Diisopropyl Ether: Story of a Chemical with Deep Roots and Modern Curiosity
The Path from Discovery to Ubiquity
Chemistry textbooks often pass over the odyssey of diisopropyl ether with barely a note, but this compound traces a winding route through the annals of industrial chemistry. Ether itself holds a notorious spot in history, powering some of the earliest anesthesia procedures and chemical extractions. Diisopropyl ether—known in labs as DIPE or isopropyl ether—emerged as chemists started tinkering with variations of basic ethers, searching for less volatile, less reactive cousins to the original diethyl ether. By the mid-20th century, the field saw diisopropyl ether taking on extraction duties, especially in separating rare earth elements for the nascent nuclear and electronics industries. The reason? Its unique structure and solvent power that often proved superior or gentler than diethyl ether, giving chemists more finesse in tricky separations.
Getting to Know Its Character: Physical, Chemical, and Technical Features
All ethers carry that faint, recognizable odor that many lab workers joke about. Diisopropyl ether keeps that tradition going, albeit with a slightly less pungent punch. Clear, colorless, and liquid at room temperature, it leaves a slick, light feeling on the skin. The boiling point hits a comfortable range for users who need to distill or recycle it—somewhere just shy of 70 degrees Celsius. These properties often tempt those who need a moderately volatile, water-insoluble solvent. Unlike its stickier relatives, diisopropyl ether floats confidently atop water, prefers organic company, and rarely mixes with polar substances. Manufacturers must pay close attention to labeling, emphasizing its flammability and tendency to form dangerously explosive peroxides after long exposure to air—one of the main headaches for storage and transport. Labels usually point out the need for tight seals, proper venting, and refrigeration if kept long term. In my experience, every good lab includes periodic peroxide testing on ether bottles to avoid surprise chemical rockets.
Making and Modifying: Behind the Scenes of Preparation and Chemistry
Rolling back the curtain on diisopropyl ether’s preparation brings you to an intersection of classic organic chemistry. The most common route takes two molecules of isopropanol and nudges them together with sulfuric acid as a catalyst, leading to dehydration and the birth of diisopropyl ether. This process calls for fine control of temperature to prevent runaway byproducts or charring. Advanced users may distill the mixture straight off to capture pure ether, which usually needs drying steps—using potassium carbonate or similar agents—to clear up unwanted water. In the lab, its chemistry isn’t all that dramatic. Diisopropyl ether resists many nucleophilic attacks, making it stable enough for a range of solvent duties. Still, it reacts violently with strong oxidizers. Over time, like many ethers, it picks up enough oxygen from the air to spawn peroxides, which can explode with minimal provocation. There’s a deep respect for this quiet danger; every chemist I know treats those old bottles in the back of the cabinet as minor time bombs.
The Many Faces and Names of Diisopropyl Ether
Walking through a chemical storeroom, one can spot multiple names on the bottles: isopropyl ether, DIPE, 2-isopropoxypropane, and a handful of trade monikers depending on supplier and region. Synonym confusion occasionally leads to supply mix-ups or improper handling, a reminder that attention to nomenclature stands as important as purity specs. For students and junior technicians, learning every alias of a compound like this often feels overwhelming, but making sense of such naming quirks separates true chemical literacy from rote memorization.
Staying Safe: Standards and Real-World Precautions
Industrial environments find diisopropyl ether both useful and unruly. Besides the ever-present fire hazard, peroxides build up slyly, especially in warm climates or those odd labs where turnover runs slow. Regulations in the United States and Europe press home the basics: store the compound in tightly closed, air-limited containers, avoid sparks, and include stabilizers or regular testing schedules. Facilities with poor ventilation or inattentive chemical hygiene see most of the horror stories—unexpected explosions from crystallized peroxides along bottle necks or inside stoppers, usually when someone grows complacent. Anyone who ignores the rule about never distilling it to dryness runs a real risk. In many teaching labs, instructors use these tales to drive home the humbling lesson that chemistry rewards caution, not bravado.
Why It Still Matters: Application in Science and Industry
Despite the risks, diisopropyl ether holds its ground in the global chemical toolkit. Extraction of rare earth elements from aqueous solutions continues in industrial metallurgy and recycling centers—companies reclaiming batteries or refining ores routinely leave footprints of isopropyl ether in their protocols. Pharmaceutical chemists lean on its ability to separate and purify drug intermediates, relying on its selectivity to improve yields. In petroleum labs, the compound helps reveal water content in gasoline blends or extract certain hydrocarbons. Organic synthesis finds use for diisopropyl ether as a non-polar solvent in reactions that need to steer clear of water or stronger nucleophiles. My own work saw it helping resolve natural product mixtures, stripping off compounds with gentle efficiency where nastier solvents threatened to destroy sensitive targets.
Ongoing Questions: Toxicity and Environmental Dose
No discussion stays honest without addressing health questions. Diisopropyl ether, like its cousins, produces drowsiness, dizziness, or worse if inhaled or absorbed in significant amounts. Most countries restrict exposure in workplace air to a handful of parts per million, backed up by vivid case reports of headaches, nausea, and, in rare cases, lasting neurological troubles. Studies point out that it carries lower chronic toxicity than some other ethers, but the risk of acute events from accidental high exposure, fire, or peroxide explosion shadows every use. Environmental agencies continue tracking spills or releases, and while diisopropyl ether tends to evaporate quickly, its breakdown in the open air happens more slowly than many hope, lingering for days or weeks, especially in cold or damp environments. Emergency rooms register few poisonings, but the compound’s volatility makes it inevitable that safety standards only work when everyone treats the risks with respect.
Charting the Road Ahead: Research, Development, and Future Applications
Innovation often circles back to time-tested materials, and diisopropyl ether fits that rule. Research groups keep probing for ways to suppress its peroxide problem. Some new stabilizers and built-in warning systems, like peroxide test strips on caps or color-changing packaging, attract interest, though high costs slow their adoption. Process chemists experiment with solvent-free or alternative extraction methods, but legacy infrastructure and cost pressures keep diisopropyl ether a staple in many workflows. Green chemistry advocates eye more biodegradable or less hazardous substitutes, yet the compound’s blend of low water solubility and high extraction power remains tough to beat at scale. Some labs trial ionic liquids or fancy deep eutectic solvents, but they must match diisopropyl ether’s effectiveness, recyclability, and price before industry pivots. The future likely holds more incremental upgrades than sudden revolutions, highlighting the chemical’s stubborn relevance across generations of technologists.
What is Diisopropyl Ether used for?
Everyday Chemistry at Work
Growing up around a family pharmacy, I picked up a few habits. Sometimes I read the back of solvent containers, curious about the odd-sounding names listed below the familiar ones. Diisopropyl ether always stood out because I rarely heard anyone talk about it outside a laboratory. Yet, this colorless solvent shapes more chemical reactions than most people imagine.
Role in Laboratories and Industry
Organic synthesis often relies on finding just the right conditions. Diisopropyl ether steps in as a solvent for Grignard reactions, which help create carbon–carbon bonds—essential links that build up molecules, including medicines and agricultural products. It’s got a pretty low boiling point and resists mixing with water, so chemists can separate layers easily when extracting or washing compounds. From my own hands-on projects, getting that clean phase separation can mean the difference between messy failure or successful experiment.
Pushing past the lab bench, manufacturers look to diisopropyl ether during petroleum refining. Refiners use it to recover and purify furfural, a chemical pulled out from agricultural byproducts and then redirected into everything from plastics to pharmaceuticals. High purity matters—contaminants easily mess with the outcome of downstream processes. Diisopropyl ether shines because it dissolves plenty of organic substances but leaves unwanted water behind. That trait saves processing time and cuts waste.
The Safety Piece
Anyone new to this solvent needs to know: it’s highly flammable and forms peroxides with exposure to air over time. I’ve seen a blown bottle or two in older stockrooms, so regular inspection always takes priority. The chemical industry records injuries—often from ignoring routine safety steps. Clear labeling, tight storage, and frequent peroxide checks protect both people and the lab. Students and teachers need practical demonstrations, not just warnings in manuals, so new generations learn to respect the risks.
Green Chemistry and the Search for Alternatives
The push for eco-friendly chemistry gets louder each year. Diisopropyl ether, while useful, leaves concerns due to volatility and explosion hazards. Research teams have started swapping it with less hazardous solvents, like methyl tert-butyl ether or bio-based alternatives. Results aren’t uniform—some reactions stubbornly resist green substitutions. Still, progress happens in small steps. Chemical supply firms now offer guidelines to avoid stockpiling old ethers, and labs can send test strips along with solvents to catch peroxides early.
Building Skills and Awareness
Understanding the role of diisopropyl ether isn’t just academic. It touches hundreds of reactions that build drugs, plastics, and specialty chemicals. Choosing it means balancing efficiency, safety, and stewardship. Chemists owe it to themselves and the wider public to stay up-to-date on safe practices. Sharing knowledge within the community matters just as much as the chemical itself. The push for safer chemistry starts with experience, solid facts, and an open conversation around what actually works.
What are the safety precautions when handling Diisopropyl Ether?
Know the Substance
Grabbing a bottle of Diisopropyl Ether in the lab comes with an unspoken set of rules. It’s a colorless liquid, almost unnoticeable until you catch its sharp smell or see the “Highly Flammable” label. I remember my first organic synthesis, barely a year out of college, and the lab manager spelled it out in the kind of plain, careful language you don’t forget: treat this stuff like it will ignite at any moment, because it just might.
Understanding Flammability Risks
Diisopropyl Ether burns at the mere suggestion of a spark. Its vapors form explosive mixtures with air and, worse, can drift across benchtops and flash back to a far-off ignition source. Real-life fires have started this way—one got out of hand at a friend’s workplace because someone switched on a piece of equipment after a spill. The biggest lesson? Always work away from open flames, hot surfaces, or any tools that could throw a spark.
Handling Peroxide Formation
Many students don't realize that ethers like this can form shock-sensitive peroxides, sometimes in as little as a few months after opening. I’ve seen vials crusted with white, crystalline deposits—peroxides waiting for a pressure change to explode. Keeping Diisopropyl Ether in tightly closed, amber glass bottles slows down this risk, but don’t rely on luck. Mark the opening date, test for peroxides regularly, and add stabilizers when possible. Dispose of old material. It’s never worth the risk to push the shelf life.
Wear the Right Protective Equipment
No one should ever handle Diisopropyl Ether in shorts and a T-shirt. Lab coat, safety goggles, and gloves are non-negotiable. Nitrile gloves hold up well here—latex doesn’t. If there’s a spill, chemical splash goggles stop a very bad day from becoming much worse. Even a small exposure can cause serious skin irritation or respiratory discomfort. I keep an extra set of gloves on hand; they take a beating with solvent work, and there’s no pride in pushing their lifespan.
Engineering Controls and Ventilation
Use Diisopropyl Ether only under a well-maintained fume hood. Vapors build up quickly, even at room temperature, and effective ventilation is the one thing that stands reliably between you and a trip to the emergency room. I’ve watched less experienced colleagues try to get by with open benches; it always ends with a sharp reminder from whoever’s responsible for safety. The simple fact is, the fume hood isn’t optional; it’s the difference between a routine experiment and a potential disaster.
Storage Rules Make the Difference
Store the solvent in small, properly sealed bottles, clearly labeled and segregated from oxidizing agents, acids, and direct sunlight. Keep it in flammables storage cabinets. Limit the quantity stored—nobody needs gallons of this on hand. It’s easy to let old bottles sit, but regular inventory checks keep surprises to a minimum. I once found a forgotten bottle tucked away behind other chemicals—lesson learned, I now stick to a calendar review and prompt disposal.
Preparedness Wins the Day
Always keep spill kits, fire extinguishers, and eye wash stations nearby. In a crisis, time matters more than theory. Knowing where everything is and how to use it beats the best protocol on paper. Training helps, but walking through the steps with real equipment does more for peace of mind and actual readiness.
Safe Habits Save Lives
People sometimes treat safety rules as background noise, but they matter most with tricky solvents like Diisopropyl Ether. The margin for error disappears quickly. Respect the risks and you get the work done—and go home safe at the end of the day.
What is the chemical formula and structure of Diisopropyl Ether?
What Is Diisopropyl Ether?
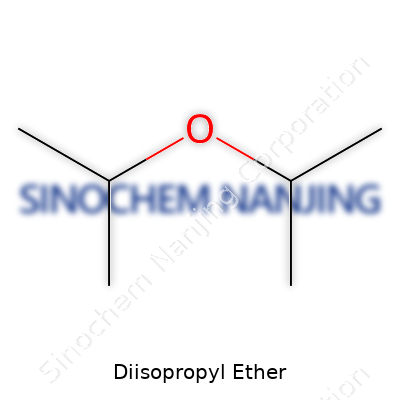
Diisopropyl ether shows up in many labs and chemical stockrooms as a useful organic solvent. Its reputation as a sturdy extraction agent in organic synthesis owes a lot to its properties: decent stability, fairly low reactivity, and a knack for dissolving nonpolar substances. The simplest definition lands on its chemical formula—C6H14O. This breaks down to six carbon atoms, fourteen hydrogen atoms, and a single oxygen atom.
Chemical Structure: More Than Just a Formula
Plenty of folks know their way around chemical formulas, but seeing the structure often makes the difference. In diisopropyl ether, two isopropyl groups sit linked together by an oxygen atom. Picture it as (CH3)2CHO–O–CH(CH3)2. Each “isopropyl” chunk looks like Y-shaped branches around that central oxygen bridge.
Instead of a straight-line structure, the molecule spreads into a loose “V” or “kite” arrangement, thanks to the angles at the oxygen atom. Having plenty of carbon and hydrogen atoms packed into those short branches makes it a low-density liquid with a faintly sweet, petrol-like smell. You catch a whiff of it in some old-school organic labs.
Why Diisopropyl Ether Matters
The real draw for chemists comes from how diisopropyl ether acts during extractions and as a solvent. It keeps itself separated from water, so folks can use it to pull nonpolar organics out of mixtures. It evaporates pretty quickly, letting chemists recover what they’re after without much residue left behind.
From my own years doing bench chemistry, I found this ether beats out some others like diethyl ether when you want less tendency for peroxide formation and a slightly higher boiling point. It handles heat a bit better and that matters if your reaction mixture gets warm. Peroxides still form over time, though, so safe storage always demands fresh bottles and regular testing.
Safety and Handling Concerns
Safety should never fall by the wayside. Diisopropyl ether can form explosive peroxides when exposed to air over long periods. In real lab practice, I always mark open dates on bottles, run tests for peroxides every couple of months, and keep solutions away from ignition sources. The ether’s low flash point means it doesn't take much to set off vapors, so fume hoods and grounded containers make a real difference.
Many industries—especially pharmaceuticals and petrochemicals—use diisopropyl ether for sample prep, purification, and even for removing moisture traces from other solvents. Yet nobody working with it can ignore its risks. Anyone storing or using it should stay up-to-date with peroxide detection methods, and disposal routines should follow local hazardous waste guidelines to avoid environmental release.
Safer Alternatives and Green Chemistry
Modern labs lean toward greener solvents where possible. Some researchers now mix in less-volatile ethers, or swap in esters and alcohols with lower health risks. That switch doesn't always match the performance diisopropyl ether delivers, especially for quick-phase separations. Still, the move toward sustainable chemistry pushes everyone to keep an eye out for improvements.
Every solvent, including diisopropyl ether, asks users to respect its capabilities—and its hazards. It stands as a textbook example of an ether’s utility, right down to the structure and formula.
How should Diisopropyl Ether be stored?
Getting Real With Chemical Hazards
Anyone working in a lab or warehouse knows certain chemicals demand more respect than others. Diisopropyl ether holds that reputation. With years handling solvents in research, I have seen how poor storage can lead to trouble. This ether ranks high for forming peroxides—unstable compounds that can cause fires or explosions without warning. Its low flash point and volatility call for more than casual shelving.
Fire Risks and Why Regular Storage Fails
I learned early that glass bottles on an open shelf with diisopropyl ether put everyone on edge. One spilled bottle, one hot bulb overhead, and you might have more than lost product on your hands. Flammable storage cabinets with self-closing doors keep containers away from sparks and high heat. These cabinets come rated for volatile liquids and often save lives as much as inventory.
Controlling peroxides, Not Leaving Things to Chance
Ethers like this one want to form explosive peroxides over time. Once in grad school, our group caught crystals forming near the lid—a nightmare moment. Nobody wants to find that during a routine check. Tight seals, metal cans, and adding antioxidant stabilizers can slow down peroxide buildup, but nothing beats marking open dates and regular testing. I make it a habit to log every opening and check peroxide strips at least every few weeks. Anything smelling sour or with cloudiness gets disposed of—by professionals only.
No Sunbathing, No Freezing: Temperature and Light Matter
Direct sunlight and temperature swings push ethers toward instability. I always pick a shaded, cool spot away from outside walls. Standard lab fridges fit the bill only if they don’t have exposed electrical parts—explosion-proof models stop vapor buildups from causing big trouble. Keeping bottles in the dark also stops light from splitting molecules and starting unwanted reactions.
Vapor Control and Spill Preparedness
Any storeroom with good airflow makes a difference. Vapors from diisopropyl ether settle low and can sneak along the floor to ignition sources far from the bottle itself. Labs with exhaust hoods or local vent ducts see far fewer incidents. Absorbent spill kits, fire blankets, and Class B extinguishers stay loaded and checked. I have seen more than one new worker try to mop up a spill with paper towels—training and clear signage save more stress than any SOP document.
Label Everything: No Guesswork Allowed
Clear, bold hazard labels help avoid mix-ups or lazy storage practices. In my experience, even experienced hands get distracted or rushed. Original manufacturer labels plus secondary labelling with open dates, hazard pictograms, and contact info for emergencies take away guessing games. Supervisors who run regular audits find fewer ugly surprises down the line.
Better Storage, Fewer Accidents
Rules exist for a reason, and no one wants to pay with their lab, business, or safety. Diisopropyl ether earns its place in the chemical hall of caution. With careful storage—in ventilated, fire-rated cabinets, clearly labeled and tracked—accidents don’t make the news. Each step might seem simple, but they add up to a safer workplace where people go home at the end of the day.
Is Diisopropyl Ether hazardous to health or the environment?
Looking Closer at Diisopropyl Ether
Diisopropyl ether finds its way into laboratories, refineries, and sometimes even into the hands of students working on chemistry projects. The smell reminds me of something harsh and sharp, a bit like rubbing alcohol, but stronger. Despite how common it appears in science settings, few outside those circles know about the risks it brings.
Health Risks Aren't Just for Chemists
Most people never realize how volatile diisopropyl ether really is. Even a curious sniff at the bottle can set off coughing. Inhaling vapors can cause headaches, nausea, and dizziness. Longer exposures could lead to more serious effects, attacking the central nervous system or irritating eyes and skin. Prolonged contact with skin sometimes causes dryness or cracking—something I learned after spilling a few drops during a hurried experiment.
The real danger hides in its tendency to form explosive peroxides. Older bottles, especially if not tightly shut or if exposed to light, can turn from a simple solvent into something much more hazardous. Stories circulate among lab workers where forgotten ether nearly caused explosions. Routine checks and a sharp attention to storage might prevent disasters, but one mistake could be catastrophic.
Flammability that Can't Be Ignored
The fire risk stands out. Diisopropyl ether evaporates at room temperature, filling the air with vapors that catch fire with the smallest spark. Friends who’ve worked in larger commercial labs remember regular drills and strict rules about static discharge, open flames, and proper ventilation. At home or in less controlled settings, things can get out of hand quickly if handled carelessly.
Solvents often bring risks, but this one calls for respect. The ability to ignite well below boiling water means that the only real safety comes from keeping it tightly sealed and far away from heat sources. Even sunlight filtering through a window can accelerate peroxide formation and evaporation.
Environmental Concerns Reach Beyond the Bottle
Spilling diisopropyl ether isn’t like dropping sugar or even acetone. Vapors travel, spreading through the air and affecting indoor air quality almost instantly. For workplaces in cities or near neighborhoods, this becomes more than an inconvenience—it poses a genuine threat to air quality and public health. Animals exposed to runoff or vapors might suffer organ damage or nervous system issues, according to toxicology studies.
Many solvents break down quickly in nature. Diisopropyl ether, though, doesn’t always disappear so fast. Its persistence can cause problems for soil and groundwater. Leaks or improper disposal let the chemical seep into places where cleanup isn’t so easy. Municipal water systems worry about industrial solvents for this reason. I’ve met folks who worked on environmental response teams and heard stories about how cleaning up after accidents demanded time, training, and substantial resources.
Looking Forward: Reducing Harm
Understanding the risks prompted many workplaces and schools to substitute safer materials or limit access. Updated labeling, mandated safety checks, and secure storage all grew in importance after a number of high-profile incidents. Some groups now push for greener chemistry in labs, swapping out hazardous substances for those less likely to pollute or cause injury.
Using personal protective equipment—like proper gloves, goggles, and lab coats—keeps the odds in favor of safety. Good ventilation and clear protocols for disposal help protect not just the user but everyone nearby and the environment outside.
Diisopropyl ether serves a role in chemistry, but only through respect for its dangers and care in its use can anyone hope to keep people and the environment safe.
| Names | |
| Preferred IUPAC name | 2-isopropoxypropane |
| Other names |
Isopropyl ether 2-Isopropoxypropane Diisopropyl oxide DIPE |
| Pronunciation | /daɪˌaɪsəˈproʊpɪl ˈiːθər/ |
| Identifiers | |
| CAS Number | 108-20-3 |
| Beilstein Reference | 603873 |
| ChEBI | CHEBI:40449 |
| ChEMBL | CHEMBL142698 |
| ChemSpider | 6825 |
| DrugBank | DB02138 |
| ECHA InfoCard | ECHA InfoCard: 0885326a-1c2d-4b1b-ae49-7e3696c9a082 |
| EC Number | 203-560-6 |
| Gmelin Reference | 52730 |
| KEGG | C01841 |
| MeSH | D006969 |
| PubChem CID | 31284 |
| RTECS number | YD2450000 |
| UNII | NQ54G60D9J |
| UN number | UN1159 |
| Properties | |
| Chemical formula | C6H14O |
| Molar mass | 102.18 g/mol |
| Appearance | Colorless liquid with a pleasant ethereal odor. |
| Odor | sweet, pleasant, ethereal |
| Density | 0.725 g/cm³ |
| Solubility in water | 1.1 g/L (20 °C) |
| log P | 2.27 |
| Vapor pressure | 32 mmHg (20 °C) |
| Acidity (pKa) | 18.1 |
| Basicity (pKb) | pKb ≈ 3.21 |
| Magnetic susceptibility (χ) | -68.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.369 |
| Viscosity | 1.9 mPa·s (at 20 °C) |
| Dipole moment | 1.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 276.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –420.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4094 kJ/mol |
| Pharmacology | |
| ATC code | V03AB06 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02, GHS07 |
| Signal word | Danger |
| Hazard statements | H225,H302,H304,H319,H336,H351,H372,H411 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P305+P351+P338, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-4-0 |
| Flash point | -28 °C |
| Autoignition temperature | 416 °C |
| Explosive limits | 1.4–21% |
| Lethal dose or concentration | LD50 oral rat 5200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat 5200 mg/kg |
| NIOSH | TTJ30000K6 |
| PEL (Permissible) | 400 ppm |
| REL (Recommended) | 0.7 ppm |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
Dimethyl ether Diethyl ether Methyl tert-butyl ether Methoxyethane 1,4-Dioxane |

