Dimethylformamide: A Practical Look at Its Journey, Uses, and Consequences
Historical Development
Chemistry has shifted the world’s gears time after time, but few substances capture the story of innovation and risk like dimethylformamide. Developed during the growth of the chemical industry in the mid-20th century, DMF gained traction when folks realized how well it could dissolve a stubborn mix of solids. Scientists looking for better plastic manufacturing tools seemed to land on DMF just as the demand for synthetic fibers took off. Its utility boomed in the postwar decades alongside polymers, dyes, and pharmaceuticals—all booming fields that needed reliable, manageable solvents. Over the decades, DMF found its way into labs, factories, and commercial processes, despite steady concerns about workplace safety and environmental harm. Stories about its use in both breakthroughs and worker illnesses underscore that its rise isn’t a clean, uncomplicated victory for industry.
Product Overview
DMF, in practical terms, is a handy go-to solvent. I think of it like the kind of adjustable wrench every mechanical shop keeps on hand—rarely glamorous, but essential for daily challenges. Every chemist learns about it in university labs, because its mix of effectiveness and relatively easy handling puts it above messier, more dangerous options. Scientists rely on it where nothing else will do, particularly when they want to work with tricky polymers or separate complex blends. The ease with which DMF mixes with water and organic chemicals gives it a role in both experiments and production lines. In my own time working with chemical formulations, DMF stood out—nothing matched its ability to unlock a stubborn reaction or separate tangled mixtures.
Physical & Chemical Properties
DMF flows as a colorless liquid with a faint smell that hints at chemicals but doesn’t overwhelm. It boils at a temperature just short of water’s, making it easier to remove from final products. Its high solubility gets a workout in many labs, since it can blend with alcohols, ethers, and hydrocarbons without breaking a sweat. What’s amazing is the range of projects that run smoother thanks to DMF’s balance between reactivity and stability. Lab notes from my early years feature scribbles about its easy mixing, but also warnings about its volatility. It evaporates fairly quickly if left out, and its low surface tension means it soaks into surfaces as fast as water, demanding cleaner setups to avoid contamination.
Technical Specifications & Labeling
Walk into any chemical supply closet and DMF sits on a shelf in brown bottles or steel drums, tagged with hazard labels. Regulatory trends require careful documentation—labels flag it for flammability and for risks to health on inhalation or skin contact. My experience with chemical labeling tells me that DMF’s packaging must carry details about purity—typically 99% or higher for most research or manufacturing settings. Over time, newer rules in the US, Europe, and Asia have pushed for more detailed labeling, keeping track of potential exposure dangers. Batch certificates and updated Safety Data Sheets crowd the workbench wherever DMF is present, responding to demands for both traceability and legal compliance.
Preparation Method
Industrial chemists started making DMF by reacting dimethylamine with carbon monoxide or by heating methyl formate with excess methylamine. Production takes skill, since heat and high pressure get involved, and both starting ingredients need careful handling to avoid runaway reactions or hazardous by-products. Some years ago, I visited a production plant where the reaction was contained in thick-walled vessels, surrounded by scrubbers to clean any escaping fumes. Waste management must work overtime, too, because the runoff can harm both air and water if allowed to leak out. Making DMF gets a company to reckon with its own environmental footprint, not just its efficiency ratios.
Chemical Reactions & Modifications
Non-experts might not see it, but DMF is more than just a bystander in the chemical world. It stands as both a solvent and a reactant, giving chemists new tools for building molecules. Its structure lets it take part in specialized reactions, like the Vilsmeier-Haack formylation, where it helps make complex rings for pharmaceuticals or dyes. As an electrophile, DMF can transfer its formyl group to other molecules, speeding synthesis for key intermediates. My own lab work with functional group modifications often relied on DMF for steps where water or alcohols would spoil an otherwise promising run. In polymer science, DMF stands out for dissolving long-chain molecules that would otherwise clump, making it a mainstay in the study of climate-protecting materials or medical devices.
Synonyms & Product Names
DMF travels under different names. Technically, it goes by N,N-dimethylformamide, but folks in the field often stick with the abbreviation. Sometimes labels mention methylformamide or DMF just to keep things clear. You might spot names like Dimethyl amino formyl, though that's rare outside dense technical documents. Working with multiple suppliers showed me that each company likes its own twist—sometimes with purely administrative codes added for tracking. The best practice is to double-check chemical identity before use, since mislabeling can lead to confusing and even dangerous substitutions.
Safety & Operational Standards
Handling DMF doesn’t leave room for sloppiness. The risks add up fast: exposure can cause skin irritation, eye damage, liver stress, or worse after long-term or high-dose contact. From the start of my career, I learned to respect the gloves, goggles, and ventilated hoods required for even small batches. Regulations set by OSHA and their European counterparts demand strict personal protective measures, monitoring of air quality, and emergency gear nearby. Plants and labs using DMF keep detailed logs of spills, exposures, and disposal—audits by authorities happen without warning, and the fines for carelessness aren’t small. In the past, DMF’s dangers flew under the radar, but public health studies and workforce activism pushed safety standards far higher. Some locals near chemical plants keep DMF on their radar, calling for better air filtration and transparent reporting.
Application Area
Industries all over the map use DMF. Textile processors count on it for spinning durable fibers, while pharmaceutical firms turn to it for medication building blocks and research trials. Its dissolving strength powers electronics, paints, adhesives, and cleaning products. Whenever someone needs to coax polymers into smooth films or coat circuits for water-resistance, DMF comes up as the trusted solvent. Over years spent supporting manufacturing teams, I noticed how well DMF speeds up productivity—projects hit their marks faster and with greater certainty. This reach means DMF is both a cornerstone of modern products and a challenging substance to replace. As companies adjust to sustainability goals, they still often fall back on DMF, not out of habit, but because alternatives either cost more or fail to deliver.
Research & Development
Research hasn’t slowed since DMF’s early days. Lab teams probe new reaction chemistry every season, hoping to fetch better yields or reduce risks at the bench. Scientists dig into green chemistry, asking if DMF alternatives can step up without losing effectiveness. Some breakthroughs use DMF for making graphene layers, batteries, or solar cell parts—areas that matter as much for their innovation as for the environmental puzzles they introduce. Universities keep DMF in their teaching labs, but also study its breakdown in water and soil. Researchers publish studies on low-toxicity encapsulation and how DMF’s polarity maps onto new solvent systems. My own academic encounters with DMF left a mark: the discussions about trade-offs shaped how I see innovation—not as a straight path, but as a balance between performance, safety, and responsibility.
Toxicity Research
People working in production facilities know about DMF’s reputation for toxicity. Medical studies link prolonged exposure to liver and reproductive damage, sparking regulations to curb vapor levels in both the plant and the community outside. Recent literature tracks biomarkers in exposed workers, building a picture of health impacts and how quickly harm might unfold. Epidemiological tracking in chemical hubs finds heightened illnesses connected to poor ventilation or leaks. I remember hearing about factory upgrades where management pressed for better air filtration, prompted by local outcry and employee health claims. The push for safer alternatives only grows as research ties DMF to broader systemic effects—the quest for a safer workplace often collides with the need to keep production running on time.
Future Prospects
DMF sits at an intersection of convenience, effectiveness, and controversy. Chemical engineers, manufacturers, and environmentalists watch the research for signals about long-term risks, regulatory trends, and emerging substitutes. The stories coming out of regulatory agencies point to a world where DMF’s dominance may slowly give way to safer, greener options. For now, companies invest heavily in recycling systems, air quality controls, and process redesigns that shrink DMF footprints without gutting reliability. If alternative solvents match DMF’s firepower without its baggage, industry will likely adapt, but technical and economic realities keep progress slow. The tension between chemical performance and human or ecological safety remains. Watching this debate unfold has shaped my sense that progress rarely comes easy—responsibility calls for honesty about costs, benefits, and the long slog of real innovation.
What is Dimethylformamide used for?
A Closer Look at a Common Chemical
Dimethylformamide, usually called DMF in labs and factories, shows up in places most folks never see. It might sound like something tough to relate to, but it actually makes a big difference in many everyday products. Working in chemical manufacturing and research, I've seen DMF’s reach firsthand, and it’s a story worth sharing—especially since safety and practicality both hang in the balance.
Why Industry Relies on DMF
Factories making plastics, fibers, or pharmaceuticals wouldn’t get far without DMF. It’s valued for strong solvent power. Mix DMF with tough-to-dissolve solids, and pretty soon, you get smooth solutions that help turn raw ingredients into useful stuff. Polyurethane coatings, like the kind that toughen shoe soles or furniture, often trace back to a DMF blend. I’ve watched DMF carry dyes through textile fibers or help spin acrylic clothing in real time—it makes fabric production possible on a massive scale.
Pharmaceutical companies lean hard on DMF, too. They use it as a solvent during manufacturing and research—especially in the tricky process of making new medicines. If a new compound refuses to dissolve for purity checks or testing, DMF usually does the job. Some might not realize that many painkillers and antibiotics spent hours or days swimming in DMF long before they reached a pharmacy shelf.
Everyday Products, Hidden Ingredient
From electronics to adhesives, DMF plays a background role. Have you ever handled “super glue” or sealed up something with a specialized adhesive? Odds are decent DMF lent a hand in producing the glues, letting manufacturers mix ingredients that don’t usually get along. Even the flexible plastics around wires, car upholstery, or rain gear often rely on DMF-based processes. I’ve toured plants where cable coatings and faux leathers are made; DMF provided the flexibility and durability needed, helping create products that hold up under real-world use.
Safety Challenges and the Search for Alternatives
This useful chemical comes with some serious baggage. Breathing in DMF fumes or skin exposure can cause health problems ranging from nausea to organ damage, as scientific studies and workplace inspections reveal. I remember safety briefings in facilities where any spill or leak became a top concern—gloves, goggles, and ventilated booths weren’t optional, and with good reason.
Safety costs money and training. That pushes companies to rethink reliance on DMF, especially since many countries now set tough limits on how much can be released into air or water. Researchers work on greener solvents. Some newer polymers come together with safer chemicals, though the change tends to be slow because DMF performs better than most substitutes in certain roles. Chemical innovation takes time, but stricter laws and better worker protections keep the push going. Companies embracing alternative solvents or closed-loop recovery systems usually find long-term savings—and avoid costly accidents or regulatory fines.
Looking Ahead
DMF delivers value, but experience teaches caution. Its story reminds us that even familiar chemicals deserve respect and scrutiny. Safer options and stronger protections for workers and communities help keep useful tools available without risking health. Choice and responsibility both matter in chemistry, whether in a factory, a lab, or a supply chain that quietly connects them to our homes.
Is Dimethylformamide hazardous to health?
The Substance at a Glance
Dimethylformamide, usually called DMF in laboratories and factories, holds a steady spot in places making plastics, synthetic leathers, and pharmaceuticals. I still recall the sharp, unpleasant smell wafting through the air during my first tour inside a chemical manufacturing plant. Back then, I spotted workers wearing thick gloves and face coverings, a sign that this chemical deserved caution. Even outside the factory world, strict workplace rules around DMF protect those handling it daily.
Health Risks: More Than Annoying Fumes
Individuals exposed to DMF often report headaches, skin rashes, and stomach upsets. This chemical finds its way through skin and lungs faster than many expect. Scientific studies have flagged that regular contact links to liver damage—some researchers call the risk “occupational hepatitis.” The World Health Organization and the US Environmental Protection Agency both classify DMF as a substance meriting strong precautions, and there’s a reason: workers exposed to enough DMF sometimes show changes in liver enzymes, an early warning of real harm.
Exposure also brings issues for reproductive health. Studies have raised red flags about fertility and potential birth defects in both men and women. A review published in Occupational and Environmental Medicine followed garment workers and chemical operators and found that those breathing or touching DMF often took longer to conceive and, in some cases, suffered miscarriages. Scientists still don’t agree on safe limits, but no one disagrees about the risks of chronic exposure.
Everyday Encounters and Community Concerns
People living near chemical plants sometimes worry about air and water quality. DMF evaporates quickly, drifting through open doors or leaky storage tanks. After local community meetings near a facility in Texas, I saw parents bring up unexplained nosebleeds and nausea in their kids. Environmental agencies now track DMF levels closely near factories, setting strict limits for what can escape into the air or groundwater.
Inside the workplace, even a few drops can cause an itchy rash if protective clothing falters. I remember one project where a small spill led to a scramble for the eyewash station and a hard lesson about following safety protocols. Gloves and goggles aren’t just for peace of mind—they prevent real trouble.
Taking Action: How to Stay Safe
Curbing risk means strict rules and cooperation from everyone on site, from plant managers to new hires. Good ventilation and closed systems trap DMF vapors. Scheduled health checkups pick up early trouble in the liver, and medical teams track workers’ health every year. Regulatory groups like OSHA and the European Chemicals Agency set strict limits for how much DMF can hang in the air.
Outside the workplace, advocacy groups push for green chemistry—researchers are searching for safer substitutes in everything from paint thinners to adhesives. Companies exploring these alternatives find the upfront cost, but many decide it’s worth it to keep their people safe and avoid lawsuits. Technology offers hope: sensors and portable air meters now spot leaks fast, reducing the time anyone spends exposed.
Shared Responsibility
No one should gamble with their health for a paycheck or profit margin. Action from leadership shapes company culture, but every individual shares a stake in safety. Reading the label, airing out the workspace, and choosing gloves that really protect may seem basic, but those habits shield people from real harm. The conversation about DMF remains relevant as long as businesses turn to chemicals to build the modern world.
What is the chemical formula of Dimethylformamide?
Dimethylformamide: At the Core of Modern Chemistry
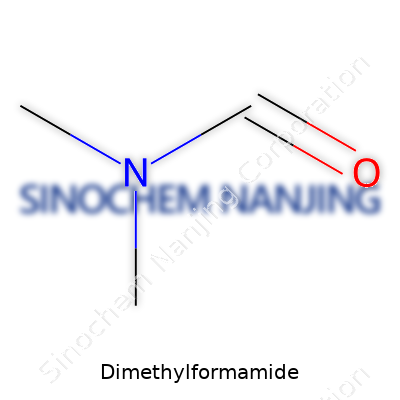
Dimethylformamide, known as DMF to most folks in a lab coat, carries a simple but vital formula: C3H7NO. Not every chemical wears its heart on its sleeve like DMF. It’s a staple in biology, chemistry, and plenty of industrial households. Growing up around a chemist, I used to hear stories about solvents so reliable they’d be called the workhorses of the lab. DMF always came up.
What Makes DMF Stand Out?
The formula C3H7NO has more weight than it seems. One carbonyl, a lone nitrogen, and two methyl groups bring it all together. That combo means it dissolves a wide mix of other substances—acids, bases, salts. Ask researchers working on polymer synthesis, they tend to reach for DMF because they trust it to get the job done with tough molecules.
It’s not just the lab folks who care. Pharmaceutics relies on DMF for medicines, fibers take their shape with it, and electronics grow cleaner crystallines because of it. The reason C3H7NO matters goes beyond shining glass flasks. Precision manufacturing depends on it. A researcher at a major university once told me, “We rarely finish a day’s work without opening that bottle.” No exaggeration—one Chinese market research report from 2023 pegged DMF’s global demand at more than a million tons per year.
Keeping the Workplace Healthy
All that usefulness brings its own headaches. DMF isn’t a friend to bare skin. I spent one summer in a pharma pilot plant where the rule was simple: splash DMF, and you drop everything and rinse, fast. Chronic exposure can disrupt liver health. The US Department of Health and Human Services puts it clearly—handle DMF carelessly, and your body pays the price. Gloves, goggles, and solid ventilation setups mean the difference between safe handling and a trip to the PCP.
There’s another snag—DMF isn’t quite green. It slips into wastewater and air, giving headaches to anyone running an industrial waste treatment plant. In Germany and the United States, tighter emission standards keep factories honest. Good policies force recycling and recovery, which doesn’t just save the planet a mess. Doing it right lowers costs in the long run, and a peer-reviewed study in the journal Environmental Science & Technology backs this up. Few businesses aim to waste money on regulatory fines or hazardous waste removal.
What’s Next for DMF?
The future for C3H7NO depends on balance. Tech companies continue to push for cleaner alternatives, but for now, precision tasks need reliable solvents. Countries with booming pharmaceuticals and electronics sectors invest in safer handling practice and greener production cycles. If you spend time in labs, proper training and monitoring makes a world of difference, not just for compliance but for everyone’s health. More labs now swap out DMF for less toxic options where possible, and some universities even set up research funds for DMF substitutes. Yet old habits die slow. DMF’s formula might look simple, but its reach goes deep.
How should Dimethylformamide be stored?
Why Dimethylformamide Demands Respect
Dimethylformamide, better known in labs as DMF, shows up in research, manufacturing, and even pharmaceuticals. People don’t usually talk about it at barbecues, but anyone who has worked in a lab knows its pungent smell and the headache that comes from working in a poorly ventilated room. Most folks shrug it off as just another solvent, though handling it without real care can lead to skin problems, headaches, and—over the long haul—much more serious health issues.
DMF evaporates easily, which means those vapors can sneak up on you. There’s no honor in simply lining up the bottles on a standard shelf and calling it a day. Safety and quality both take a nosedive if storage gets sloppy.
Picking the Right Location and Container
A container’s seal always comes first. Glass or high-quality plastic, with tight caps, keeps the substance in and air out. I remember working in an older lab where someone had kept DMF in a makeshift bottle. Months later, every cap in the chemical cabinet had turned brittle. Turns out, DMF can soften some plastics, so you just can’t grab any old bottle off the shelf and call it good. Metal containers with proper lining also work, but only if you personally check compatibility.
I’ve seen too many cases where DMF found its way onto a rusty old shelf wedged between other solvents. Flammable liquid cabinets, clearly marked, cut down the risk. These cabinets carry more than just a “safety” sticker—they can save property and lives in case of a fire. Remember, DMF likes to ignite at about 58 degrees Celsius, so it won’t take much heat to set it off. Fire-proofing isn’t just for the accident-prone; in my experience, the person who says “we’ve never had a problem” is the very one who finds out the hard way what goes wrong when you ignore good storage.
Keep the Temperature and Light Down
Warm rooms speed up vapor release. DMF belongs far away from direct sunlight and anything that generates heat. Darkness and coolness don’t just sound soothing—they stop light and heat from breaking down the solvent. If you can keep it closer to room temperature, or even a little cooler, peace of mind comes easy. If that sounds strict, well, I’ve seen firsthand what happens to a mishandled DMF supply. The solvent broke down, the results got thrown out, and someone spent a weekend neutralizing a spill instead of watching their kid’s soccer game.
Mind the Air and Humidity
Moisture creeps in if you store DMF out in the open. The substance will pull in water from the air, which can ruin purity. High humidity shortens its shelf life, and I've dealt with ruined batches that could've been saved if someone closed the door on the chemical storeroom. Always run an exhaust hood if you’re working with open containers. Not only does this keep your air clear, but vapors don’t get a chance to collect and find a spark to latch onto.
Don’t Forget Proper Labeling
Every time I see a faded label or an old piece of masking tape pasted to a bottle, I know someone’s cut a corner. Forgetting to mark the container might sound harmless, but in an emergency, it turns minor mix-ups into real disasters. Always mark containers with the substance name and the latest delivery or open date. It spares you from digging through memory or guessing—never a wise move with something like DMF.
Simple Practices Prevent Nightmares
Some folks treat chemical storage as a pesky detail. It’s not. Respect the container, control temperature and light, stay on top of humidity, and mark every bottle clearly. Good habits here make the difference between a smooth work week and one full of headaches—sometimes literal ones at that. I’ve seen what goes wrong when people skip steps. Keeping DMF safe doesn’t require luck; just diligence and the right approach.
What industries commonly use Dimethylformamide?
DMF in the Textile Industry
Walk down a busy street and just about everything made from synthetic leather and stretch fabrics has probably seen dimethylformamide somewhere along the production line. Textile manufacturers lean hard on DMF as a solvent for making polyurethane coatings and synthetic fibers like acrylic. The reason is simple: it gets the job done quickly, dissolving the polymers that wind up in our jackets, shoes, car seats, and sportswear. In large weaving and finishing halls, workers handle it daily. Health experts have linked high exposure to risks for liver and reproductive issues. Factories that haven’t switched over to safer handling systems run a real risk of harming their employees. Strong regulations and switching to safer alternatives, like water-based systems, make a difference. They protect workers and help companies avoid legal trouble.
Pharmaceuticals Lean on DMF
Chemists in pharmaceutical labs call on DMF for the tough reactions. Drug makers appreciate how reliably the solvent dissolves almost anything, especially when making active pharmaceutical ingredients for antibiotics and antivirals. The flexibility it offers has pushed newer, more effective medicines onto the market. Quality control in established firms tracks exposure at every step, but some smaller labs—especially outside major markets—can get careless, and that’s when accidents happen. Training and proper fume hoods are not options; they are essentials. Consumers rely on pharmaceutical firms not only to produce effective drugs but also to lower chemical risks throughout the process.
Paints, Inks, and Coatings Rely on DMF
Factories that churn out inks for packaging or paints for cars and appliances often pick DMF because it dissolves pigments and resins that tougher, greener solvents can’t handle. It leaves a smooth finish and deep color. Working with DMF day after day can be hard on employees, especially if protective gear isn’t standard issue. In the U.S. and Europe, strict guidelines require companies to monitor workers’ health and air quality. But in places where oversight lags, safeguards are sometimes skipped. Industry leaders need to champion changes, like closed-loop systems that catch fumes. Sticking to safer alternatives can limit heavy exposures without sacrificing quality.
Electronics: A Hidden Player
People rarely notice what's behind their phones, laptops, or flat-screen TVs. DMF helps manufacture semiconductors, lithium batteries, and flexible circuitry. Its solvent properties let manufacturers etch and rinse delicate circuit boards, removing unwanted layers without shorting the sensitive parts. I worked on an assembly line for a short time, and fumes from the chemical baths didn’t just fade away. Handling DMF takes real focus, from storage to disposal. Several Asian producers caught headlines for worker hospitalizations until stricter controls rolled in. Technology upgrades and robust ventilation systems have come into play, but without enforcement, accidents return.
Personal Experience and Solutions
After touring chemical plants for a sustainability project, I saw up close what happens when shortcuts rule the shop floor. Spills, unfiltered air, basic gloves instead of proper suits—cutting corners happens wherever margins are thin. Government agencies, including OSHA and the European Chemicals Agency, publish clear exposure limits and certify protective gear. Companies can follow suit by swapping out DMF for less toxic substitutes, where possible, and investing in safety culture. Research into new solvent systems shows promise, and a few forward-looking firms have switched over. It costs more up front, but the long-term payoff is safer workplaces and fewer environmental headaches.
| Names | |
| Preferred IUPAC name | N,N-dimethylformamide |
| Other names |
DMF N,N-Dimethylformamide Methanamide, N,N-dimethyl- Formic acid dimethylamide |
| Pronunciation | /daɪˌmɛθəl.fɔːrˈmæm.aɪd/ |
| Identifiers | |
| CAS Number | 68-12-2 |
| Beilstein Reference | 505948 |
| ChEBI | CHEBI:17741 |
| ChEMBL | CHEMBL1423 |
| ChemSpider | 5790 |
| DrugBank | DB03796 |
| ECHA InfoCard | 19ecdcbb-3fbb-4322-ace0-6a695b4a348b |
| EC Number | 200-679-5 |
| Gmelin Reference | 6137 |
| KEGG | C01016 |
| MeSH | D004046 |
| PubChem CID | 6228 |
| RTECS number | LQ2100000 |
| UNII | 4MMS62S9Y2 |
| UN number | 2265 |
| Properties | |
| Chemical formula | C3H7NO |
| Molar mass | 73.09 g/mol |
| Appearance | Colorless, transparent liquid |
| Odor | fishy |
| Density | 0.944 g/cm³ |
| Solubility in water | Miscible |
| log P | -1.01 |
| Vapor pressure | 2.7 hPa (20 °C) |
| Acidity (pKa) | 15.2 |
| Basicity (pKb) | pKb = 10.3 |
| Magnetic susceptibility (χ) | -10.1×10⁻⁶ |
| Refractive index (nD) | 1.429 |
| Viscosity | 0.92 mPa·s (at 25°C) |
| Dipole moment | 3.82 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 153.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -238.7 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1739 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H226, H319, H332, H360D |
| Precautionary statements | P210, P280, P261, P304+P340, P308+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-2-1⬡ |
| Flash point | 58 °C |
| Autoignition temperature | 445°C |
| Explosive limits | 3% - 15% |
| Lethal dose or concentration | LD50 oral rat 2800 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2,800 mg/kg (oral, rat) |
| NIOSH | NIOSH: LQ2100000 |
| PEL (Permissible) | 10 ppm |
| REL (Recommended) | 10 ppm (30 mg/m3) |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
Formamide N-methylformamide N,N-dimethylacetamide Acetamide |

