The Story of Ethyl 2-Hydroxypropionate: Insight Beyond the Surface
Tracing the Roots: How a Simple Ester Found Its Place
In everyday laboratories and manufacturing plants, few chemicals carry the lingering influence of time quite like ethyl 2-hydroxypropionate. I remember my first year working at a pilot lab, where the odd, slightly sweet odor of fresh esters marked every surface we touched. Ethyl 2-hydroxypropionate is no newcomer; its path winds back to early explorations of hydroxy acids and their role in fermentation science. Chemists in the late nineteenth and early twentieth centuries sought to break down lactic acid and manipulate its derivatives, chasing answers to food preservation and green solvent syntheses long before “green chemistry” caught hold. Its age-old cousin, lactic acid, ruled the dairy world, but the simple tweak of adding an ethyl group unexpectedly opened a new range of possibilities — from pharmaceuticals to specialty coatings.
Understanding What You’re Dealing With
Getting your head around this molecule is not an academic exercise for most workers in the field. With a molecular formula of C5H10O3, ethyl 2-hydroxypropionate easily reminds any first-year chemist of its roots with lactic acid, now given a more hydrophobic character thanks to the ethyl group anchoring one end. It's clear and colorless in most preparations, sporting a mild, ester-like scent that gives it away to anyone who has ever worked with organic solvents. The liquid form handles well at room temperature. Boiling at about 155 to 160 °C and mixing freely with many organic solvents and water, it quickly became a staple in both bench chemistry and industrial setups.
Looking Past the Label: Numbers Tell a Story
I have watched the confusion that technical specifications can cause, especially outside the research community. Yet, looking beyond stuffy labels and numbers, certain core facts remain worth knowing: it boasts a density a touch above water, around 1.07 g/cm³, and usually reaches purity levels well above 98% in commercial samples. Its refractive index and viscosity set it apart as a choice for technical coatings and advanced polymer research. Labels spell out flammability and health warnings, but they also reflect tightening regulations on solvent purity and impurity profiling. Product labeling now demands traceable sourcing and appropriate hazard communication, a change many labs have wrestled with in the last few years — it’s about more than just safety, it’s about accountability.
The Making of Ethyl 2-Hydroxypropionate
Production has evolved from rusty glassware to high-throughput, continuous reactors. A common route pairs lactic acid with ethanol under the watchful eye of a strong acid catalyst, coaxing them to yield ethyl 2-hydroxypropionate and water in a straightforward esterification. The process looks simple on paper but rewards careful balancing: keeping water away from the reaction mixture and selecting a catalyst that won’t spawn unwanted byproducts. Distillation often separates the product from a tangle of possible side reactions, and many shops work hard to minimize energy costs and waste generation. The story here is constant improvement, quicker reactions, purer product, and less environmental toll—a real challenge in industrial chemistry, not just a slogan for marketing teams.
It’s Not Just a Name: Synonyms and Assorted Aliases
Anyone who has tried to order or reference ethyl 2-hydroxypropionate in databases knows the headache of chemical synonyms. You will find it tagged as ethyl lactate through the majority of literature, but “2-hydroxypropionic acid ethyl ester” sometimes sneaks in. Pharmaceutical formulas and agricultural references cycle through names, confusing novice chemists. Clear communication matters, not just for clarity, but for worker safety, regulatory audits, and successful research.
Shifting Risks: Safety, Standards, and the Reality of Working With Organics
My early days handling solvents taught me caution should not run on autopilot. Ethyl 2-hydroxypropionate can irritate eyes and skin; inhalation will provoke coughing and headaches if you ignore proper fume hoods. Its flash point hovers in the mid-40s Celsius, which means it doesn’t take much to transform a routine transfer into an emergency. Regulatory agencies call for flame-retardant storage, grounded containers, and tightly managed inventories. Safe handling comes with proper labeling, regular air monitoring, and investment in personal protective equipment, but the bigger shift traces to improved training and a culture of no-shortcuts safety. The best teams watch their backs and learn from past mistakes.
The Reach of Ethyl 2-Hydroxypropionate: Where It Shows Up
The range of application for this compound continues to grow. In decades past, paints and coatings manufacturers reached for it as a biodegradable solvent, quietly replacing more hazardous alternatives such as toluene and xylene. Pharmaceutical labs value its gentle solvent action and relatively low toxicity, especially during synthesis and purification steps where residue matters. Food and flavor houses trust it for its pleasant odor when formulating extracts and aromas. The emergence of bioplastics, biodegradable cleaners, and safer agricultural sprays keeps extending its resume. Demand for “green” solvents isn’t just a regulatory push—real stories from end users illustrate the balance between function and health.
Where Science Always Stays Busy: Research & Development
Labs across the world probe deeper uses for this familiar ester. Polymer scientists lean on its lactate base to develop bio-based polyesters with lower carbon footprints. Green chemistry teams experiment with blends to improve paint formulations, stripping away toxic additives but keeping performance. New research explores selective derivatization routes—tweaking side groups to produce functional surfactants and drug carriers—pushing sustainable design from concept to commercial reality. In the last ten years, research into catalytic processes shaved costs and waste from traditional synthesis, supporting lower emissions targets across chemical plants. I’ve seen first-hand how much difference funding and collaboration can make, shifting work from niche curiosity to mainstream solution.
Weighing the Risks: What Toxicology Tells Us
Skepticism comes naturally to those who have seen solvent use gone wrong, and toxicological research on ethyl 2-hydroxypropionate puts concerns into context. Animal studies place its acute toxicity levels higher than many familiar solvents, which led to expanded testing for chronic effects and bioaccumulation. Calls for better occupational exposure limits grew louder as labs proved its vapor could irritate eyes and airways, especially in environments with poor ventilation. Regulatory revisions rolled out, and producers responded with improved manufacturing purity and better hazard communication. For users wondering about environmental impact, studies show rapid breakdown in soil and water—thanks to its natural origins—but monitoring remains key, especially when disposal volumes climb.
Tomorrow’s Focus: What Comes Next?
As regulatory landscapes move, I see industry rethinking solvents across every application. The next chapters for ethyl 2-hydroxypropionate will likely lean even more on its roots in renewable resources, pushing for zero-waste synthesis and biodegradable formulations. With climate goals in sharp focus, research teams lock onto green solvents like this one, not just as alternatives but as launch pads for new materials and processes. There is plenty of exciting work to do, from developing stronger occupational standards to refining catalytic methods for even milder, cleaner reactions. I expect to see new blends tailored for specialty coatings, electronic materials, and medical uses where safety and sustainability lead.
What is Ethyl 2-Hydroxypropionate used for?
Getting to Know This Chemical
Ethyl 2-hydroxypropionate, also known as ethyl lactate, has a name that sounds like it belongs only in a chemistry lab. Despite this, you’ve probably come across it in places you wouldn’t expect. With a structure rooted in lactic acid—the stuff muscles burn for during a hard workout—it’s all around us in day-to-day products.
A Solvent with Real-World Value
Cleaning paints, coatings, and inks from tools or surfaces has relied on harsh chemicals for years, but ethyl 2-hydroxypropionate offers a less toxic solution. Professional painters, for instance, use it to thin and remove old paint or varnish. It comes from fermented plant sugars, giving it a reputation as a “greener” option. Painters and printers working with their hands every day face fewer strong fumes or skin problems, since this stuff evaporates slower and lacks the bite of aggressive solvents.
I’ve been known to refinish a few old chairs myself. After working in a small basement or garage with paint strippers, I have stories about headaches and lingering smells. Switching to an ethyl lactate-based product cut down on those workday side effects. For people spending hours in poorly ventilated spaces, that’s no small thing.
Role in Food and Fragrance
My kitchen cabinet contains more than olive oil and vinegar. Food producers add ethyl 2-hydroxypropionate to some favorite packaged goods as a flavor enhancer or extraction aid. It brings a mild, sweet note and helps pull out natural flavors from herbs or spices. In the perfume world, this same gentle aroma finds a place blending citrus or floral scent mixtures.
Since it’s produced by fermentation—like yogurt or sourdough—it lines up well with clean labels and sustainable production. Anyone with sensitivities to synthetic additives often seeks out these simpler, less processed ingredients.
Helping High-Tech Manufacturing
Some people think of solvents as relics of old factories; reality tells a different tale. In microelectronics, for example, workers utilize ethyl 2-hydroxypropionate to clean circuit boards, removing grease and solder fluxes that would otherwise cause device failures. Phones, tablets, and nearly any new gadget depend on reliable cleaning to keep defects at bay. Chemical residues from earlier cleaning agents produced more electronic waste and hazardous runoff, something tech manufacturers work hard to avoid.
Lab work is another field seeing steady adoption. Extraction scientists looking for plant-based nutraceuticals or studying environmental samples benefit from ethyl lactate’s mild properties and its ability to dissolve both water-loving and oil-loving compounds. Research groups that care about safety and lower environmental impact find themselves lining up for this chemical.
Weighing the Concerns
Ethyl 2-hydroxypropionate isn’t perfect. It costs more than older, petroleum-derived solvents, and some jobs still demand those high-powered alternatives. Certain large-scale cleaning applications still favor faster-drying or more aggressive chemicals despite the positive safety profile.
Educating decision makers in manufacturing and public health about long-term exposure benefits and disposal advantages can nudge adoption. Government support for safer chemicals and strict controls on hazardous emissions make a difference, too.
Whether stripping paint, crafting food flavors, building electronics, or blending fragrances, ethyl 2-hydroxypropionate sticks around for good reason. Its versatility and reduced risk help small businesses and large industries alike move toward safer work environments and cleaner home products. By making thoughtful choices about which chemicals we use in homes or workplaces, we improve our own health as well as the world outside our doors.
Is Ethyl 2-Hydroxypropionate safe for human contact?
Looking at Human Contact: What We Really Know
Ethyl 2-hydroxypropionate, known by some as ethyl lactate, shows up in more places than many folks might think. It's tucked into food flavorings, pharmaceutical solvents, personal care items, and even paints. I’ve seen product labels list it as a greener alternative to harsher chemicals, especially for degreasing and cleaning. Its plant-based credentials often convince people that it’s gentle. That doesn’t always tell the whole story.
Across industries, safety matters come first. Lab data and experience both speak volumes. The U.S. Food and Drug Administration does classify this compound as generally recognized as safe (GRAS) for flavoring applications. So, short-term and low-level exposures in food look safe for the average person. European regulators back this up, allowing its use in food and pharma at certain levels. Big bodies like these rarely greenlight ingredients without hard evidence to back their decisions.
Still, signals from material safety data sheets (MSDS) shouldn’t get ignored. For workers handling the pure, concentrated compound, the reports flag risks—mostly skin and eye irritation. Spilling it on your hands or getting a splash in the eye can cause a burn or rash, especially for those with sensitive skin. It evaporates pretty quickly, which can also dry out exposed skin. Manufacturers recommend gloves and eye protection for a reason.
How Does It Stack Up Against Alternatives?
People often ask if ethyl lactate is less risky than what it replaces. Traditional degreasers and solvents—think toluene, acetone, or methylene chloride—leave a much rougher legacy. Prolonged exposure to them can damage organs or set off lasting skin issues. Ethyl 2-hydroxypropionate, in comparison, rarely leads to chronic health effects based on published research so far. No reports link it to cancer or birth defects in humans under standard use patterns. Low toxicity gives it an edge, especially in closed or poorly ventilated spaces.
Experience in my own home shop backs this up. Swapping harsh solvents for ethyl lactate on paintbrushes and tools, my hands felt less dried out and rough. Still, one day of skipping gloves taught me that even “green” chemicals can leave skin irritated after enough contact. Watching office custodians handle cleaning agents with clear safety data sheets but no gloves brings this issue home: labeling something as safe doesn’t mean it’s risk-free in every use.
What Practical Steps Make Contact Safer?
Relying only on product labels doesn’t cut it. Reading both technical sheets and independent testing results gives a fuller picture. Whenever handling concentrated liquid, simple steps—like wearing well-fitted gloves and rinsing splashes off quickly—really cut chances of irritation. For those with allergies or open cuts, extra caution goes a long way. Even small amounts can sting broken skin.
Advocating for better workplace training helps as well. Companies that stock shelves or use large drums in manufacturing often forget refresher safety sessions. Reminders that skin isn’t a perfect shield matter more than a one-and-done onboarding. Workers have a right to up-to-date safety info, especially if products change formulas. All of us benefit when manufacturers publish exposures studies and regular updates, showing what new data reveals about chemical contact.
Choosing Wisely in Daily Life
Ethyl 2-hydroxypropionate answers calls for greener chemistry, especially for people worried about fossil-derived or highly toxic alternatives. Treating these chemicals with respect—reading the research, following handling advice, and using the right gear—offers the safest results. Healthy skepticism mixed with good habits makes a real difference, whether you’re cleaning a kitchen or mixing paints on the job.
What are the storage requirements for Ethyl 2-Hydroxypropionate?
Why Proper Storage Practices Matter
Anyone who’s stepped into a chemical storeroom knows the uneasy feeling of containers stacked too close or labels worn thin. Ethyl 2-hydroxypropionate—also called ethyl lactate—gets plenty of use in labs, food industries, and even as a biodegradable solvent, but treating its storage lightly opens the door to accidents. Volatile vapors, slippery floors, or irritation just from fumes are real risks. Stores that skip proper precautions wind up fighting leaks, regulatory fines, and frequent disposal headaches.
Key Storage Conditions for Ethyl 2-Hydroxypropionate
Cool, dry, and well-ventilated sums up the basics. In practice, this means picking a storage area where moisture stays low. Humid rooms turn this ester cloudy and can slowly start breaking down the content. My time in process safety training pressed the lesson: keep hygroscopic chemicals far from sinks, water lines, and condensation-prone surfaces.
Sunlight speeds up decomposition for organic compounds like this one. Aim for a shaded spot, or better yet, a closed cabinet. Heat ramps up evaporation—ethyl lactate’s vapors create not only a mess but a flammable hazard if they collect in small spaces. Any storeroom that houses flammable solvents should include a working exhaust system and avoid placing containers near heaters, steam pipes, or sunlight streaming through windows.
Container Choices
Metal cans corrode and glass breaks easily, so most facilities stick with HDPE or polypropylene bottles. These plastics resist attack and can handle some minor bumps. Years of working around chemical packaging taught me to check for tightly fitting caps—ethyl lactate has a habit of sneaking past loose lids, and the fruity scent isn’t a welcome sign. Label every container with the full product name, concentration, and date received. Faded labels invite mistakes.
Drums or tanks on a warehouse scale belong outdoors, but only under cover and off direct ground contact to avoid temperature swings from the floor. Pallets with a non-slip surface help. For bench chemists and quality-control labs, gallon jugs or smaller bottles do the job, stored on shelves fitted with lips in case of breakage.
Segregation from Incompatible Materials
Acids and strong bases spark rapid reactions with ethyl lactate, sometimes creating heat and pressure. Combining cleaning agents that contain ammonia or bleach in the same cabinet is risky. Separate solvents—especially oxidizers and peroxides—by at least a cabinet or two. Correct separation might save thousands in clean-up and prevent chemical burns.
Spill Management—It’s Not Just About Absorbents
I’ve seen small spills turn into big headaches if floors aren’t level or drains are nearby. Keep chemical-resistant mats close to the storage space and assign spill kits with pads that can handle esters. People working with ethyl lactate should use gloves and goggles even for small transfers. A slip mat at the door can be the difference between a minor cleanup and a panic dash.
Regular Checks and Training Matter
It pays off to review storage areas once a month. Scan for leaking lids, crystalline deposits that signal slow reaction, or containers shifting from their assigned spots. Every team member, not just safety officers, should know where the material sits and how to handle emergencies. Good habits stick when they’re part of daily workflow, not just annual training sessions.
The Takeaway from Everyday Experience
Low-key vigilance—not fancy technology—keeps both people and product safe. Tidy storage, sensible segregation, and proper ventilation protect workers and minimize waste. What seems basic often keeps accident tallies at zero. That’s solid peace of mind for anyone managing chemicals, whether it’s one bottle or a storeroom full.
What are the chemical properties of Ethyl 2-Hydroxypropionate?
Understanding Ethyl 2-Hydroxypropionate
Ethyl 2-hydroxypropionate, also known as ethyl lactate, pops up in labs, factories, and even on ingredient lists for food and cleaning products. It’s a clear liquid with a gentle smell, and it mixes right into water and most organic solvents. As someone who’s handled everything from bleach to strange esters, I’ve found ethyl lactate a lot less intimidating than most strong-smelling chemicals. You don’t need goggles just to be in the same room, but you still see respect for what it can do.
Key Chemical Properties
The backbone of ethyl lactate’s usefulness lies in its structure. It’s an ester—created by hooking up lactic acid with ethanol. There’s a hydroxyl group lurking on its chain, which totally changes how it behaves compared to other esters like ethyl acetate. That hydroxyl group acts like a magnet for water, helping explain why this compound dissolves well in both water and organic liquids. As a cleaner or solvent, it's tough to beat for flexibility.
Boiling point lands around 154°C, which means it sticks around longer than more volatile, punchy solvents. Labs that need precise temperature control often prefer something stable like ethyl lactate over something that boils off at room temp. The flash point—about 46°C—puts it on the safer side compared to plenty of flammable solvents, but nobody would call it entirely safe. One spill near a heat source, and things get exciting fast.
Another interesting side of ethyl 2-hydroxypropionate: the stuff isn’t just a bystander. That reactive hydroxyl group is ready to form hydrogen bonds, making it a great candidate in green chemistry as a replacement for harsher solvents. Years working alongside chemical engineers taught me that people love ethyl lactate because it breaks down fairly easily, reducing headaches around hazardous waste. Compared to classics like acetone or toluene, it offers a compromise between performance and environmental impact.
Why It’s Gained Attention
Many companies seek out chemicals with a lighter environmental footprint, especially in industries where regulations keep getting stricter. Ethyl 2-hydroxypropionate steps in here because its chemical properties let it perform tough jobs—removing grease, dissolving paints, acting as a carrier in perfumes—without leaving behind a toxic ghost. Major organizations, including the FDA and the EPA, recognize it as Generally Recognized as Safe (GRAS) when used in food grades.
In my years working around risk assessments, the rules always circled back to toxicity, flammability, and biodegradability. Ethyl lactate’s strong biodegradability gives it a real edge. It breaks down easily in the environment, so you see less buildup compared to stubborn solvents like chlorinated hydrocarbons.
Room for Improvement and Broader Adoption
Ethyl 2-hydroxypropionate doesn’t check every box. People complain about its lower evaporation rate, especially in fast-drying coatings. Some workers notice skin irritation after extended contact, though nothing compared to countless harsher industrial chemicals. Lots of manufacturers blend it with faster-evaporating solvents to strike a balance between effectiveness and safety.
Tools like ethyl lactate show that safer, greener chemistry isn’t pie-in-the-sky. Companies focused on sustainable cleaning or food production have invested in products using chemicals like this. If more businesses choose options with reasonable safety and environmental records, the whole industry keeps moving in a safer, smarter direction—with every bottle of ethyl 2-hydroxypropionate counted as proof.
How should Ethyl 2-Hydroxypropionate be handled in case of a spill or exposure?
Recognizing the Real Risks
Ethyl 2-hydroxypropionate, often seen in research labs and manufacturing spaces, doesn't attract headlines the way some chemicals do. For those who work with it, though, the threat feels real enough. Skin or eye contact brings burning or irritation. Inhalation stirs up coughing and shortness of breath. Swallowing the liquid seems unlikely, but accidents find a way. Workers in protective coatings, cleaning products, or pharmaceuticals know to take warnings seriously not because of some rulebook, but from experience. Missing one small detail brings up nasty surprises, and nobody wants to be the person who learns about the dangers the hard way.
Spills Happen — Immediate Steps Matter
Nobody hopes for a spill, but shop floors and benchtops rarely stay spotless forever. The first move starts simply: alert everyone nearby. Memories of a rushed cleanup gone wrong last a long time, often longer than the burns. Decent ventilation, windows wide open or exhaust fans running, keeps stray fumes at bay. Simple absorbents — sand or commercial spill powders — scoop up the liquid without drama. Wiping a mess with your usual mop leads to spreading the trouble; I’ve seen an experienced coworker accidentally smear solvents far and wide with just a few swipes.
Once the bulk of the spill’s corralled, it goes into a sealed, labeled container. Disposing of this waste by tossing it in a garbage can only pushes the problem downstream — that’s never ended well. Specialists handle chemical waste for a reason. Anything touched by the stuff gets scrubbed, rinsed, and checked off. These steps slow things down, but rushing through leaves invisible traces. Small habits start reducing the tally of accidents on any team.
Personal Exposure — Quick Action Saves Trouble
Direct contact turns a routine shift into an urgent washup. Skin splashes hit sooner or later, even for those who double-check their gloves. Cold running water on the affected areas for plenty of minutes, not just a quick rinse, usually keeps long-lasting burns away. Getting splashed in the eyes calls for the same urgency: to the eyewash station, lids pried wide, rinse and keep rinsing.
Breathing in the fumes gets tricky. Mild exposure still brings on headaches or dizziness. Taking a break outdoors, away from the source, and getting checked by medical staff stops mild symptoms from risking something worse. Don’t play hero — a trip to occupational health beats ignoring wheezy breathing or feeling nauseous. Swallowing the liquid or powder always calls for emergency help, and trying home remedies gets folks in bigger trouble. Bringing a copy of the substance’s safety data sheet speeds up treatment decisions in the ER. It’s just common sense, learned over the years, not something that comes from a factory training slideshow.
Building a Culture of Awareness
Training sessions and printed safety guidelines make a difference, but the edge comes from people who look out for one another. Reminders to change gloves or check eye protection work far better in a supportive crew than in a place ruled by fear or corners cut. Regular safety drills start feeling less like chores and more like shared responsibility. The best-managed teams I’ve known talk openly about close-calls and improvement, not about blame and punishment. In my experience, steady attention to these basics turns potential disasters into minor hiccups instead of horror stories on the shop floor.
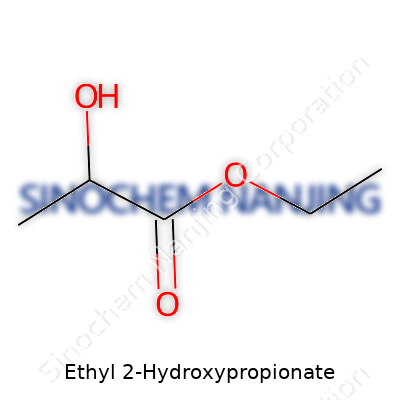
| Names | |
| Preferred IUPAC name | ethyl 2-hydroxypropanoate |
| Other names |
Ethyl lactate Ethyl α-hydroxypropionate Ethyl 2-hydroxypropanoate |
| Pronunciation | /ˈiːθɪl tuː haɪˈdrɒksiprəˈpeɪnɪt/ |
| Identifiers | |
| CAS Number | 687-47-8 |
| 3D model (JSmol) | ``` /* Ethyl 2-Hydroxypropionate */ CCC(=O)C(O)C ``` |
| Beilstein Reference | 795873 |
| ChEBI | CHEBI:32816 |
| ChEMBL | CHEMBL16133 |
| ChemSpider | 7186 |
| DrugBank | DB04248 |
| ECHA InfoCard | 100.005.664 |
| EC Number | 202-598-0 |
| Gmelin Reference | 121157 |
| KEGG | C02341 |
| MeSH | D016693 |
| PubChem CID | 12398 |
| RTECS number | OD9275000 |
| UNII | AV875T4QZ9 |
| UN number | UN2529 |
| CompTox Dashboard (EPA) | DTXSID2021551 |
| Properties | |
| Chemical formula | C5H10O3 |
| Molar mass | 104.1 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | mild |
| Density | 1.09 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.32 |
| Vapor pressure | 0.2 mmHg (20°C) |
| Acidity (pKa) | 16.0 |
| Basicity (pKb) | 8.64 |
| Magnetic susceptibility (χ) | -8.9 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.421 |
| Viscosity | 15 mPa·s (20 °C) |
| Dipole moment | 2.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | Ethyl 2-Hydroxypropionate, S⦵298 = 238.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -700.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1202 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | A16AA06 |
| Hazards | |
| Main hazards | Causes skin and serious eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 61 °C |
| Autoignition temperature | 220 °C (428 °F; 493 K) |
| Explosive limits | 3.6-18.1% |
| Lethal dose or concentration | LD50 Oral Rat 3730 mg/kg |
| LD50 (median dose) | 1950 mg/kg (Rat, oral) |
| NIOSH | SAF47820 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | No IDLH established |
| Related compounds | |
| Related compounds |
Lactic acid Ethyl lactate Methyl lactate Sodium lactate 2-Hydroxypropionic acid Lactonitrile |

