Looking Deeper into Dipropylene Glycol Dimethyl Ether: Why This Solvent Stands Out
Historical Development: Industry’s Search for a Better Solvent
Back in the late twentieth century, chemists looked for solvents that could handle demanding reactions without causing more harm than good. Petroleum-based solvents dominated the field, but folks started raising eyebrows about flammability and health risks. Researchers wanted safer alternatives that still delivered. Dipropylene glycol dimethyl ether (DPGDME) walked onto the scene as a product of those efforts, bringing together the flexibility of glycol ethers with a chemical structure built for resilience. Over time, as industries faced calls for cleaner and safer working spaces, DPGDME earned its place as a less volatile, less toxic alternative for certain applications.
Product Overview: From Laboratory Benches to Factory Floors
You find DPGDME on shelves both in research labs and on production lines. It shows up as a colorless, nearly odorless liquid, which actually matters more than most consumers realize. Strong-smelling solvents often cause discomfort and pose a greater risk of inhalation exposure. Its faint scent and stable nature let technicians work longer hours with less hassle, and industries can meet demanding emission standards without skimping on performance. My own experience working with glycol ethers taught me the value of a solvent that doesn’t overwhelm the workspace; DPGDME delivers here in ways traditional ethers and acetates sometimes fail to match.
Physical & Chemical Properties: The Right Balance
Dipropylene glycol dimethyl ether offers a solid mix of low viscosity, high boiling point, and broad solubility with both polar and nonpolar compounds. These traits matter for reactions involving sensitive or unstable components. The molecule resists hydrolysis; in plainer terms, it doesn’t break down easily under moist conditions. This chemical endurance simplifies storage and handling, and prevents reactions from spiraling out of control when water sneaks in. With a flash point comfortably higher than many legacy ethers, the risk of unexpected ignition lowers, making warehouses and labs a little less stressful. This isn’t the only property chemists care about, but it’s one of those practical factors that can make or break a solvent’s reputation over time.
Technical Specifications & Labeling: Real-World Considerations
Anyone working with chemicals soon realizes labels and data sheets are about more than just paperwork. Regulations keep tightening, and DPGDME keeps pace by meeting thresholds for purity, flash point, and boiling range demanded by chemical safety authorities. Labels reflect that, clearly outlining its composition and warning of the minor hazards present. Lab technicians and production managers gain from knowing what’s in the bottle, as ambiguities around impurities or mislabeling can result in ruined experiments, wasted raw materials, or—far worse—injuries. Having spent time in both academic and commercial settings, I’ve seen that crystal-clear technical information saves hours of headaches.
Preparation Method: Getting it Right Every Step
Manufacturing DPGDME involves etherification of dipropylene glycol using dimethyl sulfate or similar reagents. Process engineers control reaction time, temperature, and the removal of byproducts to hit purity targets and avoid dangerous leftovers. The reaction is straightforward but must be managed tightly. Waste minimization becomes significant here: anyone who’s spent time on a plant floor knows dealing with hazardous byproducts can turn simple work into a compliance nightmare. Proper design and oversight prevent unwanted side reactions, keep energy use in check, and limit the exposure of technicians to harsh chemicals. Industrial chemists still keep an eye on greener synthesis routes, hoping catalysts or alternative reagents could one day offer even safer, cleaner processes.
Chemical Reactions & Modifications: Not Just a Passive Solvent
DPGDME doesn’t just dissolve compounds and watch from the sidelines—it sometimes nudges the chemistry itself. Its strong solvating power makes it a favorite for both basic and acidic conditions, and its relative inertness can head off unwanted side products. In areas like lithium-ion battery electrolytes and certain polymerizations, DPGDME shines because it helps keep ions mobile or makes chains grow just right. Chemists who run into problems with traditional solvents often turn to DPGDME, hoping it’ll tip the balance. Some research even explores subtle modifications to the molecule’s ether groups, seeking tweaks that could make an even better fit for specific reactions.
Synonyms & Product Names: Navigating the Ethers’ World
A chemical as versatile as DPGDME pops up under a handful of names, reflecting both industry jargon and trademarked variants. Folks might see it listed as just “DMM” or “DPGDME,” with its CAS number usually included for clarity. Some big manufacturers brand it for specialty purposes, but beneath the trade names, the structure doesn’t change much. Knowing these synonyms strikes me as more than an academic exercise—it helps avoid confusion that could cause supply mix-ups, source counterfeit goods, or lead to regulatory headaches. Over the years, I’ve watched colleagues waste precious time because two different departments didn’t realize they were talking about the same molecule.
Safety & Operational Standards: Keeping Workplaces Secure
DPGDME lines up with stricter safety rules designed to reduce chemical injuries. Compared to stronger ethers, the moderate toxicity and lower volatility make accidental exposures less disastrous; spills don’t escalate as quickly, and inhalation risks drop. Still, it carries the usual flammability and organ toxicity flags common to glycol ethers, so solid ventilation and proper gloves remain standard practice. The best labs drill protocols and invest in sturdy storage cabinets. In places I’ve worked, a culture of safety got results not by relying on individuals’ memory but by building easy-to-follow handling guides right into the facility’s design. DPGDME doesn’t erase risk, but it responds well to these common-sense steps.
Application Area: Where DPGDME Makes a Real Difference
Industries flock to DPGDME for its ability to dissolve both tough resins and delicate pharmaceuticals. Paint and coating manufacturers use it to keep their products flowing smoothly and drying predictably. Battery researchers want it in their electrolyte blends, hoping for longer life and fewer fire risks. Pharmaceutical labs pull from its solvent properties during both synthesis and purification. I remember trying to find a single solvent system for a sticky resin that wouldn’t budge in anything else—DPGDME succeeded where standard glycol ethers fell short. Every application draws from its versatility, but the through-line is about optimizing both product safety and performance.
Research & Development: Scientists Seek More
Researchers keep testing new ways to tweak DPGDME for better selectivity, biodegradability, or lower toxicity. The drive comes partly from environmental pressure—solvent waste ranks high among industrial pollutants. Scientists test new catalysts, alternate feedstocks, and greener purification steps all chasing a cleaner solvent lifecycle. Collaboration between industry and academia happens more now than it did during my early days in the lab. Multidisciplinary teams move projects forward, aiming to cut costs and environmental impact without giving up the chemical virtues that made DPGDME attractive in the first place. Real-world needs keep R&D focused on actionable improvements.
Toxicity Research: Understanding Real Risks
The toxicity of glycol ethers always concerns anyone serious about workplace safety. Research on DPGDME shows a middle ground: less hazardous than older ethers but still demanding respect. Animal studies measure metabolic breakdown, looking for signs of organ stress or reproductive effects. Occupational studies track air and skin exposure, shaping guidelines that make workplace safety concrete, not just theoretical. Where safety data once lagged, now regular updates let employers and regulators set clearer thresholds. From what I’ve seen, nothing beats regular repeat tests and open data sharing—workers and managers both make better decisions with up-to-date stats on hand.
Future Prospects: Toward Safer Chemistry
Looking forward, the path for DPGDME depends on whether researchers and manufacturers can deliver versions with even less toxicity, greater biodegradability, or renewable origins. The push for green solvents won’t slow down. Regulatory authorities set steeper demands on impurity levels and environmental fate—solvent makers know these hurdles are not going away. Battery technology and pharmaceutical synthesis continue to lead the way in demanding performance beyond what older solvents can provide. From years at the intersection of research and production, I see one clear trend: only those solvents, DPGDME included, that align with health, safety, and sustainability priorities will thrive in future markets. Companies willing to share real-world data, invest in safer formulas, and look for circular production methods will keep their edge as the story of DPGDME continues.
What are the main uses of Dipropylene Glycol Dimethyl Ether?
Where This Solvent Shows Up in Real Life
Dipropylene Glycol Dimethyl Ether isn’t the catchy sort of name you’d drop at dinner, but if you dig around in laboratories or chemical plants, you’ll spot it in the thick of things. I remember a project at a specialty coatings shop, and we spent as much time choosing the right solvent as we did on pigments. With its low odor and ability to dissolve a broad range of substances, this chemical became the unsung support act, making many of those bright, glossy paints possible.
Industrial Cleaning Goes Up a Notch
Factories often get messy in ways most folks don’t think about. Grease, ink, and sticky residues don’t budge for soap and water. Dipropylene Glycol Dimethyl Ether steps up when hot water runs out of steam. Its good solvency lifts stubborn contaminants without punching holes in plastic or metal parts. Over the past decade, its use as an alternative to high-toxicity solvents has gained momentum, and that matters for workers spending whole shifts in the splash zone.
Pushing the Limits in Electronics
Electronics assembly relies on chemicals that don’t fry delicate bits. Folks soldering phones, laptops, and circuit boards lean on solvents like this one because it dries slow and steady—no streaks, no leftover chemicals corroding expensive boards. When cleaning flux from tiny joints, you want dependability. Engineers I’ve worked with opt for dipropylene glycol dimethyl ether mainly to reduce damage and boost product lifespan. Over time, this helps devices last longer, lowering waste from gadgets tossed too soon.
Pharmaceutical Moves That Don’t Get Noticed
Pill coatings and granules can get sticky. Pharmaceutical manufacturers look for solvents that dissolve both polar and non-polar compounds but don’t bring a hammer to the rest of their sensitive ingredients. I spent time working in a pill manufacturing facility—dipropylene glycol dimethyl ether cropped up in recipes meant to keep drugs stable, easy to swallow, and safer for patients. The chemical also plays a role in synthesizing active ingredients, helping chemists steer reactions without introducing harmful byproducts. Regulatory authorities pay attention here, so the solvent’s low toxicity helps it earn trust in sensitive applications.
Chasing Cleaner Paints and Sustainable Solutions
Paint and coatings producers have a balancing act: produce something glossy and durable, but keep it safe for both people applying it and the air we all breathe. Regulations on VOCs push the industry to look for safer solvent blends. Dipropylene glycol dimethyl ether shows up because it can lower surface tension, level out brush marks, and deliver solid performance without raising health flags like older solvents. This shift doesn’t just help professional painters breathe easier; it opens doors for safer DIY paints in homes and schools.
Paths Toward Safer, Smarter Industry Practices
Sticking with safer, more effective solvents means more than just swapping out a drum in the warehouse. We need regular training, better ventilation, and clear labeling so everyone on the floor knows what’s in those barrels. Encouraging manufacturers to push for greener choices—while supporting researchers hunting for even less hazardous alternatives—makes a difference. In my own experience, companies listening to feedback from chemists and field workers land on smarter, safer solutions faster. Moving toward better solvents like dipropylene glycol dimethyl ether can shrink workplace risks and improve product quality. Here, practicality and safety meet in each bottle.
Is Dipropylene Glycol Dimethyl Ether safe to handle?
Everyday Lab Life and Practical Hazards
Spending time around chemicals in the lab, you pick up a respect for the ones whose names get abbreviated, like Dipropylene Glycol Dimethyl Ether — often called DPGDME. Despite not being a household name, this clear liquid shows up behind the scenes in paint strippers, specialty cleaners, and even some battery electrolytes. Just because you don’t see its label on a store shelf doesn’t mean it skips the usual risks that come with strong solvents.
Exposure: Keeping It on Your Mind
It’s easy to forget how fast solvents enter your body through skin or lungs. DPGDME smells mild, but that doesn’t make it harmless. The liquid doesn’t burn the nostrils like acetone, which lulls some into skipping gloves or masks. That’s a mistake. Too many of us have felt the headaches after a long session in a poorly ventilated room, or the skin irritation from getting a splash and not washing up quickly. Occupational exposure limits for many glycol ethers sit low for good reason. Even basic handling, left unprotected, can bring chronic health risks with repeated exposure: reproductive effects, central nervous system symptoms, allergies. Labs run on precaution, not luck.
Who Decides What’s Safe?
Safety data sheets spell out the risks for DPGDME. The European Chemicals Agency places it in the category of irritants. The US Occupational Safety and Health Administration (OSHA) recommends handling with gloves and goggles, keeping exposure airborne below recommended levels. None of these guidelines appear out of thin air. They come from repeated studies, case reports, and a long history of workers showing symptoms after handling similar solvents. Years in the field have taught me this: Respect the data, but also respect what your nose and skin tell you.
Solutions for Safer Handling
Real safety looks like action, not just paperwork. A decent fume hood makes the difference between a quick project and a lingering chemical taste stuck in your mouth the rest of the afternoon. Proper gloves — thick nitrile or butyl, not cheap disposable kinds — keep skin from itching or drying out. Regular hand washing, not just glove use, shrugs off the accidental splash. No one wants to admit they wiped sweat from their forehead with the back of a gloved hand, only to regret it a half hour later.
Training also counts, and not the once-a-year video that everyone tunes out. Picking up the habit of checking chemical compatibility before reusing glassware, or watching out for spills on benchtops, keeps you ahead of the risks. Bigger organizations sometimes invest in real-time air monitoring. Even in a small operation, basic ventilation and good labeling go a long way.
Making Choices Based on Evidence
I’ve sat through arguments where someone claims a chemical is “safe enough” because no one they know’s gotten sick—yet. That’s not how health risks work. Chronic effects don’t pop up overnight. There’s no heroism in gambling with your future. The bottom line: DPGDME, like any solvent, earns caution. Respect it, gear up properly, ventilate the workspace, and treat its clean scent as a false friend. Safe handling grows from knowledge, vigilance, and habits built over time—not just from reading labels, but from carrying the lessons of the lab into every step of the workday.
What are the storage and handling recommendations for this chemical?
The Real Stakes of Chemical Handling
Working in a lab or warehouse with chemicals, there’s never much room for error. Some days, a single careless move can send a routine shift spiraling downhill—damage, fines, or health scares showing up without warning. Chemical storage sounds routine, but I’ve seen people cut corners and pay the price. Water-based substances, acids, solvents, and powders all bring their own headaches. One lesson I picked up firsthand: mishandling isn’t some distant worry, it sits on your shelf every day, waiting to create trouble.
Details Matter: Container Choice and Labeling
No two chemicals act alike—which means a single rule doesn’t cover all. For liquid reagents, using airtight glass or plastic containers can prevent spills, slow evaporation, and block unwanted reactions. Only original, factory-tested containers hold up to strong acids or bases over the long haul. Quick fixes with makeshift bottles or reusing water jugs turn simple tasks risky. Labels need to stick and stay legible, showing exactly what’s inside, concentration, hazard details, and date received. People skip relabeling, assuming they’ll “remember”—but in a busy setting, misidentification can land someone in the emergency room.
Temperature and Humidity: Why the Right Room Matters
Room conditions do half the work in chemical safety. A few years back, a colleague left oxidizers too close to a drafty window in July. High heat jumpstarted decomposition, leading to a nasty chemical cloud that could have ended much worse. Stuff like that proves climate control isn’t just for comfort. Aim for steady, recommended temperatures and humidity levels. Fluctuations may speed up unwanted breakdown, alter concentrations, or pop seals. Even where power outages rarely happen, backup systems and temperature monitoring alarms save headaches.
Segregation: Preventing Combos That Cause Problems
Some chemical accidents happen just because incompatible materials sit next to each other on a shelf. Acids kept beside bases, oxidizers near fuels—mixing these pairs, even by accident, can spark fires or explosions. In shared storage rooms, shelving systems built for separation work best. Put flammable liquids in metal safety cabinets designed to resist flames. Let acids have their own plastic trays, away from metals or materials they could corrode. Good practice keeps oxidizers, reducers, flammables, and toxics apart with distance and barriers, not just labels. Even in a small shop, color-coded bins and simple signage can make a big difference.
Personal Protection and Spill Plans
Nobody likes wearing goggles, gloves, and full aprons on a hot day, but skipping these steps never pays off. One splash—one chemical-soaked glove—taught me more than any safety lecture. If spills happen anyway, having clear emergency instructions on the wall, sand or spill pillows nearby, and eye wash stations ready beats fumbling around in crisis mode. Routine safety drills and visible inventories of safety gear foster a culture where accidents get managed, not ignored.
Training and Documentation: The Long View
Real safety comes down to habits. Ongoing training ensures new hires and seasoned workers follow the same playbook. Up-to-date data sheets, inventory logs, and incident reports help managers identify patterns and intervene before a small oversight becomes a hazard. Regular audits reveal gaps—missing labels, expired stock, or outdated emergency contacts. Taking notes and learning from near-misses keeps everyone diligent. Storing and handling chemicals isn’t about creating paperwork. It’s about building a system that guards people, property, and reputations with every bottle stored and every pair of gloves put on.
Can Dipropylene Glycol Dimethyl Ether be mixed with water or other solvents?
Understanding the Chemistry Up Close
Try searching for solvents that find a steady home in both the lab and the factory floor and Dipropylene Glycol Dimethyl Ether (DPGDME) quickly pops up. This solvent has carved out a space for itself among chemists and engineers, generation after generation. The big question—can you throw DPGDME together with water or other liquids and still get something useful? If you’ve ever tried to stir up a solution only to get a goopy mess, you might appreciate just how important it is to get this right.
Water and DPGDME: Not Always the Best Mix
Try pouring DPGDME into a cup of water, and you’ll quickly see that the two stay mostly separate. DPGDME doesn’t dissolve well in water. Its chemical structure acts like a shield, keeping water molecules at a distance. In the real world, this means you’ll see two layers form—the solvent won’t mix in evenly. Some people hope they’ll get a clear mixture, but the science doesn’t back that up.
I’ve seen this at work in an analytical lab. When teams need to run extractions, hoping to isolate compounds from water-based samples, picking DPGDME sometimes leads to frustration. The liquid floats on top, and you need to shake it hard to get any action. Even after all that work, the mix doesn’t stay together. This stubborn separation explains why others pick more water-friendly solvents for certain laboratory or factory jobs.
Pairing DPGDME With Other Solvents
Now, take DPGDME and try mixing it with solvents like acetone, ethers, or various alcohols, and you’ll see a different story. These solvents, with structures that “get along” with DPGDME, welcome it readily. You end up with a smooth, uniform blend, no strange clumping or layering. That’s why workers reach for DPGDME in tasks involving organic solutions—whether degreasing machine parts, stripping away stubborn coatings, or helping run a synthetic reaction. Compared to many other ethers, DPGDME often stands out for its low odor and greater safety margin.
Blending DPGDME with strong acids or strong bases creates its own set of hazards. Chemical compatibility charts show that under those harsh conditions, things might heat up fast or create volatile byproducts. Even in my own years tinkering in labs, I learned to double-check mixture charts and ask mentors before mixing any new combinations—especially when large volumes are involved.
Safety and Environmental Factors
Any time I’ve handled DPGDME, gloves weren’t just a suggestion—they were a must. This solvent doesn’t scream danger compared to some nastier chemicals out there, but it still causes irritation if splashed. Good ventilation and goggles make sense, especially during mixing. I also learned that leftover DPGDME and its mixtures, especially if contaminated with other chemicals, can’t go down the drain. Local waste rules usually ask for careful handling and specialized disposal, both to protect people and keep groundwater clean.
Solving Real-World Mixing Challenges
If a process demands a water-compatible solvent, switching to one designed for those conditions saves hassle. For jobs that can stick to organic systems, DPGDME brings a lot to the table—great dissolving power and a smoother odor profile than many alternatives. If you’re dead-set on blending water and DPGDME, emulsifiers or co-solvents can bridge the gap, though this route takes some care and testing. It boils down to matching the right tool with the right job, keeping practical chemistry and safety in focus.
What are the possible health hazards or first aid measures for exposure?
Everyday Exposures and The Risks They Bring
Most folks spend their days around all sorts of substances—cleaning products, paints, dust, fumes, or even just potent cleaners under the kitchen sink. Each of these brings its own risks for your health. Too much exposure, or a single big mishap, might mean short-term trouble like rashes, coughing, or stinging eyes, or bigger problems, like asthma, burns, or poisoning.
I remember helping a friend with yard work years back. We sprayed weed killer without gloves. By dinner, my hands itched like crazy. Turns out, skin can soak up chemicals faster than most people know. Nearly three million cases of poisoning happen yearly in the U.S. alone, most from household cleaners or pesticides, according to the CDC. Skin isn’t the only route. Breathing in strong fumes means those particles hitch a ride right into your lungs. Even food can be a culprit—think about mushrooms or berries in the wild.
Eyes, Lungs, and Skin: The Body’s Vectors for Trouble
No one enjoys that burning feeling after chopping chilies and rubbing their eyes. Multiply that by a chemical splash, and you can see how fast eye injuries become emergencies. People who work in shops or factories learn to respect safety glasses, since flying bits, dust, or even splashing liquids can damage eyesight in seconds. On the skin, harsh cleaners strip away natural oils or even cause burns. Longer exposure to things like cement or solvents might bring skin rashes, blisters, or even infections if not cleaned quickly.
The lungs face trouble every time dust, smoke, or vapors hang in the air. Painters, welders, or anyone ripping out old insulation should really use masks. Repeated breathing of fumes from harsh chemicals—paint thinners, strong adhesives, pesticides—can wreck lung tissue or trigger asthma over time. Headaches, dizziness, and nausea pop up in minutes if a space isn't aired out. It takes just 30 minutes of undetected carbon monoxide to cause confusion and permanent harm.
Solid, Smart First Aid Steps that Work
No fancy tricks needed. The main goal with most irritants or toxins: get them off, out, or away from the body. Remove any soiled clothes for sure, and rinse affected skin under cool running water for at least twenty minutes. For spills in the eyes, tilt your head and let clean, lukewarm water flow gently over the eye, blinking frequently, without rubbing.
Breathing issues? Get outside or into fresh air fast. If it's a strong smell that’s dropped you, stay low since some heavy gases linger near the floor. Anyone with trouble breathing, blue lips, or a raspy cough after chemical exposure needs medical help. In the rare case someone swallows something poisonous, never try to make them throw up. Call a poison control center or emergency line, and keep the product container handy for information.
Don’t put ointments, salves, or butter on chemical burns. That only traps the bad stuff. If pain sticks around, or if the skin changes color, blisters, or starts oozing liquid, see a doctor quickly. For larger burns or if someone seems dizzy, weak, or confused, don't hesitate—call for help.
Learning Matters More Than Luck
Accidents catch people off guard, but knowing a few good habits saves pain, long waits in clinics, and even lives. Always read product labels. Wear gloves, masks, and eye protection where it makes sense. Make it a rule to store cleaners and chemicals up high and out of kids’ reach. Ventilate your workspaces. Promote real training at home and work. Staying healthy often comes down to quick thinking and fast action, shaped by knowing what to do—long before you have to use it.
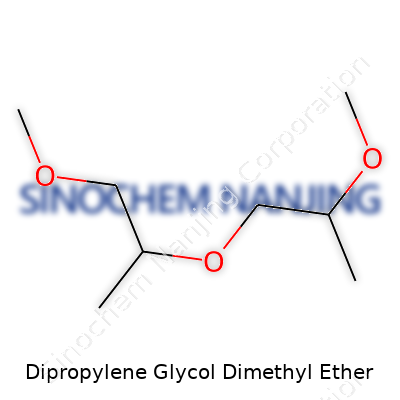
| Names | |
| Preferred IUPAC name | 1-methoxy-2-(2-methoxypropoxy)propane |
| Other names |
DPGDME 1-(2-Methoxy-1-methylethoxy)-2-methoxypropane 2,5,8,11-Tetraoxadodecane Dimethyl dipropylene glycol ether Propylene glycol dimethyl ether |
| Pronunciation | /daɪˈproʊ.piː.liːn ˈɡlaɪ.kɒl daɪˈmɛθ.əl ˈiː.θər/ |
| Identifiers | |
| CAS Number | 111109-77-4 |
| 3D model (JSmol) | `3D model (JSmol): COCC(OCCOC)OC` |
| Beilstein Reference | 1734441 |
| ChEBI | CHEBI:84887 |
| ChEMBL | CHEMBL1597845 |
| ChemSpider | 10721 |
| DrugBank | DB14197 |
| ECHA InfoCard | 100.131.749 |
| EC Number | 209-075-4 |
| Gmelin Reference | 126519 |
| KEGG | C18607 |
| MeSH | D005909 |
| PubChem CID | 81774 |
| RTECS number | JM9235000 |
| UNII | 1K6J9QN5SW |
| UN number | UN3271 |
| CompTox Dashboard (EPA) | DTXSID5020605 |
| Properties | |
| Chemical formula | C8H18O3 |
| Molar mass | 190.27 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Odorless |
| Density | 0.95 g/cm³ |
| Solubility in water | Soluble |
| log P | 0.17 |
| Vapor pressure | 0.37 mmHg (20 °C) |
| Acidity (pKa) | 19.2 |
| Basicity (pKb) | 5.03 |
| Magnetic susceptibility (χ) | -7.82×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.388 |
| Viscosity | 1.7 mPa·s (at 25 °C) |
| Dipole moment | 4.33 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 321.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -499.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4205.7 kJ/mol |
| Pharmacology | |
| ATC code | V07AB |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P280, P305+P351+P338, P403+P235 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 77°C |
| Autoignition temperature | 215 °C |
| Explosive limits | 1.3% - 10.1% |
| Lethal dose or concentration | LD50 (oral, rat): > 5,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 5,400 mg/kg (rat, oral) |
| REL (Recommended) | 0.2 mg/m³ |
| IDLH (Immediate danger) | IDLH: 400 ppm |
| Related compounds | |
| Related compounds |
Ethylene glycol dimethyl ether Diethylene glycol dimethyl ether Tripropylene glycol methyl ether |

