Diisooctyl Phthalate: Tracing the Complex Story of a Plasticizer
A Walk Through the Past: From Discovery to the Ubiquitous Bottle Cap
Few chemicals better explain the relationship between modern industry and the environment than Diisooctyl Phthalate (DIOP). I still recall vivid classroom demonstrations where drops of this clear, oily liquid turned brittle PVC piping into something you could twist without breaking. DIOP and its family of phthalates rose to fame in the 20th century as the needs of manufacturing grew, especially after World War II. The rush to make synthetic materials bend, flex, and last longer paved the road for DIOP to become a staple in everything from electrical cables to car interiors. Over the decades, research teams reported ever-expanding use cases, importing and exporting millions of kilograms yearly, until questions about its effects started cropping up.
Can One Product Be Everywhere?
DIOP’s appeal isn’t rooted in mystery. As a colorless, oily liquid, it mixes with polyvinyl chloride (PVC) to keep it soft and long-lasting. The physical properties make DIOP seem tailor-made for mass manufacturing: high boiling point, low volatility, and an ability to spread evenly through polymers. The chemical formula, with its core phthalate group bound to two iso-octyl chains, gives it properties that resist evaporation and breakdown under heat. Workers in the 1980s and ‘90s found themselves surrounded by phone cords, garden hoses, flooring tiles, and even the medical tubing used in hospitals, all softened by the same plasticizer.
What Goes Into the Bottle: Technical Details You Won’t See on a Shelf
Though most people never read a chemical label, DIOP’s trade names still pop up in procurement sheets and scientific articles. Whether labeled as Bis(2-ethylhexyl) phthalate, or packaged under a factory code, the technical side boils down to purity, moisture content, and a tight control over impurities. DIOP’s chemical preparation usually follows the same cadence: reaction between phthalic anhydride and isooctyl alcohol under acidic conditions. Researchers during the mid-century fine-tuned process conditions for higher yields, gradually reducing byproducts. For all its industrial clout, anyone who has mixed chemicals in a lab knows how easily things can go sideways—a reminder that quality at the plant level means fewer headaches in the field.
Rolling With the Punches: Chemical Behavior and Tweaks
Once in the wild, DIOP mostly stays put in plastics, but over time, it can leach out, especially where heat or repeated flexing is involved. The reactivity of the phthalate group lets chemists change DIOP into other esters, expanding its portfolio or stepping toward alternatives. Early modification work focused on making phthalates safer, less bioaccumulative, but progress takes patience. Heavy industrial use means that small tweaks at the molecular level can ripple across entire product lines, affecting both manufacturing costs and long-term product performance.
Wearing Many Hats: Where You’ll Find It (And Why That Matters)
Anyone who’s pried open a tough electronics casing or untangled a vinyl garden hose has almost definitely run into DIOP. Its roots go deep in building materials, floor tiles, adhesives, and coatings. I remember my first job on a construction site, realizing the “smell of new plastic” was more science than magic. In healthcare, the same properties that protect wires and tubes once carried over into blood bags and medical gloves, raising eyebrows as concerns over leaching and patient exposure took center stage. DIOP helped make life easier—it kept products affordable and helped push modern comforts forward. The flip side? Choices about safety and sustainability often lagged behind the sheer drive to innovate.
Keeping an Eye on Safety: Standards, Limits, and Lessons Learned
Safety concerns surrounding DIOP didn’t emerge out of thin air. Mounting data from lab tests led governments and regulatory agencies to tighten exposure limits. Reports linked certain phthalates to hormone disruption, particularly in children. Standing in a factory years ago, I watched safety teams carefully log hours and personal protective equipment use as part of updated operational standards. In the EU and US, tighter labeling rules now reflect years of toxicological research, and more companies post up-to-date Material Safety Data Sheets for transparency. Reducing direct worker exposure, improving ventilation, and limiting product categories have turned into practical steps in modern plants. For manufacturers, staying up to date with shifts in safety regulations has become a necessity rather than an option.
The Search for Something Better: Research and Development
The enormous industrial hunger for plasticizers hasn’t died out; it’s only morphed. University labs, startup firms, and global corporations have poured resources into finding less hazardous options that maintain the flexibility and workability people expect. Over years of browsing patent filings and research briefs, I’ve seen a steady stream of alternative esters, bio-based plasticizers, and clever polymer blends, but replacing DIOP’s unique performance remains a tall order. Companies that built their market share on legacy plasticizers now face the challenge of retrofitting old recipes to fit tighter environmental standards.
Unpacking Toxicity and Long-Term Impact
The story of DIOP’s health implications reads like a case study in how science and regulation can tug in different directions. Animal studies raised red flags for reproductive and developmental toxicity, prompting deeper epidemiological reviews. Some researchers tracked DIOP metabolites in urine, linking phthalate exposure to negative health outcomes, particularly in vulnerable groups. Tension grows between industries that depend on these additives and public health advocates demanding stricter oversight. In my own experience, efforts to limit school and daycare exposure have pushed innovation—not just in finding new materials but in redesigning entire supply chains. Clean-up costs, health monitoring, and legal settlements have moved the conversation away from dismissive attitudes toward deeper engagement with environmental justice.
Looking Forward: Prospects in a Changing World
The future of DIOP sits at an inflection point. Mounting public scrutiny and regulatory shifts have already spurred major changes in how it gets produced, handled, and disposed of. Still, economic realities don’t bend easily. The pressure to balance performance, cost, and safety leads to tough choices for manufacturers and product designers. Running a smaller lab project years ago, I saw firsthand how developing a new, less-harmful plasticizer could take years between idea and rollout—and the most innovative solutions often stalled when scaled to industry size. Hope lies in collaborative efforts between chemists, toxicologists, manufacturers, and community advocates to push boundaries further. Product reformulation, new safety standards, and real investment in green chemistry all carry weight. At the same time, building public trust means facing the history of industrial shortcuts and making choices rooted in both science and social responsibility. Any future with safer plastics depends not just on breakthrough compounds but on honest, open communication between everyone with skin in the game.
What is Diisooctyl Phthalate used for?
Understanding the Role of Diisooctyl Phthalate
Walk through any big box store. Take in the toys, coated wires, vinyl flooring, even the rain boots near the entrance. Chances are, many of those products wouldn’t feel the same without something called Diisooctyl Phthalate, or DIOP. You’ll find it on ingredient lists in many flexible plastics. Factories add it to soft PVC to keep it bendy, durable, and tough enough to handle the rough world of daily use.
Everyday Items and Its Hidden Influence
Flexible plastics owe much of their texture and resilience to DIOP. Extension cords and appliance wiring rely on this chemical to stay pliable and to prevent cracks, especially if they end up tangled or exposed to cold garages. Fake leather in bags and car interiors also counts on DIOP for that unmistakable soft, smooth touch that stops seats from feeling stiff and cheap. Shower curtains, pool toys, and certain sneaker soles all bring this chemical into homes without most people ever thinking about it.
Health and Environmental Concerns
Recent studies and headlines haven’t ignored phthalates like DIOP. Researchers have found links between some phthalates and hormone disruption. The science keeps evolving, but concern runs deep, especially for babies and young kids who chew on plastic toys. Plastics can leach small amounts of chemicals, and even tiny exposures raise big questions over time.
Stores and regulators have started to pay attention. The European Union has taken steps to limit or ban certain phthalates for children’s products and food packaging. In the United States, similar pressure comes from consumer advocacy groups and health experts. California’s Proposition 65 list includes DIOP among chemicals that may cause reproductive harm, which prompts product labels and pushes companies to rethink what they use.
Industry Perspectives and the Search for Alternatives
Factories want plastic that bends but doesn’t break, especially for wire coatings, construction materials, and transportation gear. For many years, DIOP filled that need at a reasonable cost with solid performance. Yet, the demand for safer materials can’t be ignored. Scientists have developed other plasticizers that claim fewer risks, like DOTP or DINCH, and bigger brands try switching—sometimes because of legal mandates, but often because shoppers ask harder questions at the checkout line.
The cost to swap out DIOP for another ingredient can be steep. Tooling and machinery already set up for DIOP might not work the same way with a different chemical. New formulas mean new rounds of testing for heat resistance, strength, and surface feel. It takes time, money, and a willingness to accept that not every substitute will feel exactly like the old standbys.
Looking Ahead
DIOP powered a plastics industry focused on flexibility and affordability. Pressure from watchdog groups, new studies, and tougher regulations now drive a search for safer choices. Plenty of rooms and cars from the past few decades have traces of DIOP in their floors and seats. As science and policy move forward, so does the quest for balance: keeping flexible plastics that work well, but with an eye toward what long-term health and safety really mean.
Is Diisooctyl Phthalate safe to use?
What People Use Diisooctyl Phthalate For
Diisooctyl Phthalate, known as DIOP, helps make plastic soft and flexible. Companies use DIOP in products like cables, flooring, toys, and even coatings for pills. Walk through a hardware store, touch that soft vinyl extension cord, and odds are DIOP played a role in its flexibility.
Concerns Over Health
Conversations about DIOP's safety have grown louder in recent years. Studies show the body doesn’t keep phthalates around for long; most break down and pass out in urine. Finding traces of phthalates in urine across the general population started raising questions.
Repeated exposure—at home or on the job—could cause trouble. Animal research points to hormone changes and fertility problems. Regulators in the US and Europe often put DIOP and its cousins in the same boat as other phthalates that show these health effects.
The Centers for Disease Control and Prevention (CDC) monitored phthalate levels in people for years. Scientists didn’t see dramatic health effects at common exposure levels, but the window between “fine” and “too much” remains unclear, especially for kids. Younger bodies absorb and process chemicals differently than adults, so stuff that seems minor to grown-ups could weigh heavier on children.
Consumer Goods and Vulnerable Groups
Phthalates including DIOP show up most in plastics meant to be flexible or coated. Items like raincoats or air mattresses might have these softeners. In toys or objects babies touch or chew, the story shifts. Children’s developing bodies face more risk from chemicals that interfere with hormone balance.
The European Union took action by restricting certain phthalates from children’s products. The US limited the use of several phthalates, including DIOP’s close relatives, in similar items. These moves came after evidence found some phthalates in blood and breast milk, raising questions worth closer review.
Worker and Environmental Issues
Factories where people handle phthalates like DIOP face separate challenges. Workers breathing or touching concentrated forms risk higher exposure. Over time, chronic exposure raises health concerns, even as modern factories run with more safety gear and better air systems than decades ago.
DIOP doesn’t just stay where it’s put. Plasticizers can leach out of finished products, especially with heat or over long storage. Once in soil or water, DIOP doesn’t break down very fast. Wildlife can run into these chemicals, and fish with DIOP in their tissue turn out less healthy.
How Safer Choices Happen
A simple solution isn’t always in reach. Industries look for replacements that offer the same flexibility without potential hormone disruption. I’ve seen companies try plant-based plasticizers. These cost more, but the price drops as factories use them in larger batches.
Recycling and safer disposal of plastic waste help limit DIOP’s spread. Label reading empowers families, letting people choose goods free of certain chemicals. These changes sound small but can cut exposure, especially where children or pregnant women spend time.
Better Regulations and Ongoing Research
Researchers and regulators keep testing DIOP and its alternatives as new data comes in. More long-term studies, honest labeling, and updated safety standards put people’s health first. Scientists still have gaps in understanding, but as families and companies aim for safer options, public health improves step by step.
What industries commonly use Diisooctyl Phthalate?
Everyday Plastics Rely Heavily on This Additive
Anyone who’s picked up a squeeze toy, stretched a garden hose, or leaned back into the vinyl seat of a car has probably gotten a little too close to Diisooctyl Phthalate (DIOP) without even knowing it. From my own experience working in a home improvement store summers during college, I saw pallets of vinyl flooring, wall claddings, and outdoor tarps—most of them had a distinct, plastic-like scent. That’s not just “new plastic.” Much of it comes from the kind of additives manufacturers blend into these products to make them softer, and DIOP fits right into this story.
DIOP belongs to the phthalate family, which gets blended into polyvinyl chloride (PVC) and other plastics. At its core, it helps plastics stay flexible, which changes everything about how products look, feel, and perform. If you ever tried to bend old-fashioned plastic without additives, you’d either snap it or get little more than a stiff bend. That just won’t cut it where flexibility means comfort, safety, or usability.
Industries Banking on DIOP
Let’s talk about construction first. Vinyl flooring, synthetic leather, window frames, and insulation use huge volumes of DIOP. Contractors like building materials that hold up to years of wear and tear, moisture, and shifting temperatures. The stuff works because it keeps the plastic supple. It’s not just sticking to one part of the build either—everything from wire insulation to waterproof coatings needs some give, and DIOP makes that possible.
Automotive makers lean on DIOP for dashboard covers, car seats, and door panels. Sitting in a car from the late ‘80s with cracked vinyl provides a clear lesson. Modern cars with flexible, long-lasting interiors owe their comfort to precisely these kinds of chemical helpers.
The medical field uses tubing, blood bags, and IV lines made from flexible PVC. Hospitals want gear that bends, twists, and stays put in unpredictable ways. DIOP can be found on that side, too, but not so much these days. With new research surfacing about phthalate health risks—especially for children and pregnant women—hospitals and manufacturers started swapping out DIOP for less controversial options. The phase-out is in progress, but some clinics, especially in regions with stricter budgets, still rely on it.
Footwear makers produce slippers, sandals, and boots that require plastic to be more than just tough—they need to be soft and forgiving. A few large-scale shoe factories still use DIOP-based plasticizers for that squishy, comfortable feel underfoot. In wet-weather regions, raincoats and waterproof boots also benefit because DIOP brings durability to the table.
Public Health and Safer Alternatives
The importance of DIOP goes beyond just technical performance. Over the last decade, experts at the CDC and EPA questioned long-term exposure, since some research connects phthalates to hormonal changes and developmental concerns. Even without all the answers, regulators and consumer advocates say, “Better safe than sorry.” This real-world concern pushed industry leaders toward using alternative chemicals. Some companies test out bio-based plasticizers and claim similar softness, but it takes time and money to upgrade equipment and materials.
As more people call for transparency and safety in manufacturing, industries keep retooling. The move away from DIOP in sensitive applications shows that markets can adapt under pressure from both customers and science. Today, choosing the right chemical in plastic production isn’t just about cost or durability; it’s about health, safety, and environmental stewardship on the factory floor and beyond.
What are the physical and chemical properties of Diisooctyl Phthalate?
Physical Traits You Notice Right Away
Diisooctyl phthalate usually comes in a clear, oily liquid form. It doesn’t have much of a color—just a faint yellow tint in some cases. You won’t smell anything noticeable from it either. What stands out is its slipperiness and how slowly it moves when you pour it, pointing to its rather high viscosity. Touching it feels a lot like handling other common liquid plasticizers: slick, a little greasy, and just dense enough to feel significant in the palm.
The liquid won’t dissolve in water. Once, I tried cleaning a small spill with just water, which did nothing except make a mess. Diisooctyl phthalate only gives in to organic solvents, like alcohols or oils. This stubborn non-mixing behavior makes it prized by folks working on synthetic polymers and PVC. If you leave it open in a warm room, the liquid takes ages to evaporate. That low volatility matters because it means products made with DOIP tend to last longer and don’t become brittle as quickly. As a side point, its boiling point sits above 380°C, which tells you it’s stable in tough conditions.
How It Behaves Chemically
Chemically, diisooctyl phthalate stands out for its stability. The molecule won’t fall apart or react with just anything nearby. I’ve seen how it refuses to break down even after years inside vinyl products exposed to sunlight and heat. People in the chemistry field know this phthalate for being good at holding up against acids and common oxidizers. It won’t change under everyday environmental exposure, barring some extreme scenarios involving intense heat or aggressive chemicals that are not easy to run into by accident.
Its structure as an ester—a product of phthalic acid and isooctanol—makes it flexible in plastic formulations, but that same structure creates trouble for environmental health. When disposed of incorrectly or left to break down in landfills, traces can work their way into water or soil. I’ve bumped into studies showing low rates of biodegradation. Microorganisms in soil don’t break DOIP down quickly, so any release in the environment tends to stick around. This raises concerns for eco-safety and justifies the growing call for tighter handling rules.
Why These Properties Matter
If you work in plastics or rubber, the physical qualities of diisooctyl phthalate—its viscosity, oiliness, and non-volatility—stand out as key factors when choosing a plasticizer. Nobody wants vinyl cables that dry out after a year, so long-lasting blends get the job. The tough chemical profile makes processing easier and helps finished goods withstand day-to-day abuse.
On the downside, that same toughness means the environment struggles to break it down. Alarming studies from the past decade link phthalate exposures to possible health risks, including hormone disruption in kids. The science around exact risks is still developing, yet enough credible voices from health and environmental agencies have flagged the need to watch and limit widespread use. After years in the lab and talking with people on factory floors, I’ve seen more companies pushing for safer substitutes, especially in toys and food packaging.
Solutions and Safer Approaches
Switching out diisooctyl phthalate isn’t easy for many manufacturers. The industry has started to use alternative plasticizers that break down faster and don’t pose similar health hazards. Switching to these options means changing recipes, re-testing products, and sometimes accepting higher costs. That said, consumer pressure and evolving regulations have pushed some of the biggest players to make this investment.
Safer waste handling helps too. In my time visiting industrial plants, real progress came after they started recycling and treating liquid waste properly. Closed-loop systems, leak detection sensors, better training for workers—it’s not glamorous work, but it all adds up to less phthalate reaching the landfill and water sources. Until science brings even better substitutes, sticking with careful use, smart design, and responsible disposal helps limit the risks tied to diisooctyl phthalate.
Are there any alternatives to Diisooctyl Phthalate?
The Challenge with Diisooctyl Phthalate
Diisooctyl phthalate, or DIOP, has been a backbone in plastic manufacturing for years. Its softening effect helps create flexible vinyl, from flooring to toys. Concerns have grown after years of research tied phthalates to hormone alterations and health risks, especially for children. Years spent working with consumer goods taught me how quickly regulations can shift when public health is on the line. Many manufacturers felt the squeeze to change formulations almost overnight.
Health and Environmental Pressures
The shift away from phthalates didn’t happen in a vacuum. Pressure from health experts pushed legislators around the world to rethink what goes into plastics near babies or food. It's easy for decision-makers to sideline risks that seem distant, but parents want peace of mind. Phthalates show up in human blood, urine, and even breast milk, and that’s tough to write off as meaningless. As a parent, nothing feels more urgent than keeping unnecessary chemicals away from family.
Choosing Alternatives: What’s Out There
Chemical firms responded with a surge of research. Over the years, I’ve spoken with engineers who watched entire labs switch gears to hunt for safer additives. Right now, people look to choices like DOTP (dioctyl terephthalate), DINCH (1,2-cyclohexane dicarboxylic acid diisononyl ester), and bio-based plasticizers like epoxidized soybean oil or citrate esters. DOTP’s lower toxicity and solid performance win it a lot of attention in consumer goods, especially in the European Union where restrictions stay tight. DINCH tends to turn up in medical supplies and kids’ products for the same reason—health standards set a high bar.
Bio-based options feel promising. I remember a case from a midsize toy company where they swapped to citrates and plant oils, landing on an FDA-accepted solution that sidestepped complicated hazard labels. Sustainable production has economic upsides too. Oil-based chemicals tend to fluctuate in price, while bio-based inputs, especially waste stream oils, keep things steadier.
Barriers and Real-World Hiccups
Switching isn’t a walk in the park. Suppliers face real obstacles—cost, availability, and compatibility. Nearly every factory manager tells the same story: new plasticizers sometimes mess with softness, transparency, or toughness. If a cable’s jacket cracks in the cold, the whole batch gets scrapped.
Some buyers find alternatives more expensive, especially at first. Few companies want to eat those costs unless forced. But disasters, lawsuits, and recalls get expensive, too. Trust takes a long time to rebuild after a product recall over something like phthalates.
How to Succeed: Teamwork and Testing
Progress often happens when everyone in the supply chain works together. Designers, chemists, and purchasing staff need room to test, fail, and try again. Regular third-party lab tests catch problems before products ship. Big brands with the resources to run large-scale tests often lead by example, helping smaller businesses learn faster.
The world’s demand for plastics isn’t dropping any time soon. Taking DIOP out of circulation gives kids, workers, and the environment a break from chemicals their bodies never evolved to handle. Direct, persistent effort—not waiting for regulations—shapes a safer future. That’s a win worth the trouble.
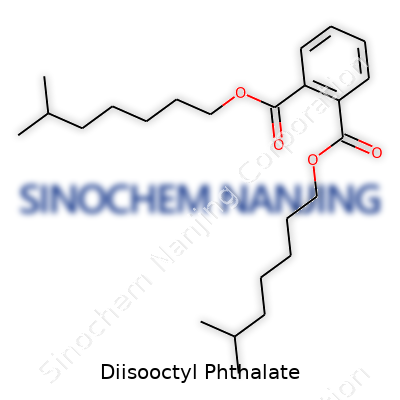

| Names | |
| Preferred IUPAC name | bis(7-methyloctyl) benzene-1,2-dicarboxylate |
| Other names |
Bis(2-ethylhexyl) phthalate DEHP DIOP Di-2-ethylhexyl phthalate |
| Pronunciation | /ˌdaɪ.aɪˈsuː.ɒk.tɪl ˈθæleɪt/ |
| Identifiers | |
| CAS Number | 117-81-7 |
| Beilstein Reference | 1461113 |
| ChEBI | CHEBI:8346 |
| ChEMBL | CHEMBL1599118 |
| ChemSpider | 7576 |
| DrugBank | DB13895 |
| ECHA InfoCard | ECHA InfoCard: 100.015.456 |
| EC Number | 204-214-7 |
| Gmelin Reference | 8982 |
| KEGG | C02450 |
| MeSH | Dioctyl Phthalates |
| PubChem CID | 8343 |
| RTECS number | TI0350000 |
| UNII | BB76Q9V6RC |
| UN number | UN3082 |
| CompTox Dashboard (EPA) | DJZFTQKIIXGBDT-UHFFFAOYSA-N |
| Properties | |
| Chemical formula | C24H38O4 |
| Molar mass | 390.56 g/mol |
| Appearance | Colorless oily liquid |
| Odor | Faint odor |
| Density | 0.982 g/cm3 |
| Solubility in water | insoluble |
| log P | 8.3 |
| Vapor pressure | 0.00001 mmHg (25°C) |
| Acidity (pKa) | pKa ≈ 2.76 |
| Basicity (pKb) | basicity (pkb) : 5.02 |
| Magnetic susceptibility (χ) | -70.5e-6 cm³/mol |
| Refractive index (nD) | 1.484 |
| Viscosity | 40-76 cP (25°C) |
| Dipole moment | 2.70 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 817.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1227.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −12,820 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D02AC09 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H413: May cause long lasting harmful effects to aquatic life. |
| Precautionary statements | H280, H315, H319, H332, P210, P261, P271, P280, P305+P351+P338, P337+P313, P403+P235 |
| NFPA 704 (fire diamond) | NFPA 704: 1-1-0 |
| Flash point | 210 °C |
| Autoignition temperature | 385°C |
| Lethal dose or concentration | LD50 (oral, rat): > 30,000 mg/kg |
| LD50 (median dose) | > 30 g/kg (oral, rat) |
| NIOSH | TNF23250* |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Diisooctyl Phthalate is "5 mg/m³ (as TWA)". |
| REL (Recommended) | 5 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Dimethyl phthalate Diethyl phthalate Dibutyl phthalate Diisononyl phthalate Diisodecyl phthalate Bis(2-ethylhexyl) phthalate (DEHP) Benzyl butyl phthalate Di-n-octyl phthalate |

