Looking at Diisopropylamine: More Than Just a Stepping Stone in Industry
Tracing the Story from the Beginning
Chemists in the early 20th century realized they could shape the world by manipulating simple building blocks, and diisopropylamine stood out for its versatility. It didn’t show up as a breakthrough in any one place, but researchers working with amines saw its value as a secondary amine that could open doors to much wider fields, from pharmaceuticals to rubber manufacturing. In my own time working with chemical synthesis in academia, diisopropylamine would crop up as a reliable partner in countless reactions, carrying small but crucial changes that bigger compounds couldn’t pull off as cleanly. This steady workhorse quality means that, even as modern labs chase after new and flashy catalysts, the tried-and-true ones like this tend to handle the backbone jobs without much fanfare.
A Hands-On Look at the Product
Diisopropylamine is not an obscure specialty chemical. It's a clear, colorless liquid with a characteristic ammonia-like smell that will wake up the senses if a flask cap isn’t secured. The formula (C6H15N) doesn’t tell the whole story—once you catch the stuff on your gloves, you'll rush to a fume hood or the sink because its odor lingers and irritates pretty quickly. Physical storage takes care to avoid its volatility, not unlike storing solvents that could escape if they're not sealed properly. That blend of convenience and caution stretches through every lab and plant floor it enters.
Physical and Chemical Behavior in the Real World
From the practical side, diisopropylamine boils around 84 °C, which means it vaporizes long before water and throws up vapors that could catch a spark faster than many expect. In lab practice, this low boiling point calls for cooling and careful handling. The solubility shows up in both water and organic solvents, so cross-contamination becomes a possibility if you’re not rigorously organized. Its reactivity revolves around the nitrogen atom, which balances stubbornness toward oxidation with enthusiasm for forming new bonds when prodded by the right electrophile. That feature made it one of the go-to choices at my bench for building enolates and other key intermediates in organic synthesis.
Technical Standards Out in the Field
Most reputable suppliers package and ship diisopropylamine according to accepted technical guidelines, such as the need for clear labeling—including hazard symbols, batch identification, and purity statistics—so researchers don’t have to guess what's inside the bottle. On real-world projects, I’ve seen how mislabeling leads to batch loss or safety hazards. These details might feel bureaucratic until you face an unexpected reaction or exposure, which is why strict adherence matters. Regulatory lists also remind teams to address transportation hazards, flammability, and compatibility, details which find their way into training sessions for anyone even indirectly exposed in shipping and storage.
How Diisopropylamine Comes into Existence
Producing diisopropylamine commonly involves heating isopropyl alcohol with ammonia over an acidic catalyst, then running the mess through separation columns to get rid of unwanted side products such as the mono- or tri-isopropyl derivatives. In practice, engineers continually tweak reaction conditions, pressures, and catalysts to favor the secondary amine. Plant operators know that efficiency here turns into cost savings and less chemical waste downstream—a concern that matters more as environmental regulations tighten year by year. A seasoned process engineer will also think about how to minimize byproducts and energy use, both for profit and for compliance with emissions controls.
The Role in Classic and Cutting-Edge Reactions
Chemists appreciate diisopropylamine for its steady turnout of reliable chemical reactions. Its most famous child, the lithium diisopropylamide (LDA) reagent, gets called up in nearly every synthetic organic chemistry lab. Made by reacting diisopropylamine with n-butyllithium, LDA turns a regular flask into a stage for enolate formation and selective deprotonation. Judging by my own time in research, the availability and predictability of reactions with diisopropylamine often steered projects toward simpler problem-solving, saving hours that would be lost with more complicated or less selective reagents. The amine group’s activeness also invites countless modifications, like acylation or alkylation, expanding its reach far beyond one-trick applications.
Name Games and Industry Language
You’ll hear diisopropylamine called many things in the lab and on documentation. Chemists might write or say 2-propanamine, N,N-diisopropylamine, or even DIPA when speed trumps formality. If you’re reading a patent or product sheet, note these alternatives to avoid missing relevant data or safety information. This mess of synonyms grows from historic naming conventions and keeps older texts relevant to current practice. Learning the common aliases saves time and headaches, especially when searching through scientific literature or international standards, which might not stick to the same word every time.
Everyday Realities of Safety and Procedures
Workers expect to be protected not only from known hazards but also from everyday lapses that catch folks off guard. Diisopropylamine can cause burns or respiratory irritation, and I’ve seen a complacent moment—like not swapping out gloves—cause a minor rash that took days to heal. The liquid flows easily and creates vapor quickly, meaning that spills don’t hang around for slow cleanup. Labs and manufacturing lines depend on proper PPE: gloves rated for organics, lab coats, safety goggles, and a decent fume-extraction system. Regulations about open flames or static in processing areas reflect real risks, not red tape fetishism, because spills here ignite much faster than more familiar chemicals. Training usually focuses on keeping exposures short, maintaining proper ventilation, and handling waste promptly, which matches personal experience as much as written protocol.
Industries That Call on Diisopropylamine
The markets taking in diisopropylamine cover everything from agricultural chemicals and rubber accelerators to medicines and even some fuel additives. In pharmaceutical labs, it's often seen building blocks for antihistamines, antihypertensives, and anesthetics. Agrochemical makers rely on it for herbicide synthesis, a controversial use since new research continually looks at minimizing off-target effects on ecosystems. Rubber processing imports it for vulcanization systems, making tire and hose production possible at massive scales. Looking at its portfolio reminds us that even low-profile intermediates influence supply chains, medical access, and manufacturing resilience, reinforcing why chemical literacy isn’t just a research concern but a broader economic necessity.
Frontiers in R&D
Diisopropylamine gets caught up in broader movements, especially green chemistry. Universities and chemical companies continue probing for less hazardous synthesis pathways—lower temperature routes and catalysts that cut hazardous waste or energy use without sacrificing purity or yield. In my own network, younger researchers lead pilot projects for continuous-flow methodologies, hoping to scale greener, safer production lines. Development of new downstream derivatives promises fresh crop protection approaches or medicines that work more selectively with lower side effects. Cross-talk between industrial and academic settings often drives changes in how the amine gets deployed, since community concerns about exposure or environmental persistence act as spurs to rethink established processes.
Digging Into Toxicity and Exposure
No one in a lab or plant can afford to ignore the toxicity research around diisopropylamine. Studies show it burns skin, irritates eyes, and messes with mucous membranes. Prolonged exposure can trigger headaches and dizziness or cause respiratory issues—so organizations set exposure limits and enforce periodic monitoring. More concerning, animal studies raise questions about chronic effects or environmental leaks that could hurt local water or soil quality, making toxicity studies and containment efforts far more than a regulatory checkbox. Even simple steps, like regular glove changes or prompt eyewash use, matter quickly once something goes wrong. Updating protocols based on toxicity findings keeps pace with growing awareness of occupational health, and industry watchdogs pay close attention to both new data and to manufacturers claiming improved safety.
Where the Future May Lead
Looking ahead, diisopropylamine probably won’t fade out of use, but expectations will get higher. Industry and researchers push for improved handling, greener production, and thorough hazard assessment. Sustainable chemistry goals highlight the need to recycle or minimize waste, even for smaller intermediates. Some labs move to continuous production or integrate digital monitoring to catch leaks or exposures before they escalate. Tightening environmental and safety standards link the fate of this chemical to broader questions—about efficiency, transparency, and workplace rights. As chemical supply chains tighten and competition mounts, each link in production comes under scrutiny not only for performance but also for footprint, so future focus will sharpen on innovations that hit both quality and safety marks.
What is Diisopropylamine used for?
Understanding What Diisopropylamine Does
Diisopropylamine sounds a bit technical, but its value shines through in many industries that keep the world moving. This chemical pops up everywhere, mainly behind the scenes. Chemists count on it because it’s a strong building block for other chemicals. Its role stretches from making medicines to supporting agriculture—you might not notice it, but its influence reaches your daily life.
Making Medicines Work
Walking past pharmacy shelves, you probably wouldn’t guess that diisopropylamine plays a part in the production of some active pharmaceutical ingredients (APIs). In labs, scientists use it during multi-step chemical reactions. Organic synthesis, especially in making drugs, demands chemicals that help form bonds between atoms. Without compounds like this, developing painkillers or treatments for chronic illnesses would slow down. There’s always a push for safer, more reliable ways to make trusted medicine, and this compound helps tick those boxes.
Agriculture Depends on Smart Chemistry
Farmers rely on crop protection to keep fields robust. Diisopropylamine forms a crucial part of this effort. Herbicide producers use it to create mixtures that target weeds without harming crops. Farmers expect these products to be safe and dependable, so people who make crop sprays work hard to balance effectiveness and environmental care. Many herbicides based on diisopropylamine get scrutiny from regulators—the Environmental Protection Agency in the U.S. and similar authorities elsewhere always keep tabs on these formulations, testing for water quality and human health impact.
Everyday Goods and Niche Applications
The uses spread even wider. Workers in the rubber industry use this chemical as a curing agent during manufacturing. It helps keep car tires and seals flexible and long-lasting. The textile world also interacts with diisopropylamine for special dye and finish production, ensuring sportswear, upholstery, and industrial materials stand up to wear and tear. Paint and coating producers sometimes turn to it for its ability to neutralize acidity in solutions, making it a handy ingredient for product stability.
Concerns and Solutions
It’s tempting to load new processes with chemicals that speed up production or enhance effect. My background in research labs taught me not to overlook the environmental and health challenges connected to industrial chemicals. Questions often arise about toxicity and safety. With this compound, most risks come down to handling in large quantities—exposure to concentrated forms requires strong safety habits: gloves, good ventilation, and regular training. Beyond the lab, regulatory bodies in many countries track how producers store, use, and dispose of such substances. Regulators tend to demand thorough testing before new uses emerge.
Sustainable manufacturing keeps pushing for greener options. Companies already swap out older, more hazardous materials for lower-toxicity alternatives. Some chemists now design reactions to use less chemical input, or to avoid generating harmful byproducts. Public pressure and smarter regulations help keep the industry honest.
Looking Forward
Diisopropylamine won’t take the spotlight on a product label, but it plays its part so much of what we rely on works as expected. Every time new safety guidelines appear or a manufacturer invests in greener chemistry, it’s a reminder that even behind-the-scenes chemicals deserve attention. People’s health and the planet’s future ride on clear standards and steady progress, both inside the lab and out in the world.
What is the chemical formula of Diisopropylamine?
Why Diisopropylamine Keeps Cropping Up in Modern Chemistry
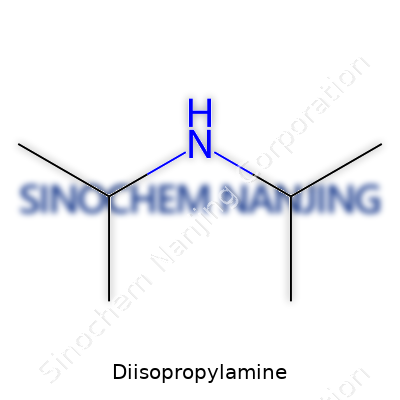
Chemists often talk shop about practical organic compounds, but few non-scientists ever hear about diisopropylamine. Those working in chemistry labs get familiar with this amine pretty early. Its formula is C6H15N. Jumbled letters and numbers, sure—but this simple compound pulls a lot of weight in research and industry.
Formula, Structure, and Relevance
Back in college, labs always had a bottle labeled “DIPA.” The strong smell gave it away long before reading the label. Its formula, C6H15N, packs three familiar atoms—carbon, hydrogen, and nitrogen—into a compact, colorless liquid that does a surprising amount of heavy lifting. Diisopropylamine gets made by adding two isopropyl groups to an ammonia skeleton. It's not fancy or exotic, yet chemists depend on it for building blocks, reactions, and protection of other chemicals.
The structure can be summed up with two branches—each a set of three carbons—hooked to the nitrogen center. A quick tally: two isopropyl groups (C3H7 each) lays out to a total of six carbons, fifteen hydrogens, one nitrogen. It’s a neat little molecule, no bigger than it needs to be. Its straightforwardness makes it easy to tweak and add to more complex chemicals.
Everyday Impact—Going Beyond the Lab Bench
Plenty of folks outside science circles never see diisopropylamine in the wild, but it's behind a lot of products and breakthroughs. Farmers use herbicides derived from it. Pharmaceutical labs convert it into intermediates for the drugs in our medicine cabinets. At first glance, the formula doesn’t seem like much. But that structure makes it a perfect fit when chemists want a non-bulky amine—something reactive, but not prone to getting in the way.
DIPA goes into everything from pesticides to corrosion inhibitors. The reason lies in that basic amine group: it grabs onto acids and helps build up new chemicals. The pharmaceutical industry relies on it to create compounds with the right biological effects. Tire makers use it to treat rubber, allowing treads to last longer and grip the road in rain.
Why Safety and Handling Matter
Anyone with a few years in a chemistry lab learns fast—personal protection trumps curiosity. Diisopropylamine’s vapors can irritate skin and lungs. Its strong smell lingers, hinting at real risks if handled poorly. The chemical’s flammability and need for good ventilation keep workers on their toes. Industry guidelines spell out glove and goggle requirements, and most shops have spill kits ready. In my experience, a slip sends the whole room scrambling, so people rarely risk shortcuts.
Looking at the Future—Balancing Utility and Risk
Every new wave of green chemistry tries to sidestep environmental hazards while keeping chemical processes efficient. Some research labs look for alternatives that keep reactions similar, but cut down on waste or toxicity. So far, DIPA still gets called up for its reliability and availability. As scientists work to design smarter, cleaner chemistry, they have to evaluate every solvent and reagent—not just its performance, but its afterlife in the environment.
Diisopropylamine’s formula, C6H15N, reflects a no-nonsense molecule—essential, practical, and just risky enough to keep both users and regulators vigilant. Even small improvements in handling and alternatives can mean safer workplaces and cleaner rivers for everyone outside the lab.
Is Diisopropylamine hazardous or toxic?
Understanding Diisopropylamine
Diisopropylamine crops up in a lot of places: industrial sites, agricultural operations, college chemistry labs. Folks use it in rubber manufacturing, herbicide production, and even as an ingredient in certain pharmaceuticals. If you’ve been near lab work or the chemical supply side of a business, you probably spotted the name before and may have caught a whiff of its strong smell—something between ammonia and cleaning solution. That potent scent isn’t just unpleasant; it’s a direct reminder that this liquid is quite volatile and shouldn’t be taken lightly.
Health and Safety Risks
Anyone handling diisopropylamine without the right gear is taking a risk. Vapors irritate the eyes, throat, and lungs. Even brief exposure can cause coughing and watery eyes. Prolonged inhalation can bring more serious trouble—headaches, dizziness, sometimes even breathing problems that don’t go away right away. When this chemical contacts skin, it burns and leaves red, blistered patches that linger well after the substance is washed off. Chemical burns aren’t something you forget quickly, and diisopropylamine has a reputation among those who work with it for just that reason.
Direct spills or splashes in the eyes raise the stakes. Immediate pain, vision loss if not flushed out promptly. Eye protection isn’t just a box-checking measure here; it’s an absolute must. In more severe incidents, high exposure without proper ventilation or respiratory protection could lead to systemic poisoning—a rare but real threat. Symptoms include loss of coordination, fast heartbeat, feeling extremely tired, and in the worst cases, collapse.
Environmental Concerns
Factories that discharge diisopropylamine without neutralizing it create hazards for both water and soil. Local waterways pick up the chemical, fish and aquatic life take a hit, and the knock-on effects for nearby farming can’t be ignored. This substance can break down in the environment over time, but while it’s present, organisms feel the impact. Accidental releases have prompted expensive cleanups—costs that sometimes land on communities living nearby.
Runoff from agricultural spraying or improper container disposal leads to contamination. Livestock and wildlife don't stand much chance against concentrated doses. Regulations have cracked down in recent years, underlining how regulators view this chemical as a red flag for environmental safety.
Mitigating the Hazards
Lessons from industry experience say plain gloves are not enough. Nitrile or neoprene gloves, chemical splash goggles, and face shields belong in any room where diisopropylamine gets opened. Ventilation systems keep air circulating, reducing vapor buildup in confined spaces. Working outside—or with open doors—might seem like a hassle, but it beats a trip to the occupational health clinic.
Spill kits and ready access to emergency showers give workers a safety net if things go sideways. Strict storage—separate from acids, away from anything that could ignite, tightly sealed containers—makes sense both for safety and cost savings over time.
Moving Toward Safer Practices
Training stands as the best defense: not just for scientists, but for anyone who moves, stores, or disposes of the material. Company leaders who treat safety as more than just a poster on the wall, backing it with real job-specific drills and easy-to-understand guidelines, see fewer accidents. Communities around manufacturing zones advocate for stricter monitoring and regular transparent reporting—not out of fear, but from the hard-earned wisdom that comes with living near chemical plants.
Finding and using safer substitutes where possible always saves trouble down the line. If a less hazardous amine can serve the same function, most responsible operations make the switch. Taking shortcuts rarely pays off, and with chemicals like diisopropylamine, playing it safe is the only smart path forward.
How should Diisopropylamine be stored?
Understanding the Real Risks
Diisopropylamine is not the kind of substance you leave on a dusty shelf and forget about. This clear, colorless liquid has a strong odor and it evaporates pretty quickly, which means it’s easy for vapors to fill a room. If you’ve handled it in a lab or in any industrial setting, you know the smell lingers on your clothes if you’re not careful. That smell hints at something deeper: its volatile and flammable nature.
Why Proper Storage Matters
I once visited a site where an unmarked drum sat under a leaky window—it turned out to be Diisopropylamine. They learned the hard way that leaks and warmth speed up evaporation. The air in the room stung the eyes. No surprise then that the U.S. Occupational Safety and Health Administration (OSHA) and the National Institute for Occupational Safety and Health (NIOSH) both stress good ventilation and secure storage for amine compounds like this.
Fires don’t always start with a bang; sometimes, all it takes is one tiny spark meeting those invisible vapors. Diisopropylamine catches fire easily and burns with a hot, hard-to-extinguish flame. You can’t always see the problem building up, but you’ll sure see the results if disaster strikes.
Good Storage Practice Is Simple, Not Easy
So what actually works? Use containers made out of steel or high-density polyethylene. Don’t trust old or mismatched lids. If there’s one shortcut I’ve seen end badly, it’s “temporary” caps and makeshift labels. Once, I saw a drum with tape instead of a proper closure leak all over the floor and the clean-up hours wasted could have gone into simply doing it right the first time.
Keep containers tight and closed, away from sunlight and heat sources—think inside a locked chemical storage cabinet, not resting by a window, not in a cluttered utility closet. Check for fire suppression, because an extinguisher won’t help if you can’t get to it fast.
Avoiding Mixing Hazards
Everyone remembers to lock away the sulfuric acid but forgets that amines and acids plus water make heat and toxic fumes. The big scare often isn’t the fire; it’s the cloud of gas that comes out when things go wrong. Diisopropylamine reacts with strong oxidizers, acids, and halogens. It shouldn’t share space or even the same secondary containment tray with them. I once worked in a facility where an accidental mixing happened after a flood—the chemical-resistant bins did their job and saved us from much worse trouble.
What Works in Real Settings
Labels should make sense. Too many sites rely on faded marker that nobody can read in a hurry. Standard printed labels with chemical names, hazard info, and expiration dates keep everyone safe. Employees should walk storage areas, check containers for rust or leaks, and make it routine, not just a paperwork drill for once a month.
No magic trick can replace training and habit. New staff need to see the safe way, not just hear a lecture. Safety data sheets (SDS) belong right next to storage, printed out and in a language workers understand. A site manager told me once that he only realized something was off because someone left the sheet open to the “inhalation hazard” section by mistake—a small stumble that may have prevented a much bigger one.
Simple Strategies Build Trust
Taking shortcuts with Diisopropylamine never pays off. The people who work near these chemicals, and the communities who live nearby plants or transport routes, all have a stake in making sure storage isn’t an afterthought. Real safety comes from habits built on facts, not just compliance. And that’s what keeps accidents from turning into headlines.
What are the safety precautions when handling Diisopropylamine?
Why Respecting Diisopropylamine Makes a Difference
I’ve spent years around chemicals in both academic labs and industry settings, and diisopropylamine always commands my attention. This clear liquid smells a bit like ammonia, and it’s more than just unpleasant — it acts fast if it spills or leaks. Years ago, I watched a fellow technician mishandle just a splash, and it sent him to the university clinic. That memory still shapes how I treat every bottle and beaker.
Proper Ventilation is More Than a Guideline
Fumes from diisopropylamine aren’t just annoying; they irritate the respiratory tract and can make people dizzy or foggy-headed without warning. My routine starts with a quick check on fume hoods — nothing fancy, but I make sure fans work and I always test airflow with a bit of tissue or smoke. Never trust a stuffy room for work involving this stuff, even if you’re only transferring a small volume from one container to another.
Gloves Aren’t Optional
Skip the latex — nitrile or neoprene gloves stand up to diisopropylamine and don’t break down on contact. Years ago, I watched someone wearing cheap plastic gloves end up with red, burning skin because the protection just wasn’t meant for strong amines. Double-checking glove integrity has become habit because it only takes a few drops to cause raw patches or worse. Long sleeves and full-length lab coats cover what gloves can’t, especially since splashes happen when you least expect them.
Eye and Face Protection Can’t Be Skipped
One accidental splash to the eye can do permanent damage. I always keep safety goggles on my face — not up on my head or dangling around my neck. If there’s even a slight chance of a splash, I reach for a face shield, not just goggles. A friend once got a droplet behind his lenses; now he won’t start work until his shield is secure. Eye washes and showers stay checked, filled, and accessible. You never want to be hunting for the eyewash when pain starts setting in.
Fire Risks and Storage
Diisopropylamine lights up easily, and I’ve seen vapors catch unexpectedly from static sparks during dry winter days. Grounding containers cuts down risk, and I store this chemical away from oxidizers and acids, never crowding shelves or stacking containers two deep. Metal cans or glass bottles with tight lids help keep vapors down. Label everything, even if it slows you down. Nothing sours a shift like grabbing an unmarked container and realizing too late what you’ve opened.
Spill Control and Emergency Response
Speed matters in a spill. I’ve trained my team to grab absorbent pads and cover liquid right away. Regular drills keep responses sharp, so no one freezes up if a bottle drops. Neutralizing agents stay on hand, as soapy water rarely does enough. Disposal follows local environmental rules because these waste streams matter for health and for the groundwater out back. Dialing for emergency help — or just shouting for backup — never makes you look foolish. Slow pride leads to more injuries than fast action ever could.
Staying Safe Creates Confidence
Confidence never comes from luck or shortcuts. Every precaution feeds into trust: trust in gear, trust in teammates, trust in routines built from hard-won experience. Safety with diisopropylamine doesn’t just protect hands or lungs — it builds a culture where people can focus on their tasks, innovate, and go home healthy. That’s worth every minute spent inspecting gloves or checking fume hoods, every single day.
| Names | |
| Preferred IUPAC name | N-Propan-2-ylpropan-2-amine |
| Other names |
N-(1-Methylethyl)-2-propanamine Diisopropylamine DIPA Isopropylamine, di- N,N-Diisopropylamine |
| Pronunciation | /daɪˌaɪsəˈprəʊpɪləˌmiːn/ |
| Identifiers | |
| CAS Number | 108-18-9 |
| Beilstein Reference | 1209276 |
| ChEBI | CHEBI:40497 |
| ChEMBL | CHEMBL50471 |
| ChemSpider | 5369 |
| DrugBank | DB14011 |
| ECHA InfoCard | 100.003.300 |
| EC Number | 204-823-8 |
| Gmelin Reference | 120585 |
| KEGG | C06147 |
| MeSH | D006967 |
| PubChem CID | 6106 |
| RTECS number | JI6175000 |
| UNII | V9V80613J6 |
| UN number | UN1158 |
| Properties | |
| Chemical formula | C6H15N |
| Molar mass | 129.24 g/mol |
| Appearance | Colorless liquid with an ammonia-like odor |
| Odor | Ammonia-like |
| Density | 0.722 g/mL at 25 °C |
| Solubility in water | Soluble |
| log P | 0.99 |
| Vapor pressure | 3.06 kPa (at 20 °C) |
| Acidity (pKa) | 36 |
| Basicity (pKb) | 3.0 |
| Magnetic susceptibility (χ) | -47.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.387 |
| Viscosity | 0.58 mPa·s (20 °C) |
| Dipole moment | 1.29 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 294.78 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −192.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4272.9 kJ/mol |
| Pharmacology | |
| ATC code | C04AX21 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H302, H314, H336 |
| Precautionary statements | P210, P260, P273, P280, P304+P340, P305+P351+P338, P310, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 52 °C (closed cup) |
| Autoignition temperature | 424 °C |
| Explosive limits | 1.1% - 8.8% |
| Lethal dose or concentration | LD50 oral rat 520 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 285 mg/kg |
| NIOSH | NJ3150000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Diisopropylamine: "5 ppm (18 mg/m3) as an 8-hour TWA (OSHA) |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Dimethylamine Diethylamine Dipropylamine Dicyclohexylamine Diisopropylethylamine |

