Diethylene Glycol Dimethyl Ether: Progress, Pitfalls, and Prospects
Tracing the Path: A Look at Historical Context
Strong chemistry foundations often start with small molecules whose names rarely leave the lab. Diethylene glycol dimethyl ether, sometimes called diglyme, isn’t flashy, but its story traces back to an era when chemists wanted better solvents to push boundaries in synthesis and separation. In the early to mid-20th century, the industry’s appetite for specialty ethers grew as petrochemical processes multiplied. Diglyme emerged out of a search for linear, higher boiling ethers that could outpace diethyl ether in stability and polarity. Its rollout played right into the hands of researchers working on organometallic reactions or electrochemistry, where old standbys just didn’t cut it. Since then, it has carved a niche as both a workhorse and a wild card—useful but demanding respect.
Getting to Know Diglyme
Diglyme’s clear, colorless appearance gives no warning about its robust solvent capabilities. With a boiling point around 162°C, it stands taller than diethyl ether and even monoglyme, which means it won’t just evaporate off when things get hot. It sports a faint, pleasant odor, almost like most ethers. It mixes with water, alcohols, and organic solvents—chemists look for this kind of flexibility because synthetic reactions rarely care for mixing limits. Its ability to dissolve both polar and non-polar compounds helps break bottlenecks in reaction design. This is why labs and production plants stick with it, especially for tough separations, delicate extractions, or as a reaction medium for lithium and sodium-based chemistry.
Specification and Labelling: Navigating the Jargon
Research settings or factory floors, there’s no getting around technical labels. Casually called diglyme or by longer formal names such as 2-methoxyethyl ether, the substance often comes flagged as a flammable organic liquid and marked with international safety codes. Chemical purity grades—lab, industrial, electronic—decide how much water or acid hangs around as an unwanted guest in the canister. Because mishaps with impurities can spoil entire production batches, those numbers aren’t red tape—they’re the insurance policy. Each drum carries hazard tags flagging irritation or toxicity issues. Legal standards place thresholds for allowable storage quantities or exposure values—steps not pushed by lawyers but by hard-earned accident history.
Making Diglyme and Tweaking Its Chemistry
Diglyme stands as a poster child for ether synthesis. Industrial setups often rely on reacting diethylene glycol with methanol in acid-catalyzed conditions, then distilling the product to get rid of by-products and residual starting material. The key to getting it right isn’t just the recipe—it’s dwell time, temperature, catalyst pick, and handling water content that test the skill of the plant operator. On the bench, diglyme participates in alkylation reactions, serves up complexes with alkali metals, and behaves as a hydrogen bond acceptor, giving chemists an edge in everything from Grignard reactions to nucleophilic substitutions. Some play with the molecule’s chain length or substitute end groups to create new variants, each opening the door to fresh reactivity or specialized solvent roles.
What’s in a Name? Synonyms and Lingo
You won’t get far in chemical safety circles without knowing aliases. Diglyme pops up as Bis(2-methoxyethyl) ether, dimethoxy diethylene glycol, or ethylene diglycol dimethyl ether. These aren’t just alternate spellings—they matter when reading safety data, scanning scientific papers, or ordering from global suppliers. Getting name and structure right avoids mix-ups that can lead to dangerous outcomes. Each label wraps a web of standards and local regulations, which calls for both attention and practical chemistry education.
In the Trenches: Safety and Handling
Through decades in labs, phrases like “respects flammable liquids” come to mind, but diglyme’s safety profile has earned that line through hard experience. Overexposure, especially through inhalation or skin contact, brings real health risks, including headaches, nausea, and longer-term organ impacts. Flammable vapor can drift and ignite from distant sparks. Chemical companies have improved warehouse conditions and lab practices over time—ventilation, solvent-resistant gloves, ground cables for drum transfers. Workers go through safety drills not because they’re told but because one slip can put entire teams at risk. Reading and respecting the safety sheet isn’t a box-ticking exercise. Industry moves in groups—no matter how much protective gear gets innovated, clear rules and mutual attention to detail hold up real safety records.
Diglyme at Work: Applications and Reach
My experience in organic synthesis taught me to respect solvents that can handle extremes. Diglyme fits the bill—high-temperature reactions, lithium-based reductions, and sodium dispersion all benefit from this solvent’s unique mix of polarity and boiling strength. Pharmaceuticals, electronics, and specialty polymers all tap into diglyme’s ability to bring stubborn substances into solution. Manufacturers making flexible resins or lithium batteries depend on clean, dry diglyme to get consistent quality. Its role in the extraction of rare earths or precious metals leverages both its solvating power and thermal resilience. All that said, downstream industries always weigh performance against toxicological profile.
Pushing the Edge: Research and Development
Recent years see research teams chipping away at diglyme’s replacement. Engineered solvents with better toxicity profiles and easier disposal draw investment, but diglyme’s unique niche in some reactions still leaves a gap. Method development in battery chemistry, electrosynthesis, and certain polymerizations checks whether new ethers or ionic liquids can beat diglyme. Yet, scalable results often circle back—other solvents lack diglyme’s fine-tuned balance or cost profile. Environmental science pushes for closed-loop systems and better solvent recycling, nudging companies to handle or reform solvent lifecycles. Diglyme now heads into cleaner syntheses, greener extraction processes, and niche reformulations tailored for shrinking its environmental mark.
Toxicity—Lessons Learned and Where Things Stand
Health data for diethylene glycol dimethyl ether draw a clear line: chronic exposure harms. Studies link its metabolism to the generation of compounds that affect organs and potentially the reproductive system. Occupational monitoring keeps its use on a short leash—biosafety teams and industrial hygienists routinely check air concentrations, even update personal equipment requirements as new data drop. Disposal regulations tighten, since improper waste management leads to groundwater and soil contamination. For smaller operations, this means stricter record-keeping and solvent separation. Medical researchers point out that real-life hazards rarely follow textbook exposure—so, practical caution wins out every time.
Looking Forward: The Road Ahead for Diglyme
Chemical industries, labs, and classrooms evolve each decade. Diglyme’s future depends on balancing its technical performance with growing expectations for workplace and environmental health. Battery makers and advanced materials researchers keep one foot in the legacy solvent camp even as they experiment with safer substitutes. Regulatory landscapes shift, demanding sharper labeling, tighter exposure limits, and comprehensive spill management. The next chapters will likely see applications fade in areas where greener chemistry outcompetes, but for certain high-performance processes, diglyme’s legacy probably continues—carefully shepherded by changing science, stricter laws, and sharper public awareness. Reducing risks means training, investment in new process design, and never settling for yesterday’s safety protocols.
What is Diethylene Glycol Dimethyl Ether used for?
A Closer Look at Its Place in Industry
Diethylene glycol dimethyl ether, often called diglyme, shows up in workplaces that deal with complex chemistry. Many people outside labs or certain manufacturing sites have never heard of it, yet this colorless liquid carries a surprising amount of practical value. Whenever I’ve worked with chemists and process engineers, diglyme pops up as a topic, especially during conversations about specialty solvents.
A Trusted Solvent for Challenging Tasks
Diglyme helps dissolve materials that give other solvents trouble. During projects in chemical syntheses, especially at the pilot scale, I’ve watched specialists reach for diglyme when they need a solvent that stays stable and doesn’t react with much else. Its most popular role falls in the manufacture of pharmaceuticals. Many drugs begin their lives as tricky molecules built up step by step. Chemists rely on solvents like diglyme to coax ingredients into reacting just right. It’s not as common as ethanol or acetone, but diglyme brings low volatility and a helpful ability to dissolve certain salts.
Electrical Applications—Not Just for Chemists
Some engineers use diglyme in the world of lithium batteries. Battery designers pay close attention to their electrolyte mix—if it doesn’t carry charge efficiently or remain stable, a battery doesn’t last long. Diglyme’s low viscosity and ability to stay liquid at wide temperature ranges gives it an edge in these applications. It carries lithium ions smoothly, making it valuable for experimental battery cells where other solvents start to freeze or break down.
Cleaner Reactions in the Lab
In organic reactions, especially those involving chemicals like alkali metals or organometallic compounds, diglyme helps keep everything on track. Many times, scientists find that reactions proceed faster or form fewer impurities because diglyme keeps reactive ingredients dissolved. In my past studies, when other ethers broke down under hot, basic conditions, diglyme kept holding on. That kind of stability draws researchers who want to avoid unwanted byproducts at all costs.
Handling with Care: Health and Safety Facts
No discussion about diglyme feels responsible without talking about its health risks. This isn’t a solvent for casual experiments at home. Exposure can be dangerous—diglyme may affect the nervous system and lungs, and over time could create real health problems. Industry guidelines recommend closed handling systems, solid ventilation, and smart storage to cut down on risk. Protective gear, including gloves and appropriate eyewear, becomes essential when working with it. Several regulatory agencies call for careful record-keeping and reporting, especially if diglyme finds its way into consumer products.
Seeking Alternatives and Sustainable Choices
Growing awareness about chemical safety has pushed scientists and manufacturers to explore greener solvents. While diglyme offers stability and effectiveness, researchers now spend time seeking less hazardous substances. Some teams have switched to alternative ethers or mixtures that break down more easily and create fewer concerns for workers and communities. Product stewardship—using solvents only for as long as truly needed and switching out as soon as viable—has helped drive this change.
The Bigger Picture
Every time industry experts talk about solvents like diglyme, the conversation touches on reliability and efficiency, but it circles back to health and safety. Balancing performance against risk feels personal—colleagues and friends work with these chemicals every day. Resources like the ECHA and OSHA databases give anyone direct access to the latest health research. It pays off to read safety data sheets thoroughly, talk to experts, and push for continued progress toward safer work environments. In the end, solvents such as diglyme power much of the progress in chemical and materials science, but safety and responsibility carry just as much importance.
Is Diethylene Glycol Dimethyl Ether hazardous to health?
A Chemical Most People Overlook
Very few folks outside labs or certain factories give any thought to diethylene glycol dimethyl ether. Step into a chemical plant or read the label on specialty solvents, though, and it shows up more often than you might guess. Plenty of research scientists or manufacturers working on electronics or engineering projects have handled the liquid, maybe without truly thinking through what it might mean for their health.
Industry Loves a Versatile Solvent
You see this chemical popping up in everything from lithium batteries to pharmaceuticals. It dissolves loads of compounds that water and more typical solvents just can’t touch. Some start-ups trying to scale newer lithium battery work look for “tried and true” solvents like this one, given the demand for quick solutions and high compatibility. With all that, people sometimes shrug off possible danger, thinking that anything used this widely can’t be too risky.
The Health Hazards That Come With It
Real risks sit attached to this clear liquid. Short-term exposure can hit the eyes, lungs, or even skin, leading to immediate irritation. Longer exposure—or high vapor concentrations in closed spaces—could lead to headaches, dizziness, or in worse cases, nervous system effects. A number of published case studies follow workers or lab techs who only realized the hazard too late, reporting kidney pain or respiratory trouble days after improper handling.
Data from the European Chemicals Agency paint an even clearer picture: animal studies turned up liver and kidney trouble along with signs of nerve damage from repeated contact. A search through the US National Library of Medicine offers up articles on reproductive and developmental toxicity in high doses. Those facts alone make me always step back before opening a bottle, no matter how “routine” a procedure might seem.
What Most People Actually Encounter
It’s probably fair to say the average office worker faces little to no risk from this solvent. For lab techs, warehouse handlers, or manufacturing engineers, the picture looks much different. A simple splash during pouring or an undetected leak from aging pipework could put someone’s health on the line. Speaking with colleagues who spend their days near chemical vats, I’ve heard more than a couple stories of hasty hand-washing or confusion over faint solvent smells. Mislabeling or inadequate training crops up more often than anyone wants to admit.
Solutions Backed By Real-World Experience
A few obvious moves work best: solid training, clear labeling, and rigid ventilation rules stop a lot of trouble before it starts. In my years of lab supervision, placing full unopened bottles in vented hoods—even during delivery—became a habit. Separate rubber gloves and face shields station-side meant fewer “I forgot my PPE” excuses. Installing leak detectors and running regular spill drills never felt like overkill.
Some labs begin swapping in greener solvents or newer alternatives with better human health profiles. Yet such changes take time, and the industry sometimes hesitates—cost and old habits matter. Regulation keeps pushing for safer labeling, controls on workplace exposure, and national reporting of incidents, which has helped awareness. Sharing incidents openly, rather than sweeping them under the rug, makes every workplace safer.
Focusing on Facts
No one can claim that diethylene glycol dimethyl ether belongs in the “safe” category. With respect for the chemical and careful routines, people can cut down on harm. We owe workers and researchers clear information, better precautions, and healthier alternatives wherever possible. That’s just common sense.
What are the storage requirements for Diethylene Glycol Dimethyl Ether?
The Realities of Chemical Space
People rarely get excited about chemical storage, but one mistake in this area and trouble unfolds quickly. Diethylene Glycol Dimethyl Ether, known in labs as diglyme, deserves some honest attention. This solvent flows clear and almost odorless, but beneath the surface lies a story of fire risks and sneakily severe health hazards. Storage isn't about ticking boxes or following basic routines—it's about keeping people and spaces out of danger.
Fire Hazards Lurking in the Drum
Diglyme sports a flash point around 85°C, which sounds safe, given that it doesn’t ignite as easily as gasoline. Still, care slips with routine, and it only takes one hot tool or forgotten heating unit to tip things into disaster. The National Fire Protection Association pins it with a flammability rating of 2, meaning flames become a real threat under the right conditions. Store this solvent in a cool, dry spot with solid ventilation. Forget exposed lightbulbs overhead. The best practice I've stuck to: use explosion-proof refrigerators or chemical cabinets designed to resist fire, especially in crowded workspaces. Ordinary shelving just creates a false sense of safety.
Tight Seals and Fume Fears
It pays off to treat every drum and bottle of diglyme like it’s ready to leak or evaporate. Vapors can irritate the nose and lungs, and it's easy to miss early on. Whenever possible, I double up on seals—use threaded lids and secondary containment trays. This doesn't just prevent costly spills; it stops fumes from finding their way into breakrooms and hallways. The U.S. Occupational Safety and Health Administration (OSHA) has strict indoor limits for airborne exposure. They set the permissible exposure level to 5 ppm over an eight-hour shift for a reason. It's smart to install chemical fume hoods or localized exhaust vents near storage and transfer points.
Rejecting Plastic: The Metal Standard
Polymers can break down slowly in the presence of solvents. Over the years, I’ve seen cracks form in cheap plastic containers, followed by silent leaks creeping onto concrete. Stainless steel or thick-walled glass brings certainty—resistant to the slow gnaw of chemicals and less likely to deform under slight heat changes. Label every vessel openly and legibly. Faded tape or smeared marker leads to confusion and accidents.
Vigilance Beyond the Container
Chemical management isn’t a solitary act. Colleagues deserve clear signage, color coding, and printed instructions near storage areas. I’ve noticed fewer mishaps in facilities committed to constant staff training. Material Safety Data Sheets (MSDS) live on walls, not in dusty drawers, so anyone can act fast if something goes wrong. Local fire departments appreciate advance notice, and agencies like the EPA expect compliance with hazardous waste rules. Waste drums need their own section, far from regular storage, so cross-contamination doesn’t become an ugly headline.
Counting the Costs of Complacency
Cleanup bills from spills and violations stack up much faster than the price of a certified cabinet or a half-hour safety meeting. The Minor irritant of routine codes and checklists beats the major disruptions when things take a turn. Diglyme’s uses in synthesis and as a solvent make it essential for research, but safety isn’t a side job. Every bottle sitting in the warehouse remains silent if respected and secured. Forgetting that turns science into hazard.
How should Diethylene Glycol Dimethyl Ether be handled safely?
Understanding the Risks
Diethylene Glycol Dimethyl Ether shows up in labs and industry as a solvent. It sounds harmless, but my time in chemical labs taught me that even widely used chemicals leave plenty of room for trouble. Skin contact, splashes, inhaled vapors—each of these can burn or irritate. This solvent passes through thin gloves and can dry out skin, leading to cracking or even systemic poisoning if exposure goes on too long.
Safe Handling: Built by Habit, Not Audits
Lab training pounded home one simple truth: safety routines stick best through constant practice, not just printed protocols. No one wants red eyes or coughing fits ruining a good research day. Proper gear counts. Nitrile gloves worked better than cheap latex. Splash-resistant goggles and a dedicated lab coat—never street clothes—create a real barrier. When curiosity once got the better of a colleague and he sniffed a cap, his headache lasted the rest of the afternoon. There’s no shortcut around chemical fume hoods. Ventilated spaces pull vapors away; breathing those fumes too long can damage nerves and organs.
The Right Containers Make a Difference
Bad habits survive in dark corners of labs and shops. Some folks pour solvents into plastic soda bottles or non-labeled jars, thinking they'll remember what’s inside. That’s a quick way to court disaster. Diethylene Glycol Dimethyl Ether needs tightly closed, chemical-resistant containers. Every label must include the name and hazard warnings. I remember a mix-up involving unmarked flasks; a minor spill quickly became a big scare. That one act of laziness cost a few hours, two ruined shirts, and the respect of a supervisor.
Fire Safety and Storage
This solvent lights up easily if given half a chance. Flammable cabinets aren’t just extra furniture—experience taught me they’re as essential as fire extinguishers. Spur-of-the-moment solutions like stashing solvents under benches invite disaster. Smoking near storage never ends well. Always ground containers when transferring the liquid, so the smallest spark doesn’t turn storage into an emergency room trip.
Spills and First Aid
Spills test the nerves of even seasoned workers. Ads and posters won’t replace knowing your eyewash stations and shower lines work. I once tripped and splashed a few milliliters on my hand. Immediate washing under running water for several minutes saved me from a chemical burn. Anything larger means clearing the area and calling for professional help. Only trained responders should handle bulk spill cleanup—the risks grow fast if fumes build up or the spill spreads near electrical equipment.
Disposal: Don’t Cut Corners
Hazardous waste bins sit in every chemical space for a reason. Pouring solvents down drains damages plumbing, pollutes water, and can even harm people miles downstream. Chemical waste needs sealed, labeled containers and should stay away from regular trash. Ignoring disposal rules attracts fines, but more important, it poisons communities slowly and quietly. Working with professionals for safe disposal isn’t just the law—it’s about respect for neighbors and the environment.
Keeping Knowledge Fresh
Safe handling grows from education, not just once but as an ongoing process. I’ve seen experienced chemists pick up new safety tricks from rookie colleagues who’ve read updated guidelines. Fresh eyes sometimes save the day. Regular safety drills, honest conversations, and a willingness to ask simple questions keep everyone a little safer. Reading up on every chemical before use, reviewing safety data sheets, and talking to colleagues about new safety practices build habits that matter as much as the thickest gloves or sturdiest containers.
What are the physical and chemical properties of Diethylene Glycol Dimethyl Ether?
Chemical Makeup and Structure
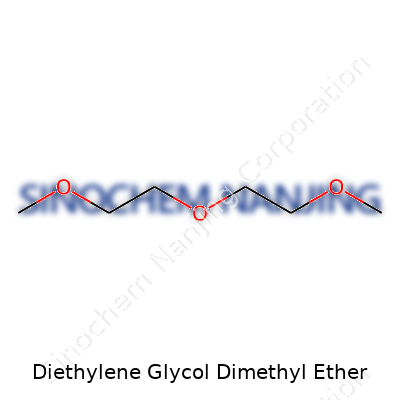
Diethylene glycol dimethyl ether, known in laboratories as diglyme, brings a combination of oxygen atoms and methyl groups into a chain-like molecular structure. The formula—C6H14O3—accounts for two ether linkages. The presence of these oxygen atoms creates spots where the molecule can form hydrogen bonds with other substances. From real-world experience handling solvents, this feature always stands out: diglyme dissolves both polar and non-polar compounds, lending it a respected spot on many chemists’ shelves.
Physical Traits
Diglyme has a clear, colorless appearance and runs smoothly like water—no stickiness or obvious odor. Light passes through it unhindered, and it's less dense than water, coming in at about 0.95 g/cm³. In practical lab settings, this makes spills easier to spot and, unfortunately, harder to clean off hands or lab benches. The boiling point sits around 162°C, higher than diethyl ether, so it doesn't easily vaporize at room temperature. Evaporation from open beakers drags on, rarely causing headaches from fumes unless you get in close.
At lower temperatures, diglyme doesn’t crystallize quickly. I’ve pulled it from a cold storage cabinet and found it still in liquid form down to about -64°C. This fluid range offers flexibility when working under shifting temperatures—a trait I found valuable during multi-step syntheses over long days.
Reactivity and Chemical Safety
Diglyme stays stable under most lab conditions. Strong acids or strong bases can chew into its ether bonds, eventually breaking the molecule apart, so storing it in glass containers with neutral pH environments makes sense. Over months of use, a chemist keeps an eye out for signs of old bottles: ethers sometimes grow unstable as they age, with the risk of forming peroxides. Simple tests—adding potassium iodide and looking for color changes—reveal unwanted buildup. Checking dates and never using bottles with crusted caps or oily layers has saved more than one bench from disaster.
Unlike many ethers, diglyme resists burning. Its flash point sits at 55°C, so it won’t catch fire at room temperature, offering some peace of mind for those used to nimbler, more flammable solvents. Still, good ventilation and eye protection remain essential. Prolonged skin contact dried out my hands quickly, probably from the compound drawing away natural oils. Gloves block that effect.
Applications Rooted in Its Properties
Pharmaceutical labs, electronics makers, and even battery developers use diglyme in large volumes. It dissolves salts and organic molecules equally well, so it often appears in electrolyte formulas. In preparing lithium-ion battery parts, this wide solubility makes mixing raw materials easier. That’s no theory—I’ve watched teams achieve consistent blends batch after batch.
Chemists handling sensitive reactions appreciate diglyme’s low water content. Many organic transformations stop working if exposed to moisture. Diglyme’s dryness preserves reactive chemicals, so yields improve, and steps run smoother.
Addressing Environmental and Health Concerns
Long-term exposure in poorly ventilated spaces brought headaches and minor nausea—a reminder that just because a solvent smells faintly, it isn’t harmless. Studies cite light toxicity with repeated exposure, so fume hoods always remain in play. Regulatory agencies continue studying potential reproductive risks, especially after animal tests showed adverse effects at high doses.
Disposal requires attention. Waste facilities need to know the full content—mixing diglyme in with general waste causes issues at treatment plants. Responsible labs collect used solvent in labeled drums, arranged for chemical collection according to region-specific rules.
| Names | |
| Preferred IUPAC name | 2-methoxyethoxy)ethoxy)ethane |
| Other names |
1,2-Ethylene glycol dimethyl ether Glyme Diglyme Bis(2-methoxyethyl) ether |
| Pronunciation | /daɪˈɛθiˌliːn ɡlaɪˈkɒl daɪˈmɛθɪl ˈiːθər/ |
| Identifiers | |
| CAS Number | 111-96-6 |
| Beilstein Reference | 1209289 |
| ChEBI | CHEBI:31398 |
| ChEMBL | CHEMBL229635 |
| ChemSpider | 15319 |
| DrugBank | DB01943 |
| ECHA InfoCard | 100.021.340 |
| EC Number | 203-973-9 |
| Gmelin Reference | 82255 |
| KEGG | C14116 |
| MeSH | D003993 |
| PubChem CID | 8177 |
| RTECS number | KL5950000 |
| UNII | C9139X6ED7 |
| UN number | UN1161 |
| CompTox Dashboard (EPA) | DTXSID2020707 |
| Properties | |
| Chemical formula | C6H14O3 |
| Molar mass | 134.18 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Sweet odor |
| Density | 0.944 g/cm³ |
| Solubility in water | miscible |
| log P | -0.36 |
| Vapor pressure | 0.49 mmHg (20 °C) |
| Acidity (pKa) | 45. |
| Basicity (pKb) | 0.21 |
| Magnetic susceptibility (χ) | -9.57e-6 |
| Refractive index (nD) | 1.378 |
| Viscosity | 0.88 mPa·s (20 °C) |
| Dipole moment | 2.21 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -532.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2985.8 kJ/mol |
| Pharmacology | |
| ATC code | D08AX06 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H302, H319 |
| Precautionary statements | Precautionary statements: "P210, P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 41 °C |
| Autoignition temperature | 160 °C |
| Explosive limits | Explosive limits: 1.5–12% |
| Lethal dose or concentration | LD50 (oral, rat): 5,350 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 5670 mg/kg |
| NIOSH | UU8790000 |
| REL (Recommended) | 1 ppm |
| IDLH (Immediate danger) | 300 ppm |
| Related compounds | |
| Related compounds |
Dimethoxyethane Diethylene glycol diethyl ether Diethylene glycol methyl ether Triethylene glycol dimethyl ether |

