The Complex Story of Methyl Butyrate: Revisiting a Simple Ester With Major Impact
Looking Back: How Methyl Butyrate Found Its Place
Methyl butyrate is an ester that hits the nose like a basket of fruit—think of pineapples and apples, a sweet hint laced with the edge of chemical oddness. For anyone who grew up near orchards or worked in the food chemistry world, the aroma is instantly familiar. This compound started making waves in the late 19th and early 20th centuries, as organic chemists picked apart the secrets of fermentation and flavor. Esterification, the process that joins an acid and an alcohol, made methyl butyrate possible both in labs and in nature, giving industries an edge in adding controlled flavor to foods, beverages, and perfumes. Its role in the fragrance and food world came not because it was unique, but because of that classic, unmistakable fruity kick combined with relatively low cost and ease of production.
What Sets Methyl Butyrate Apart in Practice?
Methyl butyrate walks into most rooms as a clear, mobile liquid, and it evaporates fairly quickly. That volatility gives it punch in flavor and scent applications, but also means handling can get tricky. With a boiling point below 103°C, any heat and most breezes will carry its smell far outside the lab or production line. For all its charm, the simplicity of methyl butyrate’s physical form means it gets used up quickly—what you smell now may vanish in ten minutes. Add in poor solubility in water and a knack for mixing well with ethanol and organic solvents, and it becomes the flavor chemist’s easy shortcut for fruity notes. It stands up as a reference point for synthetic and natural flavors, a role it earned through this balance of chemistry and sensory impact.
Digging Into Chemical Properties & Behavior
The backbone of methyl butyrate is fairly simple: four carbons in the acid group, a single methyl group tacked on, and the bond that links them. This leaves it lightweight and quick to move through the air. Like other esters, methyl butyrate doesn’t survive strong acids or bases for long, reverting to its starting alcohol and acid if conditions push it. Still, under neutral or near-neutral conditions, it’s stable enough to last through storage and shipping. In many chemical labs, this resilience gives it a place not just as a flavor chemical, but as a teaching example—students watch its classical Fischer esterification and the equal ease with which it hydrolyzes back. Working hands-on with methyl butyrate means paying attention to the little cues—its colorless appearance, that sharp, sometimes overwhelming smell, and how easily it evaporates when left uncapped.
Labeling and Technical Standards
For the food and fragrance industries, label accuracy can make or break a company’s reputation. Methyl butyrate usually shows up as “artificial flavor” or “nature-identical flavor,” though regulation can vary across countries. In some places it demands tighter traceability—high purity (often exceeding 97%), clearly marked batch numbers, and rigorous documentation. Testing for residual solvents and purity also stays at the front of any production line, with gas chromatography and mass spectrometry leading the charge. Seasoned technical staff rely on sensory panels and chemical analysis in tandem to catch off tastes arising from impurities or degradation. This blend of regulation, testing, and individual judgment helps keep the product reliable, safe, and fit for whatever end use—be it food, beverage, or aroma blend.
How It’s Made: Industrial Routes and Practical Know-How
The textbook pathway for synthesizing methyl butyrate involves butyric acid and methanol, usually catalyzed by acids such as sulfuric acid. This route stands out because the raw materials are cheap and available in bulk, and the process can be run at scale without advanced equipment. Sometimes, industries push for greener methods, like enzymatic catalysis using lipases, cutting out the need for harsh chemicals and high temperatures. Both lab and plant operators keep an eye on reaction time, temperature, and purity, since side-products creep in if the conditions slip out of range. Distillation and washing finish the job, wringing water and unreacted raw materials out of the final product. These methods have served the industry for decades, and the push for greener chemistry and better process control continues to drive new developments.
Reactivity, Derivatives, and More Than Meets the Eye
Methyl butyrate doesn’t just sit in a container waiting for use—it reacts. Strong acids or bases crack it back to methanol and butyric acid, giving it a built-in reversibility. Chemists often see it as a starting point for further modification, maybe adding halogens on the acid chain or tweaking the methyl group to alter solubility or scent. These changes provide access to a range of niche flavor and fragrance molecules, each tailored for a specific market or manufacturing need. In perfumery, such modifications expand the palette, letting creative noses blend for subtle variations in top notes or lingering trails. Lab techs know that the original ester’s volatility can lead to significant loss if storage isn’t tightly controlled, and so glass bottles with tight seals and temperature control remain the rule.
Names on the Global Stage: Synonyms and Product Branding
Spend any time sifting through old textbooks, technical data sheets, or international shipping manifests, and methyl butyrate’s aliases start piling up. “Butanoic acid, methyl ester” is its full IUPAC handle, but plenty of companies stick with “methyl butanoate” or “methyl butyrate,” depending on lineage and regulatory trends. Some old-school suppliers list it as “pineapple oil” or “pineapple ester,” leaning on that sensory association. Labels for use in the food flavor world sometimes read simply “artificial fruit flavor.” Synonyms can make safety and sourcing complex—regulators, shippers, and buyers often find themselves double-checking CAS numbers to avoid confusion or costly mix-ups.
Working Safe: Handling and Operational Rules
Methyl butyrate doesn’t explode or ignite at room temperature the way gasoline does, but its vapor can catch a spark and burn. Ventilation stands out as a real necessity, especially in production rooms where one spill could turn into a strong, lingering odor and a flammability hazard. Gloves, goggles, and lab coats help keep skin and eyes out of harm’s way, especially since esters like this can cause redness or mild irritation after repeated contact. Workers pay careful attention to material safety data sheets for storage limits and best practices, and the fact that most countries classify methyl butyrate as a volatile organic compound means air handling takes on added importance. Training sessions on spill clean-up and fire response usually factor in the specific quirks of this compound—its flashpoint, evaporation rate, and sensory warning properties form the backbone of safe use guidelines.
Finding Uses: Where Methyl Butyrate Shows Up
Ask a flavorist or perfumer about where methyl butyrate fits in the real world and the answers come easily. Most recognize it as a go-to note in fruit and dairy flavors. It softens the edge of apple in juices and candies, boosts the tropical feel of pineapple in sodas, and even helps round out rum and whiskey aromas. Chewing gum, baked goods, and dairy blends often use it to fill out natural flavors or add recognizable character at low cost. Beyond food, its use in perfume and toiletries—shampoos, soaps, and lotions—grows out of its light, refreshing profile. Increasingly, it appears in research on biodegradable solvents and sustainable plastics, riding a new wave of renewable resource interest. Still, food and beverage flavoring stand as the main commercial applications, and flavor chemists rely on its predictability both solo and in complex blends.
Pushing the Boundaries: Research and Development
Academic and industrial labs never stop searching for cleaner, safer, and more sustainable ways to make and use methyl butyrate. Some recent studies experiment with engineered enzymes and microbes capable of direct fermentation, lowering production energy needs and waste. Others target modifications for flavor releases that last longer in finished products. Environmental concerns also push researchers to examine emissions and potential aquatic impacts, since this ester can travel and linger. Cooperative research grants and company-sponsored innovation seek to balance traditional mass-production with these more nuanced, eco-friendly priorities. Not all projects bear fruit, but the push toward process intensification and green chemistry remains strong, and many universities now include methyl butyrate synthesis in coursework designed to teach both commercial scale-up and environmental responsibility.
Toxicity and Health Considerations
Methyl butyrate stays low-key in toxicology circles. High concentrations can irritate eyes and skin, and prolonged inhalation in poorly ventilated spaces occasionally causes headaches or lightheadedness. Acute oral toxicity sits at a level that counts as low-risk for typical commercial and consumer uses, but large accidental exposures or improper workplace habits can tip the balance. Long-term studies look at metabolic fate; the compound typically hydrolyzes to products the body can process. Food safety authorities in North America, Europe, and elsewhere review available studies every few years, and so far, the compound keeps its place on the list of approved flavoring agents at controlled use levels. That doesn’t excuse poor handling or overuse, but evidence supports a long record of safety under regulated use.
Looking Forward: Where Methyl Butyrate Might Go
Methyl butyrate, despite its simplicity, faces a shifting landscape. Consumer demand now tilts toward natural and sustainability-certified ingredients, and companies strive to source the compound from plant materials or through cleaner fermentation methods. Synthetic biology and green chemistry open new doors, promising the same fruit-punch scent profile grown in vats of engineered yeast or captured from agricultural waste streams. At the same time, regulatory bodies tighten requirements on purity, labeling, traceability, and environmental footprint, forcing producers to rethink supply chains and end-of-life disposal. The growing interest in personal and home care products, as well as next-generation food and beverage design, will likely keep methyl butyrate relevant. My own time in flavor chemistry showed me that the drive for bolder, safer, and more sustainable flavors never really winds down; methyl butyrate’s blend of nostalgia and flexibility guarantees it keeps popping up in labs, kitchens, and perfumeries, reinvented for new generations.
What is Methyl Butyrate used for?
Getting to Know Methyl Butyrate
Think about the smell of fresh pineapples. That crisp, fruity scent often surprises people, especially after realizing it comes from more than just juicy fruit. Here’s where methyl butyrate comes into the story. This molecule, an ester formed from methanol and butyric acid, carries that sweet, tangy aroma that shows up in all sorts of places—much further than the produce aisle.
The Appeal for Food and Beverage
The food world loves flavors and scents, chasing after ways to add depth to experiences. Methyl butyrate lands in drinks, candies, and desserts. The flavor industry uses it to capture and boost the taste of apple, pineapple, and sometimes strawberry. Chewing gum often contains methyl butyrate, making every chew just a little brighter.
Label reading teaches us a lot about what's actually in processed snacks. Seeing methyl butyrate listed next to other "natural flavors" opens up a bigger conversation about what we’re putting in our bodies. While the U.S. Food and Drug Administration lists methyl butyrate as “generally recognized as safe” for food use, consumers should stay curious and informed. Artificial and nature-identical flavors often fill in for rare or expensive natural ones. Understanding what makes them tick—health impact, sustainability, origins—builds consumer trust.
Perfume and Scent Solutions
Perfume fans get an extra whiff of this compound. Methyl butyrate shows up on ingredient lists for certain fruity fresh and floral fragrances. It doesn’t just serve as a highlight; it can soften other aromas, balance notes, and boost the "uplifting" appeal of a scent. Beyond perfume, methyl butyrate adds its agreeable smell to air fresheners, cleaning products, and scented candles. Businesses rely on this compound to infuse spaces with a lively, pleasant atmosphere.
Industrial Applications and Challenges
The solvent industry also leans on methyl butyrate, especially for removing paint or in coatings. Sometimes it even crops up in the synthesis of other fragrant esters. While these uses remain less visible to the public, their demand keeps chemical suppliers on their toes.
Challenges pop up from time to time. Methyl butyrate carries flammable properties, making storage and transport an ongoing concern for manufacturers and distributors. Plant safety protocols must stay tight to avoid accidents and health risks. Factory workers also face short-term exposure limits in occupational settings, as inhaling high concentrations may irritate the eyes and respiratory system.
Looking Toward Safer, Greener Solutions
People everywhere expect businesses to source and use chemicals responsibly. Demand for sustainable sourcing pushes chemical producers to explore greener routes for making methyl butyrate. Some manufacturers now choose fermentation over petroleum-based methods, a nod towards renewable practices. These changes aim to minimize pollution, trim down carbon footprints, and lift transparency about sourcing.
Greater public awareness of what’s in everyday products shifts industry standards. Disclosing chemical origins and safety data wins customer trust. Brands who listen—opting for renewable and less hazardous ingredients—position themselves as forward thinkers in a crowded market. For methyl butyrate, that means sticking to strong safety standards while investing in better, lower-impact production.
In Everyday Life
This molecule continues to shape the tastes and scents that define daily life. Whether sweetening food, freshening up rooms, or adding flair to fragrances, methyl butyrate finds a spot in countless routines. It always pays to stay curious about how small, hidden ingredients can have a much bigger impact than most would guess.
Is Methyl Butyrate safe for human consumption?
Methyl Butyrate in Food and Drinks
Stroll through any supermarket and your nose quickly fills with the scent of fruit and candy. Behind many of those bright flavors hides a group of chemicals called esters. Methyl butyrate lands right on that list, and you find it in artificial pineapple, apple, and strawberry flavors. Food makers love it for the punch it gives to drinks, ice creams, and sweets. This isn’t one of those obscure additives—anyone who has ever chewed a fruity gum or sipped on canned pineapple juice already tasted it.
Safety Reviews and Real Risks
My curiosity about what actually goes into everyday food started as a kid. I always asked: “If it smells so sweet, what else does it do?” Like most food chemicals, methyl butyrate has a story. Authorities such as the U.S. Food and Drug Administration (FDA) gave it the green light. They put it on their list of substances “Generally Recognized As Safe” (GRAS). Researchers working for the Joint FAO/WHO Expert Committee on Food Additives also evaluated its safety, concluding the same thing under normal food-use conditions.
For a flavor compound like this, “normal food-use conditions” means amounts so tiny you’d need to drown yourself in sodas just to get close to a harmful dose. Lab studies reported that extremely high levels caused some problems in animals, but those don't resemble how people actually eat. Each time someone tests an ingredient like this, they test at levels much higher than what we could ever eat. From my own experience working with food regulations, I’ve learned that such safety margins often stretch further than most realize.
Consumer Concerns
Even with this knowledge, people still feel uneasy about chemicals in their diets. News headlines lean into fear, not facts. I’ve seen friends get suspicious, convinced that anything with an unpronounceable name must be “bad.” The truth is that methyl butyrate actually occurs naturally in pineapples, apples, and some cheeses. While its bottled, pure form gets produced in a lab for commercial use, the molecule itself is the same.
Some folks worry about allergies or chemical sensitivities. Based on current research, methyl butyrate hasn’t shown a strong link to allergic reactions. Still, monitoring continues. Anyone with unique health conditions should always pay attention to labels and talk to a doctor or dietitian, but for most people, standard food use doesn’t present a real risk.
Transparency, Trust, and What Needs Fixing
People need more honest conversations about what's actually in their food. Food makers could do a better job of explaining why they add flavor chemicals and where they come from. Too often, ingredient lists contain nothing but mystery. I would rather see a label that says “flavors derived from pineapple” than just a number or code. Truth builds trust.
Regulators and industry groups update safety reviews as new research comes out. That system works, but making the results more accessible could clear up confusion. Plain-English summaries, public Q&A sessions with food scientists, and easier-to-understand resources on ingredient safety would close the gap people feel with chemical names like “methyl butyrate.”
People deserve both good food and honest information. Food science should work for all of us, bringing bold flavors without fear. For now, evidence says that methyl butyrate, used as intended, brings more enjoyment than worry.
What are the main applications of Methyl Butyrate in industry?
Why the Food and Beverage World Trusts Methyl Butyrate
Walk into any flavor lab or food processing plant, and someone will have methyl butyrate on the shelf. This colorless liquid smells like apples and pineapples. Ever sipped a tropical soda or tasted a chewy fruit-flavored candy? Chances are, methyl butyrate helped create those bright, juicy notes. Food scientists reach for it because the compound reproduces the natural aroma of fruits that’s hard to match through other means. The United States Food and Drug Administration classifies it as generally recognized as safe (GRAS) when used in approved amounts, which opens the door for its broad adoption across snacks, ice creams, soft drinks, and more. Fruit flavors in processed goods tend to fade during heating and storage, but methyl butyrate holds up under those conditions, so that burst of apple in a packaged pastry rarely disappoints.
Perfume and Fragrance Industry Uses
Perfumers know that even a minuscule dose of methyl butyrate adds a lift to floral and citrus notes. It helps craft the first impression you get from a new cologne, a shampoo, or even an air freshener. Since the human nose links the sweet, juicy aroma to freshness and cleanliness, companies use it as a top note in everything from laundry products to floor cleaners. Fragrance houses that develop scents for consumer products rely on methyl butyrate to brighten up the blend and make it stand out. For people who follow ingredient safety, methyl butyrate has a strong record. Researchers have not flagged it as a skin irritant or carcinogen at normal levels, which matters because repeated exposure through personal care products adds up.
Industrial and Laboratory Roles
Factories use methyl butyrate as a solvent, especially in paint removers and cleaners where they need something that breaks down grease but leaves behind less toxic residue. Unlike many alternatives, it comes from renewable sources—fermentation of sugars or synthesis from natural alcohol and acids—so it aligns with current efforts to make manufacturing greener. Scientists also rely on it for analytical research, where its distinct aroma and chemical fingerprint help test the accuracy of instruments and processes. In the classroom, methyl butyrate serves as a go-to example during lessons about esters, letting students recognize the power of chemistry to create recognizable scents out of simple building blocks.
Inevitable Challenges and Safer Handling
Demand for methyl butyrate rises every year, but supply keeps up because the process to make it is straightforward. Still, factory-scale synthesis needs careful oversight: at high concentrations, the compound can irritate eyes and airways. Workers in flavor houses and chemical plants wear masks and gloves, and air circulation systems keep fumes down. Firms in the business must commit to responsible storage and disposal because any spill risks both health and environmental damage. The chemical disperses rapidly, but repeated overuse around waterways could affect aquatic organisms’ health. I’d like to see tighter regulatory tracking and more investment in closed-loop systems, capturing vapors and recycling them before they drift into the air.
Moving Toward Sustainable Practices
Production methods for methyl butyrate now rely more on fermentation and less on petroleum sources. Brands with sustainability on their minds can choose material made from waste biomass, supporting a circular economy. Those choices don’t just shrink a company's carbon footprint. They reflect a growing expectation among consumers for transparency about where flavor and fragrance ingredients come from. Personally, I look for more companies to share their sourcing and sustainability stories, especially for everyday products. Supporting renewable chemistry brings a bigger payoff than just a pleasant-smelling soda.
What does Methyl Butyrate smell like?
The Surprising Familiarity of Methyl Butyrate’s Scent
Plenty of folks come across sweet-smelling chemicals every day and never know their names. Take methyl butyrate for example. This stuff turns up in places you’d never expect, slipping quietly into the air with a scent that most people already know. Its aroma—tart, fresh, and undeniably fruity—calls up memories of biting into a ripe apple or tearing open a pack of pineapple-flavored sweets. The scent carries a bright, juicy edge, a little like pineapple and apple rolled together with just a hint of fresh pear. Catch that first whiff and you’ll see what I mean: it smells like summer picnic baskets, the kind of smell that comes from fruit that’s just starting to give off a sugary perfume.
Where the Aroma Shows Up in Life
Food scientists count on methyl butyrate’s cheerful scent to punch up flavor in candies, gums, drinks, and baked goods. It tricks the senses into thinking dessert is packed with real fruit. Technically speaking, methyl butyrate is an ester—the same family that gives us the scents of bananas, strawberries, and other juicy snacks. It blends well with sugar notes, helping low-sugar and “natural flavor” recipes taste and smell fresh. Even in small doses, its aroma stands out. Scent panels across the globe, from flavor houses in the U.S. to food labs in Europe, confirm that this compound tops the list for recreating those genuine, mouth-watering fruit aromas.
Behind the Scenes: Natural and Lab-Made Sources
Apples, pineapples, and a handful of other fruits produce methyl butyrate in small amounts. Scientists also whip it up in laboratories using simple chemical reactions between methanol and butyric acid. That means even if a product claims to have “natural flavors,” those fruity high notes could very well come from this carefully made compound. Natural methyl butyrate often costs more than the synthetic version. For large food companies, lab-made versions keep prices in check and quality consistent. Regulatory agencies like the FDA consider methyl butyrate generally safe when used within recommended limits, based on plenty of research about its effects and its long history in food flavoring.
Scent Memories and Consumer Health
Methyl butyrate’s scent doesn’t just stop at food. Anyone who’s worked around perfumes or air fresheners comes across it, even if they don’t recognize the name. Its sharp, cheerful notes lift citrus-heavy colognes and help mask some of the less pleasant odors in cleaning products. Some people with sensitive noses pick out its “overripe” notes, especially at higher concentrations, which reminds them of spoiled fruit instead of fresh. That’s worth considering for anyone mixing fragrances or building home products. If a product overuses it, the effect skews fake, cloying, or even off-putting.
Smart Use and Safety First
Most people want what’s real and safe in their snacks and scents. Responsible companies test flavor levels before new products hit the shelves, making sure nothing slips past quality control. Consumers who prefer natural ingredients can look for clearer labeling and reach out to brands for sourcing details. Regulators keep an eye on safety, updating recommendations based on new studies. That kind of oversight protects everyone—kids digging into juice boxes, families cooking with commercial mixes, and adults rubbing on a spritz of fruity cologne.
How should Methyl Butyrate be stored?
Understanding the Real Risks
Methyl butyrate sometimes gets treated like a casual chemical, probably because it smells a bit like pineapple and flavors candies and drinks. That familiar, fruity scent hides a harsher reality though. Anyone who’s worked around solvents quickly learns: sweet smells and “natural” connections never mean harmless. Methyl butyrate catches fire easily, forming vapors that can spread tough-to-see risks throughout a workspace. Inhaling too much creates dizzy spells and headaches. Accidental spills can sneak into the air faster than you might expect. Safety calls for real attention, not just labels and checklists.
The Importance of a Proper Container
Quality storage starts with the basics: strong, tightly sealed bottles or drums built for organic liquids. Glass containers offer chemical resistance, but thick, high-density polyethylene works too. Metal cans can corrode with time from certain esters, so routine checks matter. Mismatched caps or poor seals trigger leaks and bigger headaches. The law doesn’t always spell out exactly which bottle for every chemical, but as a rule, choose one labeled safe for flammable solvents.
Label every container, even those temporary holding jars for research or mixing. I’ve seen busy labs go sour, literally and figuratively, when someone assumed an unlabeled jar was harmless. Proper labeling—the chemical name, concentration, and the date it landed on the shelf—pays off daily in safety and stopping mistakes before they gain momentum.
Fire, Light, and Fume: Environmental Traps
A few misplaced bottles near a heat source can spell disaster. Methyl butyrate ignites at only about 64°C (147°F). Heating vents, windows with steady sun, and even small appliances in the wrong corner turn an oversight into a full-blown hazard. I recall one cramped storeroom where two solvents actually shared a shelf beside a hot water pipe. The fix? Relocate both, update the risk assessment, and write a sticky note so the next shift doesn’t repeat the slipup. Stash methyl butyrate away from flames, sparks, and anything that runs warm for long stretches.
The vapor side of things gets overlooked in smaller workspaces. This chemical sends off fumes that shouldn’t stay unventilated. A well-fitted cap reduces risk, but storage in a proper fume hood or ventilated cabinet keeps air quality in check. Some labs in older buildings rely on open shelving, hoping the draft from a cracked window will do the job. It doesn’t. Investing in real ventilation pays for itself in avoided accidents, especially where flammable vapors could build.
Community and Personal Accountability
No policy or rulebook replaces personal habits. Methyl butyrate can disrupt the chemical balance of a shared space if one user gets careless. Spilled drops or drips spread through gloves and towels and easily go unnoticed. I always recommend a quick check for sticky residue before leaving a storage area—old spills can leave slick patches that both smell and burn if ignored. Training new team members to respect solvents right from the start doesn’t just follow the rules; it protects everyone in the room.
Big industrial plants install fire suppression automatically, but many research labs must improvise. Simple solutions work best. Keep small quantities on the bench, with the bulk sealed in designated fireproof cabinets. Even at home—say for flavor research or teaching—store only as much as you can use in a session and keep it well out of reach of young hands. Using a chemical, even one with a flavor connection, gives responsibility. Good storage builds that trust, bottle by bottle, shift by shift.
| Names | |
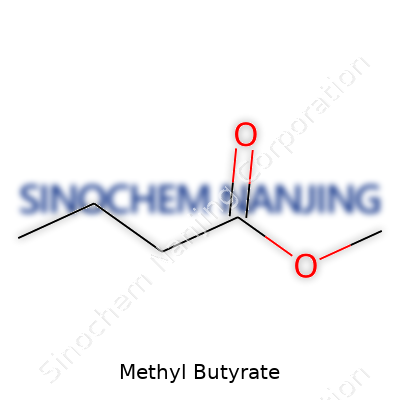
| Preferred IUPAC name | methyl butanoate |
| Other names |
Butanoic acid methyl ester Methyl butanoate Methyl n-butyrate |
| Pronunciation | /ˈmɛθ.ɪl ˈbjuː.tɪ.reɪt/ |
| Identifiers | |
| CAS Number | 623-42-7 |
| 3D model (JSmol) | `CCCC(=O)OC` |
| Beilstein Reference | 1209246 |
| ChEBI | CHEBI:7778 |
| ChEMBL | CHEMBL31860 |
| ChemSpider | 54860 |
| DrugBank | DB04256 |
| ECHA InfoCard | 100.068.262 |
| EC Number | 123-86-4 |
| Gmelin Reference | 8050 |
| KEGG | C12533 |
| MeSH | D008763 |
| PubChem CID | 7895 |
| RTECS number | EL9100000 |
| UNII | HZ4N5HTF6O |
| UN number | UN3272 |
| Properties | |
| Chemical formula | C5H10O2 |
| Molar mass | 102.13 g/mol |
| Appearance | Colorless liquid with a fruity odor |
| Odor | fruity; pineapple-like |
| Density | 0.879 g/mL at 25 °C (lit.) |
| Solubility in water | 6.1 g/L (20 °C) |
| log P | 1.96 |
| Vapor pressure | 40 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 25 |
| Basicity (pKb) | 15.10 |
| Magnetic susceptibility (χ) | -7.74×10⁻⁶ |
| Refractive index (nD) | 1.400 |
| Viscosity | 0.516 cP (25°C) |
| Dipole moment | 1.78 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 237.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -429.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2400.6 kJ/mol |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P370+P378 |
| Flash point | -10 °C |
| Autoignition temperature | 475 °C |
| Explosive limits | 1.5% - 8.0% |
| Lethal dose or concentration | LD50 oral rat 6,750 mg/kg |
| LD50 (median dose) | LD50 (median dose): 6.6 g/kg (rat, oral) |
| NIOSH | SAF22150 |
| REL (Recommended) | REL (Recommended): 5 ppm |
| IDLH (Immediate danger) | 1700 ppm |
| Related compounds | |
| Related compounds |
Methyl acetate Ethyl butyrate Butyric acid Methyl propionate Propyl butyrate |

