Digging Into Diethylene Glycol Butylether Acetate: More Than a Chemical Name
From Lab Curiosity to Industrial Mainstay
The story of diethylene glycol butylether acetate (DEGBEA) stretches back to the age when chemists wore thick goggles and carried curiosity like a badge. Early researchers started working with glycol ethers and their acetates in the 20th century. They searched for materials with unique dissolving abilities that wouldn’t evaporate too quickly or pose the same risks as earlier solvents. Over decades, DEGBEA carved out its spot across several industries, not by accident, but through a long stretch of research and a lot of lessons learned from working with similar families of chemicals. People discovered that DEGBEA had a knack for blending with both water and oil-based systems, making it more versatile than most household names in the solvent world. It’s not just the name that’s a mouthful—its impact covers a lot of ground.
Getting to Know This Compound
People often gloss over the differences between glycol ethers, but DEGBEA stands out for its unique combination of a gentle odor, stability, and flexibility. It usually turns up as a clear, colorless liquid that doesn’t shout for attention on the workbench. Its boiling point, somewhere over 240°C, keeps it from vanishing in hot processes, and it resists freezing even when temperatures dip well below zero. Chemically, DEGBEA brings together the solubility of the glycol ethers and the slow-evaporating quality of an acetate, which explains why so many professionals reach for it in tough jobs. Its balanced polarity helps it dissolve resins, greases, dyes, and inks that other solvents leave behind untouched.
What Goes Into Making DEGBEA?
Manufacturing DEGBEA isn’t all that flashy but benefits from the steady hand and sharp eye of skilled chemists. They start by reacting diethylene glycol mono-n-butyl ether with acetic anhydride. This transesterification creates the acetate group that sets DEGBEA apart from many regular glycol ethers. Conditions need to be kept tight for temperature and purity; the slightest slip can spoil the product or mess with yields. Quality control people run plenty of tests—checking water content, purity, and UV absorbance before a batch leaves a plant. All this isn’t just for show; it makes a real difference for downstream users.
Where the Science Meets the Shop Floor
DEGBEA doesn’t stay confined to flasks or spreadsheets—it moves from labs to car paint shops, printing presses, and electronics plants. Its technical specs often include a purity north of 98%, with very low moisture content. This dedication to high standards prevents unwanted reactions or defects in the finished goods. On product labels, you’ll find names like Butyl Carbitol Acetate or 2-(2-Butoxyethoxy)ethyl acetate. These show up in technical sheets and safety documents, reminding experienced workers to keep all the synonyms straight, especially if they have to handle compliance documents.
Everyday Hazards and How to Work Around Them
No industry veteran ignores the risks that come with handling solvents, and DEGBEA deserves respect. Its low toxicity gives it an edge over some nastier chemicals, but that doesn’t make it harmless. I’ve seen workers let their guard down, thinking “it’s just another glycol ether.” Eye protection and ventilation make all the difference here. The compound’s slow evaporation lulls some into a false sense of security, but skin exposure or breathing too much vapor can build up over time. Modern safety standards draw firm lines around how to handle and store DEGBEA. At the same time, ongoing research into its safe disposal and spill control continues, led by persistent toxicologists wanting to close the gaps that remain.
Applications Too Important to Ignore
In my experience, DEGBEA’s real claim to fame comes in the world of coatings and paints. It helps dissolve tough acrylic and alkyd resins, keeping paint stable and workable until it hits the surface. This matters in automotive refinishing, where a streaky or bubbled finish just won’t fly. Printers trust DEGBEA to keep inks from drying out too fast on the rollers. Electronics makers use it for cleaning circuit boards because of its manageable evaporation profile. Its ability to blend into both waterborne and solvent-based products helps manufacturers cut down on the number of solvents they keep in stock. This saves money and space, but more importantly, it reduces the tangle of safety protocols and cross-contamination risks that crop up with dozens of specialty solvents.
Looking Deeper: Chemical Reactions and Adaptations
Chemists never sit still, and they’ve found ways to combine, break down, or modify DEGBEA to suit new jobs. Because it carries both ether and ester groups, it shows up in synthesis pathways for advanced resins or complex plasticizers. Its structure resists many common degradation routes, which helps products last longer in hot or harsh conditions. That longevity pays off in paints and coatings designed to withstand weather, sunlight, or abrasion. There’s also a trend toward making DEGBEA-compatible blends that reduce the amount of harsher solvents in consumer products, which lines up with environmental regulations that grow tighter each year.
Weighing Health Risks and Gaps in Knowledge
Research into DEGBEA’s toxicity gives it a relatively clean bill of health compared to more notorious solvents. Acute exposure tends not to send people to the emergency room, but chronic effects haven’t been mapped out as thoroughly as some would prefer. Certain glycol ethers in the same chemical family raise legitimate worries about toxicity to developing fetuses or damaging blood-forming cells. While DEGBEA lacks some risky groups found in other ethers, long-term and repeated exposure still calls for caution. This is an area where more transparent data sharing and third-party research could do a lot of good, especially as companies push for “greener” chemicals without sacrificing worker safety.
Innovation and the Road Ahead
Researchers keep hunting for ways to improve on DEGBEA or swap it out for something with less environmental baggage. Stringent VOC rules and workplace safety laws keep pressure on manufacturers and scientists to either dial down emissions or come up with new molecules altogether. Some hope lies in fermentation-fed “bio-based” glycol ethers, but no one has found a drop-in replacement that matches DEGBEA’s price, performance, and availability. For now, it remains a benchmark in the world of slow-evaporating, forgiving solvents. The companies who use or sell it can’t just lean on old habits; they need to keep up with regulations, talk to the people who make and use the stuff every day, and put effort into training and monitoring that addresses the realities, not just the paperwork. People have spent almost a century with this chemical, and it’s clear that its real value comes from balancing performance, safety, and a willingness to adapt to what the world needs next.
What is Diethylene Glycol Butylether Acetate used for?
Everyday Chemistry in Action
Diethylene glycol butylether acetate sounds like a handful, but it quietly touches more parts of daily life than most folks realize. This chemical comes up often in industries that shape the way modern products look, feel, and last on shelves. Through my years covering science and manufacturing, I’ve learned how substances like this fuel progress but also demand careful handling.
The Backbone of High-Quality Paints and Coatings
Walk into any hardware store, and you’re bound to see cans splashed with vibrant colors and promises of durable finish. The secret sauce hiding inside many of these cans often includes diethylene glycol butylether acetate. Paint makers love its knack for dissolving pigments and resins. That makes paint flow smoothly, dry evenly, and stick with strength. Spray coatings on cars, furniture, and architecture depend on this chemical because it helps create that clean, streak-free look. From maintenance folks painting city benches to car factories rolling out rows of shiny vehicles, this solvent keeps the wheels turning.
Why Electronics, Printing, and Cleaners Use It
Flip over your phone, laptop, or TV remote—most have some coating or printed label. Manufacturers of electronics tap diethylene glycol butylether acetate for a couple of big reasons. For printed circuit boards, this chemical cleans surfaces before assembly and acts as part of specialty inks and adhesives. Ink makers also count on its slow evaporation, which helps give detailed prints that resist smudges.
Cleaning products targeting greasy or sticky grime often list this solvent among their top ingredients. Its ability to break down stubborn residues without leaving streaks or ugly films makes it valuable in both household and industrial cleaners. I’ve seen janitors in hospitals trust products containing this substance for tough messes, knowing it won’t damage surfaces.
Growing Role in Sustainable Formulations
Solvents like diethylene glycol butylether acetate play a crucial part in shifting toward greener chemistry. Manufacturers constantly look for ways to shrink their environmental footprint. Some waterborne and low-VOC paints have adopted it to boost performance but limit harsh emissions. Compared to older, more volatile solvents, this one gives a cleaner working environment and less air pollution. There’s a trend to phase out chemicals with known health risks, and this substance replaces harsher options when toxicity and fire hazards matter. It doesn’t solve every safety issue, but it offers a safer pathway when managed correctly, according to regulatory guidelines.
Handling Safety and Community Health
Like every industrial chemical, diethylene glycol butylether acetate comes with its own set of risks. In the factories where it’s handled, workers gear up to avoid breathing it in or getting splashed. I’ve interviewed safety officers who stress the importance of proper ventilation and protective gear. Regulations from agencies like OSHA and the EPA keep companies on their toes, making sure spills and emissions stay in check.
Some stories in the news highlight accidents tied to poor handling. From chemical exposures to storage mishaps, the lesson is always the same: clear safety plans protect both workers and the neighborhoods near industrial zones. Community pressure and watchdog organizations have pushed many companies to upgrade both containers and training protocols.
Looking Forward
The continued use of diethylene glycol butylether acetate leans on responsible stewardship. Manufacturers seeking cleaner, more efficient processes keep testing greener substitutes, while chemical engineers improve ventilation and recovery systems. For everyone from homeowners with a paintbrush to city planners choosing construction materials, understanding what goes into a product—and how it’s managed—matters more than ever.
What are the safety precautions when handling Diethylene Glycol Butylether Acetate?
Getting the Basics Right
Workshops and labs run on chemicals, but people run on habits. Bad habits show up fast when folks pour or transfer something like Diethylene Glycol Butylether Acetate (DGBEA) without stopping to ask about safety. I learned early in my career how easy it is to let familiarity creep in, until one day someone lets a solvent splash on bare skin. DGBEA won’t grab headlines like some dangerous substances, but it packs its own risks—skin and eye irritation, headache, even toxicity after big spills.
Personal Protection: No Excuses
I remember a painter who once told me he “never wore gloves, not since the ‘90s.” He paid for it with cracked hands and long-term sensitivity. DGBEA is no friend to exposed skin. Anyone who wants to avoid itching, dryness, and possible absorption into the bloodstream should throw on nitrile or neoprene gloves. A thick cotton shirt helps, but aprons or coveralls work better if there’s a chance of splashing. For eyes, goggles beat glasses, mostly because those side shields make the difference between a minor annoyance and a real emergency.
Ventilation: Keep Air Moving
I’ve seen too many small shops break out DGBEA behind a half-closed garage door. Solvents like DGBEA make the air feel heavier, and headaches often follow a full day of breathing what you can’t see. Simple fans can push fumes away, but anyone handling more than a dab needs proper exhaust systems—fume hoods, open windows, or local extraction units. These setups keep those jobs safer and cut down the odds of long-term health problems.
Handling and Storage: Respect and Routine
DGBEA doesn’t explode or catch fire at a sneeze, but flammable vapors can gather fast, especially near heat. Sturdy metal drums or plastic containers with sealed lids work best. I always make sure labels stay bright and clean, so nobody reaches for the wrong thing. A spill kit nearby—absorbent pads, neutralizers, and waste bins—unlocks a quick response instead of panic. Some businesses store volatile chemicals in an old closet; smart managers drop a thermometer inside and check it weekly, because temperature swings mean extra risk.
Clean-Up: Small Moves Add Up
More than once, I’ve found out the difference between a safe shop and a sloppy one is a mop and some absorbent sand. No one wants to chase a slick across the floor, so spills—big or small—get cleaned up right away. Soaked rags and waste find their way into proper bins marked for hazardous disposal, not ordinary garbage. Anyone who’s handled a chemical fire caused by a tossed oily rag doesn’t forget that lesson.
Training and Mindset: Make It Routine
Some folks see training sessions as a chore. Years of experience have taught me no cheat sheet or video beats taking fifteen minutes to walk a new team member through basic rules—the where, why, and how of good habits with DGBEA. Workers speak up more when they know the risks, know where the gear is, and feel like their safety matters to the people in charge.
Final Thoughts: Look After Each Other
No single precaution covers it all with chemicals like DGBEA. Wearing gloves and goggles, keeping air fresh, and treating spills with respect build habits that spread on the shop floor and back home. A safe shop costs less in lost time, fixes, and worry. People trust places where management and workers both stick to smart routines, making everyone safer in the long run.
Is Diethylene Glycol Butylether Acetate hazardous to health or the environment?
What This Chemical Does In Everyday Products
Diethylene glycol butylether acetate, often found in paints, coatings, and cleaning agents, plays a role as a solvent. Its clear liquid form seems harmless at first glance, used in factories and sometimes in workshop projects at home. It helps dissolve other compounds, making it easier for paints to glide onto surfaces or for cleaners to cut through grime. Many of us will come in contact with this chemical without even knowing its name.
What Science Says About Health Risks
Breathing in fumes from diethylene glycol butylether acetate often leads to headaches, dizziness, throat irritation, and nausea, especially if the exposure is indoors where vents do not work well. Higher concentrations and longer exposure times increase these risks. Some studies show harmful effects on blood and kidneys if people are exposed repeatedly. The evidence for long-term cancer risk from this chemical in humans remains limited, but serious eye and skin irritation appear often in reports from people who work with this solvent daily.
Personal experience in industrial environments tells me people sometimes forget to wear gloves or goggles. Shortcuts happen, especially during busy shifts when workers focus on meeting production targets. Even if companies provide protective gear, one missed step causes skin rashes or irritated eyes. Spill cleanup often gets rushed, and tiny splashes left behind stick around longer than intended.
Impacts on the Environment
Factories discharging water containing this chemical into local streams or wastewater adds to pollution. Diethylene glycol butylether acetate breaks down in the presence of sunlight, but not all of it disappears quickly. Aquatic life takes a hit as some fish and invertebrates react badly to chemicals like this leaching into waterways, resulting in toxic stress and sometimes death at high concentrations. Honeybees and birds are less affected, but indirect consequences arise when their water sources get contaminated.
The chemical’s vapor can also escape into the air and linger, contributing to urban air quality issues. Returning home after a day spent in recently painted or cleaned rooms often leads to mild symptoms—tiredness, sore throat, headaches—that fade by morning, but vulnerable people like children or those with asthma can struggle more.
What Steps Actually Help Limit Harm
Better ventilation tops my list for reducing risks indoors. Opening windows, using exhaust fans, and working outside go a long way. Gloves, goggles, and proper masks reduce skin and inhalation risks for workers handling this solvent. Simple habits, like handwashing after handling products or labeling containers clearly, also help.
Companies can reduce harmful releases by investing in water treatment and air filtration. Government rules on solvent disposal keep large spills out of rivers and landfills. Substituting with less toxic solvents also makes a difference, though some performance trade-offs exist. I’ve seen workshops phase out more hazardous chemicals just by listening to health complaints from staff. Change often starts with speaking up about symptoms, reporting spills, and seeking safer alternatives when possible.
Building Awareness Matters
Understanding the effect of chemicals like diethylene glycol butylether acetate gives workers, consumers, and neighbors the power to demand safer practices. Health and environmental risks increase without vigilance or regulation. Experience—and evidence—both say that small actions add up, whether it’s wearing gloves at the bench or choosing products with fewer toxic ingredients for the home.
What are the storage and handling requirements for Diethylene Glycol Butylether Acetate?
Getting Practical About Safe Chemical Storage
Diethylene glycol butylether acetate crops up across industries: paints, inks, coatings. Many folks outside a lab have never seen its clear liquid form or smelled its mild odor. Still, anyone who works around it knows clear steps can make a difference between a clean operation and an expensive disaster. Occupational safety isn’t a box to check—it’s a way of treating your team and community with respect.
Why Temperature and Ventilation Truly Matter
Chemicals like this aren’t ordinary. Left too hot or under direct sunlight, it can break down or trigger reactions. Regulations call for cool, dry spots, far from heat and ignition sources, and that’s not just busywork. Keeping it below 30°C is a small ask compared to an insurance claim or a ruined batch of product. I’ve seen projects hit snags just because drums sat near a back door, catching sun all afternoon. It pays to check that storage plan after the truck pulls up, not just before a surprise audit.
How Segregation Protects Plant and Workers
Not every substance mixes safely. Strong acids, bases, and agents like oxidizers can turn a boring garage into a hazard zone. Keeping chemicals apart is low-tech but effective. I’d rather lose a little shelving space than see two incompatible drums staged together. GHS labels help, but readable signage and a crew that talks through changes in stock or layout helps more. Every worker’s memory gets fuzzy, so good logs and quick walkthroughs avoid nasty surprises down the line.
Why Container Choice Can’t Be an Afterthought
Steel or HDPE drums offer enough strength without reacting with diethylene glycol butylether acetate. Any leaks or rusty seams spark short-term headaches and long-term bills. It’s not just about the main drum—trucks, buckets, pipes, and pumps all see contact. Small habits, like wiping threads and checking for cracked seals, keep things safe. Even old-timers get tripped up by tiny oversights, so double-checks and open conversations on the floor help keep everyone honest.
Spill Response: Simple Steps Make the Difference
Minor spills can happen anywhere. Cleanup materials—sand, vermiculite, or simple spill kits—don’t cost much, but someone’s got to refill them and make sure they’re in arm’s reach. I’ve watched teams freeze up over training gaps or confusion about PPE. Gloves, goggles, and aprons belong out, not locked up. The best workplaces drill these drills until nerves take over and smart moves become second nature.
Health and Air Quality Go Together
Vapor isn’t always visible. Breathing it in may dry throats, irritate eyes, and cause headaches after long exposure. A basic fume hood or even cracked windows cut risk fast, and simple air sensors take away the guesswork. Some shops resist investing in those upgrades, but ask anyone who’s been stuck in a closed room with a spill or a slow leak—clean air’s always worth it.
Record-Keeping and Ongoing Training Build Trust
From OSHA to local fire marshals, records matter. Inventories, hazard sheets, and updates should be current, not stowed in an office drawer. Monthly refreshers, not once-a-year PowerPoints, keep staff alert. People trust an operation where communication is direct and leaders roll up their sleeves to show safe habits aren’t just for visitors or show.
Experience Teaches—But Respect for Rules Keeps Us Safe
No one brags about the time they followed every protocol and nothing went wrong. Still, those routines matter. A few sharp habits can save a lot of heartache, protect a company’s future, and keep families whole. Facts matter. So does showing folks you care enough to expect the best effort, every shift.
What is the shelf life and proper disposal method of Diethylene Glycol Butylether Acetate?
Why Shelf Life Matters
Diethylene glycol butylether acetate shows up in paints, coatings, and all sorts of industrial cleaners. Some folks see a bottle sit on a shelf for years, thinking it's as good as new. That's not always true. If you use it past its prime, you risk slower evaporation or unwanted reactions. Best shelf life happens with tightly closed containers kept away from humidity and direct sunlight. On the label, you’ll often see two to three years if it’s been stored right. Past that, changes in color or smell usually say it’s time for something fresh. A trusted supplier can help with shelf life questions for any batch you’re not sure about.
Keeping Things Safe: Why Expiry Dates and Storage Matter
Chemicals stick around in storage, collecting dust because nobody tracks expiration dates. Letting old chemicals pile up might sound harmless, but it’s a shortcut to leaks, rusty drums, and even fumes sneaking out. Employees or family members who don’t know what’s in that container could handle it the wrong way. This stuff can irritate eyes, skin, or lungs if it breaks down or someone opens it without proper gear. Reliable tracking of purchase and opening dates keeps surprises to a minimum. Regular inspections spot crusty residue or bulging caps, both of which scream “dispose of me.”
Disposal: Getting It Right
Packing up leftover solvent and tossing it in the trash can cause headaches for people and the planet. Diethylene glycol butylether acetate harms water sources if it leaks out of landfill. Local wastewater treatment plants aren’t built for solvents, so pouring it down a drain sends it out into rivers and lakes. My own town has regular hazardous waste collection days through the local recycling center. If I have half a jug leftover, I book a slot and let the pros handle it. Wearing gloves, goggles, and keeping the container tightly sealed for transport keeps it all safe and above board. Some areas let you schedule chemical pickups, which works great for bigger stashes.
Environmental and Regulatory Stakes
Violating hazardous waste laws brings hefty fines. United States rules call for proper labeling, sealed transport, and tracking who handles waste. Dumping or mixing solvents with regular trash breaks state and federal law. The Environmental Protection Agency keeps a list of certified hazardous waste handlers for most regions. Countries may differ, though. Europe, for instance, puts special focus on tracking chemical movement, all the way from user to final disposal.
Looking Ahead: Common Sense Steps
Nobody likes a stockroom full of forgotten bottles, but lots of organizations still use “just in case” ordering. Updating inventories once a year, using a “first-in, first-out” rule, and sharing leftover stock between departments limits waste. Anyone buying chemicals needs safe disposal plans in place before the first drum arrives. Staff training on handling, storage, and first aid means everyone goes home healthy. Renting or borrowing instead of buying for one-time jobs also cuts down on unused chemicals. In the end, using only what you need and disposing of the rest by the book pays off for your wallet, well-being, and your local environment.
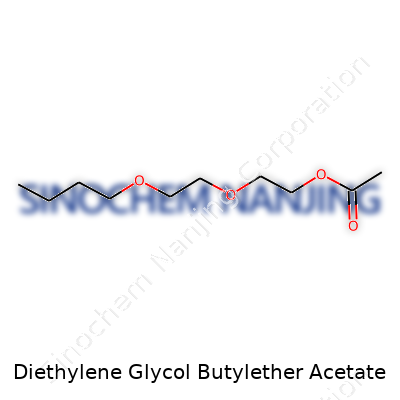
| Names | |
| Preferred IUPAC name | 2-(2-butoxyethoxy)ethyl acetate |
| Other names |
Butyl carbitol acetate 2-(2-Butoxyethoxy)ethyl acetate Butyl diglycol acetate Butoxyethoxyethyl acetate DEGBEA |
| Pronunciation | /daɪˈɛθɪliːn ɡlaɪˈkɒl ˈbjuːtɪl ˈɛθər əˈsiːteɪt/ |
| Identifiers | |
| CAS Number | 124-17-4 |
| Beilstein Reference | 2038706 |
| ChEBI | CHEBI:81839 |
| ChEMBL | CHEMBL1697827 |
| ChemSpider | 65359 |
| DrugBank | DB14177 |
| ECHA InfoCard | 100.039.250 |
| EC Number | 203-933-3 |
| Gmelin Reference | 66807 |
| KEGG | C19596 |
| MeSH | D002948 |
| PubChem CID | 8186 |
| RTECS number | KL3325000 |
| UNII | 9B35R6ATJV |
| UN number | UN3271 |
| Properties | |
| Chemical formula | C12H24O4 |
| Molar mass | 234.30 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Mild, ester-like |
| Density | 0.984 g/cm³ |
| Solubility in water | miscible |
| log P | 1.01 |
| Vapor pressure | 0.02 mmHg @ 20°C |
| Acidity (pKa) | pKa ≈ 15 |
| Basicity (pKb) | 8.2 |
| Magnetic susceptibility (χ) | -8.25×10⁻⁶ |
| Refractive index (nD) | 1.415 |
| Viscosity | 1.9 mPa·s (25°C) |
| Dipole moment | 2.45 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 435.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -773.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5377.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes serious eye irritation. May cause drowsiness or dizziness. |
| Precautionary statements | Precautionary statements: P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P403+P235, P405, P501 |
| Flash point | 75°C |
| Autoignition temperature | 230 °C (446 °F; 503 K) |
| Explosive limits | 1.1–10.5% |
| Lethal dose or concentration | LD50 oral, rat: 6500 mg/kg |
| LD50 (median dose) | 6500 mg/kg (rat, oral) |
| NIOSH | WA9475000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Diethylene Glycol Butylether Acetate: Not established |
| REL (Recommended) | 5 ppm |
| Related compounds | |
| Related compounds |
Ethylene glycol butyl ether acetate Diethylene glycol Diethylene glycol monoethyl ether acetate Ethylene glycol monobutyl ether Diethylene glycol monoethyl ether |

