Epichlorohydrin: More Than Just a Chemical Compound
Historical Development
Epichlorohydrin entered the scene in the industrial world well before environmental and health rules shaped the way companies handled and produced chemicals. The early story stretches back to the first part of the twentieth century, when scientists sought stronger and more versatile building blocks for polymers and resins. As demand heated up for synthetic rubbers and epoxy resins, epichlorohydrin gained traction thanks to its high reactivity and link to the glycerin industry. Decades of technical evolution led to improvements in both production output and product purity, with plants in Germany, the United States, Russia, and Japan all racing to sharpen their techniques. Old production relied on propylene and chlorine routes, but newer technology slowly nudged the process toward so-called “greener” methods, like using glycerol instead of petrochemicals. Of course, every new approach brings new wrinkles, especially when weighing economics against environmental controls.
Product Overview
Calling epichlorohydrin a staple in the chemical industry hardly does justice to the range of work it shoulders. This compound shapes the backbone of epoxy resins, which wind up in coatings, adhesives, composites, and an endless range of industrial parts. The chemical itself is a colorless, pungent liquid—easy to spot in a chemical storeroom and recognized immediately by anyone who’s handled reactive organics. Industry folks sometimes refer to it by one of its synonyms—ECH, glycidyl chloride, or 1-chloro-2,3-epoxypropane—but the utility remains consistent across applications. From water treatment to electronics, its print is everywhere, but that’s created headaches when downstream contamination or toxic by-products show up in local environments.
Physical & Chemical Properties
Epichlorohydrin’s strong odor and volatility announce its presence even from tightly sealed containers. The compound boils at about 117°C and comes with a density a little higher than water, so handling it calls for good ventilation and solid containment. Its most critical property is the strained, three-membered epoxy ring, which makes it hungry for reactions with nucleophiles. Chemists have long taken advantage of this high reactivity, but the same trait spells risk during storage and transport. Flammable and not especially stable in contact with strong acids, it demands rigorous control measures on site, especially where heat or ignition sources could threaten safety.
Technical Specifications & Labeling
Product labeling reflects the need for crystal-clear communication around risk and handling. Every drum or tanker carries hazard markings highlighting toxicity, flammability, and potential for causing burns. There’s never a one-size-fits-all measure for purity or composition due to varying technical requirements; leading industrial standards steer specifications based on expected downstream use, like electronics or synthetic rubber production. Transporters, warehouse handlers, and process engineers have to stay in sync, since regulations lay out strict packaging, documentation, and batch tracking to spot problems fast if something goes off-target.
Preparation Method
Plants tend to favor one of two main routes. The traditional process treats allyl chloride with hypochlorous acid, then closes the epoxide ring via alkaline dehydrochlorination. While tried and true, this route cranks out plenty of salt and chlorinated waste, pushing environmental engineers to look for tweaks or alternatives. The more modern method starts with glycerol—often a byproduct of biodiesel manufacturing—and transforms it into epichlorohydrin using hydrochloric acid and other catalysts. The new route slashes dependency on fossil feedstocks, trading it for a more sustainable supply chain. Labs keep searching for catalysts and reactor designs that save energy, cut waste, and shrink the carbon footprint.
Chemical Reactions & Modifications
Epichlorohydrin never sits still in the chemical world. Its three-membered ring draws attacks from water, alcohols, amines, and dozens of other nucleophiles. Epoxy resins start with stepwise addition of bisphenols, kicking off chain-building reactions that form the base for coatings and adhesives. On the side, the molecule adds new links onto preexisting polymers, boosting flexibility or strength without major changes to the production line. Chemists also use it to launch reactions for quaternary ammonium compounds, which power water purification and paper-processing systems. This versatility puts it in dozens of chemistry textbooks, but just as often on lists of hazardous substances that deserve care and respect.
Synonyms & Product Names
Epichlorohydrin’s aliases reflect the complicated world of chemical naming. Some know it as ECH, others as glycidyl chloride, 3-chloropropylene oxide, or 1-chloro-2,3-epoxypropane. Catalogs feature a variety of trade names, especially where producers make minor tweaks for process compatibility. While the names shift across languages and countries, safety rules demand standardized labeling to keep accident risks low and ensure compliance with international trade laws.
Safety & Operational Standards
There’s no shortcut when storing, transporting, or using epichlorohydrin. Each handler must suit up with gloves, goggles, and proper respiratory gear since exposure leads to irritation, dizziness, and—if things go really wrong—cancer risks after chronic contact. Plants rely on tight air handling, local exhausts, and fire suppression equipment at all touchpoints. National and international rules, like those set out by OSHA and the European Chemicals Agency, enforce air quality, permissible exposure limits, and emergency protocols. Continual safety drills and fast-acting cleanup routines don’t just help companies pass audits; they protect everyone who works downstream and in neighboring communities.
Application Area
Nothing highlights epichlorohydrin’s impact better than the mountain of products built on its chemistry. Epoxy resins, which depend on ECH, serve as the backbone in adhesives, paint, electrical insulation, and composite materials for everything from wind turbines to airplane parts. Paper mills use ECH-derived resins to boost wet strength. Water treatment relies on its derivatives to remove particles and disinfect supply streams. Even tire factories depend on ECH-based elastomers to achieve the right mix of grip, flexibility, and resistance. While older sectors like printing and textiles once used ECH more liberally, rising awareness about toxicity and emissions forced companies to rethink recipes and invest in closed-loop systems.
Research & Development
Labs around the world channel energy into both improving existing ECH processes and hunting for long-term substitutes. With tech pushing forward, industry is chasing cleaner routes, smarter catalysts, and better containment to cut environmental harm. University scientists and private firms often look for new uses for ECH derivatives, such as biopolymers and composites for lightweight vehicles. Efforts push beyond production methods into analytical techniques for tracking trace residues in air, soil, and water. Most chemists now view continuous monitoring and lifecycle analysis as core to developing better norms and recipes, shifting from just making chemicals efficiently to thinking about where those chemicals end up decades later.
Toxicity Research
Research on epichlorohydrin’s toxicity reveals a tough legacy in industry history. Studies point to its capacity for causing irritation, nervous system effects, and cancer after long-term exposure. Occupational health experts link it to increased risk of respiratory problems among workers who lack proper protection. Animal studies and epidemiological work underscore the need for tough standards and constant monitoring. Communities living near production plants raise local alarms when releases occur, pressing for regular health screenings and pollution controls. The evolving legal landscape forces manufacturers to invest in modern emission controls and transparent reporting. Over time, this pushes down exposure rates, but past contamination often lingers in groundwater and soil.
Future Prospects
The push for greener chemistry puts epichlorohydrin under a bright spotlight. Industry will see increasing pressure to shift toward bio-based feedstocks, improve catalyst efficiency, and install waste reduction gear. At the same time, some researchers argue for phasing out ECH entirely in sensitive applications like food contact and medical devices, trading performance for reduced risk. One thing is clear: public expectations are rising, forcing the industry to maintain both transparency and adaptability. Safer alternatives, if they deliver the same utility without the health baggage, could one day sideline ECH in a few sectors. Until then, firms must walk a narrow path, balancing the benefits of established chemistry with the cost—and responsibility—of safe production and stewardship.
What is epichlorohydrin used for?
A Loyal Workhorse of Modern Industry
Epichlorohydrin barely makes a blip in daily conversation, yet its impact stretches across industries. Pick up a golf club handle, walk into a water treatment plant, or drive a car—odds are, this chemical played some role along the way. Factories use epichlorohydrin to build products that hold electronic components together, keep pipes from leaking, and make adhesives stick for years.
What Are People Doing With Epichlorohydrin?
Epoxy resins soak up most of the world’s epichlorohydrin supply. Manufacturers blend this chemical with bisphenol-A to create hard, durable plastics. These plastics end up in paint cans, circuit boards, wind turbine blades, floor coatings, and even surfboards. Walk through a hospital, and you’ll spot its influence in the sealants lining water pipes and medical equipment. It gives coatings remarkable resilience to chemical spills, which matters in labs, power plants, and airplane hangars.
Some industries chase different qualities. Paper mills treat filter paper with epichlorohydrin-based resins to prevent soggy collapse in humid conditions. This leads to sturdy coffee filters and lab filters. Rubber goods, from gaskets to weatherproof seals in refrigerators, often trace their sturdy bounce to this same chemical. Yet, the most surprising use pops up in water purification. Water plants count on epichlorohydrin-derived resins to pull out unwanted ions and heavy metals, giving people safer water.
Safety and Environmental Hurdles
My own first experience with epichlorohydrin involved a warning sign and a pair of tight gloves. People who spend time near it rarely forget its pungent odor and strict safety rules. The United States Environmental Protection Agency classifies epichlorohydrin as a probable human carcinogen. Workers face real dangers without strong protective equipment and good ventilation.
Factories that handle this chemical must invest in robust safety systems. Leaks or spills risk groundwater contamination and unsafe air, a problem highlighted in several high-profile industrial incidents. Communities that depend on fresh water downstream from such plants keep a watchful eye. Regulatory bodies and responsible companies rely on constant monitoring and leak prevention routines to reduce long-term risks.
Demand Pressures and Future Solutions
Chemists and engineers keep looking for safer alternatives. Some companies experiment with bio-based resins. These still lag in performance, but early signs show promise for less hazardous production. Better filtration, stricter plant rules, and shifts toward closed-loop manufacturing could make a difference.
Education plays a big role in keeping this chemical’s use on the right track. When workers know what they handle and why safety steps matter, injuries shrink and production headaches ease. In communities near these facilities, public information campaigns help people track what’s happening nearby. In my own experience working around chemicals, open communication between employees and neighbors builds trust, even when risks exist.
Epichlorohydrin delivers proven results where toughness and stability make a difference. But each advance in industrial chemistry should keep workers and neighbors in mind. The challenge is clear: find ways to enjoy the benefits while cutting the danger, mess, and waste. Communities, manufacturers, and researchers share that responsibility with every batch produced.
Is epichlorohydrin toxic or hazardous?
How Real Is the Hazard?
Epichlorohydrin gets a lot of attention from professionals working near synthetic resins, water treatment chemicals, or certain plastics. The first thing worth knowing: it’s not just “another chemical.” This stuff can cause trouble. People who’ve handled it on a hot day in a stuffy room without enough protection know about brutal headaches, breathing problems, or eye irritation that follow. Some workplace stories mention workers breaking out in rashes or coming home coughing after a shift on an epichlorohydrin line.
What Science Says About Exposure
OSHA and NIOSH both put tough limits on it—1 part per million in the air across an eight-hour workday. Anyone spending time even close to that limit is still probably too close for comfort. Once inside the body, studies show epichlorohydrin reacts with DNA in cells. Long-term exposure linked to higher rates of lung cancer and gastrointestinal issues among workers set off alarm bells decades ago. Mice given doses for months grew tumors more often. That pushed agencies like IARC to label it as “probably carcinogenic to humans.”
Accidents can be dramatic. Leaks or spills can produce harsh gas, causing eyes to sting and lungs to burn. Skin contact kicks off chemical burns. A friend who spent years at a resin plant once explained how strict cleanup had to be—gloves and goggles, every time. Miss a spot, and you’d feel a chemical tingle for days.
Where We Bump Into It
Epichlorohydrin isn’t something most folks use at home. It shows up in resin and epoxy factories, as well as labs cranking out drinking water filters. Still, traces can turn up in finished goods—rubber hoses, food packaging, or the adhesives that patch leaks in water pipes. In those cases, the big risk lies with workers and the environment around factories, not the person unboxing a new kitchen sponge.
Risk Reduction: Cleaner and Safer Approaches
Everything starts with good habits. Training workers on what they’re handling, keeping up with protective gear, and monitoring air quality make a difference. In recent years, more companies swapped traditional open tanks for closed systems—shutting down vapor leaks and cutting skin contact. Government inspectors show up unannounced, checking for airborne particles and how well spills are contained. Companies failing to act face fines or risk losing insurance.
Green chemistry teams keep searching for less hazardous ways to make resins and water treatment agents. Certain industrial users have tested water-based or bio-based alternatives, hoping to trade harsh chemicals for friendlier options. Progress is slow. Epichlorohydrin does some things nothing else tackles quite as well.
Staying Alert, Not Alarmed
Everyone benefits from better access to data about risks and how to avoid them. Sometimes, workers feel pressure to finish jobs quickly and cut corners on safety. Real change depends on open discussions between management and crews, not just regulators handing out rules. A few elder workers say the sense of pride—not just in making quality products but in keeping each other healthy—remains the best defense. For communities near plants, watchdog groups and regular reporting keep releases in the news and not swept under the rug.
Handling epichlorohydrin means treating it with respect. While it fuels key products, it’s no secret it can cause harm. With honest attention to what science shows and direct effort from those using it each day, the risks can shrink, but nobody gets to ignore the hazard.
What are the storage and handling requirements for epichlorohydrin?
Understanding What’s Inside the Drum
Epichlorohydrin runs the show in everything from epoxy resins to water treatment chemicals. This clear, volatile liquid brings some muscle to industry but demands genuine respect from anyone handling it. If I ever learned anything working alongside chemical bulk handlers, it's that shortcuts have no place when storing or moving stuff like epichlorohydrin.
Decoding Storage Requirements—Why They Matter
Epichlorohydrin belongs in tightly sealed steel drums or tanks equipped to stop leaks cold. This substance releases toxic fumes even at room temperature, and anyone who’s smelled its sharp odor in a closed warehouse knows what a difference tight seals make. Humidity only makes things trickier: water reacts with epichlorohydrin and forms hazardous byproducts, including hydrochloric acid. An accidental leak isn’t just a local mess. It risks a dangerous chemical cloud, and if it touches water, corrosion eats through floors or equipment.
Keeping a storage space cool, dry, and out of direct sunlight can prevent more than one headache. A friend in chemical logistics once told me about a poorly ventilated storage closet where temperature creep left every drum swollen, and employees scrambling. Storing these drums under 30°C and away from ignition sources stops misadventures before they start.
Avoiding Mishaps with Smart Handling
Anyone lifting, pumping, or sampling epichlorohydrin needs to stick to a set, drilled-in routine. Chemical splash goggles, gloves that hold up against organics, and a proper apron don’t just look good in an audit—they spare you permanent personal injury. Respirators belong within reach, not locked away, since just a whiff can burn airways and hurt lungs for life.
Epichlorohydrin vapors cling low to the ground. If doors stay closed or air flow falls short, the space turns dangerous in a hurry. Good ventilation means more than a busted window fan; proper air exchange rates save lives. A small, closed-off room with a quick leak encourages an explosive mix before you even notice the sharp scent.
Training and Real-World Vigilance Beat Paper Protocols
Written protocols look great, but no binder ever reached into a spill. Workers who know the drill—how to neutralize a puddle, close valves, evacuate calmly—can turn a potential disaster into a routine cleanup. In smaller facilities, I’ve seen old hands pair up every harvest season to lead new folks through mock drills. Practice, not paperwork, makes for safe handling.
Fires involving epichlorohydrin throw fire departments into high alert. Water streams spread contamination and stir up more vapors. Dry chemical extinguishers do the job better, but everybody wants an end to the drama before it comes to that.
Finding Better and Safer Ways Forward
Regulators and plant managers often want to hear what’s “required.” Practical safety, though, lives in the nitty-gritty details: label every drum, check seals before each move, maintain spill kits, and keep clear walkways. Workers should review emergency plans every few months—not just at onboarding. Adding continuous gas monitors and proactive maintenance on storage tanks extends peace of mind, even for veterans. Companies that put muscle behind “see something, say something” reporting finish the year with fewer incidents.
No one turns into an expert overnight, but anyone with enough time in the field learns that attention to detail and steady respect for chemicals like epichlorohydrin keep families whole and workplaces thriving.
What is the chemical formula of epichlorohydrin?
Chemical Formula and Structure
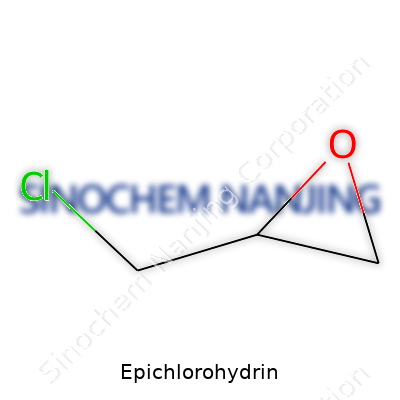
Epichlorohydrin comes with the formula C3H5ClO. Nobody really talks about chemical compounds at the dinner table, but this one shows up in the most surprising places – from your household products to the pipes that run through every city. That three-carbon backbone with an epoxide ring sets it apart, and the presence of chlorine hints at its reach in both manufacturing and public health.
Why Epichlorohydrin Matters
I once worked a summer in a water treatment plant. Each shipment of raw materials arrived with technical names and safety forms, but epichlorohydrin always sparked the most chatter in our back room. The reason? It links directly to production of epoxy resins and water treatment chemicals, but it also raises eyebrows because of possible health risks.
You find epichlorohydrin as a key ingredient in making epoxy resins, those tough adhesives and protective coatings used in cars, electronics, and even wind turbines. These resins need strength and durability, and epichlorohydrin delivers that. Municipal water workers rely on polyamine and polyamide chemicals made from epichlorohydrin to help clean up water. It binds with particles, making them easier to filter. That process can mean safer drinking water in cities large and small.
Health and Environmental Risks
Handling this chemical comes with responsibility. Countless safety data sheets highlight epichlorohydrin’s toxicity: skin and lung irritation top the list, with carcinogenic potential flagged by regulatory agencies like OSHA and the EPA. Workers in factories run the highest risk, but accidental releases – spills or vapor leaks – have made headlines, and not in a good way.
Communities near production plants have pushed for tighter monitoring. For good reason, too. Long-term exposure could lead to cancer or organ damage. EPA set strict limits for how much can show up in drinking water, pushing for less than 0.4 milligrams per liter. One study I read described how some factories switched to closed-loop manufacturing, reducing waste and airborne particles. It takes investment, but the tradeoff means fewer worries about health hazards.
Solutions and Safer Approaches
Not every solution calls for reinventing the wheel. Better personal protective equipment and more frequent training lowered incidents in one plastics factory near my old neighborhood. Simple steps like local exhaust ventilation made sure workers didn’t breathe in fumes. Some companies now use alternate chemicals for water treatment, lessening reliance on epichlorohydrin entirely—polyaluminum chloride, for example. But chemistry moves slowly. Some industries, like electronics or advanced composites, haven't found reliable substitutes yet.
Keeping an eye on environmental justice matters just as much as keeping factories running. Laws requiring better emission tracking push companies to invest in cleaner processes. Investments in greener chemistry produce new resins using bio-based feedstocks instead of legacy chemicals like epichlorohydrin. Engineers and chemists keep searching for safer methods not because regulations demand it, but because it’s the right thing to do for today's workers and tomorrow’s neighborhoods.
The Takeaway
Epichlorohydrin – C3H5ClO – shows up in more products than most people realize. It drives progress but asks us to respect its risks. Science means learning, adapting, and taking responsibility for the footprint we leave behind.
How should epichlorohydrin spills or leaks be managed?
A Chemical With Real Consequences
Epichlorohydrin carries a reputation as a tough chemical. Used to make epoxy resins, plastics, and even water treatment chemicals, it can be stubbornly persistent in the environment. Many folks working in chemical manufacturing know the smell—sharp, almost a warning in itself. Direct contact can burn skin and eyes. Inhalation brings coughing, dizziness, or worse if exposure lasts. Some studies show long-term impacts on lungs and even links to cancer. There’s no “just a little” leak with this stuff—every drop demands fast, careful attention.
Spills Don’t Happen in a Bubble
Turn the clock back to a shift I spent in a chemical plant—an odd hiss, a strong odor, the hum of the emergency ventilation. One cracked gasket had everyone moving. Not panic, but practiced action. Experience beats theory in times like these. Years of safety training made people snap to their jobs—closing valves, donning masks, rolling out absorbent pads. This didn’t fix everything, but it stopped something worse. In a real-world scenario, there’s no time for debate.
Why You Need a Clear Plan
Federal law treats epichlorohydrin spills seriously, and with good reason. The EPA puts it on the Toxics Release Inventory. OSHA slaps strict limits on exposure. In my days consulting for industrial clients, any time a bulk tank showed signs of corrosion, people learned to treat repairs as urgent, not just routine maintenance. Ignoring a small drip could risk much more than a fine.
Quick response often starts at the spill kit—alkaline detergents, neutralizers, chemical-resistant gloves, fresh cartridges for respirators. More than fancy gear, people need confidence in procedures. Trust won’t come from a binder gathering dust. Training drills—sometimes sweaty, always a little nerve-racking—help folks act when it matters most. As someone who has crawled in Tyvek suits, I know the extra effort pays off: no hesitation, no guessing.
Environmental Stakes
Epichlorohydrin travels fast if it gets loose—down storm drains, into soil, toward groundwater. I grew up near a creek that caught industrial overflow before stricter laws. Fish vanished in summer. Kids stopped wading in the water. Chemical leaks do more than threaten workers. Communities downstream pay for every shortcut someone takes upstream.
Site managers must coordinate with local fire departments, health agencies, and environmental responders. Communication lines should work in real time—cell numbers, radios, not just office phones. Cleanup isn’t a solo act. Getting outside help, especially for bigger spills, shows respect for people and nature.
Prevention Beats Cleanup
Stopping leaks before they start brings the most peace of mind. Double-walled tanks, leak detectors, regular pipe inspections—they cost less than emergencies, and they save lives. Companies that budget for maintenance rather than gambling on luck avoid painful headlines.
Open conversations about near-misses help everyone improve. Honest reporting and learning from mistakes create a culture where people feel valued. Workers watch out for each other, and that spirit finds its way into cleaner neighborhoods and safer families.
Looking Ahead
Technology grows smarter, but chemicals stay risky. Digital monitoring and remote alarms help, yet there’s no substitute for people who care and know what to do. Every season brings new hires who need a steady hand and wisdom passed down. Those stories, the lessons learned by doing, protect more than workplace reputations—they protect lives that stretch far beyond the factory gates.
| Names | |
| Preferred IUPAC name | 2-(Chloromethyl)oxirane |
| Other names |
1-Chloro-2,3-epoxypropane Chloromethyloxirane ECH Epoxypropyl chloride Glycidyl chloride Chloro-1,2-epoxypropane |
| Pronunciation | /ˌɛp.iˌklɔːr.əˈhaɪ.drɪn/ |
| Identifiers | |
| CAS Number | 106-89-8 |
| Beilstein Reference | 1207867 |
| ChEBI | CHEBI:44445 |
| ChEMBL | CHEMBL69737 |
| ChemSpider | 7714 |
| DrugBank | DB14088 |
| ECHA InfoCard | 03f48a63-4dfd-42d7-9f6a-85e1b40cff9c |
| EC Number | 203-439-8 |
| Gmelin Reference | 1755 |
| KEGG | C06381 |
| MeSH | D004826 |
| PubChem CID | 7807 |
| RTECS number | RX1400000 |
| UNII | 52EH1CT8ZZ |
| UN number | UN2023 |
| CompTox Dashboard (EPA) | DTXSID4020719 |
| Properties | |
| Chemical formula | C3H5ClO |
| Molar mass | 92.52 g/mol |
| Appearance | Colorless to slightly yellow liquid |
| Odor | Pungent |
| Density | 1.18 g/cm³ |
| Solubility in water | soluble |
| log P | 2.1 |
| Vapor pressure | 31 mmHg (25°C) |
| Acidity (pKa) | 14.2 |
| Basicity (pKb) | 13.61 |
| Magnetic susceptibility (χ) | -9.6×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.439 |
| Viscosity | 2.3 cP (25°C) |
| Dipole moment | 2.29 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 132.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -274.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1812 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS02,GHS05,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H314, H317, H331, H340, H350 |
| Precautionary statements | Precautionary statements of Epichlorohydrin: "P210, P260, P261, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P308+P311, P337+P313, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | 33 °C |
| Autoignition temperature | 320 °C |
| Explosive limits | 3.8–28% |
| Lethal dose or concentration | LD50 oral rat 90 mg/kg |
| LD50 (median dose) | LD50 (median dose): 113 mg/kg (oral, rat) |
| NIOSH | J084 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Epichlorohydrin: "5 ppm (19 mg/m3) TWA |
| REL (Recommended) | 2 mg/m³ |
| IDLH (Immediate danger) | 800 ppm |
| Related compounds | |
| Related compounds |
Glycerol Epoxy resin Chlorohydrin Propylene oxide Allyl chloride |

