate
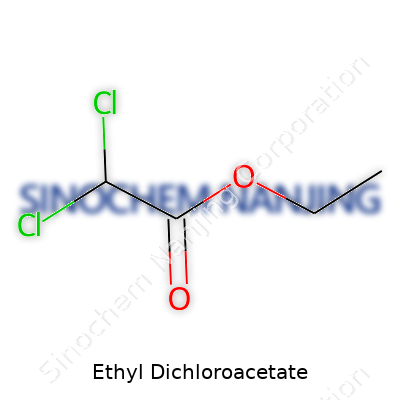
Ethyl dichloroacetate holds the chemical formula C4H6Cl2O2. Each molecule ropes together four carbon atoms, six hydrogens, two chlorines, and two oxygens. The naming sticks pretty close to its structure. The “ethyl” chunk hooks onto the “dichloroacetate” part. Drawing this compound puts two chlorine atoms on the central carbon, which turns up as a feature in its chemical and biological activity.
Picture the backbone: a two-carbon tail (the ethyl group, CH3CH2–) bonded through an oxygen to a carbonyl group (=O). The action happens at the central carbon, which sits double-bonded to oxygen, single-bonded to another oxygen (leading out to the ethyl group), single-bonded to a hydrogen, and single-bonded to two chlorines. Chemists would call that an ester of dichloroacetic acid. The skeletal formula: CHCl2COOCH2CH3.
The Relevance to Research and Industry
Researchers have paid attention to ethyl dichloroacetate for its role in metabolic investigations and medical projects. Its closer cousin, dichloroacetate itself, sometimes comes up in studies around mitochondrial diseases and certain cancers, mostly for how it tweaks cell metabolism. Ethyl dichloroacetate stands as a useful analog—scientists use it to dig into how small molecules interact with living systems or break down in processes like ester hydrolysis.
Looking at the big picture, that little pairing of two chlorine atoms changes how this molecule behaves compared to typical esters. Chlorine atoms pull on the electrons, making the molecule more reactive and changing how quickly it breaks apart inside a body. That shift matters for researchers hoping to shape new medicines or test biochemical changes. Facts on how electron-withdrawing groups such as chlorine affect metabolic fate get backed up by classic organic chemistry studies (see: Morrison & Boyd, "Organic Chemistry," 6th Edition). Chlorines influence not just reactivity, but also potential safety and toxicity.
Safety and Handling: Not Just Lab Talk
It’s easy for safety talk to fall into jargon, but anyone working with chlorinated compounds—myself included—knows that respecting chemical properties pays off. These chemicals can act as irritants or pose risks if ventilation runs thin. For ethyl dichloroacetate, keeping good airflow and protective gloves isn't just a “lab procedure”—it’s a routine. The compound can leave people with respiratory or skin sensitivity. Material Safety Data Sheets (MSDS), mandated by regulatory agencies, flag it as flammable and an eye/skin irritant.
I once watched a novice skip gloves during ester synthesis, only to end the day with tingling skin and a lecture from the safety officer. It made the “read the label” advice stick. These examples show how chemistry—formula and function—goes right from the blackboard into the hands and well-being of real people.
Opportunities for Better Practices
Companies and schools using ethyl dichloroacetate should offer clear protocols and refresher training. Digital SDS records help, but so does good signage. Engineering controls, such as fume hoods and spill kits, build extra layers of protection. Open-door conversations in the lab cut down on accidents. For wastewater disposal, the two chlorines flag the need for special waste streams. Routine environmental checks, not just annual ones, help spot leaks or mishandling before they become real problems. Responsible chemical management protects both workers and the communities around research centers.
Ethyl dichloroacetate’s structure—simple on paper—carries lessons for anyone mixing, transporting, or studying chemicals. Respect the formula, respect the molecule, and science will keep moving forward safely.
How should Ethyl Dichloroacetate be stored?
What Ethyl Dichloroacetate Really Means for a Lab
Some chemicals don’t cause much worry in a research space until they’re forgotten on a dusty back shelf. Ethyl dichloroacetate (EDA) lands in that odd spot between common and hazardous — overlooked by people who don’t study its label, and dangerous for anyone who ignores basic storage rules. Every time I walked into a storage area early in my lab work days, I’d spot at least one mystery bottle with a fading sticker or suspicious residue around the cap. You’d rather know what’s lurking there, especially when dealing with a compound like EDA that raises genuine safety issues.
Recognizing Hazards and Learning from Incidents
The MSDS and safety resources don’t mince words on EDA. Liquid at room temperature, it smells sharp and can irritate your lungs fast. Spills form vapor — breathing it in makes throats raw, and accidental contact stings the eyes and skin. A glance at its flammability and corrosiveness should prompt real caution, not paperwork for the safety binder. Researchers at several universities have learned this after vapors from poorly closed bottles led to building evacuations. Overconfidence in a chemical’s mild reputation often sets up avoidable disasters. Strict storage saves time and avoids messes that leave everyone scrambling.
Smart Storage Starts with Containers
Glass bottles with secure, vapor-tight tops give EDA the isolation it asks for. Polyethylene and polypropylene also hold up if glass isn’t available, but the bottle must seal completely to block fumes. We once discovered a leaking cap in our solvent cabinet — a small drip left a sticky film and a chemical odor for hours. It may sound simple, but double-checking a closure keeps accidents from escalating. Labels on every bottle, clear and resistant to smudges, end confusion if spills or leaks happen.
Where to Store EDA: Location is Not a Guess
EDA sits in a flammable cabinet — never on an open shelf, and never near oxidizers such as nitric acid or heavy-duty bleaches. Joining it with incompatible chemicals risks violent reactions or corrosive vapor clouds. Many modern labs use ventilated cabinets with spill trays for extra security. The importance of good airflow became obvious to me during a summer heatwave; poor ventilation made our solvent room a stuffy hazard zone. Heat and sunlight cause bottles to sweat and pressure to increase, so cabinets need a cool spot, away from all light and any heat source.
Emergency Prep and Everyday Habits Matter
Even the tightest storage plan falls apart without a little vigilance. I got into the habit of checking labels and cap tightness every Friday afternoon. Routine checks seem tedious but catch small leaks before they become expensive headaches. Eyewash stations and spill kits stay close to the storage cabinet, stocked with absorbent materials and PPE. Training goes beyond a few slides during orientation; anyone who grabs a bottle understands what to do if an accident occurs. Quick action, practiced by the team, makes a difference in emergencies.
Seeing Responsibility as Part of the Research
People sometimes joke about the fuss over chemical safety rules, but after cleaning up EDA spills myself, I know the policies have purpose. Every guideline aims to keep the lab running and everyone healthy. Respecting the dangers and respecting your colleagues means double-checking every detail. Proper EDA storage becomes a habit every researcher needs — not out of fear but because one slip ruins weeks of good science. It’s attention to detail that turns dangerous compounds into useful research partners, not hazardous leftovers.
Is Ethyl Dichloroacetate hazardous or toxic?
Understanding the Risks of Ethyl Dichloroacetate
Ethyl dichloroacetate usually gets discussed in research labs or chemical supply rooms, rather than at kitchen tables. Still, people working in production, labs, or handling shipments of chemicals might bump into it, so understanding its safety profile matters. Most people don't come across chemicals like this by accident, but for those who do, knowing the facts can save trouble.
Direct Contact and Health Effects
If ethyl dichloroacetate lands on your skin, it can burn or irritate. The chemical also causes trouble if it splashes in your eyes—redness, pain, or worse. Think of moments in a high school chemistry class, when something splattered and everyone jumped back. This chemical won't win any prizes for being gentle. Inhalation causes coughing and sore throats. Long periods of breathing it might do more harm to the lungs and airways. Swallowing it brings nausea, abdominal pain, or vomiting.
Long-Term and Repeated Exposure
Short bursts of exposure are one thing, but regular contact can create lasting issues. Some studies point out the risk of organ damage, especially to the liver and nervous system. These conclusions usually come from animal research, but in the chemical world, these red flags ask for caution. Anyone handling the stuff on a routine basis may see skin start to dry out, crack, or develop rashes. Chemicals don't just wash off; they stick around and cause slow damage if folks aren't careful.
Accidental Spills and Environmental Concerns
Ethyl dichloroacetate isn't just about personal health; spills in a lab or in transit threaten waterways and soil. It's not smart to pour leftover chemicals down the drain. Even small leaks might pollute streams or groundwater, hitting fish and insects harder than most realize. Experience in community clean-up groups taught me how everyday spills can complicate things for entire neighborhoods. Chemicals build up and hang around, so a single careless disposal lingers for years.
Practical Safety Steps
The answer isn't to panic, but to deal with ethyl dichloroacetate with respect. Gloves, goggles, and lab coats belong in every work setting where this chemical appears. Good ventilation—open windows, fans, and fume hoods—matters as much as any safety poster ever nailed to a lab wall. Clear labeling and secure storage help keep it out of the wrong hands.
In accidents, quick first aid means flushing skin or eyes with water, then getting proper medical help. No one should try to tough out exposure, especially if symptoms get worse. Spills need chemical absorbents, not just a mop or towel, and contaminated materials should go in approved containers, never regular trash cans.
Calls for Greater Communication and Training
Employers and managers can't shrug off responsibility by tacking safety sheets on a bulletin board. Real training means hands-on demonstrations, not just paperwork. Everyone in a workplace, from janitors to supervisors, should know what’s in every bottle or barrel. Small businesses sometimes skip this or rely on outdated information. Industry oversight and community awareness play a wider role, making sure rules aren’t just written but followed every day.
Summary
Ethyl dichloroacetate poses real hazards. Good habits, clear information, and early action prevent most problems. Rushing through steps or skipping protocols never pays off. Respect for chemicals, informed by experience and science, keeps workplaces and communities safer.
| Names | |
| Preferred IUPAC name | Ethyl 2,2-dichloroacetate |
| Other names |
EDCA ethyl 2,2-dichloroacetate |
| Pronunciation | /ˈiːθɪl daɪˌklɔːroʊəˈsiːteɪt/ |
| Identifiers | |
| CAS Number | [452-00-4] |
| Beilstein Reference | 1209249 |
| ChEBI | CHEBI:4478 |
| ChEMBL | CHEMBL1230546 |
| ChemSpider | 54730 |
| DrugBank | DB04124 |
| ECHA InfoCard | 100.006.764 |
| EC Number | 205-838-7 |
| Gmelin Reference | 7890 |
| KEGG | C18933 |
| MeSH | D003373 |
| PubChem CID | 8295 |
| RTECS number | KI9625000 |
| UNII | I65E371K53 |
| UN number | UN1183 |
| Properties | |
| Chemical formula | C4H6Cl2O2 |
| Molar mass | 157.02 g/mol |
| Appearance | Colorless to light yellow transparent liquid |
| Odor | Pungent |
| Density | 1.418 g/mL at 25 °C (lit.) |
| Solubility in water | slightly soluble |
| log P | 1.44 |
| Vapor pressure | 5 mmHg (20°C) |
| Acidity (pKa) | 1.26 |
| Basicity (pKb) | Ethyl Dichloroacetate has a pKb value of approximately 12.8 |
| Magnetic susceptibility (χ) | -37.5e-6 cm³/mol |
| Refractive index (nD) | 1.423 |
| Viscosity | 1.41 mPa·s (20 °C) |
| Dipole moment | 1.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 247.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -554.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1023.9 kJ/mol |
| Pharmacology | |
| ATC code | N07XX04 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. H319: Causes serious eye irritation. H315: Causes skin irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P210, P261, P280, P301+P312, P304+P340, P305+P351+P338, P337+P313, P403+P233 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 1, Instability: 0, Special: - |
| Flash point | Flash point: 110 °C (230 °F) |
| Autoignition temperature | 206 °C |
| Explosive limits | 4.8–14% |
| Lethal dose or concentration | Lethal dose or concentration (LD50, Oral, Rat): 2060 mg/kg |
| LD50 (median dose) | 2070 mg/kg (rat oral) |
| NIOSH | AM2950000 |
| PEL (Permissible) | PEL: 10 ppm |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | IDLH: 200 ppm |
| Related compounds | |
| Related compounds |
Dichloroacetic acid Methyl dichloroacetate Trichloroacetic acid Ethyl chloroacetate Ethyl acetate |

