Diethylamine: More Than Just a Building Block
Looking Back: The Early Days of Diethylamine
Diethylamine draws its roots from the adventurous days of modern chemistry, swept up with the likes of other alkylamines explored in the 1800s. Chemists searched for versatile molecules to push both industry and research forward. Once known by pungent odor long before anyone could test it with modern technology, diethylamine became a staple in the workbenches of research pioneers. Turns out, as soon as you grasp its straightforward structure—basically two ethyl groups hooked to an amine—you start to see why it caught the attention of so many. Move forward into major production eras and its role in both small-scale labs and mega-scale factories became obvious. Today, you scarcely talk about the chemistry of amines or pharmaceuticals without bumping into this compound’s legacy.
Digging Into the Stuff: What Diethylamine Looks Like
Open a bottle of diethylamine, and its fishy, sharp scent fills the air, warning to use care. This colorless liquid brings more than odor—it's highly flammable and mixes well with water, ethanol, and ether. It boils at a modest 55°C, sinks in at a density lighter than water, and evaporates just by sitting around unsealed. For most folks familiar with working around chemicals, that quick jump from liquid to vapor is a perfect test for keeping things ventilated and avoiding accidents. Its chemical formula, C4H11N, captures a lot in a small package. The molecule's shape lets it slip into reactions with acids, acyl chlorides, and alkyl halides without much ado. Look up any naming convention and you’ll see 'N,N-Diethylamine' pop up right alongside 'ethylamine, diethyl-', 'DEA', or maybe even 'secondary amine' in certain circles.
Getting Hands-On: Making and Modifying Diethylamine
Making diethylamine hasn't changed much since chemists first bubbled ammonia through ethanol. Most processes today start with ethyl chloride or ethyl alcohol and ammonia, warming things up in the presence of catalysts, letting the reaction spit out this amine along with mono- and triethylated cousins. The technical hurdles come down to controlling the ratio and separating the mix, since you get a bundle of products and nobody wants to waste costly materials. On a smaller scale, even amide reduction routes take you to diethylamine, though industrial production prefers bulk reactions. Hint: you don’t get gold medals for green chemistry just by running these old processes. Future work needs better methods, cleaner solvents, safer equipment. Tweak the molecule with acyl groups, and it turns into intermediates for dyes or drugs, or land on its hydrochloride, making transport less risky. Chemists trust the reactivity profile—simple, predictable, and handy for building more complex molecules.
Inside the Label: Technical Specs and Safe Handling
No one gets their hands on diethylamine without hearing about the hazards. Pure diethylamine demands attention—flammable vapor, skin and eye irritant, and a risk for explosive mixtures if things aren’t stored just right. Containers need tight seals, and nobody wants to sniff it for fun. Labels focus on keeping people clear about the risk—avoid open flames, ventilate well, keep well away from oxidizers. Responsible outfits run regular safety drills, keep proper gloves and goggles on hand, and monitor air quality in storage or working spaces. Most vendors stick to technical standards—purity typically hovers up to 99%, keeping residual water and ammonia in check. You might find quality specs around industrial versus lab grades, where buyers decide how much impurity they can tolerate based on their own recipes or final products.
Where the Work Happens: Research, Industry, and Beyond
Ask around in the agrochemical business, and diethylamine sits up there as a backbone—churning out herbicides, fungicides, and insecticides. The pharmaceutical sector leans on it to start syntheses that build more complicated drug molecules. Rubber accelerators, corrosion inhibitors, flotation agents for mining—all trace their roots to barrels of diethylamine. Paints, dyes, even photographic chemicals owe a nod to this amine, giving color and stability to things we often take for granted. My past work in the chemical industry threw me into hands-on trials with diethylamine-derived surfactants and specialty polymers—it shows real flexibility, especially where a base or nucleophile can open up a reaction pathway.
Toxicity—A Real-Life Risk, Not Just a Stat Sheet
Toxicologists have put diethylamine through its paces. Vapor inhalation burns throats and lungs. Spills eat at skin, sting the eyes, and can trigger headaches, coughing, and nausea even with short exposure. Chronic cases raise flags for liver, kidney, or lung distress. Trustworthy research recognizes that workplace exposure remains the major threat, since most everyday folks will never bump into this stuff. Production facilities install high-end scrubbers and proper containment because one short lapse can leak vapor quickly. Environmental chemists worry about what happens if diethylamine escapes into water or soil, noting that the profile for degradation isn’t benign. Regulators insist on air monitoring and medical checks in work environments, trying to keep exposure well below calculated occupational limits. The health data is backed by hard facts—not just anecdotal worries—underpinning strict safety and disposal rules.
The Long Road Ahead—Research and Better Ways Forward
Diethylamine gets a steady stream of attention from research teams. Ongoing studies track its reactivity on metal catalysts, its uses in emerging pharmaceuticals, and improved synthetic routes that cut waste and energy usage. One promising line involves greener chemistry—swapping fossil fuels for renewable feedstocks, using bioprocesses, or developing closed-loop systems that recapture any off-gases. Environmental groups and chemical companies see eye-to-eye on making these plants cleaner. A handful of recent papers examine biodegradable derivatives, with an eye on safer, less persistent alternatives. For the next wave of applications, smart chemists think beyond bulk commodity use, tapping into next-generation polymers, sustainable pesticides, and even custom-tailored surfactants.
What Matters Most
Diethylamine’s story isn’t just about a single molecule or chemical formula. It captures a snapshot of chemical progress—balancing practical uses against very real risks. Companies, regulators, and researchers know that marching ahead with old technologies won’t meet today’s environmental or safety expectations. Experience tells me that every corner of this industry demands teamwork: safe handling, rigorous training, and a keen eye for new advances in production. The footprint of diethylamine runs deep, from factory floors to farm fields and clinical test tubes, reflecting human drive to shape the building blocks behind so many modern materials. For all the risks and challenges, those who use it responsibly build toward a future where chemical progress meets safety and sustainability, not just productivity.
What is Diethylamine used for?
Popping up in Pesticides and Farming
Spend a little time around farms and chemical supply warehouses and you’ll spot containers labeled with names most folks can barely pronounce. Diethylamine falls into that group. Farmers and agri-businesses know it well, since this compound goes straight into the mix for herbicides. Keeping weeds down isn’t just about looks—overgrown fields mean lost harvests. Research from the National Pesticide Information Center points out that diethylamine is a key player in producing 2,4-D, a herbicide found in wide use for row crops like wheat and maize. Farmers trust it to protect yields, but it has to be handled with care and know-how for safety—basic gloves, respirators, and solid training protect people who use it.
Building Blocks in Manufacturing
Factories often buzz with smells not everyone loves, and there’s a good reason—many rely on chemicals like diethylamine to get the job done. Paints, dyes, pharmaceuticals, and rubber manufacturers turn to this compound as a starter ingredient, not something folks see in the finished goods. My own uncle worked in a carpet dye facility where diethylamine-based chemicals added bright, lasting tones to the thread. Heavy industry reviewers like Chemical Safety Facts also highlight its role in making solvents, corrosion inhibitors, and flotation agents for mining—contributing quietly to products in homes and businesses.
In Pharmacy Labs and Research
Lab techs and scientists run controlled reactions with diethylamine to build molecules needed for medicine. Companies use it to produce antihistamines, antibiotics, and pain relievers. Even though you won’t see this name at your neighborhood drugstore, its fingerprints show up in final products that keep people healthy. The process calls for tight oversight and trained professionals, since diethylamine can irritate skin and eyes and needs safe storage and handling. Regulatory agencies like the FDA and OSHA stress strict labeling, clear documentation, and regular training—they don’t make these rules up for fun.
Risk, Regulation, and Ways Forward
Diethylamine makes things possible, but it’s no friend to the careless. It’s flammable and can harm people or the environment if not used right. Workers anywhere near it need real safety measures, not shortcuts. Companies that use chemicals have learned from bitter experience—clean-ups, fines, or worse—so more are investing in better storage, leak detection, and air handling systems. City inspectors stop by more often these days, which helps keep everyone honest.
There’s hope that future production can lean more on environmental tech. Labs explore alternative sourcing and safer substitutes all the time. Researchers from leading institutions keep looking for green chemistry routes that reduce fumes and waste. Farmers can turn to integrated pest management when it works, using less chemical and more natural solutions. But nothing swaps common sense—follow protocols, use protective gear, and stay up on the current regs.
Trust Through Responsibility and Transparency
The story of diethylamine isn’t mysterious to those who work with it every day. It provides countless modern comforts, but asks for respect along the way. The best results always come from knowledge—clearly labeling containers, giving workers the right training, and updating neighbors about plant operations. That trust and transparency make a real difference, not just for business but for everyday safety and peace of mind.
Is Diethylamine hazardous or toxic?
Getting Real About Diethylamine Risks
Walk into a chemical storage room at most universities or factories, and you’ll spot diethylamine by its distinct, sharp smell. This isn’t a household name, but its use runs deep in the world of industry. I remember in the lab during my student days, we all felt a bit uneasy around it—a handful of us got headaches after brief whiffs during experiments. The lesson always drilled into us: respect the chemicals, don’t take shortcuts with safety.
What Is Diethylamine, Anyway?
Diethylamine comes as a colorless, flammable liquid. The chemical formula—C4H11N—rolls off the tongue for chemists, but for most people, this simply means "it packs a punch." Used to make pharmaceuticals, dyes, and rubber processing chemicals, it lurks behind many finished products. Yet, workers rarely talk about it unless forced by an incident.
How Diethylamine Affects the Body
Inhaling or touching this substance brings trouble, even at low doses. Short-term exposure can irritate eyes, nose, throat, and skin. More intense contact burns the skin and causes severe eye damage. The headaches, nausea, and chest discomfort follow after inhalation. Anyone who’s spilled a drop on their skin will never forget the sting—it cuts through gloves if they’re not the right type.
Diethylamine doesn’t just sit on the surface. It moves into the system quickly. High concentrations can impact the lungs and nervous system, triggering coughing, breathing difficulty, and even confusion. In some cases, fluid collects in the lungs hours later, a delayed effect that makes it hard for doctors to diagnose quickly.
Long-Term Hazard
Workers who run into diethylamine at their job on a regular basis have bigger worries. Chronic exposure could lead to liver or kidney problems. Studies point to changes in blood and organ function after repeated low-level contact. The U.S. National Institute for Occupational Safety and Health (NIOSH) sets recommended limits for a good reason.
The chemical also catches fire with ease, releasing toxic fumes like nitrogen oxides in a blaze. I’ve seen a small flask explode in a fume hood because someone overlooked basic handling rules. The hassle of dealing with fire is nothing compared to the risk from inhaling those byproducts or getting splashed during cleanup.
Spotting Solutions that Work
Chemical safety doesn’t arrive on its own. Ventilation in storerooms and labs is the first shield. Working in a fume hood and wearing face shields, lab coats, and gloves rated for amines can keep diethylamine off your skin and out of your lungs. Clear labeling and strict inventory checks also help prevent those "surprise" exposures.
Training goes a long way, not just for chemists but for everyone on site. I’ve seen new hires pick up best practices simply by watching older employees treat each bottle with caution. This culture—respect for the hazards combined with up-to-date safety gear—ends up saving lives. Reporting leaks or spills without delay makes a difference, too.
Mental Health in the Mix
Handling hazardous chemicals builds a certain kind of stress. People in chemical industries need more attention to mental health support, as accidents and repeated near-misses pile up anxiety. Making room for open discussions about fears, mistakes, or improvements changes the workplace for the better.
No one wants to land in a hospital after routine work. Diethylamine’s dangers call for more than just personal caution—it needs attention across management, training, and even mental health support for those on the front lines. Industry leaders need to support strong safety standards, updating them as science teaches us more about these chemicals’ full effects.
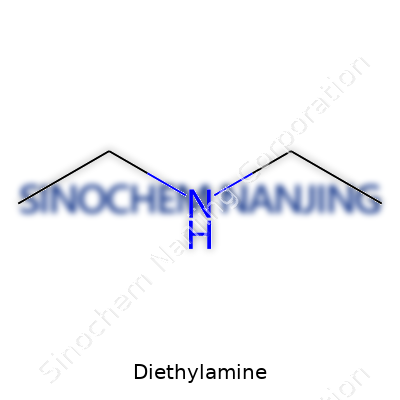
What is the chemical formula of Diethylamine?
Understanding Diethylamine
Diethylamine shows up in a lot more places than most people imagine. It’s used in producing pesticides, pharmaceuticals, dyes, and rubber chemicals. Its reputation in the science world comes from its straightforward structure: C4H11N. This formula gives it two ethyl groups connected to an amine group, and that simple makeup means it reacts easily with other chemicals. Lab workers and manufacturers usually spot it thanks to its strong, fishy odor, but it’s the simple numbers—four carbons, eleven hydrogens, and one nitrogen—that tell the real story.
Chemical Formula: C4H11N
The letters in diethylamine’s chemical formula—C4H11N—are more than a bunch of symbols. This formula guides handling rules, risk assessments, and the safe design of chemical processes. Knowing the formula lets you look up key properties such as boiling point (about 55°C), flammability, and basiciness. This one piece of information steers lab techs and safety managers toward the right protective gear, proper ventilation, and emergency protocols. In my own time working in a teaching lab, safety trainings always reminded us that a simple slip with a compound like this could lead to irritation or worse, so the formula is step one in staying alert.
Human Health and Environmental Effects
No one should take a chemical like diethylamine lightly. Exposure can irritate the skin, eyes, and respiratory system. In some settings, repeated contact leads to sensitization and headaches. That calls for thoughtful risk management, especially since some people underestimate the risk because the name doesn’t sound too threatening. Regulations expect manufacturers and labs to label chemicals with structural data, not just the name, so anyone handling it sees “C4H11N” and immediately recognizes what they’re working with. A clear label keeps people safer and helps emergency workers make quick decisions in a spill or accident.
Sourcing and Verification
Inconsistent sourcing comes up in chemical supply chains. I’ve seen new lab interns surprised when a minor change in supplier means you end up with a product that works a little differently than expected. Even a common formula like C4H11N can come with trace impurities. Trustworthy supply depends on quality standards like ISO certification, proof of purity, and batch-specific data sheets. Reliable data matters; a little confusion over what’s in the flask turns small mistakes into significant hazards, especially for research, pharmaceuticals, and agriculture.
Safer Handling Starts with Formula Knowledge
No one should walk into a lab or production floor without knowing what those chemical symbols stand for. If you’re responsible for training new staff or updating protocols, the first topic should include the real meaning behind formulas like C4H11N. Quality data, simple training tools, and clear communication make the difference between routine work and emergencies. In my own experience overseeing lab safety, even veteran staff admitted that periodic reminders on formulas and safety data helped avoid neglected steps and kept accidents down. Getting the basics—starting with the formula—prevents trouble before it begins.
How should Diethylamine be stored?
Diethylamine: More Than Just Another Chemical
Anyone who's spent time in a lab or factory recognizes that diethylamine isn’t just an everyday substance. This strong-smelling, flammable liquid brings along a real punch—unpleasant fumes, skin irritation, and even fire risk if handled without care. Practical storage plays a big part in keeping people safe and equipment intact.
Keep Diethylamine Away From Heat, Flames, and Sparks
Experience in tight, often hot, industrial spaces shows why diethylamine never belongs near open flames or heat sources. It has a low flash point, meaning it can catch fire at room temperature. One stray spark from a welding tool can trigger disaster. Always put containers in a cool, ventilated room. Don’t keep it next to workbenches, heaters, or sunny window sills—heat builds up much faster than folks expect.
Pick the Right Storage Containers
Too many people cut corners with generic containers. Diethylamine reacts with copper, zinc, and certain plastics, sometimes even corroding the inside of a drum. Go for steel drums lined with compatible coatings or polyethylene containers designed for strong chemicals. Make sure the lids seal tightly, because leaks don’t just stink—they seep into the air fast, risking exposure and corrosion.
Ventilation Matters
Anyone who's walked into a room full of chemical fumes remembers that headache, itchy eyes, or trouble breathing. Without steady airflow, vapor pockets can build up above diethylamine. Properly installed exhaust fans and fume hoods cut risk to workers. Even a cracked window barely makes a dent—strong fans or HVAC systems are essential. OSHA recommends regular monitoring for airborne concentrations, especially where there’s heavy use or transfer of the liquid.
Separate from Incompatible Chemicals
Mixing storage of acids and diethylamine brings real trouble. These mixtures release heat, toxic fumes, and sometimes even explosions. I once saw a near miss: an unlabeled jug of acid ended up next to a container of diethylamine. Only a sharp-eyed technician caught the error before something worse happened. Label all containers clearly. Keep diethylamine away from oxidizers, acids, and halogens at all times. Use designated shelves or safety cabinets with spill-proof bases to avoid cross-contamination.
Safety Gear Isn’t Optional
Direct contact with diethylamine can lead to burns, skin irritation, and breathing problems. I always stress gloves, goggles, and chemical aprons for anyone accessing storage. Spills and splashes happen when folks get careless. Emergency showers and eyewash stations offer last defense—if someone gets exposed, those seconds count for everything. OSHA and NIOSH provide specific gear suggestions; following these guidelines blocks most serious injuries.
Inventory and Emergency Planning
Leaving chemical storage unchecked invites disaster. Assign a trusted worker to check inventory dates and inspect containers for leaks or corrosion. Old supplies can turn dangerous and compromise container strength. Keep emergency numbers posted nearby, along with a clear spill protocol. Store absorbent materials and neutralizers close at hand. Practicing spill drills can prevent panic and serious damage.
Build a Culture of Safety
Routine training and open communication make all the difference. I like to ask new team members about chemical safety on their first day, and reinforce protocols every few weeks. Diethylamine doesn’t forgive mistakes. Management and workers share responsibility to keep storage safe. A strong safety culture lets everyone head home healthy at the end of the day.
What are the safety precautions when handling Diethylamine?
Understanding What You’re Working With
Diethylamine is sharp on the nose and pretty aggressive on the skin, known for catching people off guard if they take shortcuts. Used in making dyes, pharmaceuticals, and rubber chemicals, this clear liquid moves fast—vapors spread before you know it. In college, I once saw a classmate ignore the warning signs with amines. By lunch, his gloves had holes and he itched for days. No one shrugged off protective gear after that.
Personal Protective Gear Makes a Big Difference
Let’s talk gear. Lab coats, chemical goggles, and reliable gloves—preferably nitrile—form a solid first line. Latex doesn’t hold up well to amines, and I’ve seen folks take a gamble and regret it. A full-face shield matters during high splash risk. Your skin stings and eyes water even at low exposure, so skipping goggles blocks pretty basic common sense. Closed-toe shoes, nothing fancy. Long pants, always.
Ventilation Isn’t Optional
Sticking your nose over an open flask filled with Diethylamine means risking a nasty headache and coughing fits. Fume hoods weren’t designed to become drying racks; they keep those pungent clouds away from breathing space. At my first research internship, we shared one fume hood among four people and fought over it, but that was still better than nothing. Good airflow cuts down on exposure and limits problems if you spill or splash.
Smart Storage and Handling
I’ve learned there’s no such thing as being too careful with storage, especially with volatile chemicals like Diethylamine. Tight-fitting, clearly labeled containers reduce the chance of spills or accidental grabs. Keeping sources of ignition away helps—vapors catch fire easily, even from a small spark. Store it somewhere cool, separate from acids and oxidizers to keep those surprise reactions off your plate.
Clean-Up Protocols Matter Every Time
Don’t expect paper towels to save you if something spills. Mop up with absorbent material designed for solvents, and toss everything—rags, gloves, even broken glass—in a proper hazardous waste bin. Ventilate the area, and check with your institution’s chemical hygiene plan. Wash your hands even if you wore gloves. Complacency is where accidents start. At a chemical plant tour, I saw seasoned techs treat every clean-up like it would be their last of the day, no shortcuts.
Keep Emergency Measures in Reach
Knowing where eyewashes and safety showers sit saves time in a pinch. Once, a friend of mine got splashed and ran straight to the eyewash. He avoided permanent damage because he found it fast. No one wants to play memory games with emergency exits during a panic. Reviewing safety sheets and practicing response drills gets you ready for those moments when things go sideways—no one has time to read a manual mid-crisis.
Training and Respect Go Hand-in-Hand
Diethylamine isn’t something to take lightly. Good habits—like labeling, proper storage, PPE use, and spill planning—grow from solid training and not cutting corners for convenience. Trust your nose and your skin; if anything feels wrong, fix it before the small stuff becomes serious. Sometimes experience is a hard teacher, but smart preparation keeps those lessons mild.
| Names | |
| Preferred IUPAC name | N-ethyl ethanamine |
| Other names |
N,N-Diethylamine DEA Ethylamine, N-ethyl- Diethylamin |
| Pronunciation | /daɪˌɛθ.ɪl.əˈmiːn/ |
| Identifiers | |
| CAS Number | 109-89-7 |
| Beilstein Reference | 604068 |
| ChEBI | CHEBI:17547 |
| ChEMBL | CHEMBL1424 |
| ChemSpider | 682 |
| DrugBank | DB06732 |
| ECHA InfoCard | ECHA InfoCard: 100.003.142 |
| EC Number | 203-868-0 |
| Gmelin Reference | Gmelin Reference: 8417 |
| KEGG | C06102 |
| MeSH | D004037 |
| PubChem CID | 8030 |
| RTECS number | KK6825000 |
| UNII | 9T5K6G3JTV |
| UN number | 1114 |
| Properties | |
| Chemical formula | C4H11N |
| Molar mass | 73.14 g/mol |
| Appearance | Colorless to yellow liquid with a strong ammonia-like odor |
| Odor | Ammonia-like |
| Density | 0.707 g/mL at 25 °C |
| Solubility in water | Miscible |
| log P | 0.47 |
| Vapor pressure | 52.6 kPa (20 °C) |
| Acidity (pKa) | 10.98 |
| Basicity (pKb) | 3.00 |
| Magnetic susceptibility (χ) | -17.6×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.387 |
| Viscosity | 0.49 mPa·s (at 20 °C) |
| Dipole moment | 1.30 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 228.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −128.7 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2873.7 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | M01AB04 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS02,GHS06,GHS05 |
| Signal word | Danger |
| Hazard statements | H225, H302, H312, H314, H332 |
| Precautionary statements | P210, P261, P280, P304+P340, P305+P351+P338, P310, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | -28 °C |
| Autoignition temperature | 312°C |
| Explosive limits | 2–12% (in air) |
| Lethal dose or concentration | LD50 oral rat 540 mg/kg |
| LD50 (median dose) | 540 mg/kg (rat, oral) |
| NIOSH | BW6650000 |
| PEL (Permissible) | 10 ppm |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 40 ppm |
| Related compounds | |
| Related compounds |
Dimethylamine Ethylamine Triethylamine Isopropylamine Methylethylamine |

