Diethyl Oxalate: A Modern Look at an Old Chemical
Tracing the Roots: History and Early Insights
Looking back to the early chemical pioneers, diethyl oxalate has been on the scene for well over a century, long before most people thought about the industrial powerhouses that would use it today. Chemists in the nineteenth century saw the value in simple esters like this one, not just for the thrill of isolating new compounds, but for the real work they could do. By reacting ethanol with oxalic acid, chemists opened the door for new dyes, organic synthesis, and pharmaceutical building blocks. As technology advanced, diethyl oxalate made the move from curious laboratory specimen to factory-scale product, relied upon for everything from paints to medication synthesis. The demand has stayed steady in some fields, as chemistry departments and industry labs realized its practical value over and over, always finding some new corner where this simple ester could solve a problem or improve a process.
The Basics: Product, Look, and Characteristics
Walk into any lab and pop open a bottle of diethyl oxalate, and you’ll notice a colorless, clear liquid with a subtle, fruity smell. It isn’t flashy, and neither is its chemical signature—C6H10O4 breaks down into easily understood pieces, two ethyl groups attached to oxalic acid. Its boiling point falls comfortably in the 180s Celsius, staying liquid at room temperature, but watch out for its flammability. In the world of esters, diethyl oxalate stays relatively stable as long as things don’t get too rough: strong acids or bases can break it down, but careful handling keeps it in its lane. This combination of physical predictability and chemical usefulness means it finds a spot on many chemical shelves.
Spec Sheets and Real-World Labels
Chemists quickly learn to check for purity, acidity, and water content when working with diethyl oxalate. Purity matters—industry-grade products usually promise above 99 percent. Too much water, and reactions can go sideways. Labels on bottles make this clear in simple terms, pointing out hazards like flammability or the potential for skin irritation. Regulatory bodies expect clear warning phrases, not because diethyl oxalate hides extraordinary risks, but because good lab practice means not leaving anything to chance. Physical properties usually make their way onto spec sheets, including appearance, density, and boiling range.
How It Comes Together: Prepping Diethyl Oxalate
Solid practical chemistry shines through in the preparation of diethyl oxalate. The process follows an old but reliable route: blend oxalic acid and ethanol, add a catalyst such as sulfuric acid, and apply heat. The reaction proceeds with the release of water, which technicians often remove steadily to keep things moving forward. The resulting liquid is then separated, washed, distilled, and sometimes recrystallized to purify. It sounds simple, but it takes skill to get true purity, and scaling up introduces fresh challenges. Side reactions, residual water, and waste streams need control at every step.
Chemical Changes and Versatility
As a dialkyl ester, diethyl oxalate steps up as a starting material for organic synthesis. Chemists subject it to carefully planned reactions: saponification blasts it back to oxalic acid salts and ethanol, while reduction opens up new kinds of alcohols. Alkylation and condensation reactions put it at the center of syntheses for pharmaceuticals, agrochemicals, and dyes. It seamlessly hands off its ethyl groups or combines with amines and hydrazines to build heterocyclic compounds, fueling medicinal chemistry and material research. This flexibility ties directly to how useful it’s considered in R&D settings.
Other Names and Industry Terms
People rarely stick to one name. Diethyl oxalate gets called ethyl oxalate, Ethanedioic acid diethyl ester, or even just “oxalic acid, diethyl ester” on some shelves. These synonyms usually show up depending on the field or region, but their point remains—no matter what they call it, scientists know what they’re working with, and regulators keep a list of these variants to avoid confusion and maintain clear labeling.
Safety: Practical Realities and Operational Practices
No one in a modern lab treats diethyl oxalate casually. Safety glasses, gloves, and fume hoods stay standard, not just because of regulations but from the wisdom passed by lab veterans. Spills need prompt cleanup, mainly because the liquid is flammable, and skin contact leads to irritation. Working with open flames around it is a poor choice. It doesn’t take much effort to manage these risks; solid ventilation and clear labeling cut down on most dangers. Environmental concerns push labs to trap and dispose of vapors and residues, meeting current standards for emission control and chemical waste.
Where It Shows Up: Industrial, Academic, and Research Roles
Entire segments of fine chemical production count on diethyl oxalate. In the pharmaceutical field, it provides an intermediate step for drugs running the gauntlet from lab bench to clinical trial. Dyes, plastics, and agrochemicals follow similar logic—they reach for diethyl oxalate because of its unassuming but dependable reactivity. Analytical chemists use it for standardization and calibration. Even universities lean on it for teaching, letting students get hands-on with classic organic transformations. Niche uses in electronics and coatings keep it present in odd corners of industry, adding resilience to polymer structures or tweaking other esters for improved performance.
Research Horizons: Pushing the Boundaries
Researchers keep digging for new angles. The drive toward green chemistry presses for milder reaction conditions, more efficient atom economy, or renewable sources of oxalic acid. Studies have tried biocatalysts and new solvents to trim down on waste and energy, pressing toward more sustainable syntheses. Teams keep publishing new reaction schemes for rare pharmaceuticals and advanced materials, spinning diethyl oxalate into new shapes and functions. Every year, its chemical reach extends as researchers in Europe, Asia, and North America find creative uses for an old compound, inspiring eco-friendly approaches and novel products.
Toxicity in the Spotlight
Any conversation about diethyl oxalate includes a word about its effects on health. Short-term effects usually include eye or skin irritation, and the fruity smell doesn't mask the nagging concern if vapors stick in the air too long. Metabolism breaks it down into oxalic acid, which can compound risks for those with kidney troubles or in cases of large exposures. Strict industrial hygiene, detailed training, and careful storage keep incidents low. Studies on long-term exposure show little evidence of carcinogenicity, but researchers continue to track subtle effects. Responsible handling protects users and the environment, and clear communication in training rooms beats most accidents.
Looking Forward: Future Impacts and Opportunities
Diethyl oxalate holds a place at the crossroads of classic and cutting-edge chemistry. Its established uses won’t vanish, but new demands are already shaping its future. Sustainable synthesis, bio-based alternatives, and ongoing evolution in pharmaceutical needs keep it relevant. Technological pushes for lighter, stronger, more sustainable materials also hint at new uses emerging from ongoing research. Those who know the basics can leverage it in modern ways, driving innovation alongside safety and stewardship in the chemical industry. Its story blends a respect for tried techniques with curiosity about what comes next, ensuring it sticks around in laboratories, textbooks, and real-world applications for a long time to come.
What is Diethyl Oxalate used for?
Understanding the Role of Diethyl Oxalate in Chemistry
Diethyl oxalate might sound like something for specialists, but it crops up in places most folks never think about. Anyone who’s ever looked into what goes into making dyes or flavors has probably brushed past its name. In my days working in a small lab, I realized how often this compound showed up in chemical supply lists.
One of the main uses for diethyl oxalate lies in making other chemicals. Manufacturers lean on it to produce intermediates used for making pharmaceuticals, dyes, and perfumes. In the pharmaceutical industry, diethyl oxalate lends a hand with the synthesis of barbiturates, which doctors have used for years to treat seizures and sleeping disorders. The steps often start with diethyl oxalate and build up to more complex molecules. Even though newer drugs have replaced older barbiturates in most cases, those working in chemical manufacturing still count on diethyl oxalate for specialty preparations.
Diethyl oxalate opens the path for flavor and fragrance chemistry too. If you’ve got a favorite artificial flavor out there—vanilla, strawberry, raspberry—chances are good that diethyl oxalate sat somewhere on the production line to help build the mix. The chemists use it to construct esters, which give foods and perfumes their distinctive tastes and scents. Diethyl oxalate stands out among the classic “building block” chemicals for this reason.
Why Manufacturing Relies on It
The paint and coatings world takes advantage of diethyl oxalate for pigment production. Pigment makers reach for this oxalate ester to get brighter, longer-lasting colors for plastics, coatings, and even print inks. My experience with art supply companies showed how important these pigment improvements can be—even a small upgrade in color stability leads to better consumer products.
For folks on the electronics or polymer side, diethyl oxalate comes in handy for specialty solvents or as part of the chain in plasticizer manufacture. A chemist I knew worked in polymer modification, and she often mentioned how certain esters derived from diethyl oxalate improved the heat stability of her final products.
Safety and Environmental Impact
No one using diethyl oxalate should treat it carelessly. It can cause irritation if it lands on skin or if anyone breathes in the vapors. In my early years on the bench, my lab team stayed alert about keeping good airflow whenever we ran reactions involving it. Chemical manufacturers who use diethyl oxalate are already following strict workplace safety rules due to the risk of exposure.
Environmental concerns always deserve attention. Diethyl oxalate doesn’t linger in soil or water for long, but improper waste disposal can cause trouble for local waterways and aquatic life. Factory operators need reliable protocols for capturing waste and stopping any spills before they escape.
Building Safer and Smarter Practices
Solving the safety and environmental issues comes down to smart planning. Companies can invest in better fume hood systems, frequent safety audits, and stepped-up worker training. Regulators can require cleaner production techniques using less hazardous alternatives when possible—some research teams experiment with greener solvents and catalysts. Anyone involved in chemical sourcing or product development can look for suppliers who demonstrate real transparency about their safety systems.
On the scientific front, continued research into new esterification methods might eventually lead to less reliance on oxalate esters. Until then, diethyl oxalate keeps earning its place in laboratories, plants, and research projects worldwide.
Is Diethyl Oxalate hazardous or toxic?
What Folks Need to Know About Diethyl Oxalate
Diethyl oxalate sometimes pops up in the lab or in industry as a solvent or an intermediate for making things like pharmaceuticals and dyes. It's a clear, oily liquid. To most people, it sounds like something harmless—maybe just another chemical tool on the shelf. Folks who work around chemicals know tools like this one can come with risks that do not always show up in obvious ways.
Health Risks Are Real Enough
Catching a breath of fumes or getting a splash on your skin might not seem like a problem right away, but diethyl oxalate packs more punch than a strong odor. Speaking from time in labs and talking with others who’ve dealt with chemical safety, this stuff stings the eyes and burns the throat pretty sharply. Eye contact brings quick, nasty irritation. Skin absorbs it slowly, but repeated touch leaves red, itching spots and sometimes rashes.
Breathing in vapors or working in a spot without enough ventilation brings headaches, dizziness, and nausea. People who get bigger doses over time can even face lasting lung damage or kidney trouble. Not many folks realize that oxalates once inside the body can tie up with calcium and settle into tissues. This causes damage to the heart or kidneys in the worst cases. The risks of long-term exposure are never far from the minds of people who work with organic chemicals every day.
Sneaky Fire and Environmental Risks
Diethyl oxalate does not just wait around for direct contact. It lights up at relatively low temperatures, and the vapors form flammable clouds that need only a spark to blaze. Fire can spread quickly if storage and handling aren’t done right. The risk jumps up in crowded or poorly ventilated workspaces—where I’ve seen hurried cleaning jobs and spills get ignored too often.
Disposal is another headache. Dumping it carelessly ends up in groundwater or the local stream, where oxalates harm aquatic life and poison the ecosystem. I’ve seen the results in small towns where runoff twists life downstream and nobody links it to that forgotten old drum left leaking behind a shop.
Solutions Lie in Common Sense and Training
Factories and research labs need strong handling rules that go beyond basic labels. Real-world safety depends on steady habits—goggles, gloves, hoods, and spill kits right by your side. Too often, the problem isn’t ignorance but kids cutting corners or thinking it’ll never happen to them. Training that shows what the chemical does inside the body carries more weight than a wall of written protocol.
Good ventilation turns out to be the hero in most cases. I’ve cracked open a lab door to let out the sting of diethyl oxalate more often than I’d like to admit. Emergency showers and eye-wash stations need to work, not just exist on a checklist.
Responsible disposal sits at the core of environmental safety. Special waste handling services cost money, but they spare streams and soil. Nobody should pour leftovers down the drain. People deserve straight facts about the toxic side of chemistry—no scare tactics, just the reality that every chemical has a story, and good choices make all the difference.
What are the storage and handling requirements for Diethyl Oxalate?
Understanding the Real Hazards of Diethyl Oxalate
Diethyl oxalate isn’t just a quirky name on a chemical container—it carries real risks. Flammability comes right up front; this liquid catches fire at temperatures that aren’t far from a hot summer day. Vapors can linger, and, given the right nudge, start a fire. There’s also health trouble on the table, especially for folks who handle it without the right protection. Inhalation riles up the airways, splashes can sting the eyes, and long exposure takes a toll on skin. Anyone who’s worked in an older lab or chemical storeroom knows a casual approach invites trouble quickly.
Concrete Storage Requirements
Storing diethyl oxalate calls for basic but solid habits. Keep containers tight and upright, with the labels clean and clear. High shelves and flimsy stacking don’t cut it—this stuff belongs on lower racks away from walking paths to avoid spills. A cool, well-ventilated room with a steady temperature under 30°C keeps surprises in check. Fire hazards force separation from strong oxidizers, acids, and powerful alkalis. A lot of new folks treat all organic liquids as similar; that is a risky shortcut since diethyl oxalate plays badly with strong chemicals in a spill.
Metal containers rust over time, and that invites leaks. Polyethylene drums or glass bottles with solid seals last longer and give fewer headaches. No one likes handling heavy containers packed to the brim. It pays to keep stock small and rotate bottles, so older material goes out first. Enough incidents have started with people missing crusty caps or cracked seals after years on the shelf.
Safe Handling, from Gloves to Fume Hoods
Anyone transferring or measuring diethyl oxalate should put on splash goggles and decent chemical-resistant gloves. Open windows or a running fume hood pull vapors away. In stuffy rooms, vapor headaches come fast. Good labs fit work areas with spill kits and fire blankets. When a spill happens, sawdust or sand, not water, soaks it up—a careless dash of water can help vapors rise and spread out.
Transporting this liquid, even down a hallway, runs smoother with secondary containment—think of trays or carriers to catch drips. If a bottle tips and cracks in a storeroom, it only takes ten minutes before air quality tanks, and cleanup goes from simple to serious. One whiff reminds anyone that these lessons weren’t written just for procedure manuals.
A Focus on Training and Supervision
New staff sometimes figure labels are just for the look, but real training explains the why behind every rule. Safety data isn’t just a legal matter—a quick chat or refresher in a team meeting helps new and experienced folks keep habits sharp. Chemical exposure does real harm over years, not days, and regular reminders help everyone avoid shortcuts. Labs and warehouses work best with checklists for storage temperature, container rotation, and cleaning gear ready at arm’s reach.
Reducing Risks Means Better Outcomes for Everyone
Every chemical brings its own story and baggage. For diethyl oxalate, careful storage and thoughtful handling make a difference between quiet shifts and avoidable emergencies. Keeping up with good practices ensures the work benefits from this chemical without endangering the people around it.
What is the chemical formula of Diethyl Oxalate?
Diethyl Oxalate: Structure, Significance, and Uses
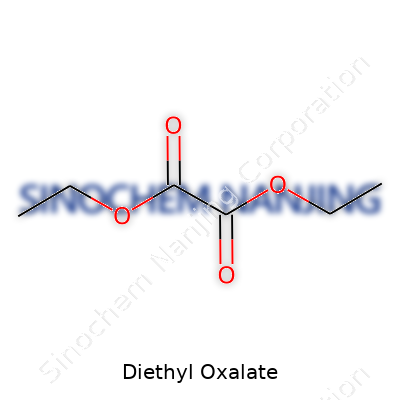
Diethyl oxalate steps up in many labs, chemistry classrooms, and even some manufacturing sectors. Its chemical formula is C6H10O4. This formula points to two ethyl groups bonded to an oxalate backbone. Chemically, oxalate itself consists of two linked carbonyl (C=O) groups, and the ethyl groups anchor to both sides through oxygen atoms. That’s the core idea: two carbon atoms, four oxygens, and two paired ethyl chains making up the C6H10O4 structure.
The importance of knowing this formula moves beyond just passing an organic chemistry quiz. Precision in chemical formulas guides lab work, quality control, and safe handling. Plenty of accidents and failed syntheses get traced back to simple mix-ups or wrong calculations. It’s easy to overlook the difference between a methyl and an ethyl group on paper, but in practice, those two extra carbons in C6H10O4 show up as a shift in boiling point, reactivity, and even toxicity.
Where Diethyl Oxalate Fits into Real-World Chemistry
Diethyl oxalate shows up as a staple intermediate in organic synthesis. Think about manufacturing dyes, fragrances, or even pharmaceuticals—the raw materials often pass through a stage involving this very compound. It acts as a building block for barbiturates, some anti-inflammatory drugs, and fine chemicals used in research. One practical example rests in the old-fashioned method of making ethyl oxamate, important for developing cancer research tools and agricultural chemicals. The trusted C6H10O4 helps build complex structures in controllable steps.
Outside the lab, there’s less talk about this compound. It doesn’t end up in consumer products much, but its backbone supports processes further up the supply chain. We rarely see all the steps involved in bringing a product to shelf, yet each chemical formula keeps that chain reliable and reproducible. Having the correct molecular structure lets workers track sourcing, manage environmental risks, and predict interactions—key moves in today’s drive for safer, greener chemistry.
Safety and Environmental Considerations
Handling diethyl oxalate calls for care. The C6H10O4 structure lends it both volatility and moderate toxicity. Mislabeling or simple forgetfulness can lead to spills or incorrect disposal, causing health hazards and polluting waterways. According to studies from the National Institute for Occupational Safety and Health, inhalation of its vapors can irritate the respiratory tract and eyes. The Environmental Protection Agency underscores the need to follow wastewater standards and containment protocols, as improper release could disrupt aquatic life or soil quality.
Personal experience in a synthetic lab drives home the impact of having chemical formulas front and center. Instructors would quiz us not only on writing formulas, but also on drawing out reaction steps. Any small slip led to wasted reagents, lost time, or scrambling for a fume hood during unexpected reactions. That level of discipline proves its worth outside school, showing up in industry audits, safety checks, and even regulatory filings.
Improving Chemical Literacy
Building a culture of clear communication around chemical formulas can lower risks. Training for both lab workers and students benefits from hands-on practice: writing, modeling in three dimensions, double-checking inventory and labels. Digital tracking tools in warehouses and educational platforms have brought error rates down. Ensuring up-to-date safety data sheets provides a safety net as well. Small investments in workflow, education, and organizational habits keep hazardous mix-ups at bay.
In a nutshell, knowing C6H10O4 means more than memorizing letters and numbers. It opens the door to effective research, successful manufacturing, and safer labs. For anyone working with or around chemicals, treating those details as non-negotiable supports health, environmental protection, and professional reputation.
How can Diethyl Oxalate be safely disposed of?
Diethyl Oxalate’s Not Just Another Chemical
Diethyl oxalate isn’t exactly a household name, but for anyone working in a laboratory, recycling facility, or even some paint factories, it pops up. It’s got a sweet smell, almost like ethyl acetate, but it doesn’t belong anywhere near a dump or sink. Pouring it down the drain or tossing it with trash can cause real harm. The stuff is flammable and releases toxic fumes, especially if it comes into contact with acid or fire. This isn’t just a regulatory headache—poor disposal can risk human health and pollute groundwater.
Why Standard Disposal Fails
I’ve spent long hours in university labs, and hazardous waste always creates panic when someone finds an old, half-labeled bottle. Stories float around about students quietly diluting chemicals and dumping them, thinking water treatment will “fix” it. That’s wishful thinking—local water plants don’t filter out chemicals like diethyl oxalate, and it doesn’t just break down harmlessly. It can reach rivers or, worse, drinking water.
The Basics of Safe Disposal
Safe disposal starts by keeping leftovers in a sealed, labeled container. Don’t mix the stuff with other solvents even if it all looks clear. One mistake and you might get a dangerous reaction. Collect diethyl oxalate waste in a cool spot, away from sunlight or heat. Chemical compatibility charts help here—it’s worth checking them before trying any disposal method.
Professional Hazardous Waste Disposal Services
Nobody wants to picture their water supply tainted with laboratory leftovers. That’s why every lab manager I’ve respected keeps the number of a licensed hazardous waste disposal company. Professional services transport the waste to a facility that breaks it down using chemical incineration or advanced treatment plants that neutralize it. These companies follow EPA and OSHA rules every step of the way, with paperwork tracking every drop from pickup to destruction. Choosing a certified company costs more up front, but it means fewer lawsuits, safer workers, and less risk for the environment.
On-Site Neutralization—A Dangerous Shortcut
Some handbooks hint at neutralizing diethyl oxalate on-site using simple chemistry, but the risk feels too high. Even with a fume hood and PPE, a small spill or mistimed step sends fumes rushing out. Having watched a friend trip the fire alarm with a minor spill during what should have been a safe reaction, I learned most quick fixes just tempt fate.
Building Better Habits
Safe disposal isn’t about ticking a box on a form or passing an audit. It means taking responsibility each time you open a bottle. Real training, clear labels, and emergency spill kits should be standard in any workspace handling stuff like diethyl oxalate. Emergency showers, eyewash stations, and clear exits matter, too. A single oversight can cause issues lasting years.
What We Can Do Next
Industry, research labs, and small businesses all face the chore of dealing with unwanted chemicals. It’s on managers and workers alike to report old stock, log every disposal, and set clear policies. Governments hand out guidance, but on the ground, daily choices matter just as much. Safer chemicals and reduced stockpiles can lessen the need for risky disposal over the long run, but until then, sticking with professional help and proper training is the best way to keep people and the environment safe.
| Names | |
| Preferred IUPAC name | Diethyl ethanedioate |
| Other names |
Oxalic acid diethyl ester Ethyloxalate Diethyloxalate Oxalic acid ethyl ester Stanioxane |
| Pronunciation | /daɪˈɛθ.ɪl ɒkˈsæ.leɪt/ |
| Identifiers | |
| CAS Number | 95-92-1 |
| 3D model (JSmol) | `3D model (JSmol) string for Diethyl Oxalate:` ``` CCOC(=O)C(=O)OCC ``` |
| Beilstein Reference | 1207052 |
| ChEBI | CHEBI:40558 |
| ChEMBL | CHEMBL188205 |
| ChemSpider | 5701 |
| DrugBank | DB14110 |
| ECHA InfoCard | ECHA InfoCard: 100.003.244 |
| EC Number | 203-743-0 |
| Gmelin Reference | Gmelin Reference: 80880 |
| KEGG | C00822 |
| MeSH | Diethyl Oxalate |
| PubChem CID | 8886 |
| RTECS number | KI8925000 |
| UNII | K6977V1P5X |
| UN number | 1162 |
| CompTox Dashboard (EPA) | EPA CompTox Dashboard (Diethyl Oxalate): **DTXSID2034153** |
| Properties | |
| Chemical formula | C6H10O4 |
| Molar mass | 146.14 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Fruity odor |
| Density | 1.078 g/mL at 25 °C |
| Solubility in water | 8.2 g/100 mL (20 °C) |
| log P | 0.97 |
| Vapor pressure | 0.39 mmHg (20 °C) |
| Acidity (pKa) | pKa = 13.09 (for the α-protons) |
| Basicity (pKb) | 13.13 |
| Magnetic susceptibility (χ) | -54.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.406 |
| Viscosity | 1.27 mPa·s (25 °C) |
| Dipole moment | 1.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 253.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -743.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1677.9 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H319 |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338 |
| NFPA 704 (fire diamond) | 2-2-1-W |
| Flash point | 63 °C (closed cup) |
| Autoignition temperature | ~320°C |
| Explosive limits | Explosive limits: 1.7–11.4% |
| Lethal dose or concentration | LD50 oral rat 2500 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 1870 mg/kg |
| NIOSH | WW5600000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Diethyl Oxalate: "5 mg/m³ (ACGIH TLV-TWA) |
| REL (Recommended) | 5 mg/m³ |
| IDLH (Immediate danger) | 400 ppm |
| Related compounds | |
| Related compounds |
Dimethyl oxalate Ethyl oxalate Oxalic acid Diethyl carbonate Diethyl malonate |

