The Story of Methyl Acetate: Chemicals in Daily Life and Industry
Tracing the Path: Historical Development
Looking back at the story of methyl acetate, the journey follows the larger history of chemistry itself. Chemists in the nineteenth century identified esters like methyl acetate while exploring the conversion of alcohols and acids. This compound made its mark in the era when the world gradually shifted from relying heavily on natural solvents—think turpentine and ethanol—to synthetic alternatives. That shift didn’t just mean progress in lab techniques; it also connected to how industries responded to public needs for safer, reliable, and less expensive solvents. Personal experience working with experienced lab technicians shows how methyl acetate always turns up in the background of most projects involving paints or coatings, making it something of a quiet workhorse, shaped by a long cycle of laboratory curiosity and industrial demand.
Understanding Methyl Acetate: What It Really Is
Methyl acetate really acts as a bridge between the worlds of alcohol and acid chemistry: a lightweight liquid, clear and quick-evaporating. Most people outside industrial labs or paint shops never think of it directly, but anyone who’s painted a room, used nail polish remover, or cleaned up sticky messes with professional products has likely benefited from its properties. This colorless liquid, with a pleasant, sweet smell, dissolves many resins and oils—traits that reward innovation in product design. It doesn’t hang around in the air or leave residue, making it a favorite where a fast turnaround matters.
Physical and Chemical Character
Working with methyl acetate, one thing becomes clear: its volatility changes the pace of any project. Boiling just above room temperature, it evaporates so fast that open containers can empty before you notice. Flammable and lighter than water, this property always draws safety discussions, especially with techs who learned tough lessons about well-ventilated spaces. Its miscibility with many common solvents, yet low water solubility, keeps storage and mixing straightforward, but the substance calls for close attention to safety data sheets. Chemically, the ester group sits front and center, rendering it reactive enough for lab work, but not so aggressive that it disrupts most manufacturing pipelines.
Standards: Technical Specs and Labeling
Regulations pay close attention to methyl acetate. From direct experience in compliance audits, labeling stands out as the main focus: clear hazard identification, flammability warnings, and precaution symbols assigned by organizations such as OSHA and GHS. Industry standards, set by institutions like ASTM, outline specifications covering purity, allowable water and alcohol residues, and signature odor thresholds. All these rules serve both to protect workers and to keep product output consistent, driving home the point that chemistry never lives in a vacuum—it’s part of a system of checks, balances, and human responsibility.
How Methyl Acetate Comes Together: Preparation Methods
Synthesis methods for methyl acetate usually stick to one main playbook. Most production lines use the reaction between methanol and acetic acid. Acid-catalyzed esterification gives high yields, especially when a dehydrating agent extracts water from the product, shifting equilibrium toward methyl acetate. Scaling up from flask to factory, distillation removes impurities, leaving a clean, fast-evaporating liquid. Being around chemists with decades of hands-on experience, you realize just how much pressure comes with balancing reaction time, yield, and cost. Each factor shapes decisions about batch size, raw material sourcing, and energy use, bringing economics as close to the reaction as the glassware itself.
Chemical Reactions and Industry Tweaks
Methyl acetate stands ready for further modification. Labs often use it as an intermediate, where hydrolysis returns it to its roots—methanol and acetic acid—or where it undergoes transesterification to adapt for industry-specific uses. Chemists find methyl acetate particularly handy for reactions that demand a gentle solvent: enough volatility to remove itself from the reaction vessel, enough chemical stability not to interfere with sensitive intermediates. The slight possibility of accidental saponification when exposed to strong base always sparks lively debate in planning meetings, underlining the ongoing learning process that good chemistry departments live by.
Synonyms: Many Names, Same Substance
Scientific traditions give methyl acetate a string of alternative names—methyl ethanoate, acetic acid methyl ester, with entries in catalogues and safety lists as MeOAc or C3H6O2. Each name traces back to the same molecular structure, but knowing the lingo helps avoid surprise results in cross-border shipments or regulatory reviews. Colleagues swapping stories from international projects highlight that language, tradition, and chemical code all merge when it comes to labeling and customs forms—a small reminder of chemistry’s global scope.
Meeting Safety and Operational Standards
Caution frames every use of methyl acetate, from small-scale laboratory tests to drums rolling across factory floors. Being flammable, storage stays limited to cool, well-ventilated locations with stringent controls on ignition sources. Workers use protective gloves and eye shields, along with vapor extraction systems—a practical routine learned the hard way after a few close calls in cramped storerooms. Safety audits focus on spill control, personal protective equipment, and emergency eye-wash stations. Proper chemical hygiene isn’t negotiable here: training, refreshed regularly, prevents incidents that leave a lasting mark on both individuals and organizations.
Real-World Uses: Where Methyl Acetate Shows Up
The chemical shows its value best in the real world: as a solvent in inks, adhesives, and coatings, and as a replacement for more toxic solvents like toluene. Its environmental advantages have grown in importance for companies aiming to shrink their ecological footprint. In the electronics sector, methyl acetate provides a safer cleaning method during assembly stages. Automotive and furniture manufacturers praise its low toxicity and high evaporation rate, both of which help speed up production without trading off safety for efficiency. As a bonus, nail polish removers with methyl acetate generate fewer customer complaints about skin irritation, based on survey responses seen in product testing groups.
Research and Innovation: Pushing Boundaries
Research teams keep extending the chemical’s reach. New synthesis techniques conserve energy, reduce emissions, and improve purity. Green chemistry specialists investigate biocatalytic production, seeking out fermentation or enzyme-driven routes that could cut fossil resource use. Focused work on recycling solvents includes methyl acetate as a candidate for capture and re-use, which would decrease pollution across manufacturing cycles. Universities partner with large chemical firms to benchmark exposure safety and find new applications, showing that the chemical’s story is far from static—it continues to evolve alongside changing priorities in science and industry.
Can We Trust It? Toxicity and Safety Data
Toxicology studies run deep because public trust in chemicals depends on hard facts and transparency. Acute exposure to methyl acetate can cause eye, skin, and respiratory irritation, but long-term studies point to low systemic toxicity compared to legacy solvents like methylene chloride or benzene. Safe limits for workplace air, based on peer-reviewed studies and regulatory review, aim to strike a fair balance between health protection and operational reality. Having worked on audits and accident reviews, most incidents follow from lapses in training rather than unknown hazards. Environmental impact gets regular review, with methyl acetate’s rapid breakdown in sunlight and low persistence leading to ongoing endorsement from safety boards—so long as companies keep containment, recycling, and downstream monitoring practices strong.
Future Prospects: What Comes Next?
Looking at the next generation of solvents and specialty chemicals, methyl acetate stands to grow in importance because of its environmental record and technical reliability. As industries phase out substances that leave long-term pollution or face stricter regulation, the need for safer choices intensifies. Market watchers predict growth in electronics, coatings, and environmentally leveraged product lines, especially as consumer preferences tilt toward products with lower toxicity and smaller carbon footprints. Research underway today—across catalytic conversion, waste stream processing, and integrated recycling—could reshape the limits of what methyl acetate can do. The same combination of curiosity and caution that shaped its early history keeps pushing the chemical forward into new areas, grounded by practical lessons learned from hands-on experience over generations.
What are the main uses of Methyl Acetate?
How Methyl Acetate Powers Industry
Methyl acetate shows up in more places than most people would expect. Every day, plenty of folks open a can of spray paint or a bottle of nail polish remover without realizing what’s inside. Methyl acetate makes those products work. It’s a strong solvent—think of it as the grease that keeps industrial wheels turning.
Paints and Coatings: Where It Shines
Walk past a house getting a new paint job or notice a car shining under dealership lights. The paint dries smooth because of solvents like methyl acetate. In the coatings world, it takes speed and consistency to keep up with modern spray systems and production lines. Methyl acetate stands out thanks to its fast evaporation and low toxicity profile. It thins lacquers just right, so the surface dries evenly and the next layer goes on without a hitch. Not all solvents can promise quick turnaround and easy handling—methyl acetate delivers both.
Adhesives: Sticking Power with Cleaner Air
Think about furniture assembly or shoe manufacturing. Glues and adhesives have to bond quickly and reliably, but harsh solvents make workspaces uncomfortable. Methyl acetate cranks up production speed, but it’s also less harsh on the nose and lungs than several older alternatives. This matters for workers spending all day gluing parts together. Cleaner air means healthier teams, which is why plenty of manufacturers make the switch.
Electronics: Making Small Parts Possible
Opening up a laptop or smartphone, you’ll find parts barely bigger than a grain of rice. Circuit board manufacturing depends on solvents that don’t leave behind residue or cause damage. Methyl acetate comes into play when cleaning delicate pieces and prepping them for coating. Its ability to break down greases while vanishing quickly gives electronics makers an edge. Industry experts have pushed for safer, low-residue options for years, and methyl acetate holds up well under tight regulations.
Personal Care and Household Products
A fresh manicure or streak-free glass cleaner leans on methyl acetate, too. Many nail polish removers and aerosols depend on it because it works fast without leaving strong odors. It’s often picked for air fresheners and cleaning products to boost performance with fewer chemicals left behind. Recent safety tests (like those reviewed by the EPA and EU) have placed methyl acetate on a better footing than tougher solvents like acetone or toluene.
Environmental Gain and Safer Handling
Methyl acetate breaks down quickly in the environment. This speed translates to less buildup in soil or water sources, reducing risks compared to longer-lasting chemicals. Businesses feel pressure these days to cut volatile organic compounds, searching for working alternatives that won’t fall foul of changing rules. Using methyl acetate can help them cut total emissions and keep up with environmental commitments.
Looking Ahead: Finding Balance
Methyl acetate shows up because it solves problems—speed, safety, and clean results. Smaller companies still struggle with high solvent prices and changing regulations, but switching to methyl acetate can often offer both performance and peace of mind. Safer alternatives lead to healthier workers and cleaner products. Having handled chemicals in both shop-floor and lab settings, I know how much daily exposure adds up. Methyl acetate gives industry a chance to make that load a bit lighter, if handled with proper respect and training.
Is Methyl Acetate hazardous to health or the environment?
Understanding Methyl Acetate and Everyday Exposure
Methyl acetate shows up in many places—factories, garages, sometimes even in nail salons. It serves as a solvent in paints, coatings, adhesives, and some cleaning products. People who work with industrial chemicals or spend hours in workshops will recognize the fruity, somewhat sweet odor. In my years around chemical storerooms and with painters, the stuff has a knack for filling a space fast. Breathing it in isn’t pleasant, either—not because of the smell, but because it leaves you feeling lightheaded or gives a sore nose with enough exposure.
Health Risks in Real Life
Short spells of inhaling methyl acetate bring on headaches, dizziness, or a scratchy throat. Go beyond that or skip decent ventilation, and the irritation kicks up to nausea or a bad cough. My old supervisor drilled into us to avoid getting vapors trapped in a small room. Mask or not, you feel the difference. For anyone managing asthma or sensitive lungs, a simple whiff could spell hours of discomfort.
It doesn’t hang around in your body for long—the liver breaks it into harmless materials pretty quickly. That’s a relief compared to other solvents that cause long-term trouble. Still, folks who work with it daily need respect for the material. Too much exposure strips oils from skin, leads to dryness or sometimes mild rashes, especially among people who tend to skip gloves. Direct splashes in the eyes burn and demand a trip to the eyewash immediately.
Environmental Concerns
Methyl acetate is volatile, so it evaporates quickly. That trait helps finish a project faster, but it also carries fumes into the open air. For big plants or auto shops, this adds to smog-forming pollution, trading indoor hazards for outdoor air problems. Over weeks, those vapors take part in reactions that build ozone at ground level, which then finds its way into public health and air quality reports.
Waterways don’t fare much worse, because methyl acetate breaks down pretty quickly in water, too. Fish or plant life barely get time to notice it before bacteria and sunlight change its makeup. That said, careless disposal from factories or spills that hit storm drains shows up as a chemical spike. Local rivers can’t handle repeated loads, so dumping never counts as a safe choice. I’ve watched environmental techs panic over drums draining near creeks, knowing the temporary jolt still matters.
Practical Solutions
Industry has moved toward better ventilation in shops and enforced mask policies. Offices that rely on cleaning crews reach for safer alternatives unless absolutely necessary. Local rules about air emissions limit how much of these chemicals can drift out of a property. People who respect safety data sheets and treat chemicals the way professionals recommend have fewer incidents—no surprise there.
Recycling becomes possible in larger workshops, reducing overall waste by distilling used solvent. Places that adopt closed handling systems (automatic paint booths, enclosed mixing stations) cut down on vapor exposure and leaks. The gear can look expensive, but compared to missed work from chemical headaches, many shops see it as a smart investment.
For health and environmental safety, nobody expects methyl acetate to disappear overnight. The key rests with education, proper safeguards, and a culture that never shrugs off exposure as “just part of the job.”
What is the chemical formula and structure of Methyl Acetate?
Getting to the Heart of Methyl Acetate
Walk through any hardware store, and you’ll spot shelves filled with paints, solvents, and cleaners. In the fine print, you might catch mention of methyl acetate. This isn’t just another obscure lab chemical. This compound shapes much of what we use, from quick-drying nail polish removers to the adhesive that holds together packaging. Scientists and manufacturers rely on its formula and structure for a reason.
Chemical Formula: No Mystique Here
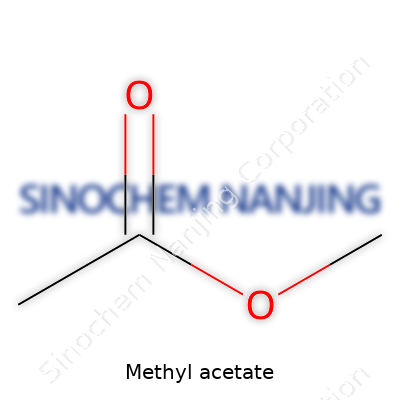
Methyl acetate goes by the formula C3H6O2. In other words, each molecule contains three carbon atoms, six hydrogens, and two oxygens. Break it down: one acetyl group bonded to a methoxy group. This isn’t just dry algebra; knowing the formula helps anyone handling this chemical to predict its behavior, assess risks, and find opportunities for safe usage.
Structure: The Real-World Building Blocks
Look at its structure, and things snap into focus. The molecule has a backbone formed by linking a methyl group (CH3) to an acetyl group (COO). The full structure, commonly sketched as CH3COOCH3, shows how the oxygen forms a bridge between two carbon atoms. This set-up creates what chemists call an ester. That ester bond shapes the way methyl acetate dissolves other compounds and the way it smells.
Why Structure and Formula Matter in Life and Industry
Real-world applications rely on this design. For anyone who has worked on a renovation project, removing old paint safely and efficiently often means picking the right solvent. Methyl acetate’s molecular size and the ester bond let it evaporate quickly, strip paint, and leave surfaces dry. Industries making coatings, inks, and adhesives turn to it because the structure delivers the right balance between drying speed and effective cleaning.
Public health gets involved, too. Its volatility means workers and hobbyists face inhalation risks. The formula lets safety experts calculate exposure thresholds, while its structure predicts how fast it leaves the body. Studies show managing air concentrations below recommended limits reduces risks for headaches and dizziness. Facilities invest in ventilation, not just because it’s code, but because experience confirms the math: safe air pays off.
Challenges and Ways Forward
Scrutiny tends to follow chemicals that see widespread use. Disposal often comes up: methyl acetate doesn’t persist in soil or water for long, but spills matter. Community experience, especially near manufacturing plants, highlights the need for faster spill response and proper waste capture. Simple collection systems and robust emergency plans can handle most leaks. Regular training keeps teams sharp.
Safer alternatives fuel debate in some circles. For certain jobs, water-based products cut down risk. But anyone in finishing trades will tell you: some tasks still need the performance methyl acetate offers. The conversation in labs and boardrooms keeps circling back to smart controls and personal protective equipment, not blanket bans.
Experience Teaches the Real Lessons
Industry veterans won’t forget the sharp, sweet scent or how fast this stuff dries under a fan. Those small details come from the molecule’s shape and formula. Over decades, understanding these basics has cut accidents and improved quality. Science, government, and everyday users make better calls when they focus on the fine structure instead of just names and numbers.
How should Methyl Acetate be stored and handled safely?
Understanding the Risks
Methyl acetate often pops up in labs and factories, showing up in paints, adhesives, and coatings. Its fruity smell might trick some into thinking it's harmless. That definitely does not hold true. People might brush off the danger because methyl acetate evaporates quickly and seems easy to work with, but anyone who’s spilled some knows the sharp fumes can fill a room in minutes. Breathing in a bit makes your head spin and eyes water, and just a spark nearby can create a real problem—since this stuff catches fire with just a little heat.
No Substitute for Good Ventilation and Common Sense
Personal experience tells me that relying on an open window or a dinky fan won't cut it. A good exhaust system helps pull fumes away from workers and out of the building fast. If you’re mixing or pouring, do it under a fume hood or in a paint booth. NIOSH recommends exposure limits for a reason; breathing methyl acetate day in and day out leads to headaches and, over long periods, even nerve damage. Proper air flow keeps both people and product safe.
Storage: Don’t Treat It Like Just Another Solvent
It might be tempting to stash methyl acetate wherever there’s space, but that can come back to bite you. Flammable liquids should go into a proper flammable storage cabinet. That means metal cabinets with self-closing doors, kept away from exits and sources of ignition. I’ve seen situations where storing it near oxidizers led to bigger issues during inspections or, worse, near-misses. Plain metal drums or tightly sealed containers are the minimum—leaky caps can turn a storeroom into a bomb.
Handling: Don’t Skimp on Safety Gear
Gloves, goggles, and even a respirator aren’t just for appearances. I’ve poured methyl acetate with thin latex gloves and watched them wrinkle up and weaken. Use nitrile gloves and goggles that wrap around the head so fumes can’t sneak in. If there’s a risk of splashing, long sleeves matter too—a quick spill can burn your skin before you even rinse off. Fire extinguishers—type ABC, preferably close by—belong near every workstation.
Training Makes All the Difference
People who know the hazards work smarter. Regular training means workers learn what to do during spills or fires, where to find the spill kit or eyewash, and how to recognize symptoms of exposure. Fact sheets aren’t just for show. OSHA guidelines spell out what needs to be covered, but nothing beats walking someone through a practice drill and pointing out real-life mistakes that have happened in other workplaces.
Getting the Basics Right
Good labeling helps prevent mix-ups. Every drum and bottle must display a clear hazard warning, not just a chemical name written in marker. Keeping incompatible chemicals apart also reduces chances of surprise reactions. Regular checks for leaks or corrosion on containers can catch trouble before it gets expensive. Spills should be cleaned right away using materials that soak up solvents but don’t ignite easily.
The Bottom Line
Having worked in places where corners got cut, I’ve learned that respect for chemicals like methyl acetate isn’t paranoia—it’s plain common sense. Smart handling, good gear, and solid habits save lives and money. When safety is second nature, accidents take a back seat.
What are the common physical and chemical properties of Methyl Acetate?
What is Methyl Acetate?
Methyl acetate ranks among the more familiar organic solvents anyone using paints, adhesives, or coatings may have encountered. Also called acetic acid methyl ester, this liquid serves a many-sided role in both industry and laboratories. Its clean, sweet odor rarely gets missed, especially by folks in manufacturing settings.
Physical Properties Worth Noting
Methyl acetate lives as a colorless liquid at room temperature. It pours easily thanks to a low viscosity and tends to evaporate with surprising speed. On hot days, its volatility becomes clear—open a tin and the scent fills the space right away. The boiling point sits around 57°C, lower than many common solvents, making it a quick-drying option that appeals to those who want a faster turnaround.
Anyone working in a spatially closed shop gets to know this compound’s flash point. Methyl acetate flashes at about -10°C, so precaution with heat or sparks pays off. Fire risk goes hand-in-hand with flammable vapors. This property deserves real respect, especially in dusty workshops or spots with poor ventilation.
Water doesn’t completely mix with methyl acetate, but the two get along better than most esters. In practical terms, if a spill happens, a fair share dissolves, the rest floats above water. That partial solubility makes cleanup different from more hydrophobic solvents. At the same time, those handling product blending find methyl acetate’s compatibility a benefit, especially for thinning lacquers or tweaking viscosity in spray paints.
Chemical Properties and Why They Matter
Methyl acetate belongs to the ester family, the result of bringing together acetic acid and methanol. It’s not just a mix; it’s a true reaction—a detail that matters in chemical recycling or industrial processing. In moisture-rich air, it slowly hydrolyzes back to its building blocks, methanol and acetic acid, which reveals something: improperly sealed storage containers can suffer, as hydrolysis shortens shelf life and may corrode tanks over time.
Another detail that stands out is its relatively low reactivity. Methyl acetate usually avoids reacting with metals or plastics, except for strong bases or acids, which pull it apart more quickly (a common headache in waste streams that see industrial chemicals mixed together). This limited reactivity suits its role in production of inks, adhesives, and cleaning fluids, where strong solvents might chew up sensitive resins.
For workers, vapor inhalation counts as a real concern. Regulatory groups advise keeping exposure levels in check. Anyone dealing with mixing, spraying, or large-scale handling should have solid ventilation and break areas clear of solvent vapor—a lesson many in factories learn early.
Toward Safer and Smarter Use
Methyl acetate’s widespread use keeps it in circulation, especially for those seeking substitutes for costlier or more hazardous solvents. It makes processes move faster, offers a reliable alternative in manufacturing, and helps modern industries keep up with customer demands. Seeking greener chemistry, companies look at methyl acetate because it breaks down more rapidly in the environment compared to other solvents like toluene or xylene.
Across the board, clear labeling, employee training, and up-to-date safety data sheets help keep hazards to a minimum. Storage in cool, well-ventilated spaces pays off, cutting down on losses and keeping risks away. In my view, the more people understand what’s in the drum or bottle they handle, the better their decisions on the floor.
| Names | |
| Preferred IUPAC name | Methyl ethanoate |
| Other names |
Acetic acid methyl ester Methyl ethanoate Methoxycarbonyl methane |
| Pronunciation | /ˈmɛθ.ɪl ˈæs.ɪ.teɪt/ |
| Identifiers | |
| CAS Number | 79-20-9 |
| 3D model (JSmol) | COC(=O)C |
| Beilstein Reference | Beilstein Reference: 1731244 |
| ChEBI | CHEBI:7926 |
| ChEMBL | CHEMBL14004 |
| ChemSpider | 5775 |
| DrugBank | DB14185 |
| ECHA InfoCard | 100.000.681 |
| EC Number | EC 203-772-9 |
| Gmelin Reference | Gmelin Reference: 76047 |
| KEGG | C01387 |
| MeSH | D008736 |
| PubChem CID | 6584 |
| RTECS number | AI0175000 |
| UNII | 9G82F0S63E |
| UN number | UN1231 |
| Properties | |
| Chemical formula | C3H6O2 |
| Molar mass | 74.08 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | fruity |
| Density | 0.93 g/cm³ |
| Solubility in water | 25.0 g/100 mL (20 °C) |
| log P | 0.18 |
| Vapor pressure | 173 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 25 |
| Basicity (pKb) | pKb = 11.5 |
| Magnetic susceptibility (χ) | -7.83×10⁻⁶ |
| Refractive index (nD) | 1.360 |
| Viscosity | Viscosity: 0.37 cP (at 25°C) |
| Dipole moment | 1.78 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 283.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -413.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -874.0 kJ/mol |
| Pharmacology | |
| ATC code | V04CX19 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P233, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | -10 °C |
| Autoignition temperature | 454°C |
| Explosive limits | 3.1% - 16% |
| Lethal dose or concentration | LD50 (oral, rat): 6,482 mg/kg |
| LD50 (median dose) | 6,468 mg/kg (rat, oral) |
| NIOSH | NA2358 |
| PEL (Permissible) | 200 ppm |
| REL (Recommended) | 200 ppm |
| IDLH (Immediate danger) | 3100 ppm |
| Related compounds | |
| Related compounds |
Acetic acid Methanol Ethyl acetate Methyl formate Methyl propionate |

