Diisobutylamine: A Practical Look at Its Role and Implications
Historical Development
Diisobutylamine has a background rooted deeply in the ongoing exploration of organic synthesis during the early-to-mid 20th century. Scientists searching for efficient pathways toward amines marked out the branched-chain alkylamines—among them, diisobutylamine. This compound captured the attention of chemical manufacturers soon after its introduction on the scene, especially as petrochemical sources grew more diverse and available in the wake of expanding refinery technology. The spread of custom chemical synthesis in the years after World War II meant researchers and industrial operators had a real interest in streamlining the process for producing substances like diisobutylamine. Over time, targeted improvements in catalysis, separation, and purification have made diisobutylamine more accessible for specialized uses.
Product Overview
Diisobutylamine stands out as an organic amine, with a branched structure. Its reputation comes from its versatility in organic transformations and intermediate chemistry. The clear, colorless liquid gives off a strong amine smell, typical in this chemical family. Manufacturers produce it with purity levels dictated by end-user demands—laboratory-grade batches differ from technical grades due to stricter thresholds for byproducts or trace contaminants. The persistent demand from sectors like agrochemicals, pharmaceuticals, and rubber processing ensures its continued place in chemical inventories.
Physical & Chemical Properties
In a typical lab or plant environment, diisobutylamine presents as a clear, volatile liquid. It resists water but dissolves well in common organic solvents. The molecule features a secondary amine nitrogen, meaning reactivity shows up in areas like acid-base behavior, alkylation, and even reduction reactions with metal catalysts. Its boiling point falls in a moderate range, making it manageable during distillation yet still subject to loss through evaporation if left exposed. As with related amines, the notable odor is hard to miss and requires thoughtful ventilation, particularly in enclosed spaces.
Technical Specifications & Labeling
Safety and regulatory bodies expect clear, accurate labeling for chemicals like diisobutylamine due to the potential hazards associated with human exposure. Industrial operators need to know concentrations, purity levels, and the relevant safety icons at a glance—critical for both compliance and in avoiding on-the-job mistakes. Exact technical specs arise from the intended application: someone working on pharmaceutical intermediates tracks trace metals or moisture more tightly than someone using it in bulk rubber additives. Transport regulations also impact how it’s packaged, with drum or bulk tanks labeled with standardized transport information to reduce risks during shipment.
Preparation Method
Organic chemists tend to use reductive amination when synthesizing diisobutylamine. By reacting isobutyraldehyde or similar branched aldehydes with ammonia or isobutylamine in the presence of a reducing agent, they steer the reaction toward the secondary amine product rather than stopping at the aldehyde or moving on to more branched tertiary amines. Industrial processes refine this pathway—streamlining separation and integrating catalysts that both protect yields and reduce unwanted byproducts. Site operators keep a close eye on clean-in-place systems and reaction byproducts, since keeping purity high makes downstream reactions perform more reliably.
Chemical Reactions & Modifications
Diisobutylamine steps into a variety of reactions, often as a base or as a precursor to more complex molecules. It picks up electrophiles readily on the nitrogen, allowing for alkylation or acylation to yield substituted amines. In my experience working with amine-based reagents, I’ve noticed how secondary amines like diisobutylamine can sometimes outcompete primary amines in selectivity, especially in reductive amination pathways. This opens up options in custom synthesis, particularly in pharmaceutical or agrochemical research. At the same time, it serves as a precursor in making numerous specialty chemicals, from corrosion inhibitors to rubber processing agents. Chemists often appreciate the molecule’s balance between reactivity and selectivity.
Synonyms & Product Names
Diisobutylamine appears under a variety of synonyms across catalogs and chemical registries. Some call it 2,6-dimethylheptan-4-amine, reflecting its structure. Many times, it gets abbreviated as DIBA, especially in the shorthand of the laboratory. This scattering of names can lead to mix-ups or overlook critical safety information. I’ve personally run into confusion when comparing suppliers, so it pays to double-check registry numbers, structural formulas, or specified CAS numbers. This kind of attention becomes even more important with similar-sounding chemicals, which might differ greatly in behavior or hazard.
Safety & Operational Standards
Handling diisobutylamine requires sensible precautions. Its strong smell signals volatility and health risks—prolonged exposure can irritate skin, eyes, or respiratory tract. In industrial and lab settings, we lean on sturdy gloves, splash-proof goggles, and local exhaust ventilation to keep contact minimal. I recall a colleague missing proper face protection during a transfer; eye exposure led to immediate discomfort and a trip to the eye wash. Standard operating protocols treat diisobutylamine with the respect given to reactive amines: keep it away from oxidizers, acids, and sources of ignition. Spill kits and neutralizing agents must sit close at hand. Safety Data Sheets provide all actionable steps for cleanup and first aid, and periodic drills are not just formality but make a real difference during accidents.
Application Area
The range for diisobutylamine stretches across sectors. Agrochemical manufacturers rely on it as a building block for active ingredients in herbicides. The pharmaceutical industry uses it as an intermediate, especially where a branched amine helps set the molecular shape and function of a drug candidate. Rubber processing benefits from additives based on diisobutylamine, which tweak physical properties and improve process efficiency. In each of these roles, the compound brings something unique—be it structural variety, reactivity, or compatibility with particular synthetic routes. Custom syntheses in research settings take advantage of the compound’s secondary amine group for tailored modifications, seeking to build complexity into product molecules. I’ve encountered it, too, in water treatment formulations and as a corrosion inhibitor in pipelines, where its balance of reactivity and stability translates into long-term performance.
Research & Development
Academic and industrial labs remain busy digging deeper into the reactivity and new uses of diisobutylamine. A lot of work focuses on greener synthetic techniques, whether by switching to milder reductive agents or pursuing enzyme-directed pathways. Research also probes selective functionalization, using modern catalysts or new solvent systems to open up new routes for value-added derivatives. One challenge is improving efficiency without boosting waste, and it’s been promising to see reports on solvent recycling and single-step conversions. In my own work with research teams, I’ve seen keen interest in partnering with universities for pilot-scale testing, translating benchtop ideas to industrial realities. This kind of collaboration doesn’t just accelerate discovery but brings practical checks on cost, safety, and performance.
Toxicity Research
Toxicological data on diisobutylamine calls for careful reading. Animal studies show that concentrated exposure irritates mucous membranes and in some cases produces systemic effects at higher doses. Still, compared to some aromatic amines or those with functional nitro or halogen groups, diisobutylamine’s profile does not point to severe chronic toxicity, yet the picture remains incomplete. A lot of workplace handling recommendations draw from general amine toxicity, erring on the side of caution due to known acute effects. I’ve seen cases where lab safety training glosses over secondary amine risks, underscoring how critical it is to review up-to-date data before starting new projects. Environmental data suggests moderate persistence in soils and waterways, fueling studies into more targeted mitigation strategies and better detection in effluent streams.
Future Prospects
Looking ahead, diisobutylamine’s prospects appear tied to shifts in chemical manufacturing, environmental regulations, and innovation in its downstream applications. As developers look for new processing aids, especially those that comply with tighter emission controls and sustainability goals, secondary amines like diisobutylamine find fresh attention. Improvements in recycling and recovery processes could give manufacturers an edge, reducing loss and boosting overall economic value. There’s another angle in new catalytic systems that promise more selective transformations, opening options for synthesizing complex molecules from basic amine precursors. I see real potential in tying research into toxicological effects more closely with regulatory changes, ensuring adoption of best practices not just for short-term worker safety but also for long-term environmental stewardship. Investments in R&D—supported by academic partnerships, smart regulation, and a willingness to rethink established routes—stand to make a difference both in how we use diisobutylamine and in minimizing its environmental footprint.
What is Diisobutylamine used for?
The Role of a Specialized Chemical
Diisobutylamine doesn’t come up in daily conversations, unless your day job involves chemistry, pharmaceuticals, or rubber. It’s not a household item you find under the kitchen sink, but it quietly supports a range of processes that affect everyday life. My early work in a chemical plant introduced me to this compound in a hands-on way, and its impact stuck with me.
Fine-Tuning Pharmaceuticals
Pharmaceutical manufacturing relies on a broad toolbox of building blocks, and Diisobutylamine makes a solid showing here. In this field, small mistakes cost more than money—they risk lives. As a key intermediate, it helps form a number of active drug ingredients, especially by easing the way toward precious nitrogen-containing compounds. The methods using Diisobutylamine save time and reduce unwanted byproducts, which helps drug makers stay on the right side of both ethics and the law. Years of process optimization in this area have taught me that safety, purity, and efficiency move companies and, ultimately, protect patients.
Lubricants and Rubber
Rubber production stands out as another area where Diisobutylamine proves its worth. My contacts in the automotive supply chain know all too well that a reliable accelerator can make or break timelines for tire manufacturing. Diisobutylamine steps in as an ingredient for age-resisting chemicals and accelerators, giving tires and seals the durability to survive rough roads and weather swings. In my own repairs, the difference between quality and cheap rubber runs deeper than looks; it’s about materials science, and additives like this one play a central role.
Pesticide and Agriculture
Agriculture carries its own burden of responsibility—balancing crop protection with environmental safety is never simple. Pesticide companies use Diisobutylamine when developing specific fungicides and herbicides. It’s not a magic bullet by itself, but it shapes chemical formulas designed to clear fields of invasive species. This chain of use has ripple effects: higher crop yield, less wasted land, steadier food prices. I grew up in a rural area where families depended on harvests. Fewer variables in farm chemicals can translate to more predictable seasons and incomes.
Engineered Resins and Beyond
The world demands stronger, lighter, and more resilient plastics for electronics, packaging, and industrial parts. Diisobutylamine matters here as well, feeding into the production of custom resins. These materials don’t just fill shelves in warehouses—they form the bodies of our tech, our cars, even simple objects like water bottles and food containers. Engineers and designers can point to advances in these materials as real improvements in product reliability and lifespan. My own workbench projects rarely tackle such advanced polymers, but I’ve seen firsthand the frustration old, brittle plastics bring compared to newer, tougher blends.
Responsibility and Innovation
Chemicals like Diisobutylamine require respect in handling due to their potential hazards. With stricter safety standards and growing scrutiny on environmental effects, producers have worked to keep exposures controlled, especially in manufacturing plants. Current best practices include closed systems and real-time sensors, a big evolution from the open beakers of my early days. Pushing for more innovation here continues to matter, whether through safer substitutes in synthesis or better recycling and waste capture.
Looking Forward
Many overlooked chemicals shape shifts in technology, healthcare, and resource management. Diisobutylamine doesn’t get the headlines, but its careful use underpins products most people rely on. As industries work through challenges like sustainability and cost, paying close attention to these supporting materials lays the groundwork for safer, cleaner, and more consistent results. Supporting transparency in sourcing and responsible production of such compounds serves not just the industries but the greater good. My experience reminds me that some of the most important ingredients aren’t just in the recipe—they’re part of the system itself.
What are the safety precautions when handling Diisobutylamine?
Understanding the Risks
Diisobutylamine lives in the chemical family where careless handling can lead to trouble for both people and the environment. It’s a colorless liquid, but don’t let looks fool you; the fumes burn your eyes, and a splash can leave your skin itchy or even blistered. Breathing vapors tends to bring headaches, dizziness, or worse in high concentrations. My years working in a university chemistry lab taught me to respect liquids like this. Once, a cracked storage cap released a strong, fishy odor that made my coworker’s eyes water instantly—a reminder that one simple mistake can lead to a nasty day.
The Value of Personal Protective Equipment
Lab coats and gloves aren’t just for show. You want chemical-resistant gloves—nitrile does well here—and safety goggles. Face shields give extra insurance. If you skip protection, even one small spill can leave your skin tingling for hours. Long sleeves cover vulnerable arms. Anyone who’s ever splashed a caustic liquid on a bare wrist knows how memorable that sting feels, and not in a good way.
Ventilation Keeps Air Safe
Good air flow helps keep fumes moving out and fresh air coming in. Fume hoods deserve their reputation. I remember opening a bottle outside the hood, thinking the smell wouldn’t reach far. Within minutes, the entire lab filled with complaints and the building manager stopped by to check on us. Lesson learned: Fume hoods keep chemical vapors out of noses and lungs. Never try shortcuts here.
Spill Response
Spills happen, no matter how careful you are. Quick action makes a difference. If liquid splashes, using absorbent pads made for chemicals, along with neutralizers, stops the spread. Sand and vermiculite mop up puddles, but never let the waste touch unprotected skin. Proper disposal matters because some chemicals react with drains or ordinary trash. Always place contaminated materials in clearly marked, sealed containers destined for hazardous waste management—never the regular bin.
Safe Storage Isn’t Optional
Store diisobutylamine in a cool, fire-safe cabinet, away from acids and oxidizers. I’ve seen cabinets where incompatible chemicals wound up crammed together, and that negligence cost the facility both money and peace of mind after a small fire. Label everything clearly, and always check for tight, uncracked lids. Even a small leak can affect indoor air quality for days.
Training Builds Confidence
Reading a safety data sheet builds a knowledge base, but real training drills procedures into muscle memory. I’ve watched folks freeze when alarms go off—so ongoing practice with spill kits and eye wash stations keeps response swift and correct. Make sure new team members shadow experienced workers before handling chemicals. Confidence comes from both information and hands-on practice.
Solutions Make a Safer Workplace
Safer habits start with awareness and the right tools. Companies and schools need a culture where staff call out shortcuts and enforce rules, rather than hope for the best. Frequent checks on protective gear, ventilation systems, and storage conditions—plus regular review of emergency procedures—prevent most accidents. Chemical safety isn’t about avoiding work; it’s about sending everyone home with their eyesight and good health intact.
What is the chemical formula and structure of Diisobutylamine?
Understanding Diisobutylamine: A Look At The Basics
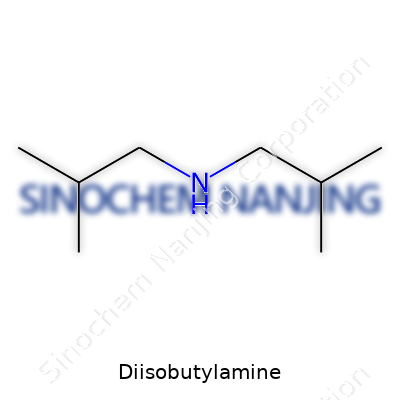
Diisobutylamine stands out in the family of organic amines. Chemically, it comes together with the formula C8H19N. On paper, this seems simple: eight carbons, nineteen hydrogens, and a single nitrogen. But looking at the molecule sheds more light on its character. The name gives away a lot—
Diisobutylamine holds two isobutyl groups tied to a nitrogen atom.The structure looks like a nitrogen atom sitting in the middle, holding hands with two isobutyl chains (each isobutyl made from three methyl groups hanging off a central carbon), leaving one spot on nitrogen open, usually for a hydrogen atom. The connectivity forms this:
- (CH3)2CHCH2—NH—CH2CH(CH3)2
That pattern creates a branched, compact layout, not a long chain. Those branches affect how the molecule mixes and reacts with other chemicals.
Why Structure Matters In Everyday Chemistry
This shape gives diisobutylamine its personality. Years spent handling organic compounds in labs give me a deep respect for how branching in a molecule can impact behavior. With diisobutylamine, the arrangement of the isobutyl groups makes it less likely to sit tightly next to its neighbors, which means it tends to be more volatile and less likely to dissolve easily in water.
Data from safety sheets and chemical suppliers confirms diisobutylamine forms a colorless liquid with a strong, fishy odor familiar in amine chemistry. Its boiling point sits up around 158°C (316°F), much higher than simple compounds like ammonia, thanks to those bulky side chains.
Why Getting the Formula Right Is Crucial
As a chemist, getting even a small detail wrong can cause headaches down the road. A misplaced methyl group changes not only the formula but also the reactivity and, crucially, the risk factors. Diisobutylamine finds its way into all sorts of specialty syntheses in the agrochemical and pharmaceutical industries. Its formula and branched structure make it a candidate for selective reactions where less-branched amines either move too quickly or can’t dodge unwanted byproducts.
Safety comes into play here. Data from the European Chemicals Agency flags diisobutylamine as an irritant, demanding careful ventilation and gloves in the lab. Mislabeling the structure could lead operators to use the wrong procedures, putting health at risk or affecting the quality of the final product. Lab memories remind me how common it is to see mix-ups between isobutyl and n-butyl groups on chemical order forms, a mistake small on paper but big in its impact on results and safety.
Community Building Knowledge and Solutions
Open sharing of accurate information inside the chemistry community lowers those risks. Peer-checked online resources like PubChem keep diagrams current, while trade associations host webinars on chemical handling and purity standards, which pushes safer and more reliable lab work. Efforts from industrial stakeholders have led to barcoding and batch tracking, which make it easier to keep tabs on the use and movement of compounds like diisobutylamine throughout their journey from factory to bench.
Clear communication remains the most reliable solution. Calling a molecule by its right name, formula, and shape allows everyone—from students to seasoned chemists—to speak the same language. This gets more important every year, as supply chains stretch out and demand grows. No room for guessing games in a business where mistakes can cost real money or safety.
How should Diisobutylamine be stored?
Understanding the Real Risks
Diisobutylamine rarely grabs headlines, but its storage raises questions worth talking about. I once worked in a lab where amines, including diisobutylamine, showed up on the shelves near everything from cleaning supplies to random equipment. The pungent odor was hard to ignore, and an old drum with a warped seal sat collecting dust. Across industries, lab techs and warehouse workers sometimes leave safety as the last item on the list. It may look like just another clear liquid, but diisobutylamine has enough punch to demand more respect.
Reliable Storage Steps
One core principle: keep the chemical in tightly sealed containers, ideally original packaging. Even a minor spill or leak can spread plenty of fumes. Gaskets and drum lids need a regular check, not just a quick glance during inventory.
Diisobutylamine reacts if exposed to acids, oxidizers, and moisture. Mixing fumes can set off fires or create toxic products. Flammable storage cabinets step up safety. Only trained staff should open these containers, wearing gloves, goggles, and sometimes even a face shield. When I handled this compound, I kept it far from acids, sulfur, and possible oxidizers — trust me, the memory of a minor splash still plays on my mind.
The storage location needs good ventilation. Any whiff that escapes means trouble isn’t far. In one poorly ventilated room, even a small mishap became a big deal after lingering odors set off complaints. Preventing vapor buildup is not just about comfort—it keeps people healthy and stops fire hazards before they start.
Temperature and Light Control
Forget open windows or exposure to sunlight. Diisobutylamine breaks down faster when hit by heat or UV rays. Temperatures closer to room level work best. I’ve heard of barrels left near heat vents or windows, only to see yellowing labels and a corrosive ring under the drum. Keep containers out of direct sun and away from sources of heat, including space heaters and engine rooms.
Fire suppression nearby isn’t optional. With a low flash point, diisobutylamine ignites easier than folks expect. Grounding metal drums against static sparks is a simple step, just like you’d do before filling a gas can. Everybody thinks it won’t happen—until one day, it does.
Protecting Workers Every Step of the Way
Training makes the difference. Each new worker at our site got a walkthrough on handling volatile organics. We practiced what to do in a spill, where to find spill kits, and how to clean up using the right absorbent. Proper labels and signage matter as much as the latest PPE. Clear communication stops mistakes.
Records help too. I kept logs on opened containers, checked electrolyte levels in scrubbers, and noted when drums reached their expiration date. Regular inspections spot rusty cans, swelling lids, or fading expiration marks—simple checks that keep everyone safer.
Moving diisobutylamine safely from one place to another means using carts or carriers fit for the load. Never skip the step of double-checking container seals or securing the load before the journey.
Solutions for Safer Operations
Proper storage sits at the center of safety culture. Follow protocols every time. Invest in real training, not just posters. Use industrial-quality ventilation, flammable storage, and up-to-date personal protection. Regulations like OSHA’s Hazard Communication Standard back up these best practices for a reason—lives depend on it.
The safest storage for diisobutylamine doesn’t take extra flair or showy gadgets. Shelf life, ventilation, temperature, and chemical compatibility all play a part. Walking the walk, day after day, proves more valuable than just writing up a set of rules.
Is Diisobutylamine hazardous to health or the environment?
Everyday Risks in the Workplace
Factories often use chemicals that sound mysterious. Diisobutylamine falls into that category. People handling it in manufacturing or labs work around real risks. I’ve worn protective gloves and goggles for chemicals with similar fumes—you have to, or your eyes sting and your head throbs. Vapors from diisobutylamine get into the air quickly, leading to dizziness, coughing, or burning in your nose and throat. Getting splashed means skin burns or rashes. A spill in a warehouse will send everyone scrambling for the nearest eyewash. For folks operating in confined areas, these vapors choke without warning. In some countries, regular exposure above 5 ppm earns a swift visit from safety inspectors.
Long-Term Health Impact
Short-term exposure leaves its mark, but repeated contact hits harder. Some studies connect ongoing exposure with liver and kidney irritation. A few lab animals showed early signs of toxicity after weeks of inhalation. While research on people stays limited, anyone who has worked around industrial amines for years knows coworkers who lost their sense of smell or developed chronic lung sensitivity. Cumulative exposure could unsettle more than just comfort. You can’t always see the damage until it’s too late, and regular check-ups don’t catch everything early enough.
Environmental Costs
Diisobutylamine spills aren’t just a headache indoors. Rinsing equipment carelessly lets the residue seep into drains and eventually rivers. In water, this chemical doesn’t break down quickly. Fish and invertebrates gulp water loaded with amines, and some die off. A single major spill took out a stretch of frogs and snails near a plant in Eastern Europe a decade ago. Soil contamination affects microbes too, and cleanup costs stretch across years, with remediation rarely perfect. Communities living downstream of chemical plants know these stories aren’t rare—dead zones in creeks are the legacy.
Facts to Remember
Diisobutylamine comes with an eight-hour permissible exposure limit according to OSHA, but daily monitoring keeps slipping through the cracks in smaller factories. The chemical produces flammable vapors, so fire hazards double with health concerns. Data from the National Toxicology Program never gives this substance a clean bill—its effects linger in ecosystems, and that should raise eyebrows.
How to Tackle the Issue
Protective clothing and good ventilation lower risks for workers, but that’s only half the job. Training builds a culture where people take spills seriously—some crews run monthly drills with spill kits and checklists on the wall. Substitution stands out as a smarter path: switch to a process using safer amines where possible, trimming the hazard out entirely. Investing in closed-loop systems works; one place I toured recycled these vapors instead of venting them outdoors, cutting both workplace exposure and emissions. Mandating tougher reporting rules for chemical leaks keeps more plants honest.
Community and Corporate Responsibility
Local governments matter here. Surprise inspections help, and so does demanding regular health screenings for nearby residents. Tougher regulations around runoff and effluent standards keep companies accountable. Buying from firms that publish safety performance helps shift the market. Public pressure nudges chemical users to clean up their act before regulators threaten shutdowns. Listening to line workers about shortcuts or bad storage practices helps everyone, including neighbors downwind.
| Names | |
| Preferred IUPAC name | N¹,N¹-Dimethylpropane-1,2-diamine |
| Other names |
N-Secondary-butyl-2-methylpropan-2-amine 2-Methyl-1-propanamine, N-sec-butyl- DIBA N-(2-Methylpropyl)propan-2-amine |
| Pronunciation | /ˌdaɪˌaɪsəˈbjuːtɪl.əˌmiːn/ |
| Identifiers | |
| CAS Number | 110-96-3 |
| 3D model (JSmol) | `3D Model (JSmol) string for Diisobutylamine:` `CC(C)NCC(C)C` |
| Beilstein Reference | 1209246 |
| ChEBI | CHEBI:85144 |
| ChEMBL | CHEMBL127438 |
| ChemSpider | 62188 |
| DrugBank | DB14006 |
| ECHA InfoCard | EC#200-871-9 |
| EC Number | 204-689-0 |
| Gmelin Reference | 8223 |
| KEGG | C06168 |
| MeSH | D002938 |
| PubChem CID | 8027 |
| RTECS number | NL4020000 |
| UNII | SWF464OT1I |
| UN number | UN2249 |
| CompTox Dashboard (EPA) | DTXSID8020403 |
| Properties | |
| Chemical formula | C8H19N |
| Molar mass | 129.25 g/mol |
| Appearance | Colorless liquid |
| Odor | Ammonia-like |
| Density | 0.728 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 0.94 |
| Vapor pressure | 3.4 mmHg (20 °C) |
| Acidity (pKa) | 10.98 |
| Basicity (pKb) | 3.31 |
| Magnetic susceptibility (χ) | -7.44×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.405 |
| Viscosity | 1.2 mPa·s (20 °C) |
| Dipole moment | 1.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 253.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -132.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5212.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H302, H314, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P337+P313, P362+P364, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0-Alk |
| Flash point | 42 °C |
| Autoignition temperature | 446°C |
| Explosive limits | 1.1% - 7.5% |
| Lethal dose or concentration | LD50 oral rat 2880 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 409 mg/kg |
| NIOSH | JN6475000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Diisobutylamine: 5 ppm (18 mg/m³) |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | 250 ppm |
| Related compounds | |
| Related compounds |
Di-n-butylamine Diisopropylamine Diethylamine Dibutylamine N-Methylaniline |

