Diisobutyl Ether: A Practical Look at a Quiet Workhorse of Chemistry
The Evolution of Diisobutyl Ether in Chemical History
Some chemicals play a modest but meaningful role across decades. Diisobutyl ether counts as one of those compounds. Early records show it emerged in the toolbox of organic chemists around the middle of the 20th century, just as petroleum refining and synthetic organic chemistry took off. Chemists back then hunted for ethers with better volatility, lower reactivity than diethyl ether, and greater selectivity as solvents. Diisobutyl ether came along as a result of that search, with its distinctive bulk and reliable properties. Over the years, it found a place not only in research settings but across specialty production lines where its unique solvent capabilities gave it a purpose that simpler ethers couldn't always match. As the chemical industry advanced, it quietly kept its footing as a high-purity solvent and reaction component, never front-page news, but always present in the scientist’s lineup.
Understanding the Compound: What Sets Diisobutyl Ether Apart
Diisobutyl ether isn’t a flashy molecule. Structurally, it’s an ether with two branched isobutyl groups surrounding an oxygen atom. That branching makes the molecule quite nonpolar, with a gentle, sweet odor reminiscent of other lower molecular weight ethers. It’s clear and colorless, evaporating fairly quickly under normal lab conditions, which helps during extractions or reactions where removal of solvent should be easy. Its boiling point lies above diethyl ether but below some heavier ethers, which works out for those running reactions at slightly higher temperatures without needing specialized cooling equipment. Its low water solubility, a direct result of its bulky structure, makes diisobutyl ether a good choice for certain non-aqueous extractions, especially where one wants to avoid water contamination. It doesn’t make for explosive peroxides as fast as dimethyl ether, but safety guides still recommend monitoring peroxide formation. In my own experience in academic labs, folks looked for it when diethyl ether proved too volatile or too reactive, and in those moments, diisobutyl ether did exactly what was needed — a stable, low hassle solvent for organic processes.
Physical & Chemical Traits That Matter on the Bench
Lab workers pay close attention to boiling point, density, and miscibility, since daily procedures often hinge on those details. Diisobutyl ether’s boiling point, sitting above 100°C, puts it in a comfort zone for many synthetic organic reactions. The low miscibility with water makes it suited for liquid-liquid extractions — you get a clear phase separation, unlike with some other solvents where water likes to sneak in and muddle things. Chemically, diisobutyl ether resists acid and base hydrolysis much better than esters, so it stands up in environments where acids or bases might get tossed around. Flammability, a shared trait with many organic solvents, calls for sober storage and good discipline in the lab, but diisobutyl ether is less volatile than smaller ethers, which reduces some handling risks. I remember old-timers in research teams appreciating its lower vapor pressure during hot summer months in under-ventilated storerooms.
Technical Specs and Why Labeling Matters in Real Workspaces
Accurate labels, clear hazard tags, and a solid Safety Data Sheet underpin every chemical’s safe use. For diisobutyl ether, labels always flag its flammability, the risk of peroxide buildup, and the potential for narcotic effects at high concentrations. Most reputable chemical suppliers align labeling with both local and GHS standards, ensuring anyone picking up a bottle knows both how to handle it safely and how to store it until use. Over the years I’ve noticed that knowing the full chemical name along with recognized synonyms — like DIBE or 2,6-dimethylheptane-4-oxane — makes it easier to track down cross-references in safety guidance and literature, reducing confusion both in the supply chain and on research logs.
How Chemists Prepare Diisobutyl Ether
Chemistry’s practical side shines through in solvent synthesis. Diisobutyl ether typically comes from dehydration of isobutanol under acidic conditions, with sulfuric acid as the classic catalyst. The process handles well on both lab and industrial scales. Purification often involves distillation, since unreacted isobutanol and trace water can knock purity below spec, especially when the solvent gets used for demanding organic syntheses. I’ve watched colleagues run these distillations under reduced pressure not only to boost purity, but to reduce exposure to high temperatures, which helps with safety and lessens the chance for peroxide formation. The greater purity achieved supports sensitive Grignard reactions and other organometallic systems.
Chemical Versatility and Refinements
Though diisobutyl ether isn’t one of those solvents famous for dramatic chemical behavior, it anchors more reactions than some might expect. In organic synthesis, it often serves as a nonreactive medium, letting reagents do the heavy lifting. It resists oxidation better than old-school ethers, provided peroxides stay under control. Its low nucleophilicity keeps reaction selectivity high in complex systems, a factor synthetic chemists respect when managing multi-step processes. Some groups further modify diisobutyl ether for specialty applications, adjusting its boiling range or purity level depending on the end use, but the bulk of demand relies on its essential, stable qualities.
Synonyms & Naming Quirks That Actually Affect the Work
Anyone who’s searched chemical inventories knows how confusing alternate product names can get. Diisobutyl ether crops up as DIBE, 2,6-dimethylheptane-4-oxane, or even as isobutyl ether in older literature. These alternate names make a difference during cross-checking hazards, ordering supplies, or combing through research articles. That’s why most labs stick with a consistent shorthand, based on CAS numbers and standardized names, to make sure everyone’s referencing the same compound. From experience, relying on proper naming conventions cuts down on expensive ordering mistakes and minimizes the risk of dropping an incompatible solvent into a sensitive procedure.
Safety Practices and Handling Expectations
Chemicals like diisobutyl ether reward scrupulous safety habits. Its low flash point marks it as a definite fire risk, especially around open flames or hot plates. The threat of peroxide formation sticks with any ether, and diisobutyl ether isn’t immune — storage in tightly sealed amber bottles, dated upon receipt and regular testing for peroxides, protects users from surprise explosions. Good labs typically limit stocks to what’s needed for a few months, rotating older bottles out of rotation. Despite its relatively low acute toxicity, inhalation of the vapor in high concentrations can cause dizziness, and prolonged skin contact may cause irritation. Labs with healthy safety cultures provide solid ventilation, gloves, and goggles, making accidents rare. These standards come not just from regulatory obligations but generations of collective hard lessons — older chemists pass on cautionary tales that stick with the next wave.
Diisobutyl Ether in Industry, Research, and Technology
The case for diisobutyl ether rests mostly on its value as a specialty solvent. In industrial settings, it’s long found use in processes like Grignard reagent preparation, where water sensitivity demands an inert and non-hygroscopic medium. Extraction of natural products, especially where phase separation needs to be crisp, benefits from diisobutyl ether, since it doesn’t drag as much water over as some alternatives. Agrochemical research, flavor and fragrance synthesis, and even certain pharmaceuticals rely on its properties during intermediate or purification stages. Teaching labs, especially at advanced levels, sometimes employ it to introduce students to non-aqueous extractions and oxygen-sensitive chemistry because it straddles ease of use, safety, and reliable performance. These aren’t headline uses that draw attention from the broader public, but they’ve kept diisobutyl ether relevant over decades.
Research and Analytical Advances: Learning from the Lab
Even if diisobutyl ether has a niche profile, research continues. Analytical chemists scrutinize it via NMR and mass spectrometry to ensure solvent purity meets ever-tightening specs for pharmaceutical or specialty synthesis. Environmental scientists developed robust methods to detect trace levels of diisobutyl ether in wastewater, a topic that’s only gotten more urgent as solvent residues in rivers and groundwater raise public health questions. Green chemistry advocates explore recycling or safer alternatives, knowing that ether solvents, while convenient, raise ecological and safety concerns. My own exposure to these research efforts showed me that every material, even a modest solvent, sits within a larger story about how chemistry shapes — and is shaped by — broader social priorities.
Toxicity: What the Studies Really Show
No solvent in this category gets a blank check for use. Toxicology reviews on diisobutyl ether tend to show low acute toxicity, but longer-term exposure data remains limited, especially compared to industrial stalwarts like toluene or acetone. Inhalation can depress the central nervous system and cause headaches, while repeated skin contact causes irritation in some individuals. Major chemical safety authorities recommend restricting exposure to controlled, ventilated areas and using protective equipment consistently. Laboratory incidents have dropped significantly since safety training became more structured and standardized — a hard-won victory fueled by studying accident reports from decades past, then integrating the lessons into modern guidelines.
Future Directions and Opportunities: Where Diisobutyl Ether Might Go
As global industry leans harder into green chemistry and sustainable production, diisobutyl ether’s future will hinge on whether it can adapt. Companies push for closed-loop handling, recycling protocols, and alternatives with less environmental baggage. Ongoing research seeks catalysts and processes for making diisobutyl ether from renewable resources, aiming to reduce dependence on petrochemical feedstocks. Specialty labs experiment with modifications that could further reduce toxicity, improve biodegradability, or change solvent selectivity. In practical terms, those who work with this ether remain keenly aware that the regulatory climate is changing, driving innovation in not just alternative solvents but safer process design. The humble role of diisobutyl ether may shift in coming decades, but its story — one of adaptation and reliability — still has chapters to write, especially if tomorrow’s chemistry strikes a better balance between workplace safety, environmental responsibility, and high performance.
What are the main applications of Diisobutyl Ether?
A Solvent That Gets Down to Work
Diisobutyl ether shows up in many corners of modern industry, but its biggest role comes as a solvent. Companies reach for it when separating useful compounds, especially in pharmaceuticals and fine chemicals. Its unique structure lets it dissolve many kinds of substances, but it stands out because it resists mixing with water. Chemists take advantage of this property to pull out target ingredients after a chemical reaction. In labs where each step can mean the difference between success and confusion, using a reliable solvent like diisobutyl ether can save hours of frustration.
Key Role in Grignard Reactions
Any chemist who has ever dealt with organometallic compounds will know that not all ethers are made equal. Diisobutyl ether pairs exceptionally well with Grignard reagents — an important class of chemicals used to build other molecules. During my university days, I learned that using the wrong solvent could stall a reaction or give poor yields. Diisobutyl ether kept unwanted side reactions at bay and made it easier to purify the final product. Trusted chemical data backs this up, since diisobutyl ether’s high boiling point and stability under basic conditions allow the reaction to proceed smoothly without unwanted heat loss.
Fuel Additive and Extraction Duties
Many people don’t realize this solvent also helps behind the scenes in the energy sector. Gasoline needs an extra boost of oxygenates to burn more cleanly, and diisobutyl ether fills this role as a blending agent, especially in markets looking to meet air quality standards. It helps lower emission levels, making it useful for both the environment and engines that have to meet tough regulations.
On the extraction side, diisobutyl ether steps in to help pull specific metals or organic compounds from mixtures. Its low water solubility means it can grab and hold onto the part you want, letting excess water drain away. From copper refining to cleaning up pharmaceuticals, it’s often the secret behind high-purity results. Working in a pharmaceutical plant, I watched as diisobutyl ether made the difference between a clean, workable medicine and a batch filled with unwanted leftovers.
Production of Specialty Chemicals
Manufacturers call on diisobutyl ether when making flavors, fragrances, and plastics. It serves both as a part of the process liquid and as an ingredient in solvents found in paints and inks. Its ability to dissolve certain resins means you find it adding value during the process and sometimes lingering in the final blend. The food and fragrance industries rely on pure, stable materials, and diisobutyl ether checks both of those boxes.
Safety and Future Approaches
Any household or factory around chemicals needs to factor in workplace safety. Diisobutyl ether carries some fire risk, just like most organic solvents, so safe storage and careful handling go a long way. With green chemistry on the rise, research teams now look into recycling and reusing solvent streams to cut down on waste and exposure. Efforts in developing nonflammable alternatives get a lot of attention, too. Until those options take over, diisobutyl ether’s mix of efficiency and stability keeps it in widespread use.
Is Diisobutyl Ether hazardous and how should it be handled safely?
Understanding the Substance
Diisobutyl Ether shows up in plenty of labs and industrial settings, usually as a solvent. It smells like most ethers: sweet and somewhat overwhelming. Many folks who work in chemistry get familiar with it early on. What hits you first beyond its sharp scent is its flammability. This ether lights up easily, much more so than water-based solvents. It evaporates fast, meaning vapors can gather in poorly ventilated spaces.
Looking at the Hazards
Touching or inhaling Diisobutyl Ether can cause trouble for your body. Short exposure might leave you dizzy, bring on headaches, or make your throat and nose sting. Contact with skin sometimes leaves itching or redness. Breathing a lot of the vapor, especially in a small, closed space, can leave you lightheaded or worse. In larger amounts, it can lead to loss of coordination or, if things go really wrong, unconsciousness.
The bigger long-term risk comes from the formation of peroxides. Over weeks or months, containers, if opened and resealed, can form unstable substances that explode if disturbed. This hazard grows when people forget a partial bottle of Diisobutyl Ether at the back of a shelf until dust gathers on the label. Many lab accidents happen for this reason.
Why Proper Handling Matters
Anyone working with chemicals knows not everything dangerous glows in the dark or emits a flaming warning. Diisobutyl Ether looks clear and harmless, but that’s not a reason to get sloppy. People have gotten lucky and gone home without drama, but luck doesn’t replace good habits. My first year in a research lab, an older technician caught me about to open a half-empty ether bottle without checking for crystal buildup at the rim. That bottle had been there for months. The lesson stuck because the risk looked invisible.
With statistics from the National Fire Protection Association and CDC, fires and chemical injuries from ethers pop up every year. Real lives get affected. The CDC notes that inhaling these vapors in high amounts may trigger central nervous system depression.
Practical Steps for Safer Use
The advice sounds simple, but these habits save more than paperwork. Store Diisobutyl Ether in tightly sealed containers away from heat and sunlight. Keep it in flame-proof cabinets if possible. Add stabilizers (like BHT) to fresh supplies so peroxide formation slows down. Always label containers with opening dates. I still use a sharpie for this; simple tools work best.
Ventilation matters—open a window, use a fume hood, don’t assume yesterday’s air will be fine today. Wear chemical-resistant gloves and splash-proof goggles every time, not just during formal lab inspections. Clothing choice makes a difference, too. Skip the shorts and sandals for cotton pants and closed shoes.
Before pouring from older bottles, check for cloudiness or crystals at the neck. If anything looks off, report it and let the professionals handle it. Don’t pour questionable ether down the drain. Disposal needs a chemical waste program—do not take shortcuts just to save time at the end of a shift.
Simple Solutions Bring Safer Workplaces
Start with training. Every new person who walks into a lab or industrial job should hear stories about what can go wrong and how to avoid it. Supervision from someone who cares about safe habits helps. Try peer reminders as well—I’ve found coworkers pointing out shortcuts makes everyone sharper.
If you don’t have access to high-tech detectors or expensive disposal gear, rely on teamwork and basic tracking of chemical usage. Assigning responsibility works. I’ve watched groups succeed just by having one person keep a logbook for solvent opening and closing. Good record keeping beats relying on memory alone.
Diisobutyl Ether, like most chemicals, rewards respectful handling and old-fashioned attention to detail. No need for fear—just smart habits and honest communication every day on the job.
What are the physical and chemical properties of Diisobutyl Ether?
What Is Diisobutyl Ether?
Diisobutyl ether shows up in labs and factories because it solves problems that come with many industrial reactions. This compound belongs to the family of ethers, meaning it contains an oxygen atom connected to two isobutyl groups. The chemical formula is C8H18O, and it has a structure that keeps it away from easily reacting with most acids and bases.
Physical Properties That Make a Difference
Diisobutyl ether looks clear and colourless. It has a sweet, mild smell, nothing overpowering. It doesn’t dissolve well in water, but it mixes easily with most organic solvents—think alcohols and hydrocarbons. This low water solubility brings real value to extraction processes, like when someone wants to separate organic materials from aqueous mixtures.
It boils at about 108°C, so it evaporates before water but not as quickly as more volatile ethers. Its melting point stays low, around -80°C, so it remains in liquid form in cold rooms and freezers. Density sits below that of water—about 0.73 grams per cubic centimetre—so it floats, creating a distinct layer when poured into a beaker of water.
In many labs, some of the physical traits that matter haven’t changed for decades. For example, the flash point is around 10°C, which means it catches fire more easily than many common solvents. So, using it requires plenty of ventilation, proper storage, and an open eye on fire risks.
Chemical Properties and Risks
From a chemical perspective, diisobutyl ether behaves much like other ethers. People count on its stability: it does not tend to react violently with acids or bases. That quality helps when making pharmaceuticals and plastics where a stable medium is needed. But time and oxygen aren’t always friendly. Over weeks or months, diisobutyl ether starts to form peroxides—dangerous, explosive compounds. Any chemist who’s worked with older bottles knows the worry. Those crystalline deposits along the cap or inside the bottle are a warning. That’s why testing for peroxides before use and discarding old bottles is standard practice.
Its non-reactive character makes diisobutyl ether useful for Grignard reactions. In these settings, other solvents tend to overreact or break down under heat; diisobutyl ether keeps things stable, letting chemists finetune reactions for higher yields.
Why Knowing the Properties Matters
Risks attached to diisobutyl ether can’t be ignored. Fires in research spaces have happened simply because someone left a bottle near a heat source. These incidents show how ignoring chemical properties invites accidents. On the positive side, people who understand solvent properties clearly decide on safe storage and proper waste handling. In places where young chemists learn the trade, properties like boiling point or peroxide risk aren’t trivia—they shape safety culture.
Real solutions start with education and regular checks. Walk into any well-run lab, and you’ll see safety labels, peroxide test strips, and solvent waste cans, all part of safe ether handling. Training isn’t just for compliance but for keeping a community safe—one bottle, one bench at a time.
How should Diisobutyl Ether be stored and transported?
Real-World Risks of Diisobutyl Ether
Diisobutyl ether, often used as a solvent in the chemical industry, brings a real edge of risk due to its flammable nature. I’ve seen what happens when someone underestimates this danger — a tiny flash in the wrong place, and suddenly every safety measure seems too little, too late. In warehouses and labs, this chemical’s low flash point is not a theoretical concern. A spark, even just from static electricity, can get very serious very fast.
Smart Storage for Safety
Keeping diisobutyl ether in steel drums or containers designed for flammable liquids is the rule with no exceptions. A friend’s factory once had a shelf collapse because they tried saving space with makeshift stacking. That shelf held solvents, and one loose cap meant a whole morning spent airing out the place and filling out incident reports. Dedicated flammable storage cabinets, with solid grounding for electrical safety, matter more than most people think until they have to deal with a spill or a fire. It shouldn’t be hidden with other supplies, and crowding only creates more risk of leaks or accidental mixing with incompatible chemicals.
Temperature and Ventilation
Extreme heat speeds up vaporization. Without good ventilation, fumes hang in the air, waiting for one hot surface or spark from equipment. I remember loading docks in midsummer feeling hotter than the surface of the sun — fans helped, but a proper exhaust system is the only way to keep those places safe. If the ether gets too warm, pressure builds up inside containers, creating an explosion hazard. Ground-level storage, out of direct sunlight and away from heat sources, is just smart practice.
Labeling and Awareness
Confusing one chemical for another may sound like a rookie mistake, but it happens. Using clear, chemical-resistant labels on every drum or tank prevents mix-ups. Safety Data Sheets (SDS) posted right inside the storage area mean anyone can check the facts on everything from personal protective equipment to first aid in seconds. It’s not just about following regulations — fast access to information has stopped more than one bad situation from getting worse, especially for new workers who may be learning as they go.
Transport: Precision and Preparation
Trucks carrying diisobutyl ether need to go beyond basic hazmat rules. It’s not just a matter of slapping on placards. Drivers have to check seals, make sure containers are secured, and double-check ventilation throughout the vehicle. I’ve seen operators get lazy about logbooks, but keeping those records up to date means tracking any leaks or container issues and acting before they become dangerous. Traffic accidents, heatwaves, or unexpected stops all add stress to the load, so route planning and regular checks pay off. Every handler should have access to spill kits, and a real plan for roadside response if the worst happens.
Room for Improvement
More training never hurts. Companies can run regular drills, not just for compliance but to make sure everyone feels comfortable handling emergencies. Investing in remote temperature monitoring and stronger ventilation might seem like extra overhead, but these improvements reduce risks and insurance costs in the long run. Inspections and maintenance checks are far from overkill — finding small leaks or weak points early keeps the crew and the community around the site safer.
Why It Matters
Ignoring these steps lands people in the hospital and puts entire communities at risk. The mix of flammable vapor, pressure, and moving vehicles means the margin for error sits razor-thin. Building a culture where everyone has each other’s backs and speaks up at the first sign of trouble is the foundation that keeps chemical storage and transport from making the news for the wrong reasons.
What is the purity specification for commercial Diisobutyl Ether?
Real Demands in Chemical Markets
Diisobutyl ether, often called DIBE in chemical catalogues, plays an unassuming but important role in the world of solvents. Its job is usually behind the scenes, where products like pharmaceuticals, flavors, and pesticides need a compound that dissolves the right chemicals or pulls water out of a solution. In these cases, purity carries more weight than price or branding. If contaminants sneak into a batch, results fall apart. Pure DIBE is no luxury—it’s a basic expectation for anyone relying on consistent performance.
What Purity Really Means for DIBE
Producers targeting commercial applications typically prepare DIBE to a standard of at least 99% purity by gas chromatography, as in Dow or BASF material safety data sheets. Contaminants—often water, alcohols, or other ethers—should fall well under 1%. Extra traces sometimes come from synthesis steps: acid catalysts or unreacted alcohol, for example. I’ve seen how a half-percent of extra water in an ether can ruin moisture-sensitive syntheses, slowing everything down or leaving behind sticky residues. Labs chasing ultra-clean reactions pay close attention to these numbers.
Facts Behind Purity Claims
Test methods matter. Chemical producers rest their numbers on gas chromatography or mass spectrometry—tools designed to expose tiny impurities down to the ppm level. Reliable suppliers show chromatograms as evidence (or will provide them if you ask). Beyond the hero number of “99% minimum,” you’ll often see extra lines in technical datasheets: Water <0.05%, Peroxides <0.005%, Acid number <0.01 mg KOH/g. If you stray into non-verified grades or bargain bins, these figures drift, increasing risks in sensitive processes.
Why Specifications Matter in the Real World
Personal experience in industry taught me that minor contaminants rarely stay quiet. If a solvent promises 99% but runs closer to 96%, side reactions creep in, products fail testing, and troubleshooting burns time and money. Years ago, using technical grade DIBE in a rushed pilot run, a batch showed a faint but persistent off-smell. Gas chromatography later revealed several out-of-place peaks that shouldn’t have been there at all—problems traced to recycled solvent with no certificate of analysis. Once we switched to certified high-purity ether, issues disappeared and yields recovered.
Improving Quality for All
Not every user sits in a spotless lab. Many small or resource-limited buyers have to trust the numbers printed on a drum or bottle. Suppliers who invest in batch traceability, regular quality audits, and detailed testing build that trust and save customers from expensive surprises. Governments sometimes step in too, setting minimum specs for solvents heading into pharmaceuticals or food production. These requirements don’t just protect companies—they make end products safer for people down the line.
Practical Ways Forward
Strengthening the chain that guarantees purity doesn’t start with new regulations. It grows from dialogue: chemical makers listen to what buyers need, document their batches, and invest in regular testing. Customers can take extra steps themselves—quick tests for water or unexpected residues before scaling up, for example. Sharing lessons from the field, between colleagues or in user forums, helps others dodge problems you already solved. With a little more transparency and less mystery, everyone benefits from consistently pure diisobutyl ether, and headaches shrink for everyone involved.
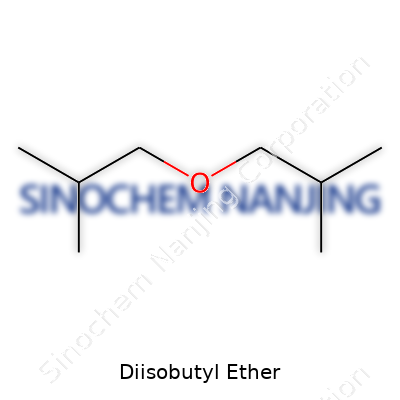
| Names | |
| Preferred IUPAC name | 1,1,3,3-Tetramethylbutyl ether |
| Other names |
DIBE Isobutyl ether Diisobutyl oxide 2-Isobutoxy-2-methylpropane |
| Pronunciation | /ˌdaɪˌaɪsəˈbjuːtɪl ˈiːθər/ |
| Identifiers | |
| CAS Number | 625-65-0 |
| 3D model (JSmol) | `3DModel: JSmol: C1CC(C)(COC(C)(C)C)C1` |
| Beilstein Reference | 1445768 |
| ChEBI | CHEBI:9419 |
| ChEMBL | CHEMBL165684 |
| ChemSpider | 57097 |
| DrugBank | DB14076 |
| ECHA InfoCard | 100.120.212 |
| EC Number | 208-870-3 |
| Gmelin Reference | 8226 |
| KEGG | C19611 |
| MeSH | D000899 |
| PubChem CID | 8173 |
| RTECS number | KI0175000 |
| UNII | N9K5A2D1G0 |
| UN number | UN1159 |
| CompTox Dashboard (EPA) | DTXSID6020142 |
| Properties | |
| Chemical formula | C8H18O |
| Molar mass | 130.23 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | ethereal |
| Density | 0.726 g/cm3 |
| Solubility in water | 2.2 g/L (20 °C) |
| log P | 2.9 |
| Vapor pressure | 3.8 kPa (at 20 °C) |
| Acidity (pKa) | 19.0 |
| Basicity (pKb) | pKb ≈ 19.5 |
| Magnetic susceptibility (χ) | -8.48×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.369-1.371 |
| Viscosity | 1.93 mPa·s (25 °C) |
| Dipole moment | 1.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 281.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -370.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4771.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H304, H336, H411 |
| Precautionary statements | P210, P280, P301+P310, P305+P351+P338, P403+P233, P370+P378 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 15 °C |
| Autoignition temperature | 410 °C |
| Explosive limits | 1.1% - 7.5% |
| Lethal dose or concentration | LD50 oral rat 5200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 14,300 mg/kg |
| NIOSH | SN2875000 |
| PEL (Permissible) | PEL: 25 ppm |
| REL (Recommended) | 250 ppm |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
Diethyl ether Di-n-butyl ether Methyl tert-butyl ether tert-Amyl methyl ether Tetrahydrofuran |

