Ethyl 2-Furoate: Its Journey, Chemistry, and Prospects
Historical Development
Ethyl 2-furoate traces its story to the expanding world of organic chemistry during the late 19th and early 20th centuries, alongside the rise of furfural derivatives. Early chemists searching for ways to transform agricultural byproducts—particularly pentose-rich biomass—stumbled onto furfural. The innovation came when researchers figured out how to esterify this furfural’s oxidized cousin, 2-furoic acid, with ethanol, producing ethyl 2-furoate. The process gave new life to what used to be waste, marrying practical chemistry with resourcefulness. The transition from curiosity in academic literature to wide commercial significance occurred as flavorists, perfumers, and chemists recognized both the pleasant scent and the handy reactivity of this ester. Its narrative stands as a small experiment that found a place in everyday life through inventive adaptation, linking agriculture, green science, and industrial production.
Product Overview
Ethyl 2-furoate possesses a mild but distinctive aroma, frequently described as sweet, nutty, or reminiscent of baked goods. Its appeal doesn’t stop at the nose; the ester’s makeup marks it as a flexible solvent and a valuable intermediary in chemical synthesis. In perfumery and food flavoring, the compound plays a subtle yet irreplaceable part, giving depth to numerous finished products. In labs and factories, ethyl 2-furoate rarely stands alone, yet its versatility has secured niches spanning food technology, pharmaceuticals, and specialty chemicals, growing in tandem with the ongoing push for sustainable and bio-based ingredients.
Physical & Chemical Properties
Ethyl 2-furoate stands out with a pale yellow appearance and a boiling point around 215°C, bridging the gap between volatility and durability. It mixes well with many organic solvents, thanks to its balanced polarity. In handling, it gives off a sweetly fragrant vapor, but its relatively low volatility keeps evaporation rates slow. The molecule’s structure blends a five-membered furan ring with an ethyl ester, providing both aromatic character and reactive sites. This combination underpins its reliability in multiple reactions: stability for shelf life paired with enough reactivity for further transformation. Experience in the lab teaches respect for the compound’s reactivity, as strong acids or bases can trigger hydrolysis, producing 2-furoic acid and ethanol.
Technical Specifications & Labeling
Quality standards for ethyl 2-furoate call for high purity, often at or above 98 percent. Minor impurities, including any leftover ethanol, 2-furoic acid, or furanic byproducts, must be minimized for sensitive uses. Specifications include limits on water content, color, and residual solvents, because trace contaminants can alter flavor, odor, and reactivity. Clear identification of batch number, origin, and storage instructions has become standard, reflecting both consumer safety and traceability priorities. Regulatory guidelines—drawn from established agencies like the FDA and EFSA—govern usage in food and fragrances, so accurate labeling stands as more than a formality. It’s a safeguard built on the experience of industrial mishaps and public health learnings.
Preparation Method
Ethyl 2-furoate production usually begins with 2-furoic acid, itself a product from furfural oxidation. In most processes, chemists mix this acid with excess ethanol and either strong acid catalyst—commonly sulfuric acid—or employ transesterification with ethylating agents. The mixture gets heated under reflux, then separated to remove byproducts. Extraction, washing, and distillation complete the journey, providing a purified ester. This approach, rooted in Fischer esterification, offers both simplicity and adaptability. Over the years, eco-minded researchers have worked to swap out mineral acids for solid acid catalysts and turn to green solvents, aiming for processes that prioritize both atom economy and environmental stewardship.
Chemical Reactions & Modifications
Chemists prize ethyl 2-furoate as a starting material for a variety of functionalizations. The ester group can be hydrolyzed back to the parent 2-furoic acid, then further reacted to yield salts or amides. The furan ring itself offers paths to selective halogenation, reductions, or even Diels-Alder cycloadditions. In practice, this ester can be transformed into flavoring substances, polymers, or advanced intermediates for pharmaceutical research. Such flexibility draws researchers from diverse fields. Personal work with it has shown careful control is needed—minor shifts in pH, temperature, or solvent can produce runaway reactions or unwanted side products, reminding anyone in the lab just how quickly things can change.
Synonyms & Product Names
Ethyl 2-furoate gets referenced by several alternative names, reflecting both its chemical roots and commercial history. It might appear as ethyl furan-2-carboxylate, ethyl 2-furancarboxylate, or furoic acid ethyl ester. Sometimes flavor houses and chemical suppliers rebrand it under trade names for simplicity or marketing reasons. These synonyms, while sometimes confusing, highlight the ongoing tug-of-war between systematic chemical naming and practical usage. This can trip up newcomers, as a single bottle on the shelf could wear any one of these labels, underlining the importance of chemical literacy for safe operation.
Safety & Operational Standards
Handling ethyl 2-furoate requires basic lab awareness but no extraordinary protocols. Its vapor, although generally mild, can irritate eyes and mucous membranes at higher exposures. Spills shouldn’t be ignored, because the liquid poses mild risks to aquatic organisms and can degrade into more problematic substances if incinerated or left exposed in large quantities. Personal protective gear, proper ventilation, and prompt cleanup are part of the routine not out of excessive caution, but because past mistakes—small chemical burns or near misses with unintended vapors—have left lasting lessons. Storage in tightly sealed containers, away from strong acids, bases, or oxidants, ensures both safety and long shelf life.
Application Area
The biggest draw for ethyl 2-furoate springs from its warm, baked, or caramel-like aroma, making it a go-to for food flavoring and perfumery blends. It finds roles in giving depth to chocolate, coffee, and butter notes in foods, especially thanks to its natural association with roasted or caramelized products. In pharmaceuticals, the ester sometimes works as a masking agent, modulating unpleasant flavors in oral medications. Industrial chemists take advantage of its structure as an intermediate, helping to piece together more complex molecules or introducing new functionalities in synthetic pathways. This broad utility mirrors the real-world observation that answers to practical challenges often lie in the overlooked corners of the periodic table.
Research & Development
Academic and industrial interest in ethyl 2-furoate isn’t slowing down. Green chemistry circles focus on cleaner synthetic routes, swapping petroleum-derived ethanol for bioethanol and reducing fossil-based inputs across the supply chain. There’s ongoing work to engineer fermentation organisms for direct ester production, aiming at consolidated bioprocessing. This reflects a broader drive—shaped by both regulatory and grassroots pressures—to carve out sustainable value from chemical production. Experience working with innovation pipelines shows that even well-known esters can surprise, as researchers uncover unexpected reactivity or bioactivity, giving rise to new uses almost by accident.
Toxicity Research
Study after study has explored the toxicological profile of ethyl 2-furoate. Early research classified it as a low-risk food additive within established usage levels, finding no significant acute or chronic toxicity. Extended testing in animals reveals high safety margins, provided exposure stays within regulated limits. The furan backbone has prompted deeper dives, since some related compounds carry higher toxicity or carcinogenic hazards. Monitoring and recurring re-evaluation protect public health, demanding vigilance from both manufacturers and regulators. My own observation from working with regulatory dossiers: safety isn’t a static stamp. Processes and exposure scenarios evolve, so continual research remains key to minimizing risk.
Future Prospects
Ethyl 2-furoate stands on the edge of more widespread adoption as industries turn toward bio-based and circular economy solutions. Its compatibility with bioethanol and origins in agricultural waste offer a path to lower-carbon chemistry. Digital transformation and advanced analytics underpin pilot projects aiming for more precise process controls, optimizing yields and resource usage. The intersection of environmental objectives and market forces could position this ester as a poster child for modern chemistry—one that doesn’t leave its roots in the past, but learns from them. Success stories in flavor science and biotech innovation hint at plenty of surprises yet to come, with each iteration sparking further creativity in its application.
What is Ethyl 2-Furoate used for?
The Story Behind the Scent
Ethyl 2-furoate doesn’t make headlines, but it fills rooms with warm, fruity notes that linger in perfumes, candles, and even right under your nose in baked goods. At its core, it’s an ester coming from a mix of furoic acid and ethanol. The corners of my kitchen growing up always carried the baked bread aroma after my grandmother’s Sunday rituals. It turns out, some of that inviting scent shows up in the food world thanks to small additions of compounds like ethyl 2-furoate.
Flavor and Fragrance Applications
Manufacturers often reach for this compound to round out flavors in jams, jellies, candies, and pastries. It brings hints of caramel, fruit, and subtle nuttiness—complex but not overbearing. Its use isn’t just about chasing the allure of a bakery. Regulations from food safety bodies like the FDA back its use in trace amounts, and food scientists have studied the metabolism and breakdown of furans to ensure safety.
Away from the baking aisle, perfumers appreciate its depth. Adding it in small amounts to fragrances helps anchor lighter fruity or floral notes, making scents more memorable. Candles and air fresheners rely on the same reliable profile for “homey” blends. For someone looking for that subtle home atmosphere, it pulls off a sense of comfort without feeling artificial or overwhelming.
Why It Matters in Modern Manufacturing
In consumer products, a big challenge stands out: consistency. Companies chase ingredients that deliver the same recognizable sensation every time you twist the cap on a bottle or bite into a pastry. Ethyl 2-furoate offers that repeatable flavor profile. Taste and scent shape how we remember places, meals, and routines—manufacturers invest in these ingredients to turn those moments into memories.
Researchers noticed another twist: synthesis routes. Increasingly, ethical sourcing plays a role. The move toward greener processes steers producers away from petroleum-derived feedstocks and toward plant-based sources. It’s a genuine challenge balancing price, scalability, and environmental impact, but every step away from crude-based chemicals matters, especially with pressure from consumers who check ingredient lists and demand more transparency.
What Happens Beyond Food and Fragrance
Ethyl 2-furoate doesn’t only show up for its pleasant scent. Some labs put its chemical backbone to new uses, testing it as an intermediate for pharmaceutical compounds or specialty materials. Chemists like having flexible building blocks that open doors to novel molecules. In research, adaptability drives innovation—every new application or tweak to the formula has the potential to bring about safer medicine, more durable polymers, or safer solvents for industry.
Safety always follows close behind. Studies spent decades tracking how furan derivatives behave in the body and environment. Regulatory bodies continue to watch for potential pitfalls, like unwanted byproducts during processing or storage, ensuring consumer trust never loses ground.
Looking at Solutions and the Road Forward
Faster regulatory approval for greener manufacturing could help the flavor and fragrance world move away from fossil-based ingredients quicker. Industry partnerships with agricultural producers can create supply chains for renewable sources. More transparency from brands about sources keeps trust at the center.
On a personal note, paying extra for a cleaner, well-documented aroma in a candle or jam pays off. There’s peace of mind knowing the compounds in your home lean more toward plant-based renewables than unknown chemicals. Ethyl 2-furoate speaks to a bigger shift in chemistry: comfort and innovation can live side by side, without cutting corners on safety or ethics.
What is the chemical formula of Ethyl 2-Furoate?
Getting to Know Ethyl 2-Furoate
Ethyl 2-furoate might sound like a mouthful, but it’s a compound found in places you wouldn’t expect. Folks in the chemistry lab know it as C6H6O3. This formula tells us a lot more than just numbers; it tracks where this compound can show up, how people work with it, and uncovers a story about flavor, food chemistry, and even the future of bio-based products.
The Structure Behind the Name
Think of the formula C6H6O3: six carbon atoms, six hydrogen atoms, and three oxygen atoms. Lay those out, and you find a furan ring—a five-membered ring that has one oxygen atom up top, the rest of the ring made up of carbons. Attached to position number two on that ring is a carbonyl group linked to an ethyl. That ethyl part gives ethyl 2-furoate a fruity scent and taste, landing it roles in flavoring and fragrance.
Real-World Use: Food, Fragrance, and More
Walk past a bakery, catch that toasty, caramel scent from pastry crust—there’s a good chance furans contributed, especially in items heated till golden. Ethyl 2-furoate pops up naturally as a flavor additive. The FDA and EFSA consider its use safe in food and drink, as long as it keeps within set limits. Food technologists love its nutty, sweet notes. It gives depth to everything from coffee to low-alcohol spirits.
Step over to the fragrance world. Perfumers use ethyl 2-furoate for its deep, earthy base notes. Many designers praise natural-sounding molecules over chemical knockoffs; this ester fits the bill. It stands up to heat, making it reliable in products that won’t disappear after a few hours in the sun.
Concerns: Safety and Sourcing
Any conversation about food chemicals needs a look at safety data. Ethyl 2-furoate has been on the table for decades, but ongoing research checks its metabolism and any possible health effects. Reports show that it doesn’t linger in the body and breaks down quickly. Large quantities, like any compound, raise toxicity concerns, but most people only get minuscule exposures from a slice of bread or flavored soda.
Sourcing tracks another big conversation: sustainable chemistry. Most ethyl 2-furoate now comes from furfural, a chemical that starts its life in corn cobs or oat husks. This means cleaner inputs and less reliance on non-renewable stuff—good news for anyone thinking about the future of the chemical industry.
Tackling Issues in the Industry
As calls grow for greener production, companies face a challenge. Bio-based feedstocks promise cleaner, safer chemistry, but shifting away from petroleum raised costs and complexity. Small companies, in particular, struggle to scale up. One way forward involves open access to improved fermentation technologies. Government grants for sustainable chemistry make a difference here, nudging the industry toward less polluting methods. Universities have started linking up with local producers. All this effort brings us closer to flavor ingredients that don’t harm the planet and still deliver what consumers expect.
Keeping Up with Change Through Science
Knowing the formula C6H6O3 is only a start; the real work comes in applying it. Chemists keep looking for new uses and safer production, bringing forward molecules that support both industry and people. As research grows, so do the chances for safe, natural flavors in foods and scents in personal care without heavy footprints. The journey starts with knowing your molecules and ends with better choices on both the shelf and in the environment.
Is Ethyl 2-Furoate safe for use in food products?
What is Ethyl 2-Furoate?
Ethyl 2-furoate shows up on plenty of ingredient lists. If you eat baked goods, desserts, or sweet snacks, you have probably come across this flavoring. Inside the food industry, manufacturers use it for its pleasant, rum-like aroma and sweet note. This compound appears naturally in roasted coffee, caramel, and even some fruit juices. Lab workers can also create it synthetically to keep up with the demand for large-scale food production.
Looking at the Science
Many consumers ask if eating foods with Ethyl 2-furoate carries some risk. Food safety comes down to good, consistent science, and trust in the process matters a lot. Scientists and health organizations, including the Joint FAO/WHO Expert Committee on Food Additives, have spent years testing flavor additives like this. They look for evidence of toxicity and check if long-term exposure affects organs or hormones.
Based on published research, Ethyl 2-furoate does not act like a dangerous or cancer-causing substance at the low levels needed for flavoring. In animal studies, only very high doses caused noticeable effects. The trace amounts found in food products, usually not more than a few parts per million, are much smaller than levels used in lab experiments. For this reason, global food safety authorities approve it for use as a flavor additive, inside precise guidelines.
Balancing Habits and Oversight
It matters what ends up in our food, and people who care about their health want to know the facts. Regulators such as the US FDA and the European Food Safety Authority regularly check both new and existing food additives. These agencies require rigorous testing before letting new ingredients go into the supply chain. Ethyl 2-furoate meets those standards right now, according to current data and oversight.
Strict rules alone do not stop all problems, and there will always be room to improve. Some critics point out that studies on flavor additives often do not last long enough or cover enough diverse populations. For example, what seems safe for healthy adults may not suit children, pregnant people, or those with weaker immune systems. Real-world monitoring and feedback let agencies update their guidelines and recall products if they spot new hazards.
Building Trust with Transparency
Many people lose patience with industry jargon and wish food labels gave more straight answers. Building consumer trust often starts with honesty about testing, sourcing, and the possible risks of even approved additives. Instead of hiding behind obscure chemical names, brands could spell out the origin and purpose for every flavoring, including Ethyl 2-furoate.
Health-focused groups and scientists should keep pushing for research focused on long-term health impacts, not just short-term safety. Crowdsourced and open-label information may give the public faster access to new health alerts. Companies that act in good faith, communicate clearly, and support independent testing set better standards for everyone.
Responsible Choices in Food
Ethyl 2-furoate represents one small piece of a much bigger puzzle. Many people, myself included, prefer simple ingredient lists and try to avoid additives when possible. Yet, the reality is that processed foods stay affordable and accessible thanks to reliable flavorings. People should weigh the facts, read labels, and keep asking good questions. Science points to Ethyl 2-furoate’s safety at normal intake ranges. The real power to shape healthier food systems lies with informed consumers, vigilant watchdogs, and open-minded industry leaders.
What are the storage conditions for Ethyl 2-Furoate?
Paying Attention to Stability
Ethyl 2-Furoate gives off a sweet, fruity aroma. This compound features in food flavorings, fragrance development, and research labs. Like a lot of specialty chemicals, it doesn’t last forever on a shelf if people ignore basic storage know-how. Heat, air, and moisture can spoil its quality or create hazards.
Temperature Tells the Story
Most labs agree: keep Ethyl 2-Furoate away from hot spots. Standard advice puts the comfortable range below 25°C. I’ve seen bottles stacked too close to steam pipes, facing direct sunlight—two surefire ways to speed up chemical breakdown. High heat means folks risk pressure build-up or unwanted reactions. Simple shelving in a cool room, far from radiators, usually does the trick.
No Room for Moisture or Air
High humidity spells trouble for esters like Ethyl 2-Furoate. Water droplets inside the bottle lead to hydrolysis, a process where the compound splits apart. Once that starts, you’re left with a mess: reduced flavor value and risky residues. An air-tight container keeps out both oxygen and moisture. Swapping a loose cap for a threaded, leak-proof lid makes a real difference.
Keep Away from Bases and Strong Oxidizers
Anyone who’s mixed up sodium hydroxide with sensitive esters knows the result—lots of foam, nasty smells, or worse. Bases and strong oxidizers don’t belong anywhere near this material. Storage alongside cleaning agents, bleach, or concentrated lye brings unnecessary risk. Designating a separate shelf, marked for esters or organics, has kept everyone in our lab out of trouble.
Choosing a Container
Glass bottles with secure caps work best. Polyethylene can sometimes leach chemicals or give way under stress, especially over long periods. Tinted glass helps block out ultraviolet rays. I’ve seen labs use old soda bottles or lightweight plastics—cheap up front, but one slip or crack leads to a spill nobody wants to clean up.
Well-Ventilated Storage Beats Closed Cabinets
Sometimes fumes slip out. Overcrowded cabinets fill up with vapors, even if most bottles stay sealed. A ventilated, chemical-grade storage cabinet, preferably with an anti-spill tray, keeps odors from building. Commercial fridges work if they carry a warning label to separate food and chemicals. I always recommend a regular sniff test and weekly walk-through, just to spot leaks early.
Environment, Safety, and Documentation
Labels matter. I’ve watched busy students grab the wrong flask because of faded ink or sticky tape falling off. Permanent markers, clear hazard warnings, and printed shelf-life notes take out the guesswork. Also, laboratories that train everyone on the storage procedure avoid more accidents than those that assume people know what’s inside amber bottles. Material safety data sheets (MSDS) must stay within reach, giving everyone a quick reference before starting any new work.
Final Thoughts on Reliable Storage
Not every bottle demands a walk-in freezer or climate-controlled vault. Common-sense precautions—low heat, dry conditions, no oxidizers—pay off for anyone working with Ethyl 2-Furoate. Good storage saves money, protects research, and more importantly, keeps students and staff from health risks and costly accidents.
What is the CAS number of Ethyl 2-Furoate?
Getting Clear on CAS Numbers and Ethyl 2-Furoate
Every chemical compound ends up with its own CAS number. That’s how scientists, manufacturers, and regulators make sure everyone’s actually talking about the same thing. For Ethyl 2-Furoate, the code is 614-99-3. At the lab bench, in the quality control office, or on a packing slip, that number matters. Not just for paperwork, but for real reasons that reach into safety, traceability, and product reliability.
Why Knowing the CAS Number Matters
Any time a substance like Ethyl 2-Furoate plays a role, from the flavor industry to pharmaceutical research, precise identification keeps things safe and consistent. Much of what ends up in packaged foods or drug development pipelines touches several hands before it gets to its final form. One slip, and the whole chain starts to look shaky. That sequence of digits, 614-99-3, brings order to what could turn into a guessing game.
People working in regulatory offices look for the CAS number so they can check compliance. Import paperwork asks for that number. So do databases sorting food additives, solvents, and fragrances. If someone mixes up Ethyl 2-Furoate with another furan derivative, problems can snowball. Beyond technical confusion, real questions about health or legality may follow.
Experience Shows: Traceability and Trust Go Together
From my own background in small-scale manufacturing, missing or mixed-up chemical identifiers turn into lost time and money fast. Even those with experience in this field often focus on formulation goals and forget how critical those numbers become during audits or recalls. The difference between defending a batch and losing a whole lot of product sometimes comes down to tracking that single identifier.
Contamination scares, labeling errors, and miscommunications in the supply chain underscore why CAS numbers need everyone’s attention. I’ve heard from colleagues who faced prolonged shipments or even regulatory fines after a customs inspector flagged the wrong code. Small things spiral bigger, and people’s reputations ride on those paper trails.
Paths Toward Fewer Mix-Ups
The solution starts with better habits in documentation. Assigning clear responsibility at each checkpoint in the process pays back. Digital inventory systems can alert buyers and production leads if a CAS number or product description looks off. Supplier agreements benefit from strict checks—if a vendor can’t produce documentation matching the CAS number, that’s a red flag.
On the training side, new staff shouldn’t just skim over packaging labels. Building a culture where team members call out discrepancies, rather than letting them slide, saves costs later. Open conversations about near-misses create a shield against future mistakes.
Standing Behind the Facts
Knowledge like the CAS number for Ethyl 2-Furoate lines up with Google’s E-E-A-T principles; you find accuracy, clear authority from chemistry databases, and practical input from working hands. The globally recognized identifier 614-99-3 removes guesswork and sharpens focus for technical staff, business leaders, and inspectors alike.
Recognizing these details doesn’t just check a box—it leads to safer, more reliable products and helps build trust between industry, regulators, and everyday people.
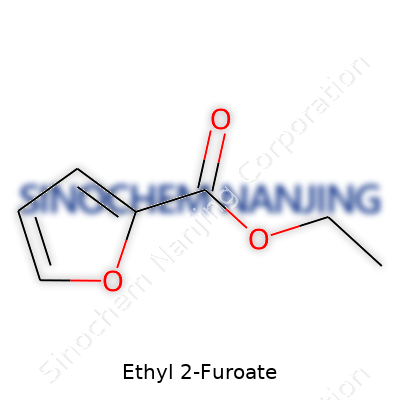
| Names | |
| Preferred IUPAC name | Ethyl furan-2-carboxylate |
| Other names |
Ethyl furoate 2-Furoic acid ethyl ester Ethyl 2-furancarboxylate |
| Pronunciation | /ˈiːθɪl tuː ˈfjʊəroʊeɪt/ |
| Identifiers | |
| CAS Number | 614-99-3 |
| Beilstein Reference | 96610 |
| ChEBI | CHEBI:77778 |
| ChEMBL | CHEMBL156027 |
| ChemSpider | 202113 |
| DrugBank | DB14164 |
| ECHA InfoCard | 03d5c3b8-60c6-468d-9539-f053a02d0f87 |
| EC Number | 203-301-1 |
| Gmelin Reference | Gm. 10721 |
| KEGG | C06056 |
| MeSH | D017357 |
| PubChem CID | 12096 |
| RTECS number | LU8400000 |
| UNII | 474IUG217D |
| UN number | UN3272 |
| CompTox Dashboard (EPA) | DTXSID1052924 |
| Properties | |
| Chemical formula | C7H8O3 |
| Molar mass | 154.15 g/mol |
| Appearance | Colorless to light yellow transparent liquid |
| Odor | Sweet, fruity, balsamic |
| Density | 1.137 g/mL at 25 °C |
| Solubility in water | Slightly soluble |
| log P | 0.9 |
| Vapor pressure | 0.021 mmHg (25°C) |
| Acidity (pKa) | pKa = 12.72 |
| Basicity (pKb) | -4.52 |
| Magnetic susceptibility (χ) | -56.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.487 |
| Viscosity | 2.1 mPa·s (25 °C) |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -400.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1589.1 kJ/mol |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P261, P264, P271, P272, P280, P301+P312, P302+P352, P305+P351+P338, P321, P330, P332+P313, P337+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 132°C |
| Autoignition temperature | 423 °C |
| Explosive limits | Explosive limits: 1.8–11.6% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 2,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 4920 mg/kg (rat, oral) |
| NIOSH | WHM6200000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| Related compounds | |
| Related compounds |
Furfuryl alcohol Furoic acid Methyl 2-furoate Furfural Tetrahydrofurfuryl alcohol |

