The Story and Significance of Epibromohydrin
Tracing the Development of Epibromohydrin
Anyone who has spent time in the chemical industry knows every substance comes with a narrative much bigger than a molecular diagram and a safety data sheet. Epibromohydrin, with the structure that draws chemists into conversation, began making waves in the mid-twentieth century, at a point where the exploration of halogenated epoxides added fuel to both pharmaceutical syntheses and polymer chemistry. The value of epibromohydrin surfaced as researchers needed ways to add both an epoxide and a bromine handle into reactions that demanded high reactivity or selective modification. Academics and manufacturers realized its potential, turning it from a laboratory curiosity to an industrial tool. Somewhere in a chemistry department or a production plant, its strategic edge became clear: it could do things a lot of standard building blocks just couldn’t pull off.
What Sets Epibromohydrin Apart
Epibromohydrin’s formula looks simple, yet behind the carbon, hydrogen, oxygen, and bromine, it holds a lot of energy ready to reshape molecules. The almost playful chemistry of its oxirane ring and the bromine sticking out like a Velcro patch lets it attach, attack, or depart, depending on the setup. As a colorless to pale yellow liquid with a sharp, irritating smell, it announces its arrival in any synthetic lab. This is not the kind of compound you leave uncapped on a bench, and experience tells you a quick waft will probably linger in your memory for the rest of the day. Boiling above water’s threshold but not so high as to require elaborate heating, it strikes a balance suited for practical chemistry. It dissolves in many organic solvents, making it a go-to intermediate when speed and flexibility really count.
Technical Edge and Labeling Expectations
Nobody buys a bottle of epibromohydrin without a glance at the label and the paperwork that comes with it. In labs that care about accuracy, you’ll see a purity range that reflects the batch, not wishful thinking. Assays tend to hover above the 98 percent mark, clarifying that most of what you pour is indeed epibromohydrin, not some obscure byproduct that will ruin tomorrow’s experiment. Density, boiling point, refractive index — these are not just numbers but useful details that prevent mishaps in scale-up or new synthesis. Packaging isn’t an afterthought, either: bottles get an extra layer of sealing, hazard labeling follows hazard communication rules that anyone with a set of goggles can appreciate, and the documentation includes every relevant warning.
Making Epibromohydrin: Routes and Choices
In practice, chemists produce epibromohydrin through routes that reflect advancements in both practicality and demand for cleaner processes. The old staple relies on brominating allyl alcohol followed by ring closure. Seeing this operation performed up close, you learn a lot about temperature control and safe storage of corrosive reagents. More sophisticated setups cut down on byproducts and streamline purification, since every waste stream is now a regulatory burden and a cost driver.
Understanding Its Chemistry
The fun — or perhaps the headache — of epibromohydrin comes in its robust reactivity. That bromine atom isn’t just waiting for a photograph; it’s a gateway for nucleophilic substitution, opening doors for a variety of analogs in pharmaceuticals, agrochemical precursors, and oddball specialty chemicals. I’ve seen more than one group leverage the epoxide ring for crosslinking reactions, especially when aiming to tweak polymer backbones on the fly. Some use it to anchor molecules, others appreciate its readiness to be replaced or opened for another functional twist. For the seasoned synthesis veteran, it unlocks transformations that would stall with more timid reagents.
What People Call Epibromohydrin
Chemists are notorious for using more than one name for the same thing. Born in publications as 1-bromo-2,3-epoxypropane, epibromohydrin also pops up as 1,2-epoxy-3-bromopropane. Once, I met an old-timer who insisted on calling it bromomethyloxirane. Regardless of who’s speaking, these names all point to the same capricious liquid, reminding us that language in science can reflect both precision and habit.
Putting Safety at the Forefront
Anyone working with epibromohydrin finds out early that respect for safety is not optional. The irritation caused by just a drop on the skin or a careless sniff signals significant occupational hazards. Local rules dictate gloves and goggles, and the industry’s shift toward closed systems reflects a collective memory of accidents that once made headlines or filled case reports. Long-term exposure concerns have drawn the attention of health professionals. In regulated facilities, routine ventilation checks and air monitoring mitigate the risk, because people simply don’t want a repeat of respiratory trouble linked to halogenated epoxides. It’s easy to forget paperwork and disposal rules look like bureaucracy, but each step reflects hard lessons learned.
Why Application Areas Matter
Many associate epibromohydrin with specialty drug synthesis, especially when selective modifications are essential. I’ve seen chemists smile after pulling novel intermediates from reactions that stubbornly resisted other reagents. Outside pharma, it has found its footing in polymer chemistry, adhesive technologies, and as a crosslinking agent, because the eager reactivity translates to tangible product improvements. Manufacturers tracking trace contaminants in foods and water may use derivatives for analytic calibration — every drop of reliability starts somewhere. The diversity of applications pulls chemists from different backgrounds into the orbit of epibromohydrin, expanding both knowledge and the scope for responsible use.
How Research and Development Responds
R&D teams—whether at chemical multinationals or small startups—look for ways to make epibromohydrin cleaner, safer, and cheaper. The push for greener pathways highlights the broader shift in the chemical industry. Organic chemists tweak conditions, hoping for reaction setups that keep yields high without saddling workers or waste streams with excessive toxicity. Regulators’ growing attention only sharpens this focus. Every breakthrough on catalytic cycles or flow chemistry for epibromohydrin production ends up shared at conferences, since even a distant competitor’s progress creates industry-wide ripples. I’ve attended talks where the most engaged questions came after presentations on sustainable halogenation.
Toxicity: Walking the Line
Anyone who handles epibromohydrin must consider the toxicological record, which holds more caution than comfort. Short-term exposure brings on eye, skin, and respiratory irritation; the stakes rise with longer or repeated contact. Animal studies suggest genetic and reproductive risks, leading many organizations to limit workplace concentrations. I recall safety officers reminding teams about the difference between productive speed and reckless shortcuts. No one wants to trade today’s productivity for tomorrow’s health crisis. The push for less hazardous analogs shows an industry willing to learn from its difficult history, though this progress often feels stubbornly incremental.
Looking Toward the Future
Epibromohydrin is not going away soon; its unique chemistry and established utility keep it in demand despite legitimate concerns. The industry faces calls for better protective methods, improved synthesis, and deeper toxicological understanding. The future likely holds new catalysis strategies, smarter containment, and cleaner downstream alternatives, giving hope to both innovators and regulators. For those at the bench or in management, each advance in handling, safety, or cost confirms the value of learning from both data and direct experience. Epibromohydrin’s story reflects the best and the most daunting parts of modern chemistry: unique utility paired with a responsibility to do better — not just for profit, but for everyone touched by the chemicals shaping today’s world.
What is Epibromohydrin used for?
Getting to Know Epibromohydrin
Epibromohydrin sounds like something out of a chemistry exam, yet it plays a quiet role in many products people rely on. This clear, colorless liquid packs a punch with its reactive structure. Its formula might not catch your attention, but its reach stretches from industrial floors to the heart of pharmaceutical synthesis. My first encounter with epibromohydrin came in a chemistry lab, during a plastics experiment, where its sharp, almost sweet smell stood out. Curious, I dug into where it ends up after the flasks and beakers get washed.
Key Uses in Modern Industry
Epibromohydrin goes straight into the creation of specialty polymers and resins. Think about strong, long-lasting coatings or electrical insulation that doesn’t give up easily. Its three-membered epoxide ring and bromine atom turn it into an ideal building block. Chemists value it because it reacts with many substances; this makes it a staple in the toolkit for creating customized materials, stronger plastics, and resins with better resistance to heat and chemicals. In the adhesives world, epibromohydrin plays a core role in improving stickiness and shelf life.
Medicine benefits from it, too. Through a range of tightly controlled processes, pharmaceutical manufacturers use epibromohydrin to make intermediates. These pieces eventually join with other molecules to craft important drugs. It supports the production of anti-viral agents and drugs targeting certain cancers. Here, the focus always lands on precision: one mistake, and the whole batch might lose value or safety. Proper handling and monitoring make sure only the right amount of epibromohydrin ends up in the final product—or, at times, none at all after the chemistry finishes.
Risks and a Responsible Path Forward
Working directly with epibromohydrin isn’t a task for the untrained. Its toxicity and volatility mean it poses real risks for skin, eyes, and lungs. Occupational studies have reported that unprotected exposure can cause headaches, nausea, and respiratory trouble. Even carrying it across a facility demands strict safety rules—ventilation, protective clothing, and constant training. Regulatory agencies like OSHA and the EPA set exposure limits and demand manufacturers log every gram that enters, exits, or gets destroyed.
The environmental angle matters a lot. Epibromohydrin spills don’t disappear but can linger in soil and water, harming plants and aquatic life. If released, its heavy atoms mean it doesn’t break down quickly. Companies moving forward have started to invest in better leak-proof containers, updated emergency procedures, and real-time monitoring. Some labs now chase new syntheses with less reliance on hazardous building blocks, hunting for greener alternatives. Research from universities and industry groups encourages recycling or safer byproducts, putting long-term health ahead of short-term gains.
What’s Next?
Industrial progress shouldn’t come with unchecked costs. Industry leaders who keep safety, worker awareness, and waste reduction as part of their daily routine help push chemistry in the right direction. Epibromohydrin’s role in building advanced materials and medicines stays important, but safety demands constant vigilance. With growing attention on both employee health and environmental sustainability, this chemical reminds us that progress and responsibility travel side by side.
What is the chemical formula of Epibromohydrin?
Getting to Know Epibromohydrin
Epibromohydrin pops up here and there in chemistry labs and industry circles. This compound draws attention thanks to its reactive nature and, frankly, the hazards it brings. Its chemical formula, C3H5BrO, turns out to be quite straightforward, but its uses and impact reach farther than most folks imagine.
Why the Formula Matters
It seems like a simple arrangement: three carbon atoms, five hydrogens, one bromine, a single oxygen. Still, knowing that formula doesn’t just satisfy trivia buffs—it gives chemists a shorthand way of predicting how the stuff will behave. Bromine’s presence makes epibromohydrin a versatile building block for synthesizing other chemicals. Whenever I walk into the storeroom and see those bottles, I remember instructors warning us about brominated compounds: sharp smells, reactive sites, something you treat with extra care.
Current Uses and Industry Relevance
Folks in different fields rely on epibromohydrin for a few key moves. The compound helps churn out specialty epoxy resins and pharmaceuticals. I spent a summer interning at a coatings manufacturer, and stories from the process chemists taught me about the persistent role of compounds like this. Epibromohydrin can serve as an intermediate in producing polymers, where its structure gives chemists the reins to tweak the final properties of various products. In research, chemists use its strained three-membered oxirane ring to open doors for synthesis routes that wouldn’t otherwise work.
Hazards and Health Warnings
People need to recognize the risks as well as the utility. Brominated epoxides give off toxic fumes. The safety data sheets flat-out state that exposure can irritate the airways, eyes, and skin. Long lab days always include gloves, goggles, and open windows. Chronic exposure could even lead to more serious effects, based on older research and my own exchanges with safety coordinators. I’ve seen scenes where improper storage nearly led to lab evacuations.
Environmental Considerations
On paper, formulas look clean—three carbons, five hydrogens, etc.—but the real-life story involves impacts beyond the flask. Industrial spills carry brominated chemicals into water sources, causing issues for aquatic life. Some studies link the breakdown products of compounds like epibromohydrin to persistent organic pollutants. Regions with high chemical manufacturing often face difficult conversations about waste and long-term public health. My trip to the local water treatment facility brought the situation front and center, seeing the care required for even trace contaminant removal.
Looking Forward: Reducing Harm and Finding Solutions
Chemists and industry leaders recognize the trade-offs between usefulness and risk. Substitution with greener chemicals where possible remains the gold standard for many workplaces. Fume hoods, spill response training, and strict inventory controls—these practices aren’t just bureaucratic boxes to tick. They’re lessons learned from past accidents, valued by those who have seen the fallout firsthand.
Research teams look for catalysts and synthesis pathways that limit harmful byproducts. Green chemistry offers hope, but swapping out entrenched reagents like epibromohydrin depends on both technical and regulatory shifts. Transparent supply chains, better labeling, and tighter shipping standards matter to everyone, whether they work in the lab or live downriver from a plant.
What are the safety precautions when handling Epibromohydrin?
Why Epibromohydrin Deserves Respect in the Lab
Epibromohydrin, a reactive chemical with a pungent edge, always brings a feeling of caution when it’s around. Anyone who’s handled it will remember the smell and the tingling caution in the air. This stuff isn’t just your average bottle on the shelf; it has some serious risks hiding in that small bottle.
Direct Contact: Skin and Eye Protection
Anyone handling Epibromohydrin quickly learns that its fumes irritate the throat and eyes. Even a small spill stings the skin. Every lab worker I’ve met puts on thick nitrile gloves and keeps a solid pair of goggles or even a face shield close by. The risk of long-term exposure—rashes, chemical burns, or worse—makes it not worth cutting corners. Long sleeves and a proper lab coat add another reliable layer, and if anything splashes onto skin, it must get washed off right away using running water.
Ventilation: Keeping the Air Clear
Working with Epibromohydrin should never be a solitary affair in a closed space. Proper exhaust hoods or fume hoods need to run during every transfer or reaction. This isn’t just a suggestion on paper. The vapors can mess with your lungs and cause headaches, dizziness, and sometimes, more severe reactions. Short cuts on ventilation can land people in a hospital faster than expected.
Respiratory Protection and Smell Test Fallacy
Some people trust their nose to warn them about danger. That’s not enough. Vapors might hang in the air, even if the concentration remains too low for your nose to pick up. Mask filters—P100 or organic vapor cartridges—play a real role here. Some places go beyond and require supplied-air respirators, especially during big transfers or cleanup jobs. The aim is never to breathe in anything, no matter how tough you feel.
Storage and Labeling: Safe by Design
It’s easy to overlook shelf organization until something leaks. Epibromohydrin demands a lock-tight, secondary containment. Simple plastic bins or drip trays stop a small drip from turning into a real emergency. No one wants this chemical near acids or bases, or near sources of ignition since it’s both reactive and flammable. Easy-to-understand labels using big, bold hazard symbols help everyone, even newcomers, know what they’re working with at a glance.
Spills, Waste, and Emergency Prep
Each lab has its own flavor of spill kit, but the best ones for Epibromohydrin feature absorbent pads, neutralizer, and full suits. Every spill turns into a practice drill, with folks suiting up and moving quickly. Good labs review emergency eyewash stations and showers so that every worker knows exactly where to move. I’ve watched drills shave seconds off response times, and those precious seconds matter when chemicals burn or fumes fill the air.
Training, Teamwork, and Fresh Habits
Lab veterans train newcomers hands-on. Safety sheets and posters on the wall only go so far if you haven’t walked through the real steps. I’ve seen the difference it makes when someone moves confidently, knowing which glove to grab or how to pop open an emergency vent. Regular safety talks and refreshers cut down on accidents. Open communication, honest questions, and people watching out for each other keep every shift running smoothly.
Stronger Culture Equals Fewer Incidents
Epibromohydrin commands respect, and a no-drama approach to safety works. With the right habits, storage solutions, labels, and emergency plans, this chemical stays in the news only for what it builds—not for what it harms.
How should Epibromohydrin be stored?
Rethinking Chemical Storage for Safety
Anyone around Epibromohydrin for long knows it isn’t the friendliest of chemicals. It’s reactive, and the possibility of exposure—skin, inhalation, or accidental mixing—sets off alarm bells for good reason. People in labs, warehouses, and even shipping yards have stories about what happens when care slips. Epibromohydrin belongs with those chemicals that, without thoughtful management, bring a real risk to health and property.
Understanding the Hazards
Epibromohydrin is flammable, vapors can catch fire easily. Getting it on your skin or in your eyes means pain and possible damage. Inhaled vapors hurt airways. The chemical itself can react with water, acids, bases or even just with itself if heat builds up—sometimes forming offshoot substances that are even harder to clean up. The CDC and OSHA both note its toxicity and stress controls to keep exposures as low as possible. Given these risks, brushing off safe storage as a tedious chore endangers workers and neighbors alike. Hard numbers back this up: U.S. fire departments respond to over 6,000 structure fires a year at industrial properties, and chemical mishandling stands as a top cause.
Best Practices: Lessons from the Field
I’ve worked warehouses and seen the domino effect when a single drum sweats or leaks. Regular audits kept surprises to a minimum. Workers checked gaskets, lids, and seals, not just once, but every time we touched a barrel. OSHA sets guidelines—sealed containers, warning signs, spill kits within reach. These are non-negotiable, but habits make or break an operation. Storing Epibromohydrin in a cool, dry, well-ventilated spot should be routine. Heat or sunlight speeds up decomposition and sends fumes through the room. That’s a recipe for trouble.
Chemical-compatible containers do the heavy lifting here—usually steel or HDPE drums rated for hazardous solvents. Ignoring compatible storage has burned teams before; pitting, rust, and weakened drum walls lead to slow leaks or sudden spills. A serious mistake: mixing incompatible chemicals in the same area. Accidentally crack open both Epibromohydrin and a strong base, and the reaction could send workers rushing for the eye wash or worse.
Housekeeping Counts
Treating chemical storage like a forgotten closet invites trouble. Daily visual checks keep storage areas honest. I remember a near miss where a small leak had stained the floor overnight. A quick check in the morning caught it before it became a cloud of irritation and anger. Properly labeled containers and clear aisle space mean, in an emergency, every second counts and nothing blocks exit routes.
Training and Emergency Planning
Training pays off more than fancy safety equipment that no one understands. Workers who know what to do, where to go, and how to use spill kits handle incidents with less panic. Periodic drills, not just one-and-done orientation, give confidence under real pressure. Up-to-date safety data sheets posted in the storage area put crucial information at hand. As recommended by EPA and OSHA, attention to detail and ongoing education majorly reduce risks linked with hazardous chemicals.
Moving Forward with Accountability
Regular audits, record-keeping, and open-door reporting let teams spot problems before someone gets hurt. The goal isn’t to just meet a checklist, but to protect the people on the ground. Every facility that stores Epibromohydrin, from small research sites to industrial hubs, plays a role in keeping workers, communities, and the environment out of harm’s way. Real safety grows out of respect—for the chemical, the job, and each other.
Is Epibromohydrin hazardous to health or the environment?
What’s the Fuss about Epibromohydrin?
Epibromohydrin pops up in the conversation any time chemical safety gets serious. Used by some industries for specialty polymers or chemical synthesis, it doesn’t show up on grocery shelves. Its presence often flies under the radar unless you work with it, test for it, or live near a facility that handles it.
What Health Impacts Stand Out?
Anyone who has dealt with strong chemicals on the job knows the drill: skin irritation, strange smells, and warnings printed in big letters. Epibromohydrin belongs to a family of highly reactive substances. Exposure can feel harsh. Simple contact may cause intense skin burns or serious irritation. That’s not a surprise, since chemicals with similar structures—like epichlorohydrin—are well-known for causing trouble. Those who spend time in labs or chemical production lines rely heavily on gloves, goggles, and strong ventilation for a reason.
Inhalation of this chemical brings headaches, dizziness, and possible long-term risks. The US Environmental Protection Agency doesn’t look the other way on these threats; toxicologists flag it for possible genetic damage. Chronic exposure in workplaces with poor protective measures could set the stage for bigger medical problems down the road.
Environmental Harm Matters, Too
Healthy land, air, and water support everything: work, play, and food. Mishandling epibromohydrin can threaten local waterways and soils. Once such chemicals slip into liquids that drain to rivers or groundwater, life downstream suffers – both human and animal. Reports from the European Chemicals Agency highlight acute aquatic toxicity. If spilled, it can kill fish and disrupt important cycles in waterways. Nobody wants to explain to their neighbors why there are warnings at the local fishing pond.
This chemical doesn’t break down quickly. The risk lingers, especially near mishaps or storage facilities that cut corners. Living near a plant handling epibromohydrin means residents keep a closer watch during storms or accidents, knowing that a spill carries risks much bigger than short-term inconvenience.
Solutions and Safer Practices
Practical prevention starts with strong regulation and day-to-day care. Strict labeling, leak-proof storage, and transparent reporting create safer conditions for workers and communities. Employers need to train staff for emergencies and never let cost-cutting chip away at safety. Regular air monitoring reduces guesswork and directly keeps people safe.
Switching to safer alternatives deserves more attention. Companies have adopted less hazardous chemicals when possible, cutting down on risk at the source. Government oversight plays a central role here. Environmentally conscious business owners also work to recycle or neutralize leftover chemicals before waste leaves their site.
Community voice changes everything. Local residents living near manufacturers should know what’s stored nearby. Public meetings, plain-language notice boards, and easy-to-read safety data sheets empower people to make smart choices.
Looking Closer at Everyday Impact
Workers and families living near industrial corridors hear about incidents and chemical releases far more often than those who don’t. Understanding the real effects of chemicals like epibromohydrin means listening to their stories, following health statistics, and learning from mistakes in the past. Long experience working with hazardous chemicals shows that most problems come from ignoring small leaks, skipping training, or hiding information from those who deserve to know.
Epibromohydrin acts as a useful reminder that risky chemicals demand respect at every step—from factory floor to the riverbank. Safety culture, tough oversight, and honest conversation hold the best promise for protecting both people and nature.
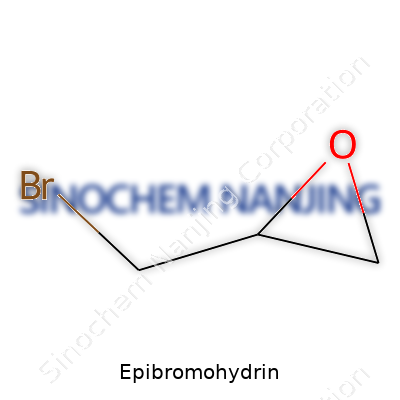
| Names | |
| Preferred IUPAC name | 2-(Bromomethyl)oxirane |
| Other names |
1-Bromo-2,3-epoxypropane 1,2-Epoxy-3-bromopropane Bromohydrin oxide Bromomethyloxirane 3-Bromo-1,2-epoxypropane |
| Pronunciation | /ˌɛpɪˌbroʊmoʊˈhaɪdrɪn/ |
| Identifiers | |
| CAS Number | 106-89-8 |
| 3D model (JSmol) | `/load ?modelID=mol:Epibromohydrin` |
| Beilstein Reference | 803749 |
| ChEBI | CHEBI:82236 |
| ChEMBL | CHEMBL169782 |
| ChemSpider | 52814 |
| DrugBank | DB08326 |
| ECHA InfoCard | 05d7b2a2-81fc-4b61-aba9-c8a49b87f4db |
| EC Number | 203-382-9 |
| Gmelin Reference | 5825 |
| KEGG | C14136 |
| MeSH | Epibromohydrin MeSH: D004824 |
| PubChem CID | 7507 |
| RTECS number | RH7760000 |
| UNII | 41S9XJ2H2D |
| UN number | UN2345 |
| Properties | |
| Chemical formula | C3H5BrO |
| Molar mass | 187.01 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | sweetish |
| Density | 1.660 g/cm3 |
| Solubility in water | insoluble |
| log P | 0.2 |
| Vapor pressure | 0.8 mmHg (20°C) |
| Acidity (pKa) | 14.54 |
| Basicity (pKb) | 1.36 |
| Magnetic susceptibility (χ) | -6.98 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4870 |
| Viscosity | 2.55 cP (20 °C) |
| Dipole moment | 2.74 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 248.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -95.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3787.7 kJ/mol |
| Pharmacology | |
| ATC code | V09AX04 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS05 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H314, H317, H335, H341, H351 |
| Precautionary statements | P210, P261, P280, P301+P310, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 3-2-2-W |
| Flash point | Flash point: 59 °C |
| Autoignition temperature | 605°C |
| Explosive limits | “1.8–10.9%” |
| Lethal dose or concentration | LD50 (oral, rat): 210 mg/kg |
| LD50 (median dose) | LD50 (median dose): 123 mg/kg (oral, rat) |
| NIOSH | NT8050000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Epichlorohydrin Epoxy propane Glycidol 1,2-Epoxy-3-chloropropane Propylene oxide |

