Looking at Dimethylsulfoxide: More Than a Laboratory Solvent
Historical Development
Talking about dimethylsulfoxide, or DMSO as most call it, means uncovering decades of scientific questions and very real stories from both medical research and industrial labs. Back in the 1950s, chemists in Russia and New Zealand started testing DMSO for its unique ability to dissolve just about anything. Researchers stumbled upon it as a byproduct from wood pulp, long before it landed on pharmacy shelves or industrial catalogs. Over time, its role grew from a chemical curiosity to a hot topic for those studying cell biology, inflammation, and even pain relief. Once the first big paper highlighted its remarkable ability to transport small molecules through skin, things took off. Scientists kept pushing, wanting to see how this clear, faintly garlic-scented liquid could help with everything from cryopreservation to arthritis creams.
Product Overview
When opening any jug of DMSO, there’s a distinct odor, something like garlic or oysters. That’s a sign of how easily it penetrates membranes—skin, plastics, and glassware alike. Folks use DMSO both as a solvent and for more experimental work. People have tried it in fieldwork, cleaning up medical devices or preparing samples, and in the clinic, rubbing it on joints and skin. Interest peaks any time researchers discover a new area where transporting molecules deep into tissues matters, or where other solvents fail. What shows up in the bottle is a liquid that attracts water out of the air, and if it freezes, it does so at room temperature. In my experience, you can't ignore those little quirks. Run a study in the wrong conditions, and the DMSO could turn solid or introduce moisture into other delicate reactions.
Physical & Chemical Properties
Anyone who has measured or handled DMSO notices right away how different it feels. At room temperature, it's a clear, nearly odorless liquid that chills quickly and freezes just above 18°C. That means a cool lab bench, or poor climate control, can leave you with a clump of solidified DMSO instead of the free-flowing liquid you expected. Its boiling point sits just below 190°C, giving it a wide range for use and distillation. It's miscible in water, ethanol, acetone, and a bunch of other polar solvents. Thanks to its polar nature, it dissolves an impressive variety of chemicals, from salts to some polymers. Its strong dipole moment and hydrogen bond accepting abilities make reactions run smoother, especially when working with tricky solutes or biological samples that need gentle handling.
Technical Specifications & Labeling
You look at commercial DMSO, and purity jumps out as crucial. Most suppliers offer it at 99.9% purity, which matters when you push for biological research or pharmaceutical work. Lab staff train to check for water content, since DMSO loves pulling in moisture. Labels usually emphasize both purity and percentage water contamination. Storage recommendations matter since DMSO corrodes some plastics and deteriorates adhesives. Anyone using DMSO on the bench keeps it away from containers made of polystyrene or with low-grade plastics, learning the hard way that leaks or breaks can ruin samples or equipment. Regulations require clear hazard labeling because DMSO drives whatever it dissolves directly into the skin. Standardization helps, with international guidelines marking DMSO for research, pharmaceutical, food, or industrial use.
Preparation Method
Industrial synthesis grew out of necessity, using a two-step process from dimethyl sulfide oxidation. The basic pathway involves recovering dimethyl sulfide from wood pulp waste or petroleum, then oxidizing it under controlled pressure and temperature—usually with air or oxygen plus a catalyst. The result is a high-yield, two-step process that produces less environmental waste than older methods from the mid-twentieth century. Experience in large-scale synthesis teaches that maintaining low temperatures and strict oxygen control delivers higher purity, while sloppy setups contaminate the final product with sulfur-containing byproducts. Most refinements aim to minimize smell and remove trace impurities, which makes the DMSO friendlier for medical or pharmaceutical work.
Chemical Reactions & Modifications
Chemists harness DMSO not just as a solvent but as a reagent. It acts as both an oxidant and a reducing agent when pushed under the right conditions. One standout example is the Swern oxidation, famous in organic synthesis for transforming alcohols to aldehydes under mild conditions. It also serves in other powerful reactions, like the Pfitzner-Moffatt and Kornblum oxidations. On its own, DMSO reacts slowly but gets into the mix with halides, acids, or bases, then delivers transformations without harsh reagents or extreme heat. In the lab, it blends with so many reactants that DMSO's presence can drive pathways you don’t expect. Environmental research keeps a close eye on how DMSO breaks down in soil or water, since it degrades into dimethyl sulfide, a more volatile compound that can contribute to odor or atmospheric sulfur cycling.
Synonyms & Product Names
People use several names for DMSO. Beyond its chemical label—dimethylsulfoxide —there's a slew of trade names, especially in pharmaceutical or veterinary products. In research, “DMSO” has stuck the longest, though “methyl sulfoxide” and “methylsulfinylmethane” still pop up in decades-old papers or patents. On the open market, topical applications for horses and pets sold as “DMSO gel” often list it plainly. In medical literature, the International Nonproprietary Name keeps things tidy, ensuring that worldwide, this molecule speaks one identity, with only minor language tweaks from country to country.
Safety & Operational Standards
Practical lab work with DMSO taught me that the warnings on the bottle aren’t just for show. Get a splash on your skin, and you sometimes taste garlic almost instantly—a surefire sign this stuff goes straight through. DMSO is considered of low acute oral toxicity in small quantities, earning it a reputation as a “safe” solvent, but complacency has caused plenty of mishaps. It ferries other chemicals across the dermal barrier without filtering, so trace solvents, pesticides, or anything dissolved ride along into the bloodstream. That’s why glove choice and countertop cleanliness set the tone for good lab practice. Regulatory agencies flag it for these concerns, with the US FDA and other global authorities setting tight guidelines for pharmaceutical-grade product, emphasizing not just operator safety but downstream risk to patients.
Application Area
DMSO's reach stretches into diverse markets and industries. Drug formulation researchers keep a bottle on hand for dissolving stubborn actives or enhancing skin absorption. Physicians and veterinarians once prescribed it off-label for joint pain and inflammation, drawing both praise and controversy. Tissue bankers and fertility clinics use DMSO as a gold standard for cryopreserving cells, blood products, or embryos, sparing delicate proteins from freezing damage. It shows up in antifungal creams and bladder irrigation solutions, and still features in industrial cleaning and paint stripping—though for industrial-grade, purity and trace contaminant thresholds relax. In material science, its unique solvent power unlocks new plastics and composites.
Research & Development
DMSO keeps inspiring new research. Scientists keep probing its role in drug delivery, nanotechnology, and pain management. Pharmaceutical firms refine its use, testing lower impurity thresholds, or pairing it with new actives for transdermal patches. Ongoing cell biology research leans heavily on its usefulness in dissolving compounds that just won’t go into water or ethanol. In stem cell transplantation, DMSO has helped set the benchmark for cell viability after thawing, supporting regenerative therapy and cancer trials. On the toxicology front, research teams continue digging into long-term effects, focusing on bioaccumulation, metabolic pathways, and how DMSO interacts with emerging pharmaceuticals. This ongoing work emphasizes precision in formulation and stricter purification strategies. At the same time, basic chemical research still mines DMSO for new oxidative transformations and material synthesis.
Toxicity Research
Toxicologists have dug deep into DMSO's risk profile for decades. While animal and early human data paint it as relatively benign at low doses, chronic or high-exposure situations reveal side effects. Skin contact sometimes leaves a temporary rash, itching, or redness. Greater concern centers on its carrier ability—transporting harmful substances deeper than expected. Some reports from animal testing suggest reproductive impacts or mild liver enzyme shifts at high doses, but these rarely reach clinical significance in standard use. Regulators and researchers continue specifying safe exposure levels, especially for populations with compromised skin or kidney function. At the same time, better lab controls, improved PPE, and more precise labeling help keep researchers and patients safer.
Future Prospects
Looking ahead, DMSO sits close to the center of innovation for any work needing efficient delivery through skin or cell membranes. Drug developers anticipate new topical formulations, smart delivery systems, and safer analogs. Research groups are examining modified versions of DMSO for better selectivity—aiming to keep the transport benefits while reducing unwanted absorption. Environmentalists eye purification breakthroughs to minimize DMSO's ecological footprint, especially in large-scale use cycles. I see materials scientists targeting solvency power for emerging polymers and as a template for custom solvents in green chemistry. As new medical applications emerge, regulatory agencies will keep turning the dial toward higher purity, tighter safety protocols, and measurable clinical benefit. In all these, DMSO’s odd mix of versatility and risk stands as both a challenge and promise, shaping the questions that future generations of scientists will chase.
What are the main uses of Dimethylsulfoxide (DMSO)?
What is DMSO?
Dimethylsulfoxide, or DMSO, started out as a byproduct of making paper. Chemists quickly discovered it does more than just sit in a factory drum. It caught the eye of researchers because of its ability to dissolve a staggering range of substances. You’d be surprised by how often DMSO pops up in both labs and clinics, even if you’ve never heard of it before.
DMSO in Medicine: Beyond the Hype
Doctors and pharmacists find DMSO useful because it passes through skin and tissue. In my early days working with biomedical engineers, I noticed DMSO always sitting on the chemical shelf, ready for action. In hospitals, the big-ticket use comes from its FDA approval for treating interstitial cystitis, a painful bladder condition. DMSO gets infused directly into the bladder and reduces inflammation and discomfort.
Outside of this approved use, you see DMSO pop up as a topical cream or gel for aches, arthritis, and even some injuries. Plenty of athletes and elderly folks have turned to these gels hoping for relief. We should pay attention, though, to the fine print: scientific studies still question how much good it really does for pain. The body absorbs DMSO quickly, and with absorption comes a garlic-like breath odor that you can’t ignore if you’ve spent time in clinical settings.
Role in Science and Research
In laboratories, DMSO keeps science moving forward. It’s a go-to solvent for dissolving both water-soluble and fat-loving compounds. If you’ve ever tried to mix a stubborn molecule into a test tube, DMSO makes the job possible. During my grad school stint, every freezer stored DMSO—for freezing biological cells. Without it, researchers risk losing their cell lines or stem cell samples. Add some DMSO before cooling, and those delicate cells are more likely to survive deep freeze.
Biologists rely on DMSO for delivering drugs in animal studies and preparing tricky compounds for analysis. Chemists respect its power for supporting reactions that wouldn’t happen otherwise. So, if a medicine or new chemical makes it to your doctor or grocer, chances are DMSO played a role somewhere along the research chain.
Handling and Safety Concerns
DMSO dissolves lots of substances. That’s exactly why folks need to keep a close eye on what’s getting absorbed along with it. Anything mixed with DMSO can ride into the bloodstream. I learned quickly not to handle it with bare hands—it’s not just about the distinctive odor, but about DMSO grabbing whatever’s on your skin and shuttling it straight in.
For regular consumers, DMSO remains a grey area. No over-the-counter products except those targeting specific medical purposes have strong safety oversight, and labeling can leave much to be desired. It’s smart to check with your doctor, especially if you’re considering DMSO for home treatment. Some skin irritation and allergic reactions do happen, so those with sensitivities should act carefully.
Moving Forward Responsibly
Science keeps unlocking new roles for DMSO, especially in drug delivery and regenerative medicine. With such a versatile tool, open discussions between researchers, regulators, and the public matter. Clear rules for handling, labeling, and medical claims would protect users and help DMSO realize its full medical and scientific potential.
Is Dimethylsulfoxide safe for human use?
Understanding What People Want from DMSO
Dimethylsulfoxide, often called DMSO, pops up in conversations among sports enthusiasts, arthritis sufferers, and a handful of folks chasing alternative medicine. This clear liquid, first made as a chemical solvent, moved beyond labs after scientists saw it might help with inflammation and pain relief. Some users swear by it. Plenty of claims surround DMSO, especially for easing joint pain or helping with minor injuries. Still, when I talk with people who've tried it, genuine curiosity always mixes with a dose of caution. The big question isn't about popularity—it's about safety.
What the Science and Doctors Say
DMSO helps some patients with interstitial cystitis—a painful bladder condition—because the FDA approved a specific form for that use. Outside of this, medical researchers keep circling back to similar points: no magic cure, no blanket green light. DMSO slips through the skin easily, which gives it potential as a transporter for medicines. Dermatologists and neurologists have looked at it for icy hot patches and nerve pain. That doesn't make it a free-for-all.
Researchers reported that skin might itch, smell garlic-like, or get irritated when exposed to DMSO. High doses sometimes bring headaches, dizziness, or nausea. A few animal studies gave more reasons to pause—eye changes at high concentrations made scientists step back, even if the research used much larger doses than regular topical gels.
What I’ve Learned from Real-World Use
Over the years, walking into co-ops and talking to folks at running clubs made clear that DMSO is no stranger to home experiments. I’ve seen horse trainers use it for their animals, and heard plenty of older adults say it helps with their knees. Not all stories end well: some people reported skin burning, while others got odd breath smells that lingered for days. One runner I met applied too much to her ankle—she got blisters that took weeks to heal.
What people don’t always see: purity can vary wildly in bottles that aren’t intended for medical use. That means buying a bottle online or from a feed store carries risk. Impurities or other solvents mixed in will end up on your skin or in your bloodstream. Several friends who double-checked the label still couldn't guarantee they were getting something safe.
Why a Doctor Matters Here
Physicians and pharmacists stay skeptical for good reason. Most professionals won’t recommend DMSO for home use unless approved for a specific condition. Pain and inflammation have many causes, and not every solution fits every person. FDA-approved drugs for joint pain, nerve issues, or arthritis go through rigorous tests for side effects and interactions. DMSO hasn’t passed most of those for general use. Mixing it with other medications, especially on the skin, could bring on toxic reactions—something doctors worry about more as people search for easier solutions.
Looking Ahead: Care, Research, and Wise Choices
DMSO holds real scientific curiosity. Some researchers keep studying its effects on inflammation, drug delivery, and nerve health. For now, those answers stay limited. Anyone considering DMSO should talk to a healthcare professional. Medical supervision trumps internet advice. In the end, safer solutions and approved treatments offer protection from risks that aren’t always obvious. If new uses for DMSO get the right research and real-world backing, maybe the conversation will shift. Until then, treating aches, pains, or chronic illness calls for tested, regulated options.
What are the possible side effects of using Dimethylsulfoxide?
Understanding the Drawbacks
Dimethylsulfoxide, often called DMSO, has built a reputation for pain relief, anti-inflammatory properties, and even experimental use in veterinary medicine. Scientists have paid attention to its power as a solvent, moving substances through skin quickly. Plenty of folks use it expecting quick results and may overlook real risks. Anyone who’s talked with people struggling with chronic pain will hear about these home treatments sooner or later.
I've watched friends reach for DMSO after workout injuries or arthritis flares, trusting online forums more than medical guidance. That choice can carry real consequences, and it’s easy to underestimate what’s at stake. DMSO has some documented side effects that simply can’t be ignored.
Physical Reactions You Can’t Brush Off
The medical world has tracked a list of unwanted reactions. For starters, many report a garlic-like taste or odor after DMSO touches their skin. That happens because it breaks down in the body and releases sulfur. While this may sound minor, the strong smell can make social life tricky. Physical discomfort often crops up—skin redness, itching, and burning top the list. At higher concentrations, DMSO burns skin and can lead to blistering.
Eye irritation matters too. If someone uses DMSO near their face, stinging or watery eyes is common. Eye complications get overlooked because many stories on the internet focus on DMSO’s supposed healing. The FDA cautions against casual use for exactly this mix of irritation and unknowns.
Dangers Below the Surface
Some effects get serious fast. People with kidney or liver issues face higher risk, since the body clears DMSO through these pathways. Abdominal cramps, nausea, and vomiting turn up in people who use high doses or take it by mouth. DMSO moves other drugs through skin, so unintended interactions enter the equation, sometimes making harmless creams toxic.
In hospitals, doctors sometimes use DMSO in bladder treatments or during chemotherapy. Even there, they see side effects like dizziness, headaches, allergic reactions, and blood pressure fluctuations. Reports from clinical trials help highlight the importance of informed dosing.
Supporting Evidence and Expert Guidance
The FDA approves DMSO for very narrow medical purposes, including treating interstitial cystitis. Self-experimentation draws real warnings from toxicologists and pharmacists. A 2019 review in the Journal of Clinical Pharmacy and Therapeutics pointed out a lack of evidence for safe home use—also noting that contamination or improper dilution brings even bigger risk.
Stories circulate about people finding relief with DMSO, but placebo effect and wishful thinking play a big role. Just because a friend used it after a pulled muscle doesn’t mean everyone gets out unscathed.
Healthy Alternatives and Safer Steps Forward
If you’re tempted by what you hear about DMSO, tap into medical guidance before you start. Pharmacists and doctors help weigh risks and look for safer pain relief. Over-the-counter creams and prescription meds come approved after rigorous testing. Physical therapy or supervised exercise often brings benefits without the downfalls.
People deserve options when managing pain and injury. The trouble with DMSO isn’t just about short-lived side effects. Once people use it without understanding the risks, they set themselves up for complications that good information could prevent. The best advice—ask questions, read trusted sources, and don’t take shortcuts where health is concerned.
How should Dimethylsulfoxide be stored and handled?
The Basics Everyone Overlooks
Dimethylsulfoxide, or DMSO, crops up in chemical labs, manufacturing, and even some medical settings. People sometimes treat it like just another bottle on the back shelf. It’s clear, it doesn’t reek, it doesn’t fume. But a seeming lack of drama hides real hazards. What’s gone wrong for others has taught me to take DMSO seriously for one simple reason—your body loves to absorb it, and it’ll drag other stuff in with it through your skin.
How I Learned the Hard Way
Years ago, I picked up a beaker with a few drops of DMSO using bare hands, thinking nothing of it. Within minutes, my mouth tasted like garlic. No one mentioned that trick in school, but ask anyone who’s actually handled DMSO. It seeps straight through, taking whatever’s on your hands along for the ride. That’s why skin contact isn’t a minor mistake, it’s a shortcut for chemicals to get where they don’t belong. Gloves—nitrile, not latex—aren’t optional. Even a pinhole in a glove is a ticket to trouble. Eye protection isn’t up for debate, either. Splashes might not burn, but DMSO acts like a highway for other substance leftovers.
Safe Storage Isn’t Optional
DMSO acts friendly in most containers, but only certain plastics stand up over time. Polyethylene or glass gets my vote. I learned early on not to trust old PVC or cheap plastic containers—sooner or later, they crack or warp. Clean, clearly labeled bottles stop coworkers from guessing at mystery liquids. DMSO may not burn with a flash, but once it hits high temperatures or open flames, the breakdown can get ugly—more toxic than people imagine.
Room Temperature Works, But That’s Not Enough
This stuff turns solid around 18°C (about 64°F). If your storage spot gets cold, you’ll find a cloudy sludge or a block of crystal that’s tough to measure. Keeping DMSO in a steady, dry spot takes guesswork out of the equation. Moisture exposure isn’t good, since even a small amount of water changes its properties and leads to accidental skin contact or dosing errors if it’s used in experiment mixes or formulations later.
Don’t Skip on Training
I’ve watched people new to labs wave off the idea of DMSO as anything special. Sadly, even veterans take “safe enough” shortcuts if the day’s been long and the bottle looks benign. Fact is, only good habits and solid training prevent problems. Safety data sheets might feel like red tape, but I’ve learned the hard way it’s better to know what to do in a spill than to google it after you’ve been exposed. Clear written protocols, routine reminders, and unambiguous hazard labeling pay off every single time.
Practical Steps Everyone Should Follow
- Keep DMSO containers out of direct sunlight and away from heat sources.
- Check your gloves for cracks or wear before pouring or pipetting.
- Use dedicated glassware and label all bottles with both name and hazard warnings.
- Store apart from strong oxidizers, acids, or substances that could react unpredictably.
- Clean up drips or residue quickly and dispose of wipes or rags using proper chemical waste procedures.
- If DMSO gets on skin, wash immediately with plenty of water—don’t wait to see what happens.
It’s not about being overly cautious. The stakes get higher with a chemical that doesn’t play by the same rules as water or alcohol. DMSO deserves a spot in the “handle with care” club, every time.
Can Dimethylsulfoxide interact with other medications or substances?
A Closer Look at Dimethylsulfoxide
Dimethylsulfoxide, better known as DMSO, pops up in medical settings, alternative therapies, and even veterinary medicine. What catches many off guard is how this chemical not only does its own thing but also opens doors—sometimes literally—through the skin or cell membranes. This ability to act as a carrier for other substances creates unique risks alongside potential benefits. Over the years, clinicians and patients alike have shared stories of good results and concerning side effects, making it vital to pay attention to more than the headline uses.
Mixing DMSO with Other Drugs—The Potential for Surprises
Combining DMSO with other medications can lead to unpredictable outcomes. DMSO changes the way drugs move through the body. It speeds up absorption when applied with other medications, pushing chemicals into deeper tissues. Someone using a topical steroid for joint pain and adding DMSO to the mix could suddenly absorb much more of the steroid than planned. That can raise side effect risks, not just in skin but across the whole body. There’s also the matter of DMSO changing how the liver breaks down certain drugs, which can mess with levels of blood thinners, sedatives, or heart medicines.
An example comes from the world of pain relief. Topical DMSO can help with swelling, but rubbing it on with lidocaine or aspirin cream might cause more of those substances to enter the bloodstream. This ramped-up delivery can make minor treatments act like strong doses. Some have ended up with unexpected drowsiness or even dangerous drops in blood pressure.
Interactions Beyond Prescription Drugs
DMSO fans often explore its use with supplements or herbal remedies. More than once, people have noticed faster effects from products like arnica or eucalyptus oil when combined with DMSO. The problem: herbal compounds aren't as well studied as prescription drugs, and neither are their interactions with DMSO. High absorption of unknown ingredients can lead to allergic reactions or toxic effects nobody saw coming.
DMSO also plays with alcohol and nicotine. People report stronger hits from patches or skin products when DMSO gets involved. That’s not just a quick buzz; it brings higher risks of nausea, dizziness, or even overdose symptoms, especially in folks with liver or kidney problems.
Why Full Disclosure with Your Doctor Helps
Many folks try over-the-counter products on a whim, not realizing that DMSO needs respect and careful handling. Talking with a doctor or pharmacist before adding DMSO to a routine makes a difference. These health professionals may flag possible trouble, especially for anyone managing several health problems or taking more than one medicine daily. Bringing up even small supplements, herbal teas, or skin products may save a trip to the ER.
For those who choose to use DMSO, keeping a detailed list of all current medications and alternative remedies supports a safer experience. Label reading and asking about interactions take a little time but prevent big mishaps. It’s old-fashioned advice, but it stands up in today’s world of easy online shopping and self-driven health care.
The Bottom Line
DMSO is not just another bottle on the pharmacy shelf. Its effects on drug delivery offer real promise but bring real risks, too. Trust and clear conversations between people and trusted health professionals remain the best allies for safety. Awareness, not worry, helps keep DMSO as an option—one that fits into full and informed health choices.
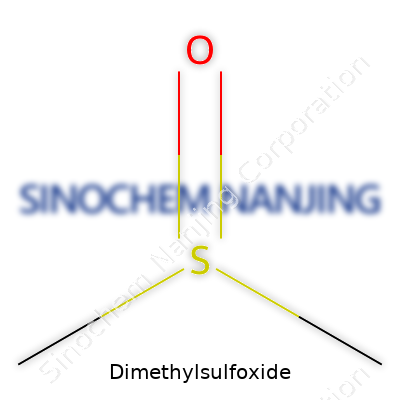
| Names | |
| Preferred IUPAC name | Methanesulfinylmethane |
| Other names |
DMSO Methyl sulfoxide Methylsulfinylmethane Dimethyl sulphoxide Sulfinylbismethane |
| Pronunciation | /daɪˌmiːθɪl.sʌlˈfɒk.saɪd/ |
| Identifiers | |
| CAS Number | 67-68-5 |
| 3D model (JSmol) | `3Dmol.js?cid=679` |
| Beilstein Reference | 0722462 |
| ChEBI | CHEBI:28262 |
| ChEMBL | CHEMBL135 |
| ChemSpider | 821 |
| DrugBank | DB01093 |
| ECHA InfoCard | 100.029.249 |
| EC Number | 3.6.4.12 |
| Gmelin Reference | 784 |
| KEGG | C02325 |
| MeSH | D005947 |
| PubChem CID | 679 |
| RTECS number | PV6210000 |
| UNII | YK6R7FD02L |
| UN number | UN1161 |
| CompTox Dashboard (EPA) | DTXSID3022662 |
| Properties | |
| Chemical formula | C2H6OS |
| Molar mass | 78.13 g/mol |
| Appearance | Colorless, clear liquid |
| Odor | Faint, garlic-like |
| Density | 1.1 g/cm³ |
| Solubility in water | Miscible |
| log P | -1.3 |
| Vapor pressure | 0.55 mmHg (at 20 °C) |
| Acidity (pKa) | 35.1 |
| Basicity (pKb) | basic |
| Magnetic susceptibility (χ) | −9.96×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.479 |
| Viscosity | 1.99 mPa·s (20 °C) |
| Dipole moment | 4.06 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 163.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -93.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1355.4 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | M02AX03 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319, H336 |
| Precautionary statements | P210, P233, P280, P303+P361+P353, P305+P351+P338, P337+P313, P370+P378 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 89°C |
| Autoignition temperature | 300 °C (572 °F; 573 K) |
| Explosive limits | 3.0–42% |
| Lethal dose or concentration | LD50 Oral rat: 14,500 mg/kg |
| LD50 (median dose) | 7.9 g/kg |
| NIOSH | NIOSH: PV6210000 |
| PEL (Permissible) | Not established |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
Dimethyl sulfide Dimethyl sulfone Methanethiol Sulfoxide Tetramethylene sulfoxide |

