Ethyl 2-Bromopropionate: Chemistry’s Flexible Building Block
Tracing the Origins: Historical Development
Ethyl 2-bromopropionate didn’t just show up on chemists’ benches overnight. Its early mentions can be traced back to the steady march of organic synthesis in the nineteenth century, when researchers worked out how to swap hydrogen atoms for halogens. Ethyl esters like this one grew up alongside the demand for tailored molecules in medicine, agriculture, and polymers. Once folks knew you could introduce a bromine atom onto a propionate backbone, a small but crucial world of downstream possibilities opened up. Over time its role shifted from specialty laboratory reagent to industrial commodity, always with its roots in making molecular building blocks richer with function and reactivity.
Product Overview: Why the Industry Cares
Ethyl 2-bromopropionate isn’t the flashiest molecule in the stockroom, but it serves where precision is needed. Its value lies in giving chemists a simple way to slip a reactive handle—a bromine atom—onto a familiar backbone. People in pharmaceutical research, for example, use it to plug in new groups or to set up complicated branched molecules. Pesticide makers count on it to create intermediates that hit the right molecular targets in bugs but not in people. Manufacturers appreciate the liquid’s manageable boiling point and its ease of handling compared to other halogenated compounds, which tend to be more delicate or riskier to store.
Physical & Chemical Properties: More Than Meet the Eye
Looking at the bottle, you see a clear, almost colorless liquid with a mild, fruity smell—a telltale sign of an ester. Its physical properties stand out for practical reasons: it boils around 140-142 °C and weighs in with a density a bit higher than water. The main chemical attraction lies with the bromine atom, snug on the neighboring carbon, making this ester ready to spring into reactions involving nucleophilic substitution or other typical organic moves. The careful balance between reactivity and stability keeps it from going off the rails during storage or transport, but still lets it run wild in the hands of a creative synthetic chemist.
Technical Specifications & Labeling: Getting the Details Right
Chemists depend on tight product specifications when planning a synthesis, since even a slight impurity can send a project off course. Bottles labeled as “Ethyl 2-bromopropionate” should list purity, which typically reaches 98 percent or higher for laboratory grades. Labels should highlight proper storage conditions—cool, dry, and well-sealed—and flag hazards. Regulations in most countries require hazard pictograms for flammability and for environmental and health concerns. That clarity on the label isn’t just bureaucracy; it’s the first line of defense against lab accidents and ruined batches.
Preparation Method: Converting Theoretical to Practical
Making ethyl 2-bromopropionate usually starts with ethyl propionate, a derivative of propionic acid. The common approach uses bromination in the presence of an acid or Lewis acid catalyst. Sometimes, folks reach for phosphorus tribromide to swap out the alpha hydrogen for a bromine atom; other times, hydrobromic acid comes into play. These routes reflect how the chemistry textbooks hit the pavement: reliable, scalable, and reasonably safe if the right precautions are respected. Each step must keep moisture and oxygen out, since the compound can break down or generate unwanted byproducts in their presence.
Chemical Reactions & Modifications: The Versatility Factor
The structure of ethyl 2-bromopropionate draws plenty of chemists thanks to the contrast between the stable ester and the lively bromine. This reactivity guides its starring role in preparing amino acids, pharmaceuticals, and crop-protection agents. Commonly it joins with nucleophiles—like amines or thiols—to trade out the bromine for a new group. Chemists have come up with ways to turn it into chiral building blocks, making molecules that only fit receptor sites found in pests or pathogens, not humans. Its ability to participate in Grignard reactions or show up as a key intermediate in multi-step syntheses keeps it in steady demand across chemistry labs.
Synonyms & Product Names: Casting a Wide Net
Almost every chemical has an alias, and this one’s no exception. In catalogues or scientific articles, you’ll also see Ethyl α-bromopropionate, 2-bromopropionic acid ethyl ester, or just plain ethyl bromopropionate. These names stand in for the same compound but can sometimes trip up less experienced researchers or lead to confusion if tracking down safety data sheets. Part of working with such chemicals involves double-checking supplier codes and CAS numbers so that a research team doesn’t wind up running the wrong reaction or violating procurement rules.
Safety & Operational Standards: Keeping the Workplace in Shape
If you spend time in a lab or plant, you realize the rules about handling chemicals like ethyl 2-bromopropionate exist for good reason. The stuff irritates skin, eyes, and lungs, and it doesn’t play well if spilled into water sources because aquatic life doesn’t stand a chance against its toxicity. Industry standards call for gloves, eye protection, fume hoods, and careful waste management. Emergency procedures stay posted for a reason. Everyone who handles ethyl 2-bromopropionate learns to respect the potential health impacts as well as the long-term environmental risks that come from carelessness. Solvent exposure can sneak up on you. Even minor exposure over months or years can pile up side effects—stressing the nervous system, liver, or kidneys.
Application Area: Where it Delivers Value
The uses for ethyl 2-bromopropionate stretch beyond just being a stepping stone to something else. Pharmaceutical research takes up a good chunk of demand, drawing on its role in building amino acid analogues or custom-tailored small molecules for drug discovery. Agrochemical companies value it because the brominated ester opens the door to reactions that standard propionate esters just can’t reach. Beyond those major industries, you’ll find it in specialty polymer synthesis or advanced materials labs, wherever someone wants to introduce a bromine handle or tweak molecular scaffolds. For a handful of chemists, it also emerges in the classroom, teaching students about ester chemistry and the clever tricks that halogen atoms can play.
Research & Development: Pushing the Boundaries
Chemists keep finding creative ways to employ ethyl 2-bromopropionate in new reaction schemes. Research journals describe using it to prepare chiral centers for asymmetric synthesis, a must for medicines that only work in specific orientations. Advances in catalysis, including transition metal-catalyzed couplings, have unlocked reactions where this modest ester serves as a platform for designer molecules. The march toward greener chemistry asks for safer, more sustainable bromination methods while reducing waste and emissions. Teams in both academia and industry chase down these improvements, knowing the payoffs ripple out through medicine, agriculture, and materials science.
Toxicity Research: Open Eyes and Cautious Hands
People want assurance that the chemicals in their workplaces don’t bring problems home. Toxicity studies show that ethyl 2-bromopropionate harms aquatic life and has the power to irritate or damage tissues after contact. Inhalation or repeated exposure can hit organ systems, so exposure limits and ventilation standards get taken seriously. Older safety studies often missed long-term impacts or didn’t account for mixtures with other solvents. Now, regulatory pressure and improved testing go deeper, checking for metabolites, environmental persistence, and the effects on sensitive groups. Research digs into possible breakdown products in the environment—because what you put down the drain today may turn up unexpectedly in a water sample years later. Only ongoing research and transparent risk assessment keep workers and communities confident that science works for, rather than against, public health.
Future Prospects: Where Ethyl 2-Bromopropionate Could Go Next
The outlook for ethyl 2-bromopropionate depends on whether chemists can balance its usefulness with rising safety and environmental expectations. Its strong position in pharmaceutical and agricultural development isn’t going anywhere soon. Innovations in catalytic methods could make its preparation safer and more efficient, maybe even swapping out bromine sources for greener options or reducing the amount of waste generated. With regulatory bodies around the globe tightening rules on hazardous chemicals, companies that adapt in advance—developing safer work practices or biodegradable alternatives—will stay a step ahead. The next big breakthroughs may well spring from outside the traditional lab: biocatalysis, machine learning-guided synthesis planning, or advanced recycling of halogenated byproducts. The key will be keeping an open mind, a careful hand, and a steady focus on both innovation and responsibility.
What is Ethyl 2-Bromopropionate used for?
Everyday Chemistry, Unseen Impact
Few people ever hear about ethyl 2-bromopropionate. Walk into a lab or chat with a chemist who’s pored over reaction tubes, and the name might pop up. In school, my high school chemistry lab felt like a puzzle, and nothing drove home the invisible reach of molecules quite like learning where these chemicals go after bubbling in a flask—sometimes into medicines, sometimes into plastics, and sometimes further. Ethyl 2-bromopropionate fits right into that narrative.
The Engine of Synthesis in Research
Chemists call ethyl 2-bromopropionate a classic building block. It’s not just jargon—it means the compound brings a certain handle that lets scientists bolt on new pieces. This chemical’s howl in the lab comes from that bromine atom. With the right nudge, that bromine can leave, letting something fresh take its place. By flipping bromine for a new group, chemists can chisel out a whole new vibe in a molecule. Keep doing that, and soon the raw material morphs into building blocks for life-changing drugs, or compounds needed in health research.
Take pharmaceutical development—every pill, every patch starts with these building blocks. Researchers reach for ethyl 2-bromopropionate because it helps craft molecules for antiviral drugs and cancer therapies, libraries of experimental compounds, and more. When you hear that a breakthrough medicine came from “organic synthesis,” there’s a solid shot that clever manipulation of halogenated esters—ethanol 2-bromopropionate among them—helped kick things off.
More than Medicine: A Route for Innovation
This chemical's story doesn’t end with test tubes and beakers. Think material science. Companies searching for new plastics, surfactants, or specialty coatings often cook up intermediates like ethyl 2-bromopropionate. Here, it lends its reactive power, offering a launching pad for inventing compounds that keep our homes cleaner or our electronics running smoother.
Through the years, I’ve seen researchers grow animated when talking about reaction “modularity.” Having flexible intermediates lets them swap, add, or subtract fragments and test new ideas quickly. Ethyl 2-bromopropionate’s value traces to just that smooth adaptability.
Looking at Risks and Responsibility
Lab work never gets far from safety talks. Ethyl 2-bromopropionate isn’t something anyone should handle without respect. It delivers strong fumes, irritates the skin, and can cause headaches or worse with careless handling. Academic settings drill into students protocols for goggles, gloves, and hoods, but stories from industry show accidents still creep in.
In light of wider concerns about chemical footprints, people now ask the tough questions: What happens when a spill leaks into local water? What waste streams do we create with chemicals like this? These aren’t questions for just big factories—small research labs play their part, too. Green chemistry strategies—such as swapping out hazardous solvents or recycling leftover reagents—help chip away at risks the compound can pose downstream.
Focusing on Future Solutions
Accountable chemistry starts with acknowledging each building block's dual nature: possibility and responsibility. Innovators are starting to redesign synthetic pathways so fewer toxic intermediates get produced, or to develop less hazardous alternatives to staples like ethyl 2-bromopropionate. Whether the goal is better medicines, safer lab practice, or leaner environmental impact, that local mindset of stewardship—one chemist, one batch at a time—makes the difference.
What is the molecular formula of Ethyl 2-Bromopropionate?
Spotlighting the Structure: More Than Just Letters and Numbers
You see a chemical’s name like Ethyl 2-Bromopropionate, and it might look like a mouthful at first. Chemists often throw around these names with easy confidence, but there’s a real structure underpinning that name. Anyone who’s ever pulled out a molecular model kit knows chemicals are not just academic—they shape medicine, agriculture, and even simple household products. The formula for Ethyl 2-Bromopropionate is C5H9BrO2, and that simple code packs in a universe of information.
Why the Formula Matters in Real Life
I’ve seen students stare at names like Ethyl 2-Bromopropionate, paralyzed by the layers in the name. The “ethyl” hints at a two-carbon chain. “2-Bromo” tells you a bromine atom sits on the second carbon of the propionate backbone. Propionate brings its own rhythm—three carbons with an acid group, minus its hydrogen because the ethyl group steps in. Getting used to these patterns builds a shortcut in your head. Even outside the lab, this instinct helps, whether reading food labels or understanding drug side effects.
Accurate formulas do more than win points on a quiz. They anchor how labs design new reactions, pick safe handling practices, and estimate impacts if a spill occurs. At factories turning out tons of chemical each week, every atom counts. A single bromine atom means different environmental hazards, reactivity, and health impacts compared to its chlorine cousin. Misidentifying a formula like C5H9BrO2 versus something similar could cost time, money, or, in rare cases, safety.
The Formula’s Real-World Reach
The molecular formula C5H9BrO2 acts as a universal language. Shipping companies, customs officers, and regulators use it to check if what’s inside a barrel matches the paperwork. Graduate students and researchers look at this string and instantly see where a compound might fit in drug synthesis, materials development, or crop chemistry.
I’ve seen how the industry relies on this shared shorthand. A formulator at a small biotech startup doesn’t need to speak the same first language as their partner in Singapore; the code C5H9BrO2 translates effortlessly. ChemSpider, PubChem, Sigma-Aldrich—all these resources list compounds by formula, making the global search for suppliers and data about Ethyl 2-Bromopropionate direct and transparent.
Doing It Right: Trust, Data, and Moving Forward
Plenty of stories highlight someone mistaking substances because of a loose approach to details. I once watched a colleague nearly mix the wrong batch because of confusion between two similar-sounding esters—one mistake, and the day was lost fixing it. Double-checking the formula and verifying with a trusted source like a chemical vendor’s data sheet saves time and limits risk. For professionals, trust builds through careful review and using reputable sources. Every batch produced, every shipment delivered depends on these habits.
Better chemical education, clearer regulatory guidance, and digital databases with up-to-date formulas help keep all of us safer and more effective. If you ever find yourself doubting, running a search for C5H9BrO2 brings you right to Ethyl 2-Bromopropionate—no guesswork, no stress. Chemistry is complicated, but a focus on formulas and details helps level the playing field for everyone working with these materials.
How should Ethyl 2-Bromopropionate be stored?
Why Proper Storage Matters in Real-World Labs
Walk into any chemistry lab and you’ll notice sharp attention paid to storage of chemicals. That’s not just about organization; it comes from hard experience. Safety issues and accidents often start with improper chemical storage. Ethyl 2-Bromopropionate isn’t especially rare in organic chemistry work, and it deserves the same level of respect as tougher reagents. It’s a clear, colorless liquid but not something to treat lightly.
Storing with Stability and Safety
Anyone who’s knocked over a bottle of volatile liquid knows how quickly fumes take over. Keeping Ethyl 2-Bromopropionate in a well-ventilated, cool area cuts down on those risks. Chemical stability depends a lot on controlling the temperature. Hot conditions can cause pressure to build inside containers, and that’s a recipe for leaks, breaks, or even releases. Cold rooms or temperature-controlled storage cabinets work best, holding the temperature typically around 2–8°C.
Shelving and containment matter too. Ethyl 2-Bromopropionate belongs away from direct sunlight, heat sources, and open flames. Flammable storage cabinets designed for liquids offer the right kind of protection. Most labs put it in its own spot, away from strong oxidizers, acids, or bases to avoid any dangerous mixing in the event of breakage.
Packaging and Labeling: Small Steps, Big Results
Original containers from chemical suppliers rarely fail, but only if people keep caps tight and seals intact. I’ve worked in places where just a poor seal led to ruined chemicals and a whole lab clouded with fumes. Always double-check caps. Store bottles upright with proper secondary containment trays to handle leaks.
Labels must stay legible, showing full chemical name, any hazard warnings, and dates. Tired old labels lead to confusion, and that’s when mistakes happen. Some techs rely on digital inventories, but nothing beats a freshly marked physical label.
Protection for People, Not Just Products
Personal experience tells me regular training makes the biggest difference. Safety goggles, gloves, and sometimes aprons make handling much safer. A few minutes of care during routine storage save hours cleaning up a mess or dealing with exposure. For example, Ethyl 2-Bromopropionate irritates skin and lungs, so no shortcuts with PPE. Good lab culture includes spill kits and eyewash stations nearby. Fume hoods help when transferring the liquid from storage to use.
Long-Term Storage and Waste Reduction
Stockpiling more than needed increases the risk of storage problems. Smaller, just-in-time orders reduce waste from expired stock, and it’s less likely a forgotten bottle will put people or projects at risk. Regular reviews of chemical inventory pay off. Anything past expiration or showing signs of discoloration or smell should get disposed of according to hazardous waste rules.
Trust in chemical supply chains and storage only goes as far as the last careful check. Building habits around safety, clear labeling, and controlled environments keeps labs running without drama. Responsible stewardship of chemicals like Ethyl 2-Bromopropionate respects both the science and everyone working behind the scenes.
Is Ethyl 2-Bromopropionate hazardous or toxic?
What People Who Handle Chemicals Should Know
Chemistry classrooms and research labs use all kinds of substances, some more risky than others. Ethyl 2-bromopropionate often pops up in organic synthesis. It helps produce medicines, flavors, and specialty chemicals. Safety questions tend to follow chemicals with unfamiliar names, and this one is no exception. Getting clear on what makes this substance tick means the difference between safe handling or a nasty accident.
Understanding the Risks
Anyone who has worked with organic bromides knows they often carry extra baggage. Ethyl 2-bromopropionate can irritate skin, eyes, and lungs. The liquid smells sharp. Accidentally spilling it on bare skin leads to a burning or stinging sensation. Even without visible burns, the exposure can cause redness or blisters in sensitive people. A whiff can leave your nose and throat scratchy or coughing for a while.
Breathing higher amounts for longer periods can trigger headaches and even nausea. The real trouble comes when careless handling leads to splashes in the eyes, which causes immediate pain and injury. Handling larger containers (think: manufacturing scale or bulk storage) multiplies the danger.
The Big Picture on Toxicity
Ethyl 2-bromopropionate gets classified as harmful but not acutely deadly. Scientists have tested it on lab animals, and those studies show clear signs of organ and nerve toxicity at higher doses. That doesn't mean one small splash spells disaster for most adults, but repeated careless use (or big spills) ramps up long-term risk. Long-term, low-level exposure could harm the central nervous system or liver.
Brominated compounds also show up in discussions around endocrine disruption, causing some experts to speak out for more careful lab practices and further research. Chronic exposure has not been studied in detail for this exact chemical, but similar chemicals sometimes linger longer in the body than many expect. It remains unwise to gamble with casual habits in the lab.
What Responsible Use Looks Like
In a decade of chemistry teaching and project supervision, I saw more eye injuries from underestimating routine chemicals than anyone wants to believe. The basics—goggles, gloves, lab coats—truly earn their reputation. Ventilated hoods matter just as much; breathing in chemical vapors day after day piles up invisible damages. Safety gear isn't about ticking regulatory boxes—it really protects your health every time.
Cleanup and disposal need attention. Waste solvents or contaminated paper towels usually fall under “hazardous waste” rules. Pouring leftovers in the sink or tossing rags in the regular trash spreads risk outside the lab. I once saw a minor spill, left unreported and cleaned poorly, eat a tiny gap in a student lab bench—for weeks afterward, the spot stank and triggered coughs, proving how even small misses can become lasting problems.
Better Approaches for the Future
For now, the best approach uses good ventilation, proper container labeling, and extra caution mixing or heating this chemical. Training goes a long way. Colleagues who practiced emergency drills handled the rare splash far better, never panicking or making mistakes under pressure. Stronger policies around substituting less dangerous reagents—when possible—could shrink the hazard list further and help new learners focus on what matters: safe, effective science.
What are the common applications of Ethyl 2-Bromopropionate in industry or research?
A Building Block in Organic Synthesis
Ethyl 2-bromopropionate often sits on the chemical shelves of university labs and large production floors alike. Its biggest draw comes from its role as a reliable alkylating agent in organic chemistry. During my early days in a graduate lab, it showed up often as a key starting point for creating a range of esters and acids, especially those tuned for pharmaceuticals or agrochemicals. In practice, the bromine atom makes the molecule reactive and lets scientists attach new chemical groups much more easily.
Pharmaceutical Research and Drug Development
Medicinal chemistry teams often reach for ethyl 2-bromopropionate when developing new drug candidates. The structure makes it a favorite tool for modifying basic molecular frameworks, which opens the door to new versions of anti-inflammatory and antiviral compounds. Take the case of beta-amino acids: using ethyl 2-bromopropionate, researchers can introduce side chains with high precision, aiming for molecules with greater activity and fewer side effects. Such fundamental steps may sound routine, but tiny tweaks made possible by this compound can decide the success of a whole drug portfolio.
The Power to Create Chiral Compounds
Chirality, the property that makes molecules act differently in living systems, plays a huge role in drug safety and effectiveness. Ethyl 2-bromopropionate’s asymmetric carbon atom lets chemists build chiral centers with better control. My colleagues working in synthesis often point out how this single molecule can simplify the process, whether the goal is building blocks for new medications or raw materials for pesticides. It’s a handy shortcut in a field where time and precision both matter.
Fine Chemicals and Advanced Materials
Outside the drug world, this reactant also finds use in the production of flavors, fragrances, and polymer additives. Its reactive nature allows chemists to stepwise build larger and more complex molecules. From a practical angle, this translates to smoother manufacturing of specialty chemicals and materials with precise performance traits. I have seen teams in the coatings industry use it to design additives that offer better adhesion or longer durability, choosing this compound for the flexibility it gives them in molecular design.
Potential Hazards and Safer Practices
Handling any brominated material calls for respect and careful planning. The reagent can irritate the skin and respiratory system and generates concerns around toxic byproducts. Labs and factories often invest in fume hoods, gloves, and precise disposal protocols. The movement toward greener chemistry highlights the need for safer alternatives, but right now, the versatility and reliability of ethyl 2-bromopropionate keep it in regular rotation. Open sharing between industry partners and academic labs speeds up the hunt for less hazardous substitutes, but for now, education around safe handling stays crucial.
Looking Ahead: Encouraging Responsible Use
Shifting regulations and rising costs push researchers to rethink common reagents. As more groups publish alternate methods—such as using biocatalysts or milder conditions—the grip of traditional halogenated materials will loosen. Still, in my experience, no single replacement matches the all-around utility ethyl 2-bromopropionate delivers for now. Careful management, ongoing research, and honest risk assessment stand as the practical path forward.
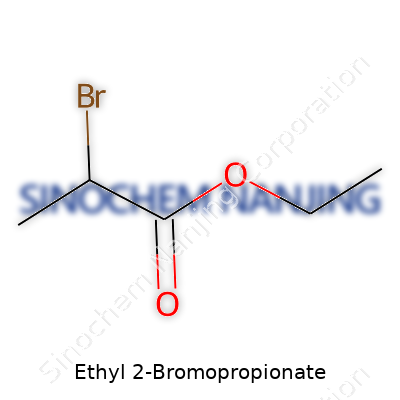
| Names | |
| Preferred IUPAC name | Ethyl 2-bromopropanoate |
| Other names |
Propanoic acid, 2-bromo-, ethyl ester Ethyl alpha-bromopropionate Ethyl 2-bromopropanoate Ethyl bromopropionate 2-Bromopropionic acid ethyl ester |
| Pronunciation | /ˈiːθɪl tuː ˌbroʊmoʊ proʊˈpiːəneɪt/ |
| Identifiers | |
| CAS Number | 535-11-5 |
| Beilstein Reference | 1209243 |
| ChEBI | CHEBI:132042 |
| ChEMBL | CHEMBL154572 |
| ChemSpider | 21170120 |
| DrugBank | DB14112 |
| ECHA InfoCard | echa.europa.eu/substance-information/-/substanceinfo/100.007.790 |
| EC Number | 205-807-5 |
| Gmelin Reference | 8553 |
| KEGG | C06358 |
| MeSH | D017217 |
| PubChem CID | 7358 |
| RTECS number | UF8225000 |
| UNII | TZJ1F9S290 |
| UN number | UN2346 |
| CompTox Dashboard (EPA) | DTXSID3076999 |
| Properties | |
| Chemical formula | C5H9BrO2 |
| Molar mass | 167.03 g/mol |
| Appearance | Clear colorless to yellow liquid |
| Odor | sweet |
| Density | 1.422 g/mL at 25 °C (lit.) |
| Solubility in water | Slightly soluble |
| log P | 0.9 |
| Vapor pressure | 3.3 mmHg (20 °C) |
| Acidity (pKa) | pKa = 25 |
| Basicity (pKb) | – |
| Magnetic susceptibility (χ) | -9.54 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.435 |
| Viscosity | 1.595 mPa·s (20 °C) |
| Dipole moment | 2.20 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -473.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2003.8 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P309+P311 |
| NFPA 704 (fire diamond) | 1-2-1 |
| Flash point | 80 °C |
| Autoignition temperature | 437 °C |
| Explosive limits | Lower explosive limit: 2.6%, Upper explosive limit: 12.5% |
| Lethal dose or concentration | LD50 oral rat 2900 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 2,075 mg/kg |
| NIOSH | CN9650000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Ethyl propionate Ethyl 2-chloropropionate Methyl 2-bromopropionate 2-Bromopropionic acid Ethyl bromoacetate |

