Looking Deeper Into Dimethyl Maleate: More Than a Building Block
A Glimpse Back: The Path to Dimethyl Maleate
Dimethyl maleate has a longer story than most folks reckon. This chemical, a colorless liquid with a mild, fruity scent, takes its roots from the ongoing march of organic chemistry through the 1900s. The broader chemical industry, fueled by demands for new plastics, coatings, and additives, led to increased interest in fine-tuned esters and intermediates. Maleic acid and its derivatives, including dimethyl maleate, quickly found starring roles thanks to their reactivity and versatility. Innovation in catalytic processes, like those using palladium or copper, has improved the yields and purity of dimethyl maleate. This transformation mirrored the growth of the plastics and chemical manufacturing world more generally—always chasing efficiency and safety, always adjusting to new environmental rules.
Understanding the Chemical
Anyone who’s worked with dimethyl maleate knows that physical and chemical properties guide its real-world uses. As a clear liquid, it doesn’t raise many eyebrows in the lab. It boils at just over 197°C, melts around -4°C, and loves to dissolve in organic solvents like alcohols and ethers. The two ester groups open up a world of options for chemical reactions, since they react cleanly and predictably. In my experience, shelf storage rarely proves tricky as long as handlers respect flammability and keep it away from oxidizers. The clear labeling, typically showing “dimethyl (Z)-butenedioate” or “trans-Dimethyl maleate,” helps avoid mix-ups with similar-sounding substances like its cousin, dimethyl fumarate, which has sharply different biological effects.
Getting to Dimethyl Maleate
Chemists usually prepare dimethyl maleate with a straightforward strategy: start with maleic anhydride and add methanol using a little acid catalyst. Warming up the mix pushes the reaction forward, giving high yields with few side products. Some labs switch to direct esterification from maleic acid, but anhydride methods tend to be cleaner at scale. One thing that stands out from hands-on work is how sensitive this reaction can be to water. Even small amounts throw off yields, so careful drying and attention to process make a big difference. This feels typical for organic chemistry: success rides on preparation and patience.
Reactivity and Cultural Role in Chemistry
Dimethyl maleate doesn’t just sit on the shelf. Its double bond and ester groups serve as “handles” for a wide menu of reactions. In synthesis, it’s like a Swiss army knife—ready for Michael additions, Diels-Alder cycloadditions, transesterification, or reductions. Nobody in polymer chemistry forgets the crosslinking power of maleic-based esters. Years spent trying to tweak acrylic resins or latex systems taught me that a small amount of dimethyl maleate can tune flexibility, adhesion, and even aging resistance. In the world of plant protection and agrochemicals, it slips into the picture as an intermediate for active compounds where careful control of stereochemistry spells the difference between a useful additive and an unwelcome impurity.
The Bigger Picture: Names and Labels
Dimethyl maleate answers to many names, a point that’s led to more than one mis-shipment in busy warehouses. On bottles it might be “trans-Butenedioic acid dimethyl ester,” “maleic acid dimethyl ester,” or the formal IUPAC tag, “dimethyl (Z)-butenedioate.” Chemists use these interchangeably in discussion, but paperwork and regulations demand strict care to avoid confusion with related compounds—especially dimethyl fumarate, whose toxicity profile lands it in a very different risk zone. Standard labeling, globe-style pictograms, and hazard statements keep everyone on the same page, saving labs and logistics chains from expensive mistakes.
Safe Handling: More Than Just a Checklist
Having spent long hours around open drums and glassware, it’s clear that comfort with chemicals never breeds carelessness. Dimethyl maleate brings moderate acute toxicity—skin contact can sting, vapors irritate eyes and lungs, even at levels that seem mild. Inhaling vapors shouldn’t happen, but labs without exhaust fans or spill kits gamble with their safety. Once, a rushed transfer without gloves left a painful rash on a colleague’s arm—a reminder that “mild” hazards deserve respect. Standard gear—nitrile gloves, goggles, good ventilation—cuts risks to size. In an age where responsible waste and spill management anchor environmental credibility, training and clear protocols mean more than just paperwork; they keep teams healthy and regulators satisfied.
Where It Matters: Application Footprints
Most people never see dimethyl maleate, but plenty use products shaped by its chemistry. It shines brightest in the manufacture of adhesives, synthetic resins, and copolymers, which hold together everyday items—from packaging to automotive coatings. Any chemist building specialty esters or working on new crop protection compounds will cross paths with this chemical sooner or later. In plastics, small doses shape properties like UV stability, flexibility, and resistance to cracking. Pharmaceutical chemists see it less as a medicine and more as a scaffold or intermediate, while the fragrance and flavor sector taps it for specialty blends, always watching for regulatory barriers and sustainability concerns.
Innovation in Research: What’s Next
Research keeps picking at the limits of what dimethyl maleate can do. Teams have explored eco-friendlier catalysts and greener synthetic pathways, aiming to shrink waste streams and energy use. New modifications—like forming cyclic derivatives for advanced materials—open fronts in both polymer science and drug discovery. Thousands of academic citations reveal its utility as a building block or probe in synthesis, catalysis, and green chemistry. In practical terms, every graduate student seeking a reliable dienophile or functionalized ester has tinkered with dimethyl maleate at some point. My own brief foray into polyesters for advanced coatings owed a debt to its predictability and gentle reactivity.
Ongoing Concerns: Toxicity Under the Microscope
Conversations about safety can’t stop at gloves and goggles. Toxicology research gives a fuller picture. Acute exposure leads mostly to irritation, while long-term or high-dose studies highlight mild effects on the liver and kidneys in animal models. Occupational settings rarely bump against exposure limits, but the story changes in enclosed or poorly ventilated spaces. Environmental persistence stays low because this compound breaks down quickly, but accidental spills into waterways could still harm aquatic organisms. Regulators lean on studies showing no clear carcinogenicity signal in animal experiments, but routine health surveillance remains a smart move for workers in plants and research labs. Using it “safely” means more than knowing the numbers; it means building habits that shield teams from slips, splashes, and cumulative exposures.
Peering Into the Future: Where Does Dimethyl Maleate Go Next?
Dimethyl maleate might not grab headlines, but the drive toward greener chemistry means it’ll keep finding new uses. Pressure to reduce VOCs and improve life-cycle performance for plastics and coatings means more demand for well-understood, versatile intermediates. Novel applications in renewable polymers and specialty drug syntheses keep chemists searching for ways to build off its well-mapped molecular skeleton. Academic research pushes for catalysts and processes that use less energy and produce less waste. For industry, tight supply chains and the call for transparent safety data push suppliers to keep up with demand for quality and responsibility. For every new regulation on chemical safety or product labeling, the chemical industry sharpens its approach—less pollution, more transparency, better tracking of health outcomes for both workers and end-users. Dimethyl maleate’s story gets written alongside broader changes in chemistry: safer process design, life-cycle analysis, and the hunt for innovation that actually serves both people and the environment.
What is Dimethyl Maleate used for?
More Than a Chemical Name
A lot of folks have never heard of dimethyl maleate, but this colorless liquid shapes more parts of our daily lives than you’d guess. In the manufacturing world, this stuff brings flexibility and resilience to materials people use every day. I’ve run into its impact while working alongside teams in coatings and plastics—fields where what goes into a formula can make or break a final product’s durability.
How Factories Put It to Work
On the factory floor, companies rely on dimethyl maleate to produce resins and plastics that stay strong against wear and tear. Grab a piece of automotive trim, a garden tool handle, or some electrical insulation—likely, at some stage, dimethyl maleate played a part in making that item tough enough to handle sun, water, or abuse. People downstream in the supply chain, from molders to painters, count on it to improve flexibility without turning materials brittle. Unlike some additives, it seems to find a middle ground between making something strong and keeping it workable for manufacturers.
Vital Role in Production for Decades
Decades ago, makers of polyester resins and plasticizers started turning to dimethyl maleate for its straightforward reactivity. Resin formulations depend on it because it bonds well with other ingredients, making the end product more reliable. Over time, as products needed to last longer outdoors—on house siding, garden equipment, or car parts—this compound ended up in more recipes. I’ve seen colleagues in R&D labs debate new mixtures, always coming back to dimethyl maleate for cost and consistency. No one wants recalls over cracking plastic.
Impact on Sustainability and Safety
Environmental regulations push industries to clean up raw materials and figure out safer reactions. Dimethyl maleate stays in the game because it breaks down in ways that don’t stack up persistent toxins, unlike some legacy additives used decades ago. In terms of workplace safety, it brings its own hazards—as most chemicals do—but workers trained on handling procedures can stick with established safety practices and protect themselves. Plants that set up the right ventilation and PPE routines rarely report serious issues.
Tackling Supply Chain Challenges
During the supply crunches of recent years, companies relying on specialty chemicals felt the pinch—dimethyl maleate included. Price swings and shipment delays hit not just the big resin makers but the small shops ordering by the drum. Strong supplier relationships and local storage buffer some of the shock, but not all. From my view, industries built around just-in-time delivery need to think ahead, perhaps keeping a closer eye on alternate suppliers and local production options. Panic buying and overstocking won’t cut it; planning and partnership make a bigger difference in quality and continuity.
Room for Innovation
Beyond its familiar uses, some research teams experiment with dimethyl maleate in greener plastics, adhesives, and water treatment projects. I’ve heard from a few chemists who see promise in tweaking its chemistry for biodegradable plastics, already a hot topic as consumer pressure rises for less waste. Those who figure out more efficient reactions—using less energy and fewer byproducts—set themselves up for a competitive edge. For anyone thinking chemistry doesn’t touch everyday life, this is one of those crossover compounds that keeps proving otherwise.
What are the safety precautions when handling Dimethyl Maleate?
Understanding the Risks
Working with Dimethyl Maleate, a chemical often used in making resins and polymers, isn’t like dealing with regular household cleaners. It’s a liquid that can irritate your skin and eyes, and when inhaled, it can bother your lungs. I remember a former coworker who once neglected gloves, thinking he’d just pour a quick sample—he ended up with a nasty rash that lasted days. That experience hammered in just how quickly chemicals can get under your skin, literally and figuratively.
Proper Personal Protective Equipment
The first step for handling Dimethyl Maleate centers on solid personal protective gear. Rubber gloves matter here, backed up by goggles or even a face shield if you’re pouring or mixing. Lab coats protect arms from splashes, and closed shoes mean your feet won’t get exposed. This isn’t just about “following the rules”—it’s about getting home in the same condition you arrived in. If I’ve learned anything in the lab, it’s that one shortcut can bring headaches for weeks.
Good Ventilation Isn’t Optional
Dimethyl Maleate comes with fumes that can seriously irritate your nose and throat. I spent one afternoon working in a space where the exhaust fan stuttered, and by the end of it, everyone was coughing. That lesson stuck. Good air flow isn’t just a box on a safety checklist—it’s real protection. Use a fume hood or, in bigger spaces, make sure exhaust fans pull those vapors away quickly. Opening a window doesn’t cut it for chemicals like this.
Storage and Handling
Many people tuck away chemicals wherever there’s space. With Dimethyl Maleate, sloppy storage invites trouble. Keep the container sealed tight and store it in a cool, dry area away from sunlight and anything flammable. I’ve seen containers left half-open, making a room smell like a glue factory and raising the risk for anyone who comes by. Labels should face outward, clear and easy to read, and older chemicals rotated to the front.
Spill Management
Accidents catch you off guard. Leaks or spills from Dimethyl Maleate aren’t just messy—they pose health risks. Keep spill kits handy, including absorbent pads and neutralizing agents. Staff should know exactly where these kits sit and how to use them. We once had a new hire try to mop up a spill with paper towels; chemicals soaked through to the skin and caused an allergic reaction. That day, our team refresher about spill response made all the difference going forward.
Safe Disposal
Getting rid of leftover Dimethyl Maleate by dumping it down the drain jeopardizes water systems and people’s safety. Chemicals like this need special disposal, often as hazardous waste, collected by professionals. Written logbooks and hazard bins keep things organized—cutting corners leads to fines and contamination that can stretch for miles.
Training and Preparedness
Many safety problems stem from poor training. It pays to go over emergency procedures regularly. Make sure everyone on staff knows not just the location but the function of eyewash stations and safety showers. Practicing mock scenarios before something happens builds confidence and prevents panic. For me, practice drills turned dry instructions into muscle memory.
Toward a Safer Workspace
Respect for chemicals like Dimethyl Maleate doesn’t grow out of fear—it’s about common sense and looking out for each other. Focusing on protective gear, good ventilation, careful storage, spill readiness, safe disposal, and solid training forms a safety net. Real safety comes from habits formed every day, not just during inspections. I’ve learned that firsthand—preparation makes all the difference between a close call and a disaster.
What is the chemical formula of Dimethyl Maleate?
Chemistry in Daily Life: The Role of Dimethyl Maleate
Dimethyl maleate pops up more often than most folks imagine. The chemical formula, written as C6H8O4, reflects its structure: six carbon atoms, eight hydrogens, four oxygens. This isn’t some obscure substance locked up in a lab. You’ll find it in adhesives, resins, and certain coatings. Scientists appreciate its reactivity, and industry counts on its ability to cross-link and modify polymers, especially when there’s a need for flexibility or resistance to degradation. For those who like to tinker, that formula means each molecule has a backbone made up of carbons and a pair of ester groups—the same type of connection that gives fruits their smell and plastics their toughness.
The Facts about Formula and Structure
The numbers in C6H8O4 aren’t random. If you look at Dimethyl maleate, each ester group (COOCH3) comes off of a maleic acid core. Maleic acid itself carries some history, used since the 1800s in dyes and synthetic fibers. Chemists swapped hydrogens for methyl groups here, giving us dimethyl maleate—changing both the solubility and the performance in physical settings. The two methyl esters hang on either side, making the molecule less polar, easier to dissolve in organic solvents, and more suitable for industrial chemistry than basic maleic acid.
Why the Formula Matters for Safety and Manufacturing
Experience in a small manufacturing plant taught me the importance of the right formula. The difference between C6H8O4 and a similar-sounding compound could mean equipment corroding, workers suffering from skin contact, or batches that never set up properly. Knowing exactly what’s in the barrel saves time, money, and stress. For smaller producers, especially, reading the formula isn’t just for show. C6H8O4 tells an operator what kind of gloves to wear, how much ventilation a space needs, and whether a batch can be trucked alongside food products or not (hint: it shouldn’t).
Environmental and Health Watch
Handling Dimethyl maleate raises some concerns. The chemical formula points toward hazards—esters can act as irritants to skin and eyes. Factories near waterways monitor runoff, because compounds built on the maleic acid skeleton may linger and affect fish or plant life. The more people understand what C6H8O4 means, the better choices they make about treatment and disposal. Regulatory agencies set exposure limits for workplace air. Good labeling and education protect workers, and a clear chemical formula starts a conversation about risk and best handling practices.
Paths Forward in Research and Manufacturing
Teams in specialty chemical development look for ways to use dimethyl maleate to make products more sustainable. Technicians ask if there’s a plant-based version, or if the process uses less energy under the same C6H8O4 formula. Keeping close to the basics—the molecular weight, the chemical bonds—lets manufacturers tweak recipes for safer, greener goods. In academic labs, understanding the functional groups and layout of dimethyl maleate lets researchers tackle questions about new plastics, coatings, or biodegradable alternatives.
The Value of Knowing Your Chemistry
Whether you work in a factory, a research institute, or you’re just curious about what goes into common goods, it pays to know why C6H8O4 means dimethyl maleate. That little formula acts as a key. It unlocks a better understanding of how chemicals show up in jobs, products, and the environment—and leaves everyone better prepared to deal with the realities of modern manufacturing.
How should Dimethyl Maleate be stored?
Why How You Store Chemicals Like Dimethyl Maleate Actually Matters
Working in a lab taught me early that not all chemicals behave themselves. It might look like a harmless clear liquid, but dimethyl maleate reacts if given a chance. Handling and storage go beyond ticking boxes — you keep people safe, prevent costly spills, and protect your data. Messing up with storage isn’t something you just clean up; sometimes it means replacing equipment or people getting hurt. No complicated protocols solve all problems, but simple, consistent storage routines make a huge difference.
Know the Trouble Spots
Dimethyl maleate has some very definite preferences. It prefers cool, dry places. Moisture means hydrolysis, leading straight to messy byproducts. In ordinary warehouses, humidity climbs, especially in summer. If you’ve ever opened up a chemical drum after a humid day, you know how fast rust and mold spread. High temperatures do more than just spoil the chemical; pressure might build up inside containers, seals could fail, and then vapors could escape. Dimethyl maleate vapors will irritate your nose and eyes in no time. Further exposure brings headaches and worse, so ventilation matters.
Containers Do the Heavy Lifting
Metal plays well here, but only if the drum or canister has a coating resistant to corrosion by esters. Label all containers thoroughly. Inconsistent labeling can lead to using the product incorrectly or, even worse, mixing it up with something much more dangerous. Take it from anyone who’s spent an afternoon sorting through unlabeled jugs — frustration is the least of your worries.
Never use food containers for storage. Cross-contamination horror stories usually start with something as simple as a repurposed soda bottle. Dimethyl maleate gets absorbed by skin and harms if ingested. Dedicated, clearly marked storage solves this quickly.
Shelving and Segregation Prevents Accidents
Keep this chemical off the floor and far from acids, bases, and oxidizers. In one mishap, dimethyl maleate caught up with a bleach spill in the same cabinet. Corrosive fumes filled the room. Hazmat cleanup and hours lost — all because someone shelved them together. Simple organizers help here. Assign shelves by chemical families: esters with esters, acids alone, oxidizers in a locked cabinet. No fancy equipment required; even color tape makes a difference for quick visual cues.
Don’t Forget Real Ventilation
I once worked in a storage room where “ventilated” just meant a hole punched in the wall. That didn’t cut it, especially with volatile liquids. Mechanical exhaust fans actually move the fumes out, not just stir them around. Every time a drum gets opened, invisible vapor escapes, and slow buildup can set off alarms or, worse, affect people’s breathing. OSHA facts back this up: good ventilation cuts incident rates where volatile chemicals get stored.
Routine Checks and Common Sense
Do visual checks regularly: look for swelling containers, leaks, or strange odors. A sticky drum means the seal might have failed. Every professional organization — from OSHA to the European Chemicals Agency — points to leaky drums as a major source of accidents. Instead of formal audits, walk through each week and make notes. Ask employees what they see and smell; first-hand experience often spots trouble before any checklist does.
Small Changes Keep You in Control
Sometimes, the best solution means retraining staff. The best chemical storage process in the world gets undermined by a single person treating it as routine. Bring everyone together for annual hands-on sessions. Explain past incidents and what simple changes could prevent a repeat. People quickly buy in when they know their health and job security depend on it.
Keep storage simple, visible, and ventilated. Chemical safety seems intimidating, but it depends much more on daily habits than complicated procedures. That’s true for something as technical-sounding as dimethyl maleate — and just about anything else that lives in the back room.
Is Dimethyl Maleate hazardous to health or the environment?
Everyday Exposure and Worker Risks
In chemical plants and laboratories, dimethyl maleate shows up often as a reactive ingredient. This clear, oily liquid has a sharp odour and mainly acts as an intermediate for making plastics, resins, and pesticides. Everyone involved with its handling—from chemical technicians to industrial cleaners—needs to understand its risks. Even quick, routine tasks can expose workers to its vapours or accidental spills.
My time in a polymer manufacturing facility proved the importance of strong safety habits. Colleagues there never skipped gloves or goggles. Direct skin contact with dimethyl maleate can cause redness or burns. Breathing in the fumes brings headaches, dizziness, or throat irritation. High concentrations have triggered lung problems or nausea for unlucky workers. Chemical Safety Sheets place strict limits on safe airborne levels. Loading dock areas always kept windows open, and local exhaust fans ran year-round.
Lingering Effects on Water and Air
This chemical doesn’t just disappear after use. Disposal drives the bigger problem. Dimethyl maleate dissolves readily in water and leaks into soils or drains if storage tanks fail or drums get knocked over. Rain brings runoff into rivers, affecting aquatic life. Fish and aquatic insects, exposed at even low doses, sometimes develop abnormalities or cannot reproduce properly. At larger scales, this upsets the food web far from the original factory.
Airborne vapours drift beyond the fence line. Odours reach nearby homes, making residents anxious. Short-term exposures may bother people with asthma or allergies. Local authorities have flagged emissions near old manufacturing sites and pushed for tighter controls. Simple upgrades, such as covered containers and better venting, help slow the spread.
Understanding the Human Impact
Beyond the immediate plant fence, dimethyl maleate’s risks expand to ordinary people. Trucks carry it across highways and railroads. Spills on the road put emergency crews at risk, especially if wind carries vapours before cleanup can begin. Local hospitals may see a spike in breathing complaints after transport accidents. This links to public health regulations that restrict transport times or require special training.
Households almost never encounter this chemical directly. Yet, products that use it in manufacturing—like paints or certain adhesives—can emit tiny traces after opening. Most brands run their own safety tests, and government regulators monitor limits, so household risks generally remain low. Still, residents living near manufacturing centres face higher risks from plant accidents or long-term releases. Listening sessions between businesses and neighbours sometimes uncover gaps in communication and trust.
Paths Toward Safer Handling
Some companies have started replacing dimethyl maleate with safer alternatives. Switches take time, especially when changing recipes for established products. In the meantime, regular safety drills, spill kits on hand, and updates to storage facilities keep dangers in check. Training makes the biggest difference—new staff who understand the risks rarely make the same mistakes as those kept in the dark.
Government agencies keep updating guidelines as research uncovers more about chronic effects and ecological harm. Companies adopting new leak-prevention technologies have seen a drop in workplace accidents and environmental releases. The science grows, but the drive to protect people and nature never stops for those closest to the job. My experience tells me no rulebook or technology really works without a culture of care and clear communication in the workplace.
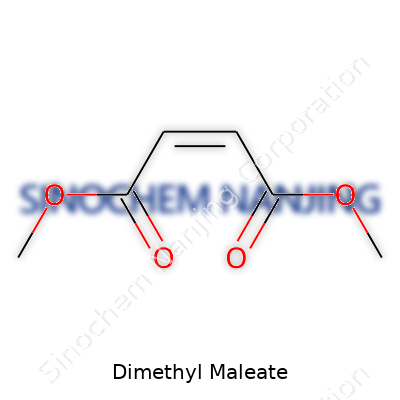
| Names | |
| Preferred IUPAC name | Dimethyl (Z)-but-2-enedioate |
| Other names |
Dimethyl cis-butenedioate Maleic acid dimethyl ester trans-Butenedioic acid dimethyl ester Dimethyl (Z)-butenedioate cis-Dimethyl maleate |
| Pronunciation | /daɪˈmiːθɪl məˈleɪət/ |
| Identifiers | |
| CAS Number | 624-48-6 |
| Beilstein Reference | 1206666 |
| ChEBI | CHEBI:64043 |
| ChEMBL | CHEMBL20297 |
| ChemSpider | 54607 |
| DrugBank | DB13925 |
| ECHA InfoCard | 03d9e3f1-353c-4c56-97a8-1b0acf74a53c |
| EC Number | 203-653-1 |
| Gmelin Reference | 82241 |
| KEGG | C01142 |
| MeSH | D008950 |
| PubChem CID | 10140 |
| RTECS number | PM3675000 |
| UNII | 2T1UN55V7X |
| UN number | UN1162 |
| CompTox Dashboard (EPA) | DMTCGXJMVYUVNS-UHFFFAOYSA-N |
| Properties | |
| Chemical formula | C6H8O4 |
| Molar mass | 144.17 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | Fruity |
| Density | 1.084 g/mL at 25 °C |
| Solubility in water | slightly soluble |
| log P | 0.87 |
| Vapor pressure | 0.26 mmHg (25 °C) |
| Acidity (pKa) | 6.1 |
| Basicity (pKb) | 1.94 |
| Magnetic susceptibility (χ) | -9.93·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.415 |
| Viscosity | 1.619 mPa·s (20 °C) |
| Dipole moment | 1.53 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 170.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -452.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3332 kJ/mol |
| Pharmacology | |
| ATC code | D07XA01 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P305+P351+P338, P333+P313, P337+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 2-2-2-0 |
| Flash point | 83 °C (181 °F; 356 K) |
| Autoignition temperature | 355 °C |
| Explosive limits | Explosive limits: 1.1–8.1% (in air) |
| Lethal dose or concentration | LD50 oral rat 2870 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 1,560 mg/kg |
| NIOSH | NIOSH: ON8050000 |
| PEL (Permissible) | PEL: 5 ppm |
| REL (Recommended) | 6 mg/m³ |
| IDLH (Immediate danger) | 800 ppm |
| Related compounds | |
| Related compounds |
Maleic anhydride Dimethyl fumarate Maleic acid Diethyl maleate |

