Diphenylamine: A Look Beyond the Science
Historical Development
Diphenylamine came out of late nineteenth-century organic chemistry efforts, springing to life at a time when dye manufacture was pulling all sorts of aromatic amines out of coal tar. Its history weaves through everything from early explosives to modern-day agriculture. Chemists first recognized it as a byproduct when searching for ways to stabilize nitrocellulose in munitions. It wasn't long before the same molecule found work stabilizing rubber, turning up in analytical chemistry, and later, protecting apples in cold storage. Interest in diphenylamine tracks bigger trends. During wars, explosive factories paid attention to its antioxidant abilities. Later, as food markets grew more commercial, storage chemists found new uses for its chemistry.
Product Overview
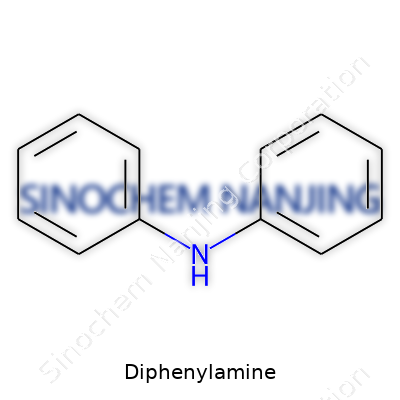
Diphenylamine shows up as a white to pale-yellow crystalline solid. Most people involved in handling it recognize a faint, aniline-like odor. The molecule carries two phenyl groups hung off a simple nitrogen. This structure sets it apart for its resilience against oxidation and its ability to bind with acid gases and free radicals. Chemists tend to value diphenylamine because it can work as both stabilizer and reagent. Formulators reach for it in finished goods ranging from lubricants to agricultural sprays, each time taking advantage of its chemical bite.
Physical & Chemical Properties
With a melting point resting around 54°C and a boiling point substantially higher, diphenylamine keeps its shape under room conditions. The powder doesn’t dissolve well in water, but slips easily into most organic solvents. Its low vapor pressure means inhalation risks take a back seat to skin exposure. The aromatic rings lend a degree of stubborn chemical stability, but the amine group opens doors to reactivity that suits tailoring for different jobs. In storage, the compound stays solid and dry unless exposed to strong acids or oxidizers.
Technical Specifications & Labeling
Quality matters where diphenylamine is concerned, especially when destined for food storage or pharmaceutical labs. Producers measure purity by gas chromatography, watching for trace amounts of aniline and biphenyl as impurities. Regulatory bodies require clear hazard labeling under GHS guidelines, flagging the molecule for its aquatic toxicity and possible chronic effects on health if used carelessly or in large doses. In trade, barrels move under UN codes that caution against spillage and dust formation.
Preparation Method
Engineers generally rely on the reaction of aniline with phenol under acid catalysis to synthesize diphenylamine. The process heats the mixture, causing a substitution where one phenol unit links up with the amine. Yields have steadily improved as heat transfer and catalyst recovery get better in modern plants. Some older processes relied on coupling chemistries with chlorobenzene, though residue handling became a bigger concern. Each method reflects a battle between cost, purity, and environmental safeguards.
Chemical Reactions & Modifications
Diphenylamine stands out for the way its amine group opens paths to further changes. Nitration produces powerful antioxidants, sulfonation leads to water-soluble dyes, and alkylation sets the stage for proprietary lubricants. In the lab, the compound can act as a reducing agent for peroxides. The aromatic ring resists most simple substitutions at room temperature, so chemists often turn to catalytic strategies when building more complex derivatives. Each tweak changes how the molecule interacts in its end market.
Synonyms & Product Names
Over decades of use, diphenylamine has picked up more than a few handles: DPA, N-phenylaniline, and N-benzylaniline all point to the same molecule. Industry buyers may see references to “anti-scald diphenylamine” in produce storage or “DPA” in explosive-grade stabilizers. The array of chemical synonyms reflects its trek through multiple markets, sometimes leading to confusion without careful labeling or supply chain checks.
Safety & Operational Standards
Handling diphenylamine takes respect for its hazards, even if the solid doesn’t leap out as dangerous at a glance. Exposure through skin or inhalation over time links to toxic effects in laboratory testing. Workers run gloves and goggles, with dust-control protocols in place to avoid accidental exposure. Emergency plans factor in the fire hazard if the molecule meets a strong oxidizer. Storage keeps the barrels dry, sealed, and separated from food contact areas at all times. Many countries enforce safety data sheets detailing medical response, disposal procedures, and required signage.
Application Area
Use cases for diphenylamine stretch wide. In the fruit business, post-harvest teams apply it to apples and pears as a spray to stave off scald, reducing waste and saving millions of pounds of fruit each year. Munitions plants include it in storage mixes to keep propellants from breaking down, preventing explosions before they can threaten workers or civilians. Rubber engineers rely on its antioxidant activity to stop their materials from crumbling under sunlight and ozone. Analytical chemists, tracing nitrate and nitrite concentrations in water or explosives, use it as a colorimetric indicator. Pharmaceutical researchers sometimes start with diphenylamine as a building block on the way to antihistamines and anticonvulsants. Its fingerprints show up across agricultural, technical, and research fields, each time bringing a blend of stability and reactive potential.
Research & Development
Horizon work on diphenylamine always orbits around two goals: unlocking more selective chemical changes for specialty products, and driving down toxicity or environmental leaks. Academic labs hunt for greener synthetic routes that ditch heavy metals and lower waste streams. Industry groups fund studies to replace older, less selective processes with more targeted functionalizations. In agronomy, researchers track the breakdown products after fruit treatments, making sure residues drop below safety benchmarks before hitting the store shelf. Patent offices keep busy as companies file claims for new DPA derivatives that solve old problems in rubber stability or pharmaceutical design.
Toxicity Research
Scientists flagged diphenylamine’s toxicity profile decades ago. Short-term exposure in animals at high doses triggered nausea, liver damage, and reproductive effects. Environmental health specialists monitor its runoff in orchard regions, checking for risks as the molecule degrades into possibly more toxic fragments in the soil and water. Regulatory bodies around the world publish strict limits on residue levels in food and discharge limits in wastewater. Occupational safety officers require annual refresher training for plant workers, reflecting how easy it is to become complacent with long-familiar chemicals. Ongoing toxicological studies help update those limits, shifting recommendations as science improves.
Future Prospects
The story of diphenylamine points to a disconnect between the utility of tried-and-true chemicals and the pressure to find greener replacements that minimize human and environmental risks. Demand for naturally derived antioxidants in foods and rubbers points away from DPA, though cost and performance keep it relevant. Farmers need alternatives that safely extend shelf life on fruit without lingering residues. Explosives engineers test safer stabilizers, but reliability often brings them back to diphenylamine’s record. R&D teams see a future where synthesis runs leaner, exposure risks drop, and end-users trust the safety data behind every application. Open scientific forums and regular regulatory updates stand as the best route to making sure history’s workhorse keeps pace with modern standards without leaving environmental stewardship behind.
What is Diphenylamine used for?
A Chemical With Many Uses
Milk, apples, rubber, explosives—diphenylamine connects to each in a way that seems straight out of a trivia contest. Scientists first started working with this compound in the late 1800s. Diphenylamine, or DPA if you’re in the know, pops up in farming, fruit storage, even in labs around the world. I remember flipping through an old chemistry catalog and seeing it listed with uses ranging from gunpowder stabilizers to apple preservatives. The variety feels endless, and that’s what draws attention: one chemical, so many jobs.
Keeping Apples Fresh—and Safe?
Americans buy more apples than any other fruit. Grocers want those apples to look perfect: no browning, no scalding on the skin. Diphenylamine helps with that. Growers spray apples with DPA to block a natural, but pretty ugly, process called “storage scald.” The fruit goes into cold storage for months, with DPA acting like a chemical shield between the fruit and the damaging effects of oxygen. Not all countries think it’s a safe bet. The European Union banned it in 2012 due to concerns about possible byproducts—nitrosamines formed from DPA might raise cancer risks. Here in the US, the FDA still allows it, and apple testing in recent years shows DPA levels within the current safety limits. As a regular apple eater, I know most folks don’t want to worry about every bite, but transparency matters. Clear labeling and regular independent testing would help shoppers choose what feels right for their own families.
Saving Explosives From Disaster
Old-timey black powder, modern propellants, even fireworks—energy stored in a tiny space brings danger. DPA shows up again here, but this time as a stabilizer. Nitrocellulose in gunpowder breaks down over time, releasing chemicals that turn the whole thing unstable. DPA mixes in as a safety net. Instead of the nitrocellulose eating itself up, DPA takes the hit, keeping ammo safer for longer. Hunters, police, and soldiers all count on their ammunition staying intact in storage. Everybody feels safer when explosions only happen on purpose. This stabilizing trick keeps aging stockpiles safe, but the process creates its own waste and needs tight control, since accidental contamination could mean trouble.
Rubber, Antioxidants, and Lab Work
Every driver feels the benefits of DPA, even if they don’t realize it. Tires spend their lives exposed to sunlight and oxygen, which break down rubber fast. DPA gets into the rubber during manufacturing, working as an antioxidant to slow that damage. It pops up again in some lubricants and industrial oils, fighting the slow creep of oxidation there too. Occasionally, environmentalists raise a flag about chemicals leaching into waterways, especially with higher production. Regular monitoring of runoff and stricter waste management at factories would help address these environmental worries.
Looking Ahead
Diphenylamine won’t vanish soon. It brings longer shelf lives to apples, safer ammunition, and longer-lasting tires. Like with any chemical, getting real facts about risks and enforcing tough standards matter more than ever. Over the years, I’ve seen science help us solve problems, but the best solutions always come with honest, public discussion and a willingness to reevaluate choices as more facts come in. Keeping that attitude leads to both innovation and safety—for our food, our roads, and our future.
Is Diphenylamine hazardous to health?
Background of Diphenylamine
Diphenylamine shows up in more places than most people realize. It gets used in everything from stabilizing explosives to preventing fruits like apples from turning brown during storage. Over the years, it’s become an industrial staple. Anytime a chemical pops up in agriculture, food storage, and manufacturing, questions about its safety come up—rightfully so.
Health Risks from Exposure
Those who handle diphenylamine directly at work face greater risk than the general public. Skin contact can bring on rashes, and inhaling dust or fumes can leave workers coughing or feeling short of breath. Intake in large quantities—though rare—may affect the liver or kidneys. The U.S. Environmental Protection Agency classifies it as a Group D chemical, meaning not enough evidence exists to say if it could cause cancer in humans, but some animal studies hint at possible long-term risks.
The use of this chemical on fruit drew attention from health agencies in both the United States and Europe. Apples, since they’re often coated with a diphenylamine-based preservative, sparked debates about chronic, low-dose exposure. Data from the European Food Safety Authority shows that residue levels in fruit typically fall below established safety limits, but the agency still chose to restrict its use further just to be careful.
Why Monitoring Matters
I grew up around fruit orchards, and any tool that keeps apples fresh longer means less waste and more food for local markets. That said, parents deserve confidence that their kids’ snacks aren’t carrying unintended risks. While most authorities say tiny amounts washed onto fruit don’t pose a major threat, people with higher exposure in factories or packing sheds face a different set of challenges. Regular monitoring, protective gear, and up-to-date training really make a difference for them. Stories of sore skin and lung troubles highlight the gap between what looks safe on paper and what actually happens when gloves or respirators aren’t used right or ignored.
Finding Better Solutions
Turning away from chemicals like diphenylamine isn’t easy overnight. Farmers and storage operators need dependable ways to keep produce from spoiling before it hits shelves. As research into natural preservatives and improved storage technology advances, growers will get more options. Transparency is key. Consumers want simple labels and straight answers—so tracking and publishing residue tests protects both shoppers and honest growers.
On a larger scale, international cooperation has shaped the current rules. Differences between U.S. and European rules sometimes leave exporters scrambling to follow different requirements for each market. Clear, science-based rules help everyone—less confusion for farmers, more trust in the food supply, and less opportunity for unsafe practices.
Practical Steps Forward
If workplace contact with diphenylamine can’t be avoided, regular medical checkups help catch issues before they get serious. For shoppers, washing fruit under running water and peeling when possible takes care of most chemical residues. Pushing for ongoing research and public data gives peace of mind. Experience shows that health and safety keep moving forward when people ask tough questions and demand better answers. Keeping a close eye on diphenylamine just means keeping those standards high.
What is the chemical formula of Diphenylamine?
Knowing More Than Just the Formula
Diphenylamine gets written in the books as C12H11N. That simplicity hides a deeper story. Spotting the arrangement—two benzene rings hugging a single nitrogen—brings to mind its connection to both the lab and the real world. Remembering endless hours in a university chemistry lab, I watched that same molecule move from powder to solution, catching its sharp, almost sweet scent. It feels like one of those building blocks that people outside a science class would never think twice about.
Why Formula Details Matter in Practice
The formula, C12H11N, holds more than an academic answer on a test. In industry, detail shapes everything from safety protocols to product development. Take agriculture—apples and pears in storage often rely on diphenylamine to keep them from browning too soon. That reliable nitrogen bridge does the job by scavenging free radicals. Food scientists and quality control teams have to get the specifics right because missteps could mean health risks or spoilage.
Pushing even deeper, nitrocellulose cartridge powder depends on diphenylamine as a stabilizer. Its chemical structure, resisting easy breakdown, makes it a loyal defender against unexpected reactions. Not just chemistry trivia, this detail means stability and life span for ammunition, which means safety for the end user. I've spent time with shooting sports enthusiasts who track shelf life, and every one knew that minor ingredient choices change everything.
Potential Risks Needing Smart Solutions
Anyone handling diphenylamine faces some real risks. Breathing in dust, skin contact, or leaking storage tanks bring exposure issues at work sites and factories. Agencies like OSHA keep a close watch on permissible exposure levels because, as research points out, accidental contact may cause headaches or skin reactions, and larger doses could prove much worse. In the early 2000s, I worked summer jobs in chemical storage—a reminder that safety culture grows from real awareness of each molecule’s quirks, not just labels on containers.
Waste management poses other headaches for companies. Reports from the EPA highlight cases where careless disposal of diphenylamine-tainted water affected groundwater in rural communities. Local governments stepped in, often late, leaving lasting tension between farmers, regulators, and neighborhoods. Avoiding such messes demands stronger rules and honest training, not shortcuts or trusting luck.
Smart Handling and Forward Thinking
Addressing these risks begins with straightforward education—training workers to recognize symptoms of overexposure or to use equipment suited for handling materials like diphenylamine. Auditing old storage sites, updating containment systems, and pushing for responsible disposal act as next-level solutions. Community outreach helps, too. In one rural Midwest town hit by illegal dumping, neighbor-led public meetings led to better emergency planning and stronger ties with first responders.
Bringing Chemistry Closer to Home
Looking past the whiteboard, diphenylamine’s chemical formula shapes the way people store food, build safer products, and protect each other from harm. Understanding the molecule shows the impact of knowing what’s inside the barrel, the bag, or the bottle. Learning about basic formulas doesn’t just help scientists; it arms neighbors, families, and workers with the power to make better choices, every single day.
How should Diphenylamine be stored?
What Makes Diphenylamine Different
Diphenylamine plays a big role in both laboratories and industries thanks to its use in dyes, pesticides, explosives, and antioxidants. This compound offers value in many applications, but misuse or poor storage brings risks to health and the environment. I’ve worked in spaces where chemical safety almost felt like an afterthought. Thankfully, experience—and a bit of research—can teach anyone what it takes to treat chemicals like this with the seriousness they deserve.
Safety Starts with Knowledge
Storing any chemical isn’t about ticking boxes; it comes down to protecting people and places from preventable accidents. Diphenylamine poses dangers worth understanding. The compound can catch fire, release harmful fumes, and even irritate skin or lungs after exposure. Its solid form might seem harmless at first glance, but all it takes is one mistake to change that.
Common-Sense Storage Starts with the Right Container
My own work with chemicals showed me the headaches that come from using the wrong containers. Plastic might melt under stress, or a lid could warp after a few weeks. For diphenylamine, I stick with glass or sturdy metal containers. No leaky plastic, no unnecessary surprises. Each container gets a clear label—not just a scribble or shorthand, but full identification and hazard warnings that anyone, even visitors, can understand at a glance.
Control Temperature, Avoid Trouble
Heat and chemicals rarely mix well. I’ve watched more than one workshop suffer from storing sensitive materials near radiators or sunlight. For diphenylamine, room temperature works best, away from direct sun and all heat sources. Too much warmth and the risk of decomposition goes up, which could send toxic fumes into the air or create flammable vapors. Cool, dry shelves work better than crowded cabinets or trucks left in the sun.
Keep Chemicals Apart
No matter where I’ve worked—school labs or industrial plants—I came to respect the danger of storing incompatible chemicals together. Diphenylamine can spark trouble if it touches strong acids, oxidizers, or nitrites. A good system separates materials by type, with clear signage and space between each group. Simple, reliable, and safer for everyone involved.
Prevent Moisture at All Costs
Humidity and chemicals rarely end well together. Dampness may not seem like a big deal, but it can cause clumping, spoilage, or even increased hazards. I’ve seen colleagues use silica gel packets or tight-sealing lids on all diphenylamine containers. It’s an extra step that pays off over months—less mess, fewer accidents, and longer shelf life.
Emergency Planning Isn’t Optional
An overlooked step in many storage routines involves planning for a spill or accidental exposure. It’s never fun to imagine the worst, but running drills and keeping cleanup supplies handy made all the difference where I worked. Fire extinguishers suitable for chemical fires, gloves, goggles, and proper ventilation turn a potential crisis into something manageable.
Learn from Experience, Not Just the Label
Years in the field show me what the textbooks don’t mention: people make mistakes, equipment fails, and shortcuts tempt us. Careful storage of diphenylamine—for the sake of health, safety, and the law—matters every single day. Routine checks, clear training, and shared responsibility put everyone in a safer spot.
What are the safety precautions for handling Diphenylamine?
Why Safety Matters with Diphenylamine
Diphenylamine pops up in industries like agriculture, rubber, and dyes. I’ve seen folks brush it off as just another white powder, but my time around chemical storage rooms taught me that looks can be deceiving. Inhaling or touching this stuff brings a risk of skin irritation, breathing trouble, or headaches. Long exposure may affect your liver or cause longer-term health problems. Nobody wants a day at work ending with a medical incident, so paying attention to safety rules matters.
Personal Protective Gear: The Basics
I learned early to never work with chemicals empty-handed. Wear solid gloves, preferably nitrile rather than latex. Permeation happens faster than most people guess—hot weather and sweat don’t help. Don a long-sleeved lab coat and a set of goggles to protect your eyes. If there’s any chance of dust or fumes, I grab a respirator mask that's rated for organic vapors.
Ventilation Isn’t Optional
Cracked windows and fans do only so much. Proper fume hoods or vented workspaces mean the difference between a stingy throat and safe breathing. In one shop I worked in, we skipped the hood for quick jobs; all it got us was complaints about coughs and a sharp chemical stench. Now, any handling or mixing of diphenylamine means checking that the air system runs smooth and the hood pulls airflow the right way.
Storage: Don’t Get Casual
Leaving bags or jars open makes accidents pretty likely and shortens the shelf life. Store diphenylamine in a tightly sealed, clearly labeled container. Keep it somewhere cool and dry, away from acids and oxidizers—they really don’t mix. I’ve seen what happens when these things combine: smoke, heat, and a mad dash for the fire extinguisher. Stacking chemicals in a cluttered cabinet risks spills and confusion, so give each substance a proper, organized home.
Clean-Up and Spill Response
Spills can sneak up quickly, especially if you’re measuring in a rush. A minor scatter calls for a disposable scoop, paper towels, and a small amount of water to damp down dust, followed by a sealed bag for cleanup waste. Larger spills mean roping off the area, alerting co-workers, donning a dust mask, and bringing out the spill kit—never your bare hands or a vacuum cleaner.
Disposal Has Rules
Dumping diphenylamine down the drain or tossing it with everyday trash brings legal trouble and harms local water supplies. I always check the local environmental guidelines. In several labs, disposal meant collecting the chemical waste in containers labeled for hazardous pick-up. Your workplace safety officer or local authority usually has guidelines and supports safe methods. It’s not about red tape—it’s about protecting your neighbors and yourself from unnecessary risks.
Education and Training: Keeping It Fresh
Procedures change over time as we learn more about chemical risks. Regular safety briefings or online courses, even if they sound dull, prevent old habits from sticking around. Reading safety data sheets makes a difference too. I scan them before using any unfamiliar product. Sharing these basics with newcomers helps create a culture that keeps mistakes from repeating themselves.
Respecting diphenylamine’s hazards means everyone gets home healthy, and the job site avoids preventable setbacks. It’s common sense and good practice, drawn from the everyday reality of working where chemicals play a part.
| Names | |
| Preferred IUPAC name | N-phenylaniline |
| Other names |
N-phenylbenzenamine DPA Anilinobenzene N-benzenylaniline Aniline, diphenyl- N-Phenylbenzeneamine |
| Pronunciation | /daɪˌfiː.nɪlˈæm.iːn/ |
| Identifiers | |
| CAS Number | 122-39-4 |
| Beilstein Reference | 1202453 |
| ChEBI | CHEBI:16219 |
| ChEMBL | CHEMBL1401 |
| ChemSpider | 855 |
| DrugBank | DB02624 |
| ECHA InfoCard | 100.003.710 |
| EC Number | 204-539-4 |
| Gmelin Reference | 136072 |
| KEGG | C06587 |
| MeSH | D003822 |
| PubChem CID | 8310 |
| RTECS number | KN7520000 |
| UNII | 9FQ9IRL8F4 |
| UN number | UN1320 |
| Properties | |
| Chemical formula | C12H11N |
| Molar mass | 169.23 g/mol |
| Appearance | White to faintly colored crystalline powder |
| Odor | faint aromatic odor |
| Density | 1.16 g/cm3 |
| Solubility in water | 0.28 g/L (20 °C) |
| log P | 3.5 |
| Vapor pressure | 0.001 mmHg (25°C) |
| Acidity (pKa) | pKa = 0.79 |
| Basicity (pKb) | pKb = 9.17 |
| Magnetic susceptibility (χ) | -70.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.684 |
| Viscosity | 1.97 cP (20°C) |
| Dipole moment | 2.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 43.5 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -6350 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H312, H332, H373, H410 |
| Precautionary statements | P210, P261, P264, P270, P271, P272, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P330, P363, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-2-🐢 |
| Flash point | 129°C |
| Autoignition temperature | \["430°C (806°F)"\] |
| Explosive limits | Explosive limits: 0.7–2.8% |
| Lethal dose or concentration | LD50 oral rat 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2000 mg/kg (oral, rat) |
| NIOSH | JN6475000 |
| PEL (Permissible) | 10 mg/m3 |
| REL (Recommended) | 1 mg/L |
| IDLH (Immediate danger) | 50 mg/m3 |
| Related compounds | |
| Related compounds |
N-Phenylaniline Triphenylamine Phenylenediamine Aniline Naphthylamine |

