Dimethyl 1,3-Propanediol: Navigating an Evolving Chemical Landscape
From Lab Bench Curiosity to Industrial Mainstay
Dimethyl 1,3-propanediol didn’t emerge from the shadows of chemistry all at once. Chemists started with simple glycols before halfway through the last century, and this compound arrived as part of the push for better intermediates in polyester and coating chemistry. Manufacturers soon realized those two methyl groups on the 1,3-propanediol backbone changed the way resins cured, the flexibility of plastics, and how well they stood up to the sun and water. Innovation in organic synthesis and refining made volume production possible, shrinking costs and opening the door to research teams aiming for next-gen coatings, cosmetics, and elastic fibers.
Physical & Chemical Touchpoints You Can't Ignore
Dimethyl 1,3-propanediol comes as a clear, nearly odorless liquid sitting comfortably at room temperature. Pour it out and it doesn’t evaporate like acetone or ethanol. Its boiling point climbs into the mid-200s Celsius, so it hangs around—an asset in coatings and polymer reactions where you don’t want volatility working against you. The two hydroxy groups at the ends offer a reactive handle, making it keen to join up in esterification or etherification reactions. Those methyl side groups, though, don’t just hang out as decoration—they add a layer of steric bulk that makes the molecule less likely to load up on water through hydrogen bonds, which increases its hydrophobic tail in larger polymers, and boosts resistance to hydrolysis.
Technical Standards Must Earn Trust
Markets and regulators pay attention to specs for good reason. Purity rules for dimethyl 1,3-propanediol sit above 98 percent for industrial applications, and makers measure water content by Karl Fischer titration to assure longevity in finished resins. Labels outline chemical identity, batch, and recommended storage—all shaped by accident data and safe handling history rather than empty checkboxes. Gel permeation chromatography, NMR, and IR have taken over as the go-to techniques for confirming products, leaving little room for misidentity. Real world application comes down to trust in each drum’s consistency, since one out-of-spec batch can turn a major coatings facility’s day upside down.
Manufacturing Paths Write the Environmental Story
Two main methods have become widespread. Most commercial output flows from hydrogenation of the corresponding dimethyl esters or via the hydroformylation of unsaturated precursors followed by reduction. Each carries its own waste profile and carbon footprint, which plays bigger every year as sustainability stakes climb. Some chemists have explored metabolic and enzymatic pathways—using engineered microbes to feed on sugars or other renewable feedstocks, but scaling has proven trickier than pitching a new lab process. As buyers prioritize green credentials, process chemists have their hands full squeezing out solvents, stepping up recycling, and running life-cycle analyses that stand up to real scrutiny.
Why Synonyms Matter on the Factory Floor
Dimethyl 1,3-propanediol travels under a handful of names, from 2,2-dimethyl-1,3-propanediol to neopentyl glycol—the latter rolls off the tongue in the industry, even though some might use both terms in a technical document. If you’re not paying attention, those naming quirks can trip up purchasing managers, confuse import labels, and muddle HAZMAT inventories. Overlooking label detail gets expensive fast as different countries hold different CAS registry numbers or trade stamps. Being clear about synonyms isn’t just for paperwork; it’s tied directly to worker safety and avoiding supply chain mistakes.
Reactivity That Fuels A Whole Industry
Neopentyl glycol’s diol backbone makes it crave reactions with acids, anhydrides, and isocyanates—giving rise to esters, urethanes, and even high-end polyester resins that form tough, weatherable coatings. Its methyl branches push back on oxidative degradation—an absolute win compared to traditional glycols, especially in paints facing sun, rain, and temperature swings in outdoor use. Scientists have tweaked its basic structure, attaching specialty groups: halogens for flame retardancy in electronics, or larger alkyl chains for lubricity in advanced grease formulations. Each tweak spins off a new thread of performance, steering R&D into uncharted territory.
Rules, Safety, and Getting It Right
Moving lots of flammable, reactive organics isn’t work for amateurs. Industry guidelines for neopentyl glycol shape how large players store, transfer, and use the chemical. Splash goggles and gloves mark only the baseline, and ventilation needs addressing in any operation handling kilogram batches. Long-haul transport—by rail, road, or ship—requires bulletproof documentation. In my own work environments, slip-ups with labeling or mixing up containers threatened both process yield and safety outcomes. Institutional memory—the lessons from decades of near misses—drives stricter auditing and worker onboarding today. Inspection regimes ask the right questions: Is spill control robust? Are hazard sheets updated and reachable? Does everyone know extinguishing requirements if things go south? Not all facilities hit every mark, but ongoing attention pushes the bar higher.
Application Spurs Innovation—in Surprising Corners
Most folks who’ve worked in coatings or polymer R&D know neopentyl glycol as a backbone for high-durability alkyds and polyesters. That application dominates industrial demand, as it creates building blocks for paints that shrug off corrosion and yellowing—essential in the construction and auto world. Outside of that, its component esters sweeten up as flavors and fragrances, though carefully regulated. I’ve watched formulators use it to boost the dimensional stability of engineering plastics, while cosmetic chemists fiddle with its derivatives to control viscosity and texture in creams and lotions. Each new use demands a performance tweak, and cross-industry collaboration becomes the norm as consumer and regulatory groups call for safer, longer-lasting, and greener products.
Research Keeps Exposure and Health in View
Safety conversations start and end with real-world human data. Decades of handling haven’t flagged neopentyl glycol as acutely toxic, but any glycol must be handled with respect. Animal studies suggest moderate irritation with high doses. Notably, longer-term bioaccumulation and metabolic breakdown inside living systems receive more attention nowadays, with modern toxicologists prioritizing developmental and reproductive endpoints that often fly under the radar. In regulated workplaces I’ve observed, regular refresher courses and continuous monitoring shrink the risk, but smaller producers sometimes struggle to keep up. Academic and industry research needs to keep closing data gaps—especially as regulatory agencies in Europe, the US, and Asia sharpen scrutiny around chronic exposure, environmental persistence, and vulnerable worker populations.
The Road Ahead: Demanding More from a Familiar Molecule
Neopentyl glycol’s story isn’t set in stone. Pressure from downstream users builds higher standards for carbon neutrality and circular economy wins. Markets want raw materials that don’t just enable strong, sun-resistant plastics but do so without amplifying waste. The race now focuses on process improvements—waste heat recovery, solvent minimization, and catching value from byproducts that used to ship as landfill. Companies invest in biotechnology plays that swap fossil feedstocks for renewable sugars, though scalability hurdles remind us that change is slow and incremental. If regulators accelerate phaseouts of certain solvents or additives, forced innovation may rewrite process recipes altogether. Chemistry teachers already pitch the molecule’s branching structure as an example of how small structural changes can punch above their weight in physical properties. That lesson lands every time a chemist finds a fresh application or a greener process route.
What is Dimethyl 1,3-Propanediol used for?
How Industries Are Using Dimethyl 1,3-Propanediol
More manufacturers have started turning to Dimethyl 1,3-Propanediol as a backbone ingredient for making certain plastics and resins. This glycol-based chemical carries a reputation for reliability in producing polyester resins, which show up in anything from coatings to the finish on desks and cars. Many in the business of packaging want plastics to last longer and have fewer weaknesses over time, so they rely on the qualities this compound brings. It lets finished goods resist breaking and cracking, even after repeated handling or exposure to tough elements like heat or moisture. From personal experience managing supply chains, I’ve seen requests for this chemical tick upward whenever a client needs stronger, longer-lasting components, whether for outdoor signage or food-safe containers.
Another big area of use comes from the coatings industry. Dimethyl 1,3-Propanediol helps produce paints and varnishes that dry evenly and hold up against daily wear. If you’ve ever noticed how some surfaces get sticky or tacky with humidity, the right type of chemical base can make a difference. With this diol in the mix, surfaces tend to keep their finish and resist “yellowing,” which keeps everything looking cleaner over time. Automotive suppliers and furniture makers often highlight these advantages, since nobody wants their products fading or looking rough just months after purchase.
Why the Details Matter
Some folks might brush it off as just another chemical, but there’s more at stake here. Unlike some alternatives, Dimethyl 1,3-Propanediol offers a safer profile for both workers and end users. Regulatory agencies in the US and Europe keep a sharp eye on harmful byproducts, so companies favor ingredients that keep them compliant. In my own work with sustainability teams, questions always come up about eco-labeling and toxicity. This glycol typically gets the green light for low emissions and limited hazardous waste during processing.
The ripple effects appear downstream, too. Storage, shipping, and disposal all cost less when a product leaves behind fewer harmful residues. Businesses that pay attention to these costs often come out ahead, whether they’re reducing fines or improving their brand image with consumers. The shift away from more volatile chemicals runs parallel to what we’ve seen across industries—healthier materials win both in the market and in regulatory reviews.
Room for Improvement
Even as the outlook looks positive, no single ingredient solves every challenge in manufacturing. Sourcing has its own hiccups—raw material prices can swing and global supply chains remain fragile. Energy use stands out as a concern; some resin plants still run on outdated systems that burn up more fuel just to keep production lines moving. I’ve watched teams struggle with how to make practices greener without letting costs spiral. This often comes down to investing upfront in newer equipment or smarter process controls, which pays back over time but takes vision and resources.
Some researchers explore bio-based versions of 1,3-Propanediol, aiming to cut reliance on fossil resources and shrink the overall environmental footprint. Companies looking to stand out could benefit by pushing for these renewable options, provided quality and strength remain solid. Supply partners also play a key role in sharing information about best practices, helping everyone in the chain move toward safer and more sustainable production.
In fast-moving sectors, keeping pace with regulations and customer preferences shapes the future more than raw chemistry does. Materials like Dimethyl 1,3-Propanediol represent a step forward, but real progress depends on a broader commitment to responsible sourcing, transparency, and innovation.
Is Dimethyl 1,3-Propanediol safe to handle?
What’s in Dimethyl 1,3-Propanediol?
Dimethyl 1,3-propanediol doesn’t turn up in the grocery aisle, but it finds its way into coatings, polymers, or specialty chemicals. The substance looks like a clear liquid with a faint odor. You might spot someone in a lab coat pouring it into a reactor, thinking it works like any other industrial additive. This compound has real uses but also real risks, especially if the basics of safe handling get ignored.
Everyday Concerns in the Workplace
If you’ve ever watched a training video on chemical safety, you know many workers treat liquids with healthy caution. Dimethyl 1,3-propanediol belongs right there on the list of substances that deserve gloves and goggles, along with solid training. Direct skin contact might turn up with redness and irritation. Breathing in the vapors in a closed space without proper ventilation can irritate the nose and lungs. The liquid itself can harm eyes—a splash is just as bad as touching your face after exposure.
Trusted Sources: What Science and Regulators Say
Occupational safety regulators in the US, Europe, and Asia publish chemical safety sheets for a reason. The scientific literature calls for practical measures—good ventilation, protective clothing, eye protection. The Globally Harmonized System (GHS) classifies dimethyl 1,3-propanediol as causing possible skin and eye irritation. Long-term studies on humans stack up a little thin, but the structure of this molecule means it’s wise to avoid getting it on your hands or in your eyes.
Real-World Experience: Learning from Mistakes and Successes
In chemical plants, complacency often leads to near misses. Years of working near chemicals taught many technicians not to cut corners. Safety culture relies on real stories—a mishap involving a leaking container leads to improved labeling or better storage. More than once, I’ve seen a forgotten glove or mask end with someone heading for the eyewash.
The good news: proper personal protective equipment and routine training outsmart most risks. The Material Safety Data Sheet (MSDS) takes a few minutes to read, but following it makes life easier. Plenty of workers build their own muscle memory around these tools.
Shared Responsibility and Safer Solutions
No chemical swaps out common sense. Supervisors need to ensure workers understand the hazards and report close calls. Clear labeling, addressable spills, and good communication all make workplaces safer. Manufacturers can help by designing clear instructions, distributing PPE without hassle, and encouraging prompt reporting of issues.
More companies are looking at safer alternatives, especially if research backs them up. For now, as long as dimethyl 1,3-propanediol plays a part in manufacturing, vigilance stays front and center. A healthy respect for any chemical—not just this one—keeps people in good shape for the next shift.
What are the physical and chemical properties of Dimethyl 1,3-Propanediol?
Looking Past the Chemical Name
Dimethyl 1,3-Propanediol. Just reading that, most eyes glaze over. It sounds like something reserved for only the most specialized labs. In reality, this molecule jumps into everyday products—paints, plastics, coatings, and even household items. What puts this diol in the spotlight is its set of physical and chemical properties. Let’s look at what gives it its unique reputation in the world of chemicals, without the technical jargon that draws blanks from most people.
Getting a Feel for Its Physical Traits
This compound shows up as a clear, almost syrupy liquid at room temperature. Anyone who has handled similar glycols like propylene glycol knows the sensation: slippery, viscous, and just a touch heavier on the fingertips than water. It doesn’t change into a gas easily; its boiling point hangs out well above 200°C, letting manufacturers use it in high-temperature settings without losing half their product to vapor. Most everyday folks never think about this, but in paint or plastic factories, a chemical that sticks around instead of evaporating too quickly cuts costs, limits waste, and keeps workplace conditions stable.
It’s surprisingly cooperative with water. This means it dissolves easily, which helps when mixing it into solutions or blends. Imagine stirring sugar into your coffee—no clumps, no endless stirring. That solubility matters for anyone designing smoother, more reliable coatings or fibers.
Chemical Personality: Reactivity and Safety
Dimethyl 1,3-Propanediol doesn’t like to catch fire, which brings peace of mind—in factories, flammable liquids spell disaster. Its “flash point”—the temperature where it can catch fire from a spark—sits relatively high for a chemical liquid, and its low volatility means there’s not a lot of vapor hanging in the air waiting to ignite. Safety officers always keep an eye on this; it saves headaches in risk management and insurance costs.
Its most noteworthy trick chemically comes from its two alcohol groups. These “-OH” groups turn it into a workhorse for making polymers like polyesters and polyurethanes. Any chemist who’s worked in a lab making plastics knows how valuable a reliable diol can be for chain-building reactions. These groups react well, helping link up small molecules into tough, flexible plastics. It stands out for resisting yellowing and breakdown when exposed to light or heat, gotten first-hand experience with a failing plastic water bottle will know why that matters.
Why It Shows Up in So Many Products
Companies appreciate chemicals that don’t overcomplicate their processes. Dimethyl 1,3-Propanediol doesn’t play hard to get. Its physical stability, willingness to mix, and ease of handling tick boxes in the production line. It resists picking up water from the air, which is handy in protective coatings meant to last outside, under heat, rain, and sunlight. From sports gear to building materials, consistent performance under stress counts for a lot.
Room to Do Better
Sourcing safer chemicals always raises concerns about long-term safety for workers, communities, and the environment. Research over the years suggests dimethyl 1,3-Propanediol doesn’t pose major health dangers under proper working conditions, but regular review and improved protective equipment form the backbone of a safer industry. Building more sustainable production methods would make a big difference. Companies pivoting to renewable feedstocks—plant-based rather than petrochemical—could help reduce the carbon footprint. Choosing approaches that use less energy or create fewer byproducts streamlines operations and shrinks environmental impact.
Looking Ahead
Dimethyl 1,3-Propanediol keeps earning its spot in manufacturing because it combines chemical reliability with physical ease of use. I’ve seen firsthand that chemicals with these traits don’t just serve science—they push industries forward by enabling better products for people. In chemistry, as in life, the simplest traits sometimes do the most heavy lifting.
How should Dimethyl 1,3-Propanediol be stored?
Not Your Everyday Pantry Chemical
Dimethyl 1,3-Propanediol goes far beyond what comes out of a home toolkit. It shows up in industrial plants for making coatings, resins, and plastics. I’ve spent time in specialty chemical warehouses, and I’ve seen what can go wrong when folks get careless about chemical storage. It’s not just about keeping the drum lid tight. Safe storage means thinking about people and the environment first. That part comes from experience, not a textbook.
Location Really Matters
Setting aside a proper spot for storage has more to do with safety than convenience. Keeping chemicals like Dimethyl 1,3-Propanediol away from direct sunlight and heat sources helps prevent breakdown or reactions over time. Factory workers I’ve talked to hate dragging big containers around because warehouses jam random pallets together. Giving these containers a special section, away from traffic and steam pipes, says you respect the risks.
Steel Drums and Smart Seals
Four years back, I watched a floor manager open a drum, only to find crusted-up product from air sneaking in. A simple oversight – the gasket on the lid had dried out. For Dimethyl 1,3-Propanediol, keeping containers airtight is a must. Moisture can sneak in and the chemical can absorb water, changing its properties and causing headaches later in the production process. Stainless steel or HDPE drums usually stand up best, and checking seals feels tedious until that one emergency makes you wish you’d done it every time.
What’s Nearby Shouldn’t Be Ignored
Storing this chemical away from acids, strong oxidizers, and open flames isn’t just a rule – it keeps the workplace standing another day. Cross-contamination from a leaky neighboring drum can spark reactions no one wants in their building. I’ve seen supervisors map out which aisles hold what. That kind of planning takes extra time but pays off in the long run, since insurance claims and downtime are a bigger pain than walking a few extra steps.
The Right Temperature
Factories often forget that not every chemical likes sitting hot and humid. Manufacturers recommend 20 to 25 degrees Celsius for Dimethyl 1,3-Propanediol because temperatures outside this range risk making the product less stable. I’ve seen batches harden or go cloudy at higher temperatures. Factories trying to save on cooling later spend more sorting out drums that won’t pour or mix the way they should.
Paperwork That Matters
Those material safety data sheets some folks tuck in a drawer actually serve a purpose. During audits or spill and exposure incidents, up-to-date labels and records make a difference as emergency crews work out what’s what. Staff training needs to be more than a box on a safety checklist. Refresher sessions once every quarter keeps everyone sharp, and makes the difference between a routine day and a disaster.
Staying Safer, Working Smarter
Chemical safety often comes down to habits, not high-tech tricks. Clear walkways, working ventilation, correct containers, and staff who get regular hands-on training keep products like Dimethyl 1,3-Propanediol where they belong—and people safe. Order in chemical storage signals respect—for the science, the factory floor, and the humans running it.
What is the typical purity of commercial Dimethyl 1,3-Propanediol products?
Why Purity Levels Matter
A lot of folks overlook chemistry until the end product fails. That's when purity suddenly matters. In my last job working with polymers, I learned this the hard way. The quality of a monomer, like Dimethyl 1,3-Propanediol, ripples through every batch. Fail to check purity, you’ll see it in foggy plastics or coatings that don’t cure right. Businesses that cut corners at this step spend more money fixing mistakes down the line.
Numbers You Can Trust
Most manufacturers offer Dimethyl 1,3-Propanediol at a minimum purity of 98%. That’s the standard I’ve found in every tech data sheet worth reading. Some companies hit the market with product closer to 99% or higher. Extra purity costs more, but it makes sense for industries like pharmaceuticals or electronics where quality drives everything.
I’ve seen some small shops get tempted by off-spec batches advertised at cut rates, sometimes sitting at 96% or 97%. Those few missing percentage points often come from residual solvents or related diol impurities. In polyurethane or polyester synthesis, a stray contaminant can cause discoloration, or products that break down faster than you’d expect. Real-world consequences pop up fast, especially in automotive or medical devices.
Trace Impurities and Quality Control
Purity numbers don’t tell the whole story, though. Customers want to know about trace metals and residual reactants. In one batch, our team ran into copper traces, and suddenly every test panel corroded twice as quickly. What’s in the other 1-2% of commercial material ends up driving the work for the analytical team. Responsible suppliers back up claims with certificates showing limits for water content, color (often measured with the APHA scale), and sometimes even odor.
Years ago, I visited a supplier who insisted on running GC-MS (gas chromatography-mass spectrometry) on every lot. The investment paid off. No customer ever sent a batch back. For anyone mixing resins or compounding plastics, those outlier specs save time and headaches. I always tell newer chemists: purity claims aren’t just a checkmark; they’re guardrails for the entire supply chain.
What Drives the Purity Race?
Over time, demand for higher purity only grows. Regulations get tighter every year, especially in Europe and Japan. Environmental agencies in these countries enforce strict thresholds for unintended contaminants, especially organics that could off-gas or leach out. Larger clients started requesting third-party lab results alongside standard documentation. In big chemical plants, that meant dedicating whole shifts to running purity tests.
As tighter specs became the norm, some factories upgraded their distillation setups. Old glassware gave way to stainless reactors with better control over pressure and temperature. Downtime shrank, and so did the risk of contamination. That investment didn’t just satisfy audits—it let teams spend time improving formulations instead of scrambling to replace bad batches.
Onward With Better Transparency
I don’t see the purity push slowing down. Clients expect more than a printed COA. They ask what methods back up numbers. Whether you’re a small batch adhesive formulator or a major automotive player, trust grows when suppliers share not just figures but stories behind those figures—where the numbers come from, and how they prevent failures down the road. Dimethyl 1,3-Propanediol stands as a good example of how every percentage point and every trace impurity carries weight in the real world.
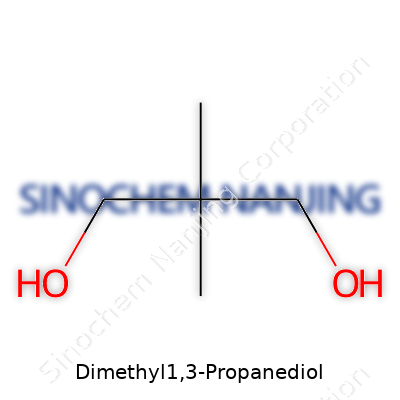
| Names | |
| Preferred IUPAC name | 2,2-Dimethylpropane-1,3-diol |
| Other names |
1,3-Dimethoxypropane 1,3-Propanediol dimethyl ether Dimethyltrimethylene glycol |
| Pronunciation | /daɪˈmɛθɪl waɪn θriː ˈproʊpeɪnˌdaɪˌɒl/ |
| Identifiers | |
| CAS Number | 126-30-7 |
| Beilstein Reference | 1361164 |
| ChEBI | CHEBI:34467 |
| ChEMBL | CHEMBL16298 |
| ChemSpider | 12399 |
| DrugBank | DB14096 |
| ECHA InfoCard | 13b4bfba-bdd7-41fd-8f8a-b0e67ba713ca |
| EC Number | 203-498-1 |
| Gmelin Reference | 124147 |
| KEGG | C16444 |
| MeSH | D003825 |
| PubChem CID | 8855 |
| RTECS number | EK2275000 |
| UNII | Z980A8X3T8 |
| UN number | Not regulated |
| CompTox Dashboard (EPA) | DTXSID8057590 |
| Properties | |
| Chemical formula | C5H12O2 |
| Molar mass | 134.18 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | Odorless |
| Density | 1.03 g/cm3 |
| Solubility in water | Soluble |
| log P | -0.49 |
| Vapor pressure | 0.01 mmHg (25°C) |
| Acidity (pKa) | 14.51 |
| Basicity (pKb) | 9.27 |
| Magnetic susceptibility (χ) | -61.08e-6 cm³/mol |
| Refractive index (nD) | 1.428 |
| Viscosity | 15.8 mPa·s (25 °C) |
| Dipole moment | 2.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 318.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -643.65 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3465.3 kJ/mol |
| Hazards | |
| Main hazards | Causes serious eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 122°C |
| Autoignition temperature | 252 °C |
| Lethal dose or concentration | LD50 Oral Rat 6500 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 16,600 mg/kg |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.005 ppm |
| Related compounds | |
| Related compounds |
Trimethylolpropane Neopentyl glycol Glycerol Ethylene glycol 1,4-Butanediol |

