Understanding Dipropylene Glycol Methyl Ether Acetate: Value, Safety, and the Road Ahead
Tracing Roots and Evolution
Every modern solvent comes from a story of pressing needs and persistent refinement. Dipropylene Glycol Methyl Ether Acetate, known by many as DPM Acetate, emerged out of decades of work in the glycol ether family, which keeps showing up wherever fast, effective, and manageable solvents are required. Demand for safer and more versatile alternatives to old harsh solvents began rising after tighter environmental rules and growing worker-safety demands pushed chemists and manufacturers to look for something that could deliver performance but leave some of those hazards behind. What once started as a niche product found its spot in everything from paint formulations to specialty coatings, and that evolution never happened in isolation—an entire ecosystem of research, regulation, and application shaped each change along the way.
What Does DPM Acetate Really Bring?
DPM Acetate looks like a clear, colorless liquid and carries a mild odor. What that really means to me is that, even after spending time in both paint shops and coating laboratories, it feels less punishing on the senses than many alternatives. The liquid itself boils at a moderate temperature, so it doesn't evaporate madly or hang around forever. Its moderate polarity and balanced evaporation rate make it attractive to formulators. I’ve seen people reaching for it in thinner blends when they wanted to control drying times without sacrificing finish. It dissolves a wide range of resins, which opens doors for product formulators who can’t quite get there using older, harsher chemicals.
Labeling, Specifications, and the Tricky Balancing Act of Standards
Clear information on technical specifications matters because people need to work safely and get predictable performance. Labeling requirements in North America, for example, come shaped by OSHA’s HazCom rules and the GHS framework. Labels print clear hazard icons and list out physical properties—things like flash point, boiling point, and recommended storage conditions matter in daily practice. Pure DPM Acetate usually is not classified as a high-hazard solvent but it still requires respect: keep the containers sealed, control ignition sources, and avoid unnecessary contact. Technical data sheets often run a bit dry, but those numbers come from actual lab work; viscosity, vapor pressure, and water solubility affect how a batch will handle or blend in a plant.
How the Chemistry Comes Together
Making DPM Acetate happens by etherifying dipropylene glycol methyl ether with acetic acid, a process built on straightforward organic chemistry. This sort of reaction calls for conditions that most industrial facilities handle every day: controlled temperatures, foam management, and a cleanup of byproducts. Modifications come into play when custom properties matter; maybe the end user wants more tail in the evaporation curve or a different interaction with resins. These tweaks rely on careful changes to feedstocks, catalysts, or reaction time. Consistency isn’t just a matter of pride—products may need to hit the same performance marks every run, or else jobs, coatings, and equipment could all suffer.
Names Matter: Synonyms and Confusion in the Marketplace
One of the issues that keep coming up in industrial sectors involves confusion over synonyms. DPM Acetate pops up under different names, like Dipropylene Glycol Monomethyl Ether Acetate or 1-(2-Methoxy-1-methylethoxy)-2-propanol acetate, depending on the supplier or the standard. It’s easy to see how someone in a warehouse or a purchasing office could misread or mix labels, leading to costly order errors or, worse, improper handling on the job. This highlights the ongoing need for clear communication down the supply chain, from the original chemical synthesis to the final blending stage.
Living with Safety, Not Just Talking About It
Handling DPM Acetate never means throwing care out the window just because it poses fewer acute hazards than old-school heavy hitters like toluene or xylene. I remember the early days of my career when safety talks sometimes skipped over “safer” solvents. That led to unnecessary skin contact and headaches for some of the crew. Even with lower toxicity, regular use in closed environments, poor ventilation, or hot conditions can raise the risks—at the very least eye and skin irritation, or dizziness for someone dealing with spills. Modern standards call for local exhaust ventilation, chemical-resistant gloves, proper storage, and training. Seeing the shift from the “tough-it-out” approach to the zero-incident safety culture has made a big difference to workplace health.
Where Does It Go: Real-World Applications
You see DPM Acetate in product lines across automotive paints, industrial and architectural coatings, inks, and specialty cleaners. In practice, its big selling point is the ability to dial in open time for paints and coatings—painters want enough workability to avoid roller marks and streaks, but they don’t want to wait forever for things to cure. Print shops use it in ink formulations for its solvency and moderate evaporation, keeping print heads clean without gunking up the works. I’ve watched chemists rely on it for water-based blends that need a little extra solvency punch. Beyond manufacturing, most people never realize how much the look and durability of their car, phone, or furniture depends on these behind-the-scenes formulations.
Pushing the Limits: R&D and Toxicity Questions
Lab teams keep digging into alternatives all the time. Toxicity profiles matter more today than ever. Research shows DPM Acetate has lower acute toxicity and doesn’t show signs of major mutagenicity or reproductive hazards at occupational exposure levels. Chronic studies look good so far, but researchers still monitor for subtle and long-term effects, especially on vulnerable populations. Green chemistry trends drive R&D groups to look for even safer, even lower VOC substitutes—sometimes with plant-based feedstocks. Process chemists experiment with the recycling of solvent streams, hoping to close the loop on waste and emissions. Tighter standards push companies to tweak molecules and processes to meet stricter air-quality rules in places like California, the EU, and Japan.
The Way Forward—Future Prospects and Solutions
Demand for DPM Acetate doesn’t look likely to drop as long as coatings and electronics keep progressing. But, the chemical industry faces headwinds: rising regulatory hurdles, community resistance to traditional manufacturing, and mounting pressure to address micro-exposures and cumulative health risks. Smart solutions will likely come from rethinking the way solvents are produced, distributed, and recycled. More process innovation means higher purities, less waste, and more flexible blends. Transparency throughout the supply chain will be key—clearer labeling, reliable safety data, and consistent standards between countries. Training workers to handle solvents with respect and investing in new research, rather than patching up old processes, keeps risk down and opens the door for greener alternatives. My experience tells me that innovation happens fastest where manufacturers, regulators, and end-users keep the conversation focused on real-world needs rather than sticking with “good enough.” DPM Acetate stands as a snapshot—of where chemical engineering meets everyday life and the never-ending push for better, safer, and more sustainable choices.
What is Dipropylene Glycol Methyl Ether Acetate used for?
The Life of a Solvent
Dipropylene Glycol Methyl Ether Acetate, known in chemical circles as DPM Acetate, shows up where most folks rarely look. If you have walked through a house with fresh paint on the walls or visited an auto body shop, you have probably gotten a whiff of this solvent without realizing it. DPM Acetate slips quietly into common products: paints, coatings, inks, and cleaning agents. For years, people in manufacturing have leaned on it for one main reason—it plays well with both water and oil. That opens a door to all sorts of creative chemical blends.
On the Paint Line
Freshly sprayed lacquer, smooth automotive enamel, durable industrial coating—many roll out better and dry more evenly with help from DPM Acetate. In my own days painting a rental house, some of the best results came from paints staying ‘wet’ long enough to work the tricky edges. This chemical slows down how fast paint dries, fending off brush marks and lap lines. That means less stress and rework. Finished pieces look good, so fewer materials and labor hours go to waste.
Behind the Label: Printing and Inks
The world of package printing might look high-tech, but a lot of it comes down to chemistry. DPM Acetate dissolves pigment and resin nicely, giving printer operators more control over viscosity and drying times. High-quality print jobs rely on this balance—if the ink dries too fast, you get clogging; too slow, and you get smudges. In one shop I worked at, managing these properties meant hitting deadlines or burning through overtime. Reliable solvents mean smoother runs, less scrap, and more uptime.
Formulating Cleaners
Janitorial supply aisles offer row upon row of cleaning sprays—some for glass, others for greasy counters, a few for both. Many of the streak-free and degreasing claims owe something to DPM Acetate. This solvent breaks up sticky residues and then evaporates, leaving less need to double-wipe surfaces. Offices and hospitals rely on quick-drying cleaners to keep floors safe and shiny. I have seen maintenance staff favor products with this solvent for both efficiency and effectiveness.
What About Safety?
Some solvents carry nasty health baggage. DPM Acetate avoids a lot of those headlines—but that still doesn’t mean you ignore safety data sheets. Workers deal with real risks if they breathe too much solvent vapors or skip gloves. According to the European Chemicals Agency, exposure limits matter. Proper ventilation and personal protective equipment lower the chance for headaches, skin irritation, and the longer-term issues that come from chronic contact.
Facing Environmental Questions
Big manufacturers can’t ignore regulatory rules. DPM Acetate lands in a safer category than old-school options like toluene, which caused more environmental headaches. Even so, plant managers and supply chain teams need to keep inventories in check, minimize spills, and follow disposal rules. Regulatory agencies encourage the use of less volatile organic compounds to limit air pollution. There’s always room to switch to low-impact chemicals, though finding alternatives with the same performance takes patience and investment.
Going Forward
As industry standards evolve and consumer awareness grows, companies look for safer, more sustainable solvents that still deliver results. It pays to be informed—both for buyers making choices and for workers handling these products every week. Readers can ask manufacturers for more details before bringing a chemical into their workplace or home. Demand for new, lower-emission paints and cleaners will keep growing, and that pressure can lead to smarter, safer chemistry for all.
Is Dipropylene Glycol Methyl Ether Acetate hazardous to health?
Common Uses and Exposure
Dipropylene glycol methyl ether acetate pops up in a lot of places—paints, inks, cleaning agents, and coatings. It’s a popular choice for manufacturers because it helps dissolve or thin other substances without a harsh chemical smell. If you work in printing, auto refinishing, or even household cleaning, you have probably come across it. Most folks don’t think twice about these ingredients, but I’ve noticed a rising interest in their actual impact on our well-being.
What Science Says About Safety
Safety data from the Environmental Protection Agency and the European Chemicals Agency agree that dipropylene glycol methyl ether acetate isn’t considered a major toxin. It doesn’t carry the same danger labels as some other solvents. In animal studies, it has taken high doses to see significant health changes. For someone handling a paint can at home, the risk doesn’t look huge.
That being said, these sources do flag some risks. Breathing in the vapor can irritate your nose and lungs, especially in a room that doesn’t have enough fresh air. Splashes on the skin can lead to dryness or mild irritation, which I’ve seen pretty often in folks who handle paint thinners or degreasers at work. Letting any solvent stay on your skin can be a bad habit, and gloves go a long way here.
Long-Term and Occupational Risks
Most concern comes up in jobs with repeated or long-term exposure. Factories or large workshops can trap fumes indoors, so hourly workers might end up breathing these vapors shift after shift. Government health reports show that headaches, dizziness, and nausea hit hardest in poorly ventilated spaces. That lines up with stories I’ve heard in industrial spraying and screen printing.
Nobody sees severe organ damage at consumer-level or brief exposures, but workers in high-exposure industries can see health decline if they skip the basics—ventilation and protective gear. Chronic exposure to a lot of solvents, even mild ones, puts extra strain on the nervous system and liver over time. This is where regulations step in, setting limits for how much can hang around in the air.
Raising Safety Standards
Most companies know the drill: use a decent extractor fan, give workers masks, and rotate jobs to cut down single-person exposure. I’ve worked with teams who kept open windows and wore gloves, and health complaints dropped sharply. Labels have started explaining risks in plain language, which arms people with knowledge they need on the job or at home. Product makers update safety sheets regularly, giving both bosses and workers the right facts before each use.
What Could Improve Health Outcomes
Access to better air flow remains the biggest fix for headaches and sore throats. Workplaces that set up fans and vent fumes outside protect workers’ lungs every day. Store all solvents in tight-sealed containers away from heat, and never pour them down a sink. Training sessions help everyone understand why gloves and goggles matter even for common products. If someone feels lightheaded or sick, heading out for fresh air makes a real difference.
Governments and industry watchdogs run regular checks in high-use workplaces, which keeps everyone accountable. As research continues, up-to-date limits can get tighter, and safer substitutes might show up, lowering any health concerns even more.
What industries commonly use Dipropylene Glycol Methyl Ether Acetate?
The Paints and Coatings Business
Many people don’t think twice about what goes into that can of wall paint sitting in the garage. In my years working with contractors and paint crews, one chemical pops up in supply orders again and again: Dipropylene Glycol Methyl Ether Acetate, or DPM Acetate. Coatings manufacturers use this solvent to keep paint flowing smoothly from brush to surface, and to extend drying times so professionals can avoid streaks and lap marks. DPM Acetate helps paints settle before hardening, which matters a lot on a crowded job site or a hot summer day.
Industrial-scale painters often need something that can keep up with busy schedules. DPM Acetate fits because it toughens a paint film and gives painters some breathing room, even with fast-paced rollouts. It also keeps paint from thickening up too much during application, which means fewer headaches for workers and more consistent results for clients.
Electronics Manufacturing
Circuit boards and electronic parts go through rigorous cleaning and coating stages in factories across the globe. DPM Acetate’s low volatility and stable nature make it a solid choice for removing stubborn residues, fluxes, and greasy layers from delicate surfaces. I remember talking with a process engineer who swears by this solvent for achieving spotless results before soldering.
Manufacturers aim to avoid leftover contaminants, which can disrupt circuits or cause failures. Factories turn to DPM Acetate because it lingers longer than many alternatives, offering better control over cleaning time without risking damage or corrosion. In a world obsessed with shrinking electronics and tighter tolerances, choosing the right solvent shapes the reliability of the end gadget.
Printing and Ink Formulation
Printers value DPM Acetate for its ability to dissolve pigments and resins, leading to vibrant and even color transfer onto paper or plastic sheets. Commercial printers usually juggle jobs with tight deadlines, so they look for inks that stay open long enough to avoid clogs and downtime. This solvent delivers that flexibility, without the harsh fumes of some older chemicals.
The printing industry adopted DPM Acetate as a safer way to balance performance with worker safety. It tames the evaporation rate, so ink doesn't dry out inside the press or splatter during a big run. Anyone who’s ever worked in a print shop knows how frustrating equipment breakdowns can get—choosing the right solvents goes a long way toward keeping the presses rolling and the air a bit less noxious.
Automotive and Aerospace Applications
Assembly lines for cars or planes use plenty of surface treatments—paints, adhesives, cleaners. DPM Acetate shows up in degreasing baths and in coating formulations for car bodies, aircraft interiors, and plastic trims. Its strong solvency tackles tar, grease, or oil without stripping away base materials.
Regulators keep an eye on chemical safety, especially in spaces where people work closely with solvents. DPM Acetate’s relatively low toxicity has pushed it forward as environmental rules phase out harsher alternatives. Maintenance crews and quality inspectors rely on it to pre-treat surfaces before painting or gluing, knowing that a little extra control during prep often pays off with stronger adhesion and longer service life.
Room for Cleaner Choices
The growth of DPM Acetate across so many sectors points to real needs: safer workplaces, efficient production, products that stand up to the real world. Companies now face pressure to cut emissions and reduce exposure risks even further. Investing in closed-loop recycling, automating handling, and picking safer blends keeps people healthy and business running smoothly. Factories often review ventilation and training programs to keep solvent use as safe as possible.
How should Dipropylene Glycol Methyl Ether Acetate be stored?
Why Attention to Storage Matters
Dipropylene Glycol Methyl Ether Acetate, a mouthful if you try to say it fast, shows up in paint thinners, industrial cleaners, and coatings for a reason. It does a fine job breaking things down and blending materials. But the same qualities that make it helpful in a factory or workshop can turn risky if the storage is a mess. Growing up in an area with lots of small paint shops, I saw more than once what happens when barrels leak or sit next to a hot radiator longer than they should. Losses don’t just hit the business—workers and neighbors pay for it through health problems or chemical fires.
Storing Smart, Not Just Safe
Ventilation isn’t something you notice until the odor creeps in or you start to feel dizzy at work. Dipropylene Glycol Methyl Ether Acetate brings noticeable fumes. So, stashing containers in a stuffy utility closet or an unventilated basement ramps up the risk of headaches and long-term harm. Keeping storage areas open to fresh air, ideally with exhaust fans that move the fumes up and away from people, makes a world of difference. The stuff can build up and hang around—it tends not to just “fade away” on its own.
Direct sunlight heats metal drums and plastic containers, creating pressure inside and pushing vapor out into the room. Fires can ignite from stray sparks or a forgotten cigarette butt. Shade matters. Roofs with overhangs or storage sheds with no windows slow down temperature spikes and cut the chance of a mishap, something every neighbor would appreciate.
Labeling and Training: More Than Red Tape
Labels look boring, but the amount of confusion they dodge is remarkable. I walked through warehouses and took shortcuts plenty of times, and I saw workers reach for containers thinking they grabbed paint, only to spill solvents all over the floor. Labels that spell out contents, hazards, and expiration save people from making the kind of mistakes that insurance won’t always cover. Training seems repetitive at first glance, but folks who hear stories and learn what can go wrong end up keeping each other safer.
Leaning on Good Practices
Spill containment trays catch drips before they soak into the ground or leak onto a walkway. Drains should not run directly into public sewers where chemicals find their way back into our water. Simple shelf choices—no stacking heavy drums on lighter ones—plus inspection schedules keep the storage area trustworthy. A neighbor of mine kept old coffee cans to hold leaking liquids “just in case,” and it never did any good. Only sturdy, sealed containers meant for chemical storage do the job right.
Fires still pose a threat, so keeping fire extinguishers and sand buckets close by saves response time. Local codes usually require them, but out in the country or in small garages, people skip this step. I learned after seeing one stubborn fire in a neighbor’s shop that common sense beats shortcuts every time.
Safeguarding Health and Environment
Safe storage of Dipropylene Glycol Methyl Ether Acetate protects more than a warehouse. It shields everyone in the area—workers, customers, even pets—from harm. The chemical does its job best when treated with respect, not fear or forgetfulness. Double-checking lids, reading labels, and setting up basic safety measures lower the odds of a crisis, and those steps matter every day, not just after an accident.
What is the chemical formula of Dipropylene Glycol Methyl Ether Acetate?
Digging Into the Chemical Formula
Dipropylene Glycol Methyl Ether Acetate carries the formula C8H16O4. You won't bump into it at the local grocery store, but spend time in manufacturing or lab settings and it pops up again and again. The chemical bulks up commercial cleaning solutions, paints, inks, and coatings. It's not just about performance either. The chemical structure matters a lot—every bond and atom count shapes how it works in real-life situations. C8H16O4 breaks down to eight carbon atoms, sixteen hydrogens, and four oxygens. Not a flashy lineup on paper, but pretty powerful once you understand what it brings to the table.
The Reason It Matters to Industry and Daily Life
Factories use this solvent because of its stability and versatility. It’s not overly reactive with other chemicals, so you see less breakdown and more consistent results. Safety data show that, compared to many other solvents, it’s not as aggressive or volatile. That doesn’t make it harmless, but practical people favor it since it carries a lower risk of fire or health effects for workers if used responsibly. I've spoken to painting contractors who prefer products containing Dipropylene Glycol Methyl Ether Acetate simply because it dries at the right rate and smells less pungent than rivals. Less odor equals happier workers and better relationships with customers who don't want their house smelling like a chemical plant.
Safety Shouldn’t Be an Afterthought
Still, safety habits matter. The Centers for Disease Control and Prevention lists possible symptoms from high exposures: headache, nausea, dizziness. Respirators, gloves, ventilation—those all matter in the real world, especially in stuffy workshops or storage rooms. One mistake I made in my early lab days was thinking a more "benign" chemical didn't need much care. Reading up on the Material Safety Data Sheet afterwards drove the point home—every solvent deserves respect.
Environmental Impacts Keep Us Honest
Regulations around the world keep tightening. Dipropylene Glycol Methyl Ether Acetate doesn’t hang around for ages in the environment, but wastewater from industrial sites can still carry it out to streams and rivers. Water treatment plants can catch some, but not all. Looking at emission inventories, the Environmental Protection Agency tracks around a million pounds released annually in the United States. These numbers raise the bar: industry leaders face the pressure to capture solvents before they escape into the air or water.
Practical Solutions for Safer Use
What works in one factory might flop in another, but smarter design and updated filters cut down on waste. Closed mixing systems, for example, trap vapors so they don’t escape and reduce health risks for workers. Companies have started trialing alternative solvents with shorter life cycles in nature, but so far, few options match the balance of safety and performance. Continuous training makes a big difference—workers who see why precautions matter step up and follow best practices. On-site recycling is another fix. Collecting spent solvent, cleaning it, and reusing it keeps materials out of the environment and cuts costs for companies.
More Than Just a Formula
C8H16O4 isn't a random collection of atoms—it's a backbone for many things we use every day. The challenge is balancing performance, safety, and environmental health. Clear data, like what comes from the EPA and CDC, helps us steer toward smarter choices. New solutions always take time, but every upgrade in handling or cleanup nudges us closer to safer workplaces and cleaner surroundings.
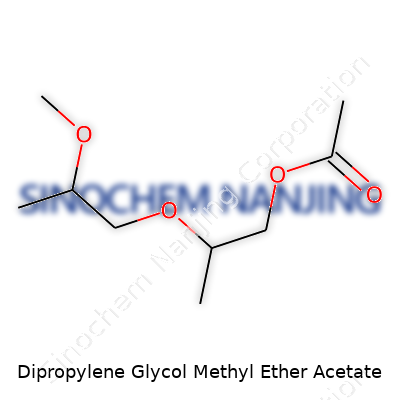
| Names | |
| Preferred IUPAC name | 1-methoxy-3-(2-methoxy-1-methylethoxy)propan-2-yl acetate |
| Other names |
DPM Acetate 1-(2-Methoxy-1-methylethoxy)propan-2-yl acetate Dipropylene glycol monomethyl ether acetate Propylene glycol methyl ether acetate PGMEA Dowanol DPM Acetate |
| Pronunciation | /daɪˌproʊ.piːˈliːn ˈɡlaɪˌkɒl ˈmɛθ.əl ˈɛθ.ər əˈsiː.teɪt/ |
| Identifiers | |
| CAS Number | 88917-22-0 |
| Beilstein Reference | 1365531 |
| ChEBI | CHEBI:88190 |
| ChEMBL | CHEMBL1651957 |
| ChemSpider | 147301 |
| DrugBank | DB14183 |
| ECHA InfoCard | 100.047.801 |
| EC Number | DPGMEA EC Number: "252-104-2 |
| Gmelin Reference | 11177 |
| KEGG | C14323 |
| MeSH | D005898 |
| PubChem CID | 11637 |
| RTECS number | KL5950000 |
| UNII | AU44GYD8HQ |
| UN number | UN number: "UN3272 |
| Properties | |
| Chemical formula | C10H20O4 |
| Molar mass | 190.24 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Mild ether-like |
| Density | 0.957 g/cm³ |
| Solubility in water | miscible |
| log P | 0.36 |
| Vapor pressure | 0.05 mmHg @ 20°C |
| Acidity (pKa) | pKa > 13 |
| Magnetic susceptibility (χ) | -8.67 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.416 |
| Viscosity | 0.95 mPa·s |
| Dipole moment | 3.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 324.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -726.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4549.7 kJ/mol |
| Pharmacology | |
| ATC code | D02AE51 |
| Hazards | |
| GHS labelling | Warning, H227, H336, P210, P261, P271, P304+P340, P312, P403+P233 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P243, P261, P271, P280, P301+P312, P305+P351+P338, P337+P313, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | ≈ 75°C |
| Autoignition temperature | 190 °C (374 °F) |
| Lethal dose or concentration | LD50 (oral, rat): 5,190 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 5,000 mg/kg |
| NIOSH | RV3500000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 150 mg/m³ |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Dipropylene Glycol Dipropylene Glycol Monomethyl Ether Propylene Glycol Methyl Ether Acetate Ethylene Glycol Monomethyl Ether Acetate Tripropylene Glycol Methyl Ether Acetate |

