Understanding Dimethyl 1,3-Propylene Glycol: Past, Present, and Future Applications
A Look Back at Its Emergence
Long before chemists began using complex computational simulations, the story of dimethyl 1,3-propylene glycol started in crowded laboratories where people were as likely to break new ground as they were to spill reagents. Early synthetic organic chemists showed real curiosity toward glycol derivatives. As the chemical industry grew throughout the 20th century, interest in this family of compounds deepened. Processes developed for laundry detergents and plastics found new use. Advances in distillation and purification meant that by the 1970s, dimethyl 1,3-propylene glycol emerged from relative obscurity into mainstream production lines. These shifts marked new possibilities for manufacturers exploring polymers, resins, and lubricants. Today, the substance has become somewhat of a quiet workhorse, tucked behind more headline-grabbing chemicals, but its role in industry stands firm.
What Makes Dimethyl 1,3-Propylene Glycol Stand Out?
Working with dimethyl 1,3-propylene glycol, I’ve come to appreciate its character. It doesn’t have that sharp, biting odor some glycols announce themselves with. In the lab, its clear, syrupy appearance and viscous flow reveal just how well it plays with solvents and other organics. Its chemical formula, C5H12O3, and structured presence as (CH3O)2C3H6O, places it in a sweet spot: low enough volatility for stable storage but just reactive enough to mesh with diverse applications. Melting and boiling points give manufacturers flexibility in processing. Solubility in water and alcohols provides good options for blending or dissolving it as needed. That balance—neither too reactive nor too inert—makes it a favorite for resin chemists and coating engineers alike.
Technical Details and Labeling Practices
Chemists and plant operators alike benefit from clarity in how a substance like this gets labeled and specified. Working with bulk supplies, I’ve seen confusion over similar-sounding names create real-world safety headaches. Industry calls it by several synonymous names—dimethyl propane-1,3-diol or dimethyl trimethylene glycol being the most common. Regulatory and safety data sheets typically describe its hazard profile, flammability, storage needs, and personal protective equipment requirements. Batch purity, moisture content, and assay limits matter most to downstream users, especially those building something that ends up in biomedical coatings or specialty adhesives. Accurate technical specs don’t just help engineers; they also help protect workers and support regulatory compliance—which, in the wake of tightening chemical management rules, becomes ever more important.
Manufacturing and Synthesis: Hands-On Know-How
Making dimethyl 1,3-propylene glycol calls for both method and patience. In practice, most factories rely on multi-stage synthesis, often harnessing the reaction of epichlorohydrin with methanol using acid or base catalysts. Oxygen exclusion stands as a must for avoiding unwanted byproducts. In my experience, purification in this context isn’t just about yield—it’s about safety and reliability downstream. Distillation columns must work efficiently to minimize trace contaminants, especially if suppliers aim for pharmaceutical or food-grade purity. Operations teams routinely perform monitoring and adjustment to keep reactions running smoothly and materials within spec.
Chemical Modifications and Reaction Profile
Dimethyl 1,3-propylene glycol’s backbone lets chemists take it in multiple directions. In my own lab work, I saw how the hydroxyl groups open doors to ether or ester formation. Epoxidation, dehydration, or cross-linking via polycondensation can turn a base molecule into something destined for polyurethane foams or heat-resistant coatings. Direct modifications, such as alkylation, create specialty intermediates for surfactant and lubricant production. These transformations tend to be clean and well understood, which is probably why industry adoption remains strong. Choices in catalysts, reaction conditions, or downstream handling all weigh heavily on practical outcomes and economic efficiency.
Safety and the Changing Landscape of Industrial Practices
Anybody handling dimethyl 1,3-propylene glycol outside the safe cocoon of a fume hood knows respect for its properties comes first. Exposure risks in highly ventilated labs drop well below those in production settings crammed with tanks and piping. Safety records show a fairly mild toxicity profile, but mishandling concentrated vapors or skin contact brings enough risk that gloves and goggles become more than a formality. The industry has moved toward closed systems and better training, which sharply reduces the risk of occupational asthma or dermatitis. Recent regulations stress risk assessment and emergency readiness across the supply chain, forcing companies to stay sharp, continuously update safety protocols, and upgrade leak detection or air-quality systems. Worker training on chemical handling remains a primary defense against avoidable accidents.
Markets and End Uses
Once you get outside the lab, the question becomes: where does dimethyl 1,3-propylene glycol actually show up? I've seen it blended into high-performance coatings, especially those used to provide weather-resistance or chemical durability for bridges, aircraft, or industrial floors. Its properties also fit it well into lubricants, offering viscosity without gumming up at low temperatures. As a building block for specialty polyurethanes, it contributes to cushions, insulation, and sometimes even biodegradable plastics. The pharmaceutical sector taps it for its solvent potential in some formulations. These days, most R&D efforts circle around sustainability—how to make the extraction, synthesis, and eventual disposal friendlier to the planet.
Research, Development, and Toxicity Studies
Decades of research paint a reassuring picture: dimethyl 1,3-propylene glycol shows low acute toxicity in mammals, both in oral and skin exposure routes. Long-term data on chronic exposure stay in short supply, spurring further interest from toxicologists. My time in collaborative studies highlighted its relatively slow breakdown in soil and water, raising longer-term environmental persistence questions. Regulators and health experts want more comprehensive toxicity data, especially with wide-ranging use in consumer and industrial products. Over the years, improvements in analytical techniques have made it much easier to detect trace residues and degradation products, feeding back critical information to plant engineers and environmental managers alike.
Looking Ahead: Innovation and Responsibility
Years of working with chemicals like dimethyl 1,3-propylene glycol convince me that responsibility must sit at the center of every discussion about its future. New regulatory frameworks ask tough questions about sourcing, emissions, and waste handling. With global pressure mounting for greener, less energy-intensive production cycles, researchers look at renewable raw materials or enzymatic routes to trim down carbon footprints. Materials scientists continue searching for advanced formulations that stretch physical performance without compromising worker safety. Larger companies face public and investor scrutiny over supply chain transparency; small specialty firms wrestle with the cost of compliance and upgrades. That push for sustainable chemistry turns every step—from molecule design to product launch—into a test of both scientific creativity and social responsibility. In the years to come, progress will depend not just on technical breakthroughs, but also on a shared commitment to health, safety, and long-term environmental care.
What is Dimethyl 1,3-Propylene Glycol used for?
The Quiet Helper in Manufacturing
Dimethyl 1,3-propylene glycol slips quietly into the backbone of many important products. The average person doesn’t walk into a store and ask where to buy it, but chances are they use something related to it daily. In the chemical world, this compound helps craft the things people actually recognize—coatings, inks, flexible plastics, cleaning sprays, and more.
Why Manufacturers Care
Chemicals like this one shape how paints go on walls, how plastics bend and last, and even the way scent lingers in a cleaning product. Factories need a workhorse that mixes well with both water and oils, makes other ingredients flow smoothly, and doesn’t break down during tough processing. Dimethyl 1,3-propylene glycol checks those boxes. In paint production, it keeps pigments even, so colors don’t streak or clump. Polyurethane makers turn to it for its role as a building block: it gives flooring and foam cushions the bounce and strength customers want.
Printing companies count on dimethyl 1,3-propylene glycol for its solvent action. It helps disperse ink evenly on paper and boosts drying time, so freshly printed flyers or books don’t stick together. Even the rubbery feel in some sealants and adhesives often owes something to it.
Personal Care and Cleaners
Most people never check the fine print on a shampoo or a deodorant stick, but here, too, this glycol finds a job. Since it combines easily with different ingredients, manufacturers use it to stabilize perfumes, lotions, and sprays. Imagine a fragrance that hangs in the air for hours—part of that comes from the way this compound tethers volatile scent molecules. In soaps or cleaning sprays, its presence means active ingredients dissolve better, making them more effective on stubborn dirt.
Safety and Environmental Questions
With every new ingredient, concerns about health and environmental safety come up—rightly so. Dimethyl 1,3-propylene glycol rates low in toxicity for humans, according to research gathered by agencies like the European Chemicals Agency (ECHA). Factories still follow tight guidelines to limit exposure in production halls. As for the planet, this glycol breaks down fairly easily in the environment. Still, wastewater systems aren’t perfect, and manufacturers face pressure to tighten controls and cut down on any runoff.
Looking for Sustainable Choices
Industries can’t afford to walk away from useful glycols overnight, but the push is on for greener options. Researchers aim to tweak the chemical structure so it comes from renewable sources, or to design alternatives that keep performance strong without making disposal harder. That way, the products people depend on—from paints to deodorant—can keep improving, but with an eye on long-term safety.
Why All This Detail Matters
For consumers, staying informed about what ends up in daily essentials helps drive better buying decisions. People ask more questions than ever about their purchases, and that keeps companies honest. For those of us who want paints that roll on smooth or printed books that don’t smudge, it’s worth caring about the chemistry behind the scenes. Dimethyl 1,3-propylene glycol plays its part—a reminder that familiar comforts often rely on not-so-familiar ingredients.
Is Dimethyl 1,3-Propylene Glycol safe for use in cosmetics?
Walk down a drugstore aisle, flip over a moisturizer, and there’s a good chance you spot an ingredient like Dimethyl 1,3-Propylene Glycol. It hides in plain sight on many ingredient lists. Product designers often seek out this compound because it keeps textures silky, helps other ingredients blend, and prevents formulas from drying out too quickly. Still, safety questions pop up every time a technical-sounding name finds its way into the stuff people put on their skin. Most folks ask the same thing: is this stuff safe?
What Is Dimethyl 1,3-Propylene Glycol, Anyway?
It’s a mouthful, but it’s basically a synthetic alcohol. Chemists blend it into things like creams, lotions, and makeup to create a smooth experience. Picture a lotion that spreads nicely over your hands, sends a little cooling tingle, or doesn’t feel sticky — Dimethyl 1,3-Propylene Glycol could be behind that. It also plays well with preservatives, surfactants, and other cosmetic staples. It won’t naturally turn up in a field or river, but it’s not new to the formulation world either.
Safety Checks and Oversight
Back in the early 2000s, safety panels like the Cosmetic Ingredient Review (CIR) dug into all sorts of glycols and their cousins. Health regulators in the US and Europe keep a tight eye on synthetic ingredients. As part of the process, researchers tested Dimethyl 1,3-Propylene Glycol for things like skin irritation, allergic reaction, toxicity, and the potential for build-up in the body. They checked animal studies, compared different glycols, and looked at how these substances break down over time.
Those findings show most folks tolerate this ingredient well when it is used on skin or hair, especially at levels normal in retail products. Major regulatory agencies, including the US Food and Drug Administration and the European Commission, group it with similar glycols and place fewer restrictions on its use, as long as it doesn’t go near sensitive membranes or internal use. I’ve spoken with cosmetic chemists who point to its long safety record in rinse-off and leave-on products.
Potential Concerns and Sensitive Skin
Some people notice redness or itching after using products with these types of solvents. But scientists tend to trace most of those reactions to heavy use or mixing with other strong chemicals. Dermatologists routinely see more problems from fragrances or preservatives than from ingredients like Dimethyl 1,3-Propylene Glycol. Personal experience backs that up for me — as someone with touchy skin, reactions show up more with harsh soaps or powerful exfoliants.
There’s always a need to stay alert for real allergies. Patch testing helps. If someone breaks out after trying a new product, a dermatologist can help figure out the real cause. Careful product selection always matters, especially for folks prone to eczema or rosacea. But for millions, this glycol forms part of their daily skincare or hair care routine without trouble.
Better Products, Smarter Use
Cosmetics brands know consumers watch ingredient lists more closely these days. They tweak formulas, remove known irritants, and keep testing new blends for gentleness. Brands that want loyal customers run careful safety checks and act quickly if any reports suggest a product isn’t as gentle as it should be. More transparency would help confused shoppers feel better about ingredient names they can barely pronounce.
Reading up, talking with health pros, and listening to your own skin will always trump blind trust or blind suspicion. For the vast majority, Dimethyl 1,3-Propylene Glycol still plays a background role, making products more pleasant to use — and so far, science gives it a pretty clean bill of health.
What are the physical and chemical properties of Dimethyl 1,3-Propylene Glycol?
The Simple Science of Structure
Dimethyl 1,3-propylene glycol carries the scientific label, but you might meet it under other names in the lab. This molecule holds two methyl groups and an odd little backbone of three carbon atoms connected by oxygen atoms—making it a diol. That means it comes loaded with two alcohol groups, boosting its ability to mix with water and a stack of common organic solvents. In plain language, it isn’t fussy about its company, sliding easily from water to alcohols like ethanol or even acetone. Unlike big bulky molecules, it doesn't throw off a powerful smell or leave behind a streak of residue after evaporation. Liquid at room temperature, colorless, and practically odorless, this glycol brings a friendly face to those who mix chemicals all day.
Physical Traits You Can Trust
Anyone who’s worked in a lab knows certain chemicals demand caution. Dimethyl 1,3-propylene glycol doesn’t fight back. It feels oily, but slips faster between your fingers than mineral oil. Its boiling point rises north of 200°C, so you can cook it up in reactions without it skipping off as vapor too early. It loves water, blending in drop by drop, and laughs in the face of freezing points that trip up rival glycols—dipping below -30°C before turning solid. Viscosity sits somewhere in the middle—thin enough to pour, thick enough to avoid splashing all over your bench. Its transparency isn’t just for show; this quality helps in quality control and analytical work.
Chemical Personality: Reactive, But Predictable
The two alcohol groups act as the main handle for chemical work. Reactions involving esters, ethers, or even plastic production know how to take advantage. The methyl ends keep the molecule from getting too sticky with hydrogen bonds, so it avoids clumping and snags less moisture from the air. This stability means you won’t see it decompose or change much unless you push it with serious energy or strong acids and bases. No surprise explosions, no runaway color changes, no mysterious odors. Safety data back that up—skin contact or a quick sniff doesn’t trigger panic, though you shouldn’t sip it for fun either. Most regulations treat it as a low-toxicity compound compared with harsher glycols or alcohols.
The Why Behind Its Importance
Anybody in manufacturing, pharmaceuticals, or polymer chemistry has likely come across dimethyl 1,3-propylene glycol. Because it stays liquid, resists freezing solid, and moves easily between water and organics, it gives engineers and scientists a flexible tool for mixing, reacting, and stabilizing more complex products. In the past, I’ve relied on it as a balancing act in solubility puzzles while designing drug delivery solutions. It doesn’t just solve technical headaches—it keeps costs in check due to stability and easy storage.
Pushing Toward Safer, Smarter Use
Placing a chemical with a lower toxicity profile at the center of everyday applications means workers face fewer risks, and environmental managers worry less about disposal. Still, no chemical is perfect. Vigilance over exposure limits and spill control remains a must. Developing more transparent labeling, and ensuring better worker training, should cut down on accidental misuse. Simple changes, like improved ventilation or better gloves, go a long way for anyone working with glycols, keeping both people and product safe without slowing down science or industry.
How should Dimethyl 1,3-Propylene Glycol be stored and handled?
Understanding the Hazards
Dimethyl 1,3-propylene glycol doesn’t show up in everyday conversations, but it often turns up in manufacturing labs, coatings shops, and synthetic processes. Anyone working with it knows that the real test comes not just from theoretical safety data but from hands-on experience and awareness. The dangers don’t always announce themselves with big warning signs. Instead, leaks, vapor exposure, or fire risks build up quietly if the basics go ignored. From my time around industrial chemicals, I know ignoring storage guidelines leads to hefty cleanup days, hospital visits, and, occasionally, lost jobs. That stuff lingers far longer than the faint, sweet smell it sometimes carries.
Key Storage Measures for Dimethyl 1,3-Propylene Glycol
The chemical sits happiest in rooms where air moves steadily. It doesn't take fancy engineering to achieve good ventilation—cracked windows and a sturdy fan block accidental inhalation. Many ignore this until a spill fills the air with headache-inducing fumes. Storing it in a dry, cool place halts unnecessary pressure buildup inside drums or bottles. I once witnessed an entire batch ruined because storage temperatures climbed over 30°C, causing cracked seals and a mess that ate away three workdays. Keeping containers sealed and away from heat sources or open flames isn’t up for debate, especially since it’s flammable under the right conditions. Flammable liquids and heat play poorly together, every time.
Safe Handling Practices
I can’t stress enough: personal protective equipment doesn’t earn respect just by hanging on a hook. Gloves that fit properly, splash goggles, and long sleeves stop the chemical from sneaking onto skin or into eyes. Chemical burns and irritation never seem like a big deal until the pain sets in hours later. Washing hands after use, even after short jobs, prevents accidental ingestion or skin absorption. I’ve watched experienced techs, thinking they had it all under control, rub their eyes with unwashed hands—an instant ticket for a medical evaluation.
Spill control speaks to a shop’s discipline. Proper absorbents and immediate cleanup keep slips and chemical exposure from snowballing. In small workspaces, clear labeling of containers makes a real difference. I’ve seen containers swapped, contents mixed, and reactions occur—an expensive reminder that good labels are just as important as strong locks.
Training and Emergency Prep
Reading the safety data sheet counts for a lot, but hands-on drills and safety talks drive the message home. Workers who practice what to do in case of splashes or fires keep panic from taking over in the real moment. The right eyewash and shower stations in arm’s reach matter—not across the warehouse where seconds can mean lasting injury. My experience has taught me that the best teams talk openly about near misses. They adjust workflows to close gaps, and that’s a practice everyone benefits from.
Looking Ahead: Smarter Solutions
Digital inventory systems, smart sensors to track temperatures, and routine equipment checks build a culture where mistakes get caught early. Some companies are taking on green chemistry approaches to limit reliance on hazardous solvents altogether. Responsibility stretches well beyond the storage room door—it covers transport, disposal, and community safety. People who take stewardship seriously reduce risk for everyone, not just themselves.
Proactive steps, informed by real-world experience and guided by sound science, keep harm at bay and workdays predictable. That earns trust from workers, managers, and everyone the business touches. Chemical safety isn't fancy, but it remains essential at every step.
Is Dimethyl 1,3-Propylene Glycol environmentally friendly or biodegradable?
Real Impact Behind Chemical Labels
Dimethyl 1,3-propylene glycol might sound like one of those obscure compounds only specialists care about, but its story weaves into everyday products. It crops up in manufacturing, resins, paints, and even personal care. With concern for planet health growing, people want straightforward answers about environmental safety and breakdown in nature. In my experience reading chemical safety data and talking to people in the chemistry field, curiosity often turns into worry unless you cut through technical jargon.
The big question many folks bring up is: Does this stuff hurt the environment, and will it break down on its own or just hang around stubbornly?
Understanding Biodegradability
The word “biodegradable” sparks hope for eco-conscious shoppers and industries alike. If something breaks down in water and soil fast enough, staying harmless, it earns environmental brownie points. According to studies such as those from the European Chemicals Agency, dimethyl 1,3-propylene glycol proves pretty persistent. This isn’t the good kind of persistent either. Standard biodegradation tests show only partial breakdown over 28 days. That means if this compound gets dumped or leached into the environment, it tends to stick around longer than naturally occurring substances or truly biodegradable chemicals like ethanol or certain plant-derived ingredients.
I remember the disappointment reading that report, expecting better from a compound used in so many “clean” formulations. Instead of vanishing quietly, traces will linger in bodies of water or soil unless proper wastewater treatment steps in to help break it down. Toxicity testing shows relatively low risk for fish, but aquatic life doesn’t always get the last word—persistent waste builds up over time, making long-term effects far harder to predict.
Environmental Friendliness Beyond a Label
Marketing doesn’t always paint the whole picture. Manufacturers tout low volatility and low acute toxicity as marks of safety. While that’s good for worker safety and air quality indoors, it does not answer what happens ‘downstream.’ With enough run-off or poor disposal habits, even mild chemicals can pile up in riverbeds or drainage canals. People can’t just rely on product packaging; checking sourcing, producer disclosures, and independent reviews helps fill in the gaps.
I worked with a small sustainability group to review chemicals used in cosmetic factories. Even though some suppliers called their formulas “green,” a closer look at the ingredients like dimethyl 1,3-propylene glycol showed the term sometimes meant “less dangerous,” not “environmentally healing.” Recyclers and treatment facilities have to use extra filtration or special biological treatments to handle such persistent compounds, raising operational costs and complexity.
Looking Toward Better Solutions
To really solve this, chemical manufacturers and regulators should push for alternatives proven to break down quickly and safely. Researchers suggest biosourced glycols or new plant-based polymer blends may step into this role with less environmental baggage. Companies could improve labeling transparency, calling out persistence and real life-cycle impact instead of hiding behind technical terms. Routine and independent third-party testing keeps everyone honest, letting buyers and local officials see exactly what enters their water and soil.
For industries still using dimethyl 1,3-propylene glycol, better waste management and on-site treatment will reduce accidental emissions. This means setting higher standards and rewarding those firms investing in green chemistry. The conversation shouldn’t stop at risk; pushing for genuine biodegradables built from scratch helps build trust and gives both factories and consumers the peace of mind they deserve.
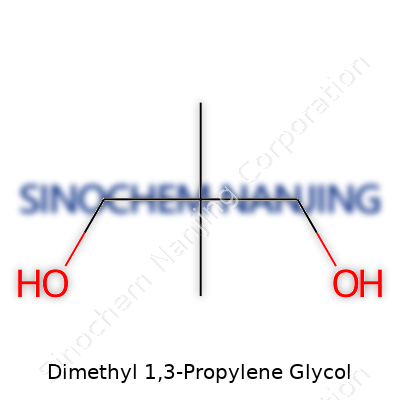
| Names | |
| Preferred IUPAC name | Dimethyl 3-hydroxypropane-1,1-dioate |
| Other names |
1,3-Propanediol dimethyl ether Dimethyl propyleneglycol 1,3-Dimethoxypropane Propanediol, 1,3-dimethyl ether Dimethyl-1,3-propanediol |
| Pronunciation | /daɪˈmiːθɪl wʌn θriː ˈprəʊpɪliːn ˈɡlaɪkɒl/ |
| Identifiers | |
| CAS Number | 100-79-8 |
| Beilstein Reference | 2201259 |
| ChEBI | CHEBI:132754 |
| ChEMBL | CHEMBL15829 |
| ChemSpider | 20915 |
| DrugBank | **DB14118** |
| ECHA InfoCard | ECHA InfoCard: 03-2119457554-30-0000 |
| EC Number | 203-498-7 |
| Gmelin Reference | 86297 |
| KEGG | C19407 |
| MeSH | D017174 |
| PubChem CID | 10962 |
| RTECS number | HU5950000 |
| UNII | ZR43714F9T |
| UN number | UN1993 |
| Properties | |
| Chemical formula | C7H16O4 |
| Molar mass | 134.18 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Mild, characteristic |
| Density | 1.02 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.8 |
| Vapor pressure | 0.01 mmHg (20°C) |
| Acidity (pKa) | 14.5 |
| Basicity (pKb) | 5.10 |
| Magnetic susceptibility (χ) | -54.0e-6 cm³/mol |
| Refractive index (nD) | 1.422 |
| Viscosity | 13.3 mPa·s (25 °C) |
| Dipole moment | 2.67 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 416.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -642.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4205.5 kJ/mol |
| Pharmacology | |
| ATC code | No ATC code |
| Hazards | |
| Main hazards | Causes serious eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319 |
| Precautionary statements | P210, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 154°C |
| Autoignition temperature | Autoignition temperature: 371°C (700°F) |
| Lethal dose or concentration | LD50 (Oral, Rat): 3750 mg/kg |
| LD50 (median dose) | LD50 (median dose): 5,040 mg/kg (oral, rat) |
| NIOSH | PU8750000 |
| PEL (Permissible) | Not Established |
| REL (Recommended) | 10 mg/m3 |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Dimethyl 1,2-propylene glycol Dimethyl 1,4-butylene glycol Ethylene glycol dimethyl ether Diethylene glycol dimethyl ether Propylene glycol |

