Diethylenetriamine: A Commentary on Its Role in Industry and Research
Reflecting on the Development of Diethylenetriamine
Walking through the timeline of industrial chemistry, diethylenetriamine (DETA) makes for an interesting story. Its introduction in the early 20th century gave chemists a new tool for exploring polymer technology. Produced mainly by reacting ammonia with ethylene dichloride, DETA became popular as industries searched for more effective polyamines to build resins and as curing agents for epoxy systems. Industrial shifts and growing plastics demand pushed DETA from laboratory curiosities into large-scale production. This step represented not just innovation but a shift in thinking—a move toward chemicals that could serve many purposes and solve a whole batch of practical problems across manufacturing sectors.
Getting Familiar: What Is Diethylenetriamine?
DETA stands as a clear to pale-yellow liquid, and anyone who’s worked with amines knows its distinctive, amine-like smell will hit right away. Its molecular structure, with three amine groups, not only gives it high reactivity but also allows it to form a broad range of derivatives. This versatility found it working anywhere from chelation chemistry to the formation of polyurethane foams. The presence of these amine groups offers lots of possibilities for chemical modifications—chemists have turned to DETA to build adhesives, oil additives, and water treatment agents. If you have spent any length of time fighting stubborn scale in water systems, you can appreciate the value of a molecule able to grab metal ions and wrap them up.
Properties That Matter Day In, Day Out
Talking about physical and chemical properties, DETA boils around 207°C, a point not easily forgotten in process engineering where overheating can be costly. Its high solubility in water and organic solvents means process teams can blend DETA straight into a variety of systems without wrestling with emulsifiers. The substance’s density—about 0.95 g/cm³—lets it fit well into industrial flows. A strong base by chemical definition, it reacts swiftly with acids and can be hazardous in unskilled hands. I’ve learned to respect its corrosive bite; gloves, goggles, and fume hoods stay close. Chemical reactivity springs from the primary and secondary amine groups, helping DETA participate in a slate of organic syntheses.
Labeling, Technical Specifications, Safety: Non-Negotiables
Clarity in labeling and technical standards separates the safe user from the regretful. Diethylenetriamine gets listed with the proper chemical identifier—N-(2-aminoethyl)ethane-1,2-diamine—on packaging. Labels warn of health risks and regulatory requirements for handling, and storage follows strict guidelines to avoid exposure or unwanted chemical mixtures. From my own experience, operational standards must mandate airtight containment, chemical-resistant surfaces, drench showers, and a real understanding of emergency steps. Safe operations don't come from checking boxes; they come from rigorous training and constant reinforcement of proper technique.
Preparation: A Legacy of Industrial Knowhow
The traditional route for preparing DETA uses ethylene dichloride and ammonia, passing the mixture over a catalyst at elevated temperature. This process yields a mix of ethylene amines including mono-, di-, and tri-ethylene compounds, requiring further separation steps to isolate DETA. Distillation takes over as a workhorse, separating by boiling points. Improvements to this process have allowed for less waste and more selective yields over time, showing just how much chemical engineering adapts to reduce environmental impact and increase efficiency.
Chemical Reactions and Modification: The Workbench Expands
Chemists who work with DETA appreciate the molecule for its broad reactivity. The three amine groups allow for stepwise alkylation or acylation, forming intermediates for surfactants and textile treatments. DETA's chelating ability gives it a place in coordination chemistry, helping bind metal ions for separation or purification. Its reactivity with epichlorohydrin creates epoxy resins with tunable hardening rates and mechanical properties, a boon for construction adhesives and coatings engineers. This flexibility has built DETA’s reputation among those looking to tweak polymer characteristics or build novel additives.
Synonyms and the Jungle of Names
Anyone scanning a chemical catalog or research paper might see DETA described by many names: 2,2'-Iminodi(ethylamine), Diethylene triamine, Pentamethylenetriamine. Synonyms crop up from historical patents, brand naming, or translation from other languages. I’ve seen confusion mount in online ordering, occasionally getting chemicals under alternate names due to incomplete catalog listings. For safety and clarity, leaning on the CAS number—111-40-0—cuts through ambiguity, a practice widely encouraged for anyone navigating the semi-chaotic world of chemical sourcing.
Tough Standards for Safe Handling
Regulatory control and safety standards keep DETA’s risks in check. Its corrosivity commands respect: direct skin or eye contact leads to burns, and inhalation risks respiratory irritation. Companies lean on standards such as those from OSHA in the US, enforcing limits on workplace exposure and specifying safe handling procedures. Engineering controls—ventilated hoods, closed transfer systems—run on established chemical safety rules, alongside consistent monitoring. Safety Data Sheets (SDS) must go beyond paperwork by shaping habits, from regular PPE use to thorough first aid and spill response training.
Where DETA Works: Beyond the Factory Gate
DETA’s application footprint stretches from specialty polymers to oilfield chemicals. It forms polyamide resins for paints, coatings, and inks—a sector I’ve seen grow alongside the shift to more durable, solvent-resistant materials for public infrastructure projects. Water treatment specialists trust DETA-based chelating agents to control metal ions in municipal and industrial systems. The same chemistry that helps remove scale can bind trace heavy metals, offering tools for in-situ environmental remediation. In the oilfield, DETA derivatives stop corrosion, clean pipelines, or modify the flow of drilling fluids, where unpredictable well conditions reward chemical ingenuity.
Research & Development: Not Just for Big Labs
Laboratories keep finding new ways to modify DETA into agents with unique functions—flocculants, biocides, even novel catalysts. Work on DETA-based dendrimers hints at applications in drug delivery: adding branches to the basic DETA core yields molecules that can carry medication or imaging agents with specific targeting. Universities and industry research groups have already started testing these derivatives for their ability to improve water solubility or enhance encapsulation. The fast pace of research, plus open scientific sharing, means today’s promising DETA modification might show up in tomorrow’s medical implants or biodegradable plastics.
What We Know and Don’t Know About Toxicity
Toxicity studies show that unprotected exposure to DETA can cause skin burns, eye damage, and possible sensitization. DETA does not appear to be mutagenic at normal exposure levels, but chronic or repeated exposure raises questions about long-term respiratory or systemic effects. Animal models indicate oral toxicity, but occupational risks usually come from inhalation or dermal contact. Regulatory oversight maps exposure limits based on up-to-date toxicology, but caution stays paramount—chemical burns from amines take little time to set in but a long time to heal. I’ve seen best practices evolve, with greater emphasis on secondary containment and medical surveillance, especially in high-throughput operations or large-volume manufacturing.
Looking to the Future: A Molecule With Momentum
The future of DETA lies in continued process innovation and finding greener, more sustainable production routes. Demand trends push for lower-emission, energy-efficient manufacture. With environmental regulations tightening and customers demanding transparency, DETA’s sector will need to adapt, aiming for closed-loop production or biobased alternatives. Research also points to DETA’s role in advanced materials for energy storage and conversion, tapping into the rise of electric vehicles and renewable power. It’s not only about chemistry; it’s about understanding the ripple effects in workplace health, environmental impact, and new technology launches. Chemists and engineers willing to combine safety with creativity will keep DETA relevant well into the next generation of industrial progress.
What is Diethylenetriamine used for?
What Is Diethylenetriamine?
Diethylenetriamine, often called DETA by folks in chemical plants, is a clear liquid with a strong ammonia-like smell. People who have spent any serious time around industrial work know how important it is in making everyday items. DETA comes from the world of amines, a group of chemicals that play a role in everything from cleaning agents to fixing up cars. That ammonia odor hits hard, letting you know it means business.
Industrial Glue and Resins
Anyone who’s worked on construction sites or with manufacturing machines will have run into epoxy resins at some point. DETA gets mixed in as a curing agent. Without a curing agent, epoxies would just stay sticky and weak—no good for floors, tools, or coating pipes. Epoxy adhesives, protective paints, and even the stuff holding wind turbine blades together use DETA. Pipelines count on that toughness, and so do DIY enthusiasts patching up garage floors after winter has chewed them up.
Chemical Synthesis
DETA acts as a chemical workhorse. Companies use it as a building block for other chemicals—flocculants for water treatment, textile softeners, and oil additives. For water plants, having a steady supply of flocculant helps stick unwanted particles together, making water cleaner and safer. People working in water towers or mining operations pay close attention to these processes. There’s a comfort knowing clean water isn't just luck—it’s the result of tried-and-true chemistry like what DETA brings.
Corrosion Inhibition
Pipes carrying oil or water can suffer rust over time, but corrosion inhibitors keep things running longer. DETA gets blended into these protective formulas, slowing down metal breakdown. Maintenance crews in factories and power plants have come to rely on these inhibitors. Losing a pipe line isn’t just a hassle—it can mean costly repairs, lost product, and plenty of stress. The investment in proper chemical treatment, with ingredients like DETA, goes a long way toward keeping operations smooth.
Personal Experience in the Workplace
Spending time on the floor in a plant, you notice how even small leaks or chemical spills become a big deal. DETA needs careful handling: gloves, face shields, and fans humming to keep the air moving. There’s a lesson in that level of caution. Chemicals like DETA are helpful, but they demand respect. Folks who skip safety gear rarely do it more than once. One whiff will remind them quickly.
Safety and Environmental Concerns
DETA stings the eyes and skin. Long exposure without protection leads to headaches or worse for anyone getting too relaxed about safety rules. While modern companies use special handling systems and train workers well, not every place is as careful. Chemical runoff can harm fish and plants. Regulators and environmental groups often keep a watchful eye on places storing or using a lot of DETA. Investing in good training and proper waste controls keeps workers and neighborhoods safer. My time visiting small and large plants showed that the best-run operations took these steps seriously.
Finding Better Solutions
Newer technologies keep pushing for safer, greener ways to achieve the same industrial results. Alternatives to DETA are under study, but few offer the same flexibility. For now, DETA keeps proving useful—so long as companies follow good safety plans and watch for leaks or spills. More research and tighter environmental controls help reduce harm. Building more awareness about best practices, training, and safety equipment makes hands-on workers and communities less vulnerable, and that’s a positive step forward in any industry.
Is Diethylenetriamine hazardous or toxic?
What Diethylenetriamine Actually Does
Diethylenetriamine shows up in all sorts of factory settings—paint hardeners, resins, even some lubricant manufacturing. People who work with industrial glues or rubber processing chemicals bump into this substance far more often than anyone strolling down the street. Its utility keeps it stocked in many chemical storerooms, but its risks go hand-in-hand with that access.
Health Risks That Stand Out
My direct personal experience in a lab setting taught me that safety with chemicals really deserves attention, even when a compound doesn’t scream “poison” at first glance. Touching liquid diethylenetriamine can cause skin and eye burns. The fumes sting and irritate lungs, nose, and throat. Chemical suppliers label this material as corrosive for good reason—a splash does not belong on bare skin or near eyes.
Breathing in its vapors for a short time can bring on coughing, tightness in the chest, and shortness of breath. If this exposure drags on, it damages deeper parts of the respiratory system. Inhaling more than a trace invites headaches, nausea, and sometimes more severe symptoms. Chronic exposure can show up as allergic skin reactions long after the first incident. Medical studies from Europe point to ongoing occupational asthma in workers handling it unsafely, and that risk proves stubborn even with brief contacts.
Environmental Hazards
Diethylenetriamine doesn’t just vanish after use. Contaminated water can harm fish and other aquatic life. Unlike some organic chemicals, this one dissolves readily, making spills tricky to clean up. Without quick action, it seeps into surface or groundwater. Waterways downstream from factory leaks have measured notable harm to organisms once thought able to tolerate most pollutants.
How Much Exposure Is Too Much?
The US Occupational Safety and Health Administration (OSHA) and the National Institute for Occupational Safety and Health (NIOSH) set strict air limits for workplace exposure. Even small amounts mean workers must wear gloves, splash goggles, and sometimes powered respirators. Laundry staff who clean chemical-soaked uniforms keep spaces well-ventilated and tightly manage contaminated laundry—lessons hard-won in the chemical trades.
Scientists haven’t found strong proof that diethylenetriamine causes cancer in humans so far, but that doesn’t make it harmless. The chemical meets enough risk criteria that experts advise treating it with the same respect given to more notorious industrial toxins. Better to take proper care now than wait for new studies to reveal harm years later.
Smarter Handling and Solutions
Relying on good ventilation and prompt spill cleanup makes a real difference. Even in small shops, using closed transfer systems and regular air monitoring brings peace of mind. In my own workshops, storing it in clearly labeled, corrosion-proof containers reduced confusion and cut down on minor accidents. Emergency eye-wash stations don’t just gather dust in places that take chemical risks seriously—they save vision when accidents strike.
Companies can adopt greener substitutes where possible. Alternatives continue to pop up, with some plant-based or less persistent amines doing much of the same work. Pushing for updated safety training and investing in modern protective equipment amounts to more than regulatory box-ticking. It means getting everyone home at the end of each day—intact and healthy. Attention to the details, honest risk assessment, and proper planning form the backbone of responsible chemical use around diethylenetriamine or any similar compound.
What are the storage and handling requirements for Diethylenetriamine?
The realities of Diethylenetriamine in the Workplace
Working with Diethylenetriamine, a clear, colorless liquid with a strong ammonia-like odor, brings some straight-up challenges. This organic compound serves essential roles in the manufacture of epoxies, chelating agents, fuel additives, and more—jobs that drive modern manufacturing forward. Its value in industry can’t be overstated, but ignoring its safety demands comes with real risks. I’ve seen colleagues underestimate how fast a situation with this chemical can get out of hand, especially in smaller shops.
Key Facts About Storage: Taking Safety Seriously
Diethylenetriamine catches fire easily. Store it in tightly sealed containers made from materials like stainless steel or certain plastics—steel and polyethylene prove robust against its corrosive tendencies. Place drums or IBCs in a dry, cool spot, far from any direct sunlight, sparks, or sources of open flame.
Humidity causes trouble because this liquid absorbs water from the air, so a dry environment always makes sense. Keep storage spaces well-ventilated. It wasn’t until I started inspecting some older warehouses that I saw how quickly vapors can build up in a stuffy room, even with what looks like a modest stash of chemicals. Leaks or spills create hazards, so reliable secondary containment (think spill pallets or bunds) keeps unexpected accidents in check.
Never pile Diethylenetriamine next to incompatible substances. Keep away from acids, oxidizers, and carbon dioxide. Once, a careless drum placement next to a cleaning solution set off a bad reaction—damage that never needed to happen. Signage can't prevent every mistake, but clear labels help everyone recognize the risks at a glance.
Handling Challenges: People Make the Difference
Handling Diethylenetriamine takes real care—it can burn skin and eyes on contact, and the fumes hit your nose and throat hard. Personal protective equipment becomes non-negotiable here: splash goggles, chemical-resistant gloves, aprons, and a solid face shield. Even a quick splash can send someone to the emergency room. Workplaces skip these steps to save time, but the cost catches up fast.
Proper ventilation means more than just a cracked window. Reliable local exhaust or fume hoods maintain air quality and cut down the risk of workers inhaling toxic fumes. Routine training makes the biggest difference in my experience. It keeps everyone sharp and helps new team members stay safe, especially in fast-paced settings or during night shifts.
If a spill occurs, responders should grab assigned kits—absorbents like sand or vermiculite—and clean up using methods that prevent vapor clouds or further spread. No one likes to practice spill drills, but the payoff shows when someone handles a leak swiftly, without panic.
Addressing Gaps with Smarter Solutions
Investment in better monitoring, like fixed gas detectors, often gets overlooked—yet these tools can spot vapor leaks long before they endanger a crew. Regular inspections of containers and valves catch corrosion or wear, defeating problems before they start. In my view, companies that encourage staff to report near-misses create a culture where people own safety together, not just in theory but in daily routines. Regulatory guidelines from OSHA or the European Chemicals Agency give a solid starting point, but boots-on-the-ground experience makes standards stick.
Diethylenetriamine isn’t going anywhere in industry, but small steps—thoughtful storage, daily vigilance, honest training—are what shield people and property from danger.
What is the chemical formula and molecular weight of Diethylenetriamine?
What’s Behind the Formula?
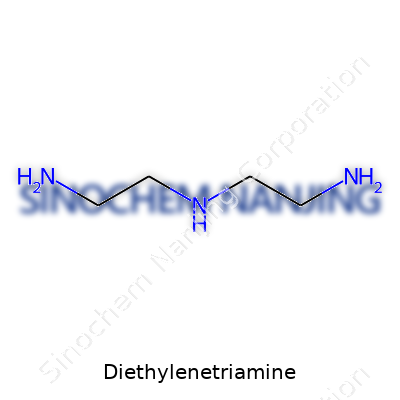
Diethylenetriamine, often called DETA in labs and factories, carries the chemical formula C4H13N3. Every molecule brings together four carbon atoms, thirteen hydrogens, and three nitrogen atoms. Its structure looks like two ethylenediamine units linked by a nitrogen atom, giving it flexibility for chemical reactions. The molecule unfolds like a simple backbone, not packed or complex, allowing for easy interaction with acids, metals, and polymers.
Crunching the Numbers: Molecular Weight
Anyone handling chemicals cares about weight. Getting the right formula means knowing exactly what goes into every reaction or mixture. For DETA, the molecular weight lands at 103.17 grams per mole. This number doesn’t just help in calculations—it’s a tool for dosing, ordering, and keeping reactions balanced in both research and industrial settings.
Why Knowing These Details Helps
Manufacturers, lab techs, and environmental specialists rely on simple numbers like formulas and weights. If you try to synthesize epoxy resins or make corrosion inhibitors, DETA comes up often. My time working on water treatment projects showed me that tracking quantities and purity cuts down on waste, keeps costs down, and staves off unpredictable outcomes.
The chemical’s three nitrogen atoms give it a very reactive character. This means it forms bonds with lots of different partners—good news for making adhesives, bad news if mixed without care. Mistaking the formula or weight might lead to toxic byproducts, runaway reactions, or faulty end products. In one instance at a facility where I assisted, simple miscalculation by a new team member led to an epoxy batch that wouldn’t harden. That kind of loss can be traced back to basic math gone wrong.
The Safety Angle
Every industrial chemist or safety manager learns quickly that even common chemicals demand respect. DETA is no exception. It’s known to irritate the skin and can cause harm if inhaled or swallowed. A standard material safety data sheet calls for gloves, goggles, and proper ventilation. I’ve had team members require first aid after casual contact due to underestimating its potency. Getting familiar with exactly what’s inside, right down to the atom, isn’t a paperwork burden—it’s self-protection.
Looking Toward Better Handling and Transparency
Mistakes often hide where the facts aren’t clear. Chemical plants and research labs keep running trainings, but fresh eyes may miss subtle points. Using digital systems to track each component and every batch’s math helps—the days of scribbled notes and faded labels should be left behind. Sharing info across teams, between shifts, and with regulators isn’t just red tape: it means cleaner audits, better insurance rates, and fewer close calls.
People trust the work when they know everyone’s looking at the right figures for chemicals like DETA. That trust builds a company’s reputation and keeps people eager to work at the bench or in the control room. Science becomes safer, cleaner, and more reliable when every molecule is fully accounted for from start to finish.
How should Diethylenetriamine spills or exposure be managed?
Understanding the Risks on the Ground
Walking into a chemical plant, you quickly sense that handling substances like diethylenetriamine (DETA) isn’t just about ticking boxes. Anyone who’s ever been involved in emergency drills knows DETA’s strong ammonia-like odor doesn’t give much warning before irritation sets in. Even a small spill can sting your nose, redden your eyes, and burn your skin. People who work closely with it recognize the need for a fast, organized response, long before reading an official protocol.
Protecting the People First
No job or cleanup effort matters if you leave the crew exposed. In my experience, those massive yellow chemical suits aren’t just for show — DETA can seep through ordinary clothing and stick to your skin long after you think you’ve washed it off. Respirators with organic vapor cartridges help, but goggles and gloves make the critical difference, since it only takes a little on the skin to cause blisters and persistent irritation.
Ventilation Makes a Difference
Most modern work sites invest in strong ventilation systems, and not by chance. Letting fumes linger turns a small incident into a big problem. I’ve seen how quickly managers shut down all nearby equipment and flip on emergency fans the moment DETA spills. Directing fresh air across the site—quickly—protects more than just those in the hazmat suits. It puts distance between the chemical and everyone else.
Containment and Cleanup Techniques that Work
My old plant supervisor drilled into every newcomer: “You don’t let it spread. Handle the puddle, not the whole floor.” Keeping the spill from reaching drains or soil makes all the difference. Sand, inert absorbents, or even clay work well. They soak up the liquid and make it much easier to sweep up without splashing. Rushing to mop doesn’t help—dry absorbent puts a lid on the chemical right away. Bagging used materials in clearly labeled, chemical-resistant containers stops the mess from ending up in the landfill or seeping into the water supply.
Looking After Long-Term Health
A splash might not sting now but can set off delayed rashes or even asthma. Anyone exposed deserves a medical check right away. Occupational medicine doctors track exposures and watch for problems down the line. Even if you rinse off or take a hot shower on-site, trace amounts could show up a day later. Trained staff keep logs so patterns get spotted quickly if something starts affecting crews over time.
Training and Communication Reduce Mistakes
What sticks with me most from years on the floor is that reaction speed and common sense save more people than a two-inch manual. The best teams talk through every step before setting out. They check in if anyone feels light-headed or itchy. Trust builds when leaders care enough to pause and check safety gear before acting tough or trying to hurry. Regular drills, clear signage, and easy-to-read emergency plans keep staff sharp, so nobody panics when they hear “spill.”
Why Strong Management Matters
Site managers who set clear routines and provide strong training cut down on spill rates and lessen injuries. Modern hazard communication standards—required by OSHA and global best practices—ensure that every worker from the loading dock to upper floors knows which containers hold DETA, what alarms sound like, and where exits lead. Spending money upfront on training and equipment pays back every time a close call turns into nothing more than a lesson learned.
Safe handling of chemicals like diethylenetriamine isn’t just a checklist—it’s a lived commitment. Workers bring that safety mindset into every shift, and everyone gets to go home in one piece if leaders value clean practices over cutting corners.
| Names | |
| Preferred IUPAC name | 2,2'-Iminodi(ethylamine) |
| Other names |
1,4,7-Triazaheptane DETA Diethylene triamine N-(2-Aminoethyl)ethane-1,2-diamine |
| Pronunciation | /daɪˌɛθ.ɪˌliːnˈtraɪ.ə.miːn/ |
| Identifiers | |
| CAS Number | 111-40-0 |
| Beilstein Reference | 63549 |
| ChEBI | CHEBI:28689 |
| ChEMBL | CHEMBL14278 |
| ChemSpider | 5464 |
| DrugBank | DB01992 |
| ECHA InfoCard | 03e5e51c-384a-4dd1-bae1-9c81e4d2424b |
| EC Number | 203-865-4 |
| Gmelin Reference | 11700 |
| KEGG | C00527 |
| MeSH | D004034 |
| PubChem CID | 8030 |
| RTECS number | XR9350000 |
| UNII | 690IHT43NR |
| UN number | UN2071 |
| Properties | |
| Chemical formula | C4H13N3 |
| Molar mass | 103.17 g/mol |
| Appearance | Clear, colorless to yellow liquid |
| Odor | Ammonia-like |
| Density | 0.954 g/cm³ |
| Solubility in water | Miscible |
| log P | -2.04 |
| Vapor pressure | 0.38 mmHg (20°C) |
| Acidity (pKa) | 8.89 |
| Basicity (pKb) | pKb = 3.3 |
| Magnetic susceptibility (χ) | -54.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.483 |
| Viscosity | 3.08 mPa·s (20 °C) |
| Dipole moment | 2.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 232.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -63.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4181.7 kJ/mol |
| Pharmacology | |
| ATC code | V03AB54 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS07 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H314, H317, H319, H412 |
| Precautionary statements | P280, P261, P264, P272, P273, P302+P352, P321, P332+P313, P362+P364, P305+P351+P338, P310, P303+P361+P353, P304+P340, P312, P501 |
| NFPA 704 (fire diamond) | 3-1-0 |
| Flash point | > 94 °C |
| Autoignition temperature | 385 °C |
| Explosive limits | 1.8% - 10.8% |
| Lethal dose or concentration | LD50 oral rat 1080 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 1080 mg/kg |
| NIOSH | WR3400000 |
| PEL (Permissible) | 1 ppm (parts per million) |
| REL (Recommended) | 6 mg/m³ |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Ethylenediamine Triethylenetetramine Tetraethylenepentamine Piperazine Amines |

