Diethyl Methylmalonate: More Than Just a Synthesis Building Block
Historical Development: Foundations Rooted in Organic Chemistry
Chemists first looked at malonic acid derivatives like diethyl methylmalonate over a century ago. It makes sense. The thirst for creating increasingly complex molecules from simple origins sparked a lot of breakthroughs, especially in the twentieth century. Once researchers figured out the versatility of malonic esters in carbon-carbon bond formation, compounds such as diethyl methylmalonate stopped being just curiosities. They turned into workhorse reagents, serving not only in academic labs but showing up in major pharmaceutical and agrochemical syntheses. I remember thumbing through old organic chemistry texts and seeing these malonate esters popping up over and over, proof of their deep roots in chemical invention and industrial growth.
Product Overview: A Vital Player in Synthesis
The thing about diethyl methylmalonate is that it looks unassuming. In a bottle, it’s a nearly colorless oily liquid with a mild odor, not giving away its utility at all. Its main draw comes from its chemical backbone—central malonic structure flanked by two ethyl esters, with a methyl tag hanging off it. This subtle change unlocks access to an enormous range of transformations, letting chemists introduce complexity where needed. As a mid-stage player, it isn’t something you spot in a final drug tablet or a crop protection canister, but in the hands of someone working at the bench, it’s almost indispensable.
Physical & Chemical Properties: Practical Implications in Real-world Use
Measured in the lab, diethyl methylmalonate holds a boiling point that hovers a bit over 200°C. That means it can handle gentle heating, but doesn’t force chemists into burdensome cooling situations for most procedures. Its density falls close to that of water, so it doesn’t float or sink to extremes if it spills, which is always something to keep in mind. Solubility tends to favor organic solvents—think ether, alcohols, and chloroform. Water just doesn't mix with it well, which shapes choices during purification. And the compound tends to stay stable under normal storage if kept out of intense light and away from oxidizers.
Technical Specifications & Labeling: Not Just Another Bottle on the Shelf
Diethyl methylmalonate demands careful labeling. Those ethyl ester groups can react—especially if there’s acid or base around. Container materials and storage precautions matter, since stray water can hydrolyze the ester, turning a valuable reagent into a lab headache. Labels must note purity, batch number for traceability, and date received or repackaged, since age can affect performance in demanding syntheses. In professional settings, barcodes have replaced hand-written notes, but the bottom line stays the same: one badly labeled bottle can mess up months of research.
Preparation Method: Chemists’ Ingenuity on Display
Making diethyl methylmalonate usually follows a straightforward path, starting with the alkylation of diethyl malonate. A methyl group from a methyl halide, under basic conditions, attaches right where the action happens—the central carbon. From experience, timing and purity of reagents make all the difference between a clean reaction and a series of side products that complicate everything. Each batch becomes not just a chemical conversion but a lesson in practical lab skills: controlling temperature, using the right glassware, and being patient with purification steps. At large scale, the synthesis shifts a bit, accommodating for efficiency and waste management—concerns that only grow once production jumps from grams to kilos.
Chemical Reactions & Modifications: Opening Doors for Creative Synthesis
If there’s one thing that keeps diethyl methylmalonate relevant, it’s the way those ester groups and the reactive methane carbon are primed for substitution. Tackling this molecule, a chemist can create an enormous range of acids, ketones, and even new heterocycles. The classic malonic ester synthesis allows the building of substituted acetic acids through straightforward hydrolysis and decarboxylation. What drives home its usefulness for me is how even a modest lab setup can use this compound to make building blocks for drugs, dyes, and agrochemicals. Indirectly, diethyl methylmalonate threads through more products and processes than most people would imagine—from active pharmaceuticals like barbiturates to intermediates for vitamins.
Synonyms & Product Names: Navigating the Terminology Jungle
Anyone who’s ordered reagents knows the frustration of mismatched names. Diethyl methylmalonate sometimes turns up as Methylmalonic acid diethyl ester or Diethyl 2-methylmalonate. These aren’t just academic distinctions—they matter on procurement forms, customs declarations, and even regulatory paperwork. Getting them confused, particularly where shipping regulations or safety data are involved, means delays and extra costs. Over time, chemists get used to flipping between CAS numbers, IUPAC names, and plain English, but newcomers can easily fall into jargon traps.
Safety & Operational Standards: Staying Safe in the Lab
Working with diethyl methylmalonate is straightforward most days, but safety isn’t something to take lightly. Vapors can irritate, spills make slick surfaces, and those same reactive esters mean the compound burns if exposed to open flame or strong oxidizers. Labs handling larger quantities keep spill kits and robust ventilation for a reason. Gloves, goggles, and lab coats become routine—not just for compliance, but because nobody wants to risk long-term health just to save five minutes of cleanup. Safety data sheets—if actually read—can save careers and lives, making them as important as the molecule itself. As regulations tighten, protocols around storage, spill response, and waste disposal get reviewed every year, ensuring bad habits don’t creep in as staff changes.
Application Area: A Quiet Backbone Across Multiple Industries
Diethyl methylmalonate stands as a gateway to countless downstream products, so its applications ripple across industries. In pharmaceuticals, you find it in the toolkits for synthesizing anti-convulsants, barbiturates, and intermediates for APIs where precision in carbon chain length matters. For agrochemicals, it enables cost-effective construction of herbicide and pesticide precursors, especially when a methyl group’s position spells the difference between safety and toxicity. Even specialty chemicals—notably flavors, fragrances, and dyes—benefit from the versatility of this compound. Talking to colleagues in contract manufacturing, I’ve seen the surprise when an obscure intermediate traces its lineage back to this humble ester. Its role may not be glamorous, but the impact on global supply chains is real and far-reaching.
Research & Development: A Catalyst for New Chemistry
R&D keeps pushing the boundaries for diethyl methylmalonate. New coupling reactions, greener synthetic routes, and automated multi-step processes all benefit from its reliable reactivity and commercial availability. I’ve watched research teams streamline the synthesis of complex chiral centers using variants of malonate chemistry, shrinking steps from seven down to three or four. There’s also work ongoing to recycle waste streams and replace hazardous reagents, in an effort to reduce the environmental footprint. Academic publications—and even the patent literature—continually refresh interest in known compounds like this by uncovering new routes for old transformations. The drive for drug discovery and agricultural innovation creates a steady demand for such platform molecules.
Toxicity Research: Balancing Utility and Responsibility
Most work involving diethyl methylmalonate takes place in locations where safety and health guidelines carry legal weight. Short-term exposure tends to irritate skin and eyes, and accidental ingestion or inhalation poses greater risks. Animal studies over the decades show acute toxicity at high doses, but routine use under proper ventilation and meticulous handling keeps such incidents rare. Ongoing research explores chronic effects, bioaccumulation, and breakdown products. People working with this reagent must respect it, both for their health and for those handling waste downstream. Regulatory frameworks evolve alongside new scientific data, so anyone using diethyl methylmalonate stays alert for updates from toxicity studies and workplace hazard assessments.
Future Prospects: Diethyl Methylmalonate in a Changing World
As synthesis moves toward more sustainable and efficient pathways, diethyl methylmalonate is well-positioned to remain a core player. New regulations around waste create pressure for cleaner production and greener reagents, and this chemical adapts well to such shifts. Efforts to improve atom economy, reduce solvent volumes, and recover byproducts all draw on the experience gained through decades of working with malonate chemistry. As artificial intelligence and automation reach chemical synthesis, the role of standardized, versatile reagents becomes even more pronounced. In my view, as long as the hunger for pharmaceutical innovation and custom molecule creation persists, compounds like diethyl methylmalonate will quietly underpin the future of synthesis. Those seeking to improve sustainability, safety, and speed in the lab constantly revisit the classics, and this molecule stands ready for whatever comes next.
What is Diethyl Methylmalonate used for?
In the Lab: Not Just Another Chemical
Diethyl methylmalonate tends to show up in chemistry circles where people want to build something new. I see this compound regularly featured in discussions around creating fine chemicals, specifically for pharmaceuticals and agricultural products. It’s not a name you’d hear tossed around at a family barbecue, but its fingerprints are on the developments that keep grocery shelves stocked and medicine cabinets full.
The Secret Sauce for Drug Synthesis
Drug developers trust diethyl methylmalonate for creating the backbone of a variety of medicines. The molecule carries a special structure with two ester groups and a methyl group, making it perfect for adding complexity to organic frameworks. Chemists use it to construct barbiturates, antihypertensives, and anticonvulsants. Look up some of the classic drugs in the anticonvulsant class and you’ll find they were built from diethyl methylmalonate or one of its close relatives. It wasn’t all about big companies, either—university researchers tinkered with this compound in labs, learning how minor tweaks could lead to different therapeutic effects.
A Workhorse in Agrochemical Creation
All of us want farmers to grow enough food without spraying too many harsh chemicals. Protective agents for crops help with that goal, and these agents often come from chemical chains involving diethyl methylmalonate. Herbicides, fungicides, and certain insecticides trace their origins to reactions that start with this chemical. Chemists can use it to build new molecules capable of targeting pests without harming the plants or nearby animals—a win for sustainable farming.
Chemical Reactions that Demand Specificity
If a synthetic chemist wants precise control, diethyl methylmalonate delivers. It’s great for the malonic ester synthesis, one of the classic techniques for making substituted acetic acids. Simple reactions with strong bases like sodium ethoxide pull off a hydrogen, letting the molecule form new carbon-carbon bonds. This opens the door to customized chemicals and intermediates.
Risks and Safety—Real-World Concerns
Diethyl methylmalonate doesn’t belong in the hands of amateurs. I’ve talked to safety officers who stress goggles, gloves, and proper ventilation. Breathing in the vapors can irritate, and spills create headaches fast in unprepared labs. Responsible chemists lock away all reagents and manage waste according to strict guidelines. The process matters just as much as the product.
Moving Forward: Opportunities for Greener Synthesis
Sustainability has finally caught up to specialty chemicals. Some experts want chemists to find bio-based pathways to diethyl methylmalonate, or at least cut down on wasteful byproducts when making it. A key area of current research involves catalysts that let you build and modify this molecule without relying so much on harsh conditions or toxic solvents. With the right collaboration between academia and industry, every step toward safer synthesis means stronger trust in the products that come from this chemical.
What is the chemical formula of Diethyl Methylmalonate?
Why the Formula Matters
People who work with organic chemistry or applied sciences know a chemical’s formula tells more than just atoms lined up together. Diethyl methylmalonate pops up as both a tool and a building block, and recognizing what makes up its structure helps anyone—students, chemists, folks in manufacturing—make better sense of its role and risks. Its chemical formula, C8H14O4, describes a molecule that’s got eight carbon atoms, fourteen hydrogen atoms, and four oxygen atoms all working together.
Following the Carbon Skeleton
The core of this molecule traces back to malonic acid, a simple structure with potential. By swapping two of its hydrogens for ethyl groups and keeping one methyl group attached, the molecule gets unique properties. The structure looks a bit like this: CH3C(COOC2H5)2CO. You have a methyl (CH3-) on the α-carbon, along with two ethyl ester groups. That’s what the “diethyl” part is about: two ethyl groups sitting where once were acidic hydrogens in malonic acid. It’s not just a pretty arrangement on paper; these changes affect reactivity and how the compound behaves in living things and the lab.
Real-World Relevance
Labs depend on diethyl methylmalonate to build more complex molecules. This stuff plays a quiet but key role in making pharmaceuticals, perfumery ingredients, and agrochemicals. Take barbiturates—some of the old-school medicine for anxiety and seizures. Their synthesis starts off with molecules like this one. It’s not flashy, but without it, plenty of other creations would never see the light.
Its formula, C8H14O4, also signals certain handling needs. It’s flammable, and as an ester, it wants to break down in the presence of water over time. That means people storing or transporting diethyl methylmalonate watch for spills and keep containers sealed. Chemists lean on its specific reactivity, exploiting that methyl group when they need selective reactions. In short, learning this formula isn’t just a test question—it points toward safety, storage, and proper use worldwide.
Spotting Concerns and Smart Handling
Misunderstanding chemical formulas always leads to trouble. One small slip—thinking it’s a simple acid or confusing its structure—can mean botched batches or worse, dangerous reactions. I’ve seen new chemists forget that oxygen-rich esters like this release vapors that irritate eyes and skin. Safety data sheets for substances with C8H14O4 spell out what’s needed: splash goggles, gloves, real ventilation. Teachers who emphasize the meaning behind each atom help students understand why the formula looks the way it does and why it matters once bottles hit the benchtop.
Moving forward, as industries get more complex, companies and labs have to focus even more on training and transparent labeling. Some push for digital tracking—QR codes on bottles linking to up-to-date hazard data. Others suggest regular refresher courses so no one forgets what those eight carbons and four oxygens mean in practical terms.
Clear Communication Builds Trust
Being able to explain exactly what C8H14O4 stands for helps break down barriers between academics, industry, and the public. When researchers, workers, and suppliers understand the chemical, those new to the scene learn respect for the hazards and possibilities. Sharing clear information about diethyl methylmalonate doesn’t just help in the lab—it supports safety and smarter innovation everywhere this molecule travels.
How should Diethyl Methylmalonate be stored?
Understanding Why Proper Storage Matters
Chemicals like Diethyl Methylmalonate don’t ask for much, but respect for safe handling goes a long way. Many of us overlook small details until a leaky container or a strong odor turns up one day in the lab or storeroom. That’s a problem that’s easy to avoid if you take preparation seriously from the start. I’ve seen it save time, budget, and hassle—plus, it’s the only way to comply with rules that keep people out of harm’s way.
Getting Real with Hazards
Diethyl Methylmalonate packs a punch most folks don’t notice at first. The clear liquid may seem harmless, but shifting vapors or accidental skin splashes can ruin a good day. Flammable properties add another layer; anything from a spark nearby to a warm heat source raises the risk. Forgetting a cap or using the wrong container spells double trouble. These aren’t small concerns—I’ve seen entire shelves cleared in a panic because somebody cut corners.
Smart Storage Makes All the Difference
A chemical like this thrives on predictable care. Store it tightly closed in a container made to resist chemical attack. Glass works best in my experience, though some high-grade plastics fit the bill. Keep it in a cool, dry place far from sunlight or any source of heat. It can break down over time, especially if humidity and temperature swing all over the place. Shelves with ventilation and spill trays help keep drips from turning into disasters.
Don’t put it near acids or bases—cross contamination can spark dangerous reactions. In every lab I’ve worked at, we treated this liquid like it belonged in its own apartment, always grouped with compatible chemicals and kept at eye level to avoid reaching up and risking a spill. Most accidents I’ve witnessed happened because someone tried squeezing just one more bottle on a crowded shelf.
Label Everything and Know Your Inventory
Illegible labels cause real problems. I’ve seen folks waste an afternoon because someone wrote the name once in fading marker. Clear, printed labels showing chemical name, purchase date, and hazard warnings spare you confusion in the long run. Never mix old and newly received stock; always rotate so nothing hides out on a forgotten back row, slowly degrading.
Track quantities and check for evaporation or leaks every month. Mold or strong smells mean something’s wrong. Fast action cuts losses and keeps you on the right side of safety regulations.
Protecting People Comes First
Wear gloves, goggles, and a lab coat whenever you handle the bottle. I learned early that even careful folks slip up—one missed step, and chemical burns or breathing problems can follow. Working in a well-ventilated space, or better yet, a chemical fume hood, limits vapor buildup.
Emergency showers and eyewash stations belong nearby. At every proper facility I’ve visited, these tools made a difference when someone acted fast after an accident. Policies only work if people rehearse responses until they feel second nature.
Simple Steps Prevent Big Problems
It doesn’t take fancy technology or expensive storage. Consistency matters more. Good habits, appropriate containers, and steady checks let Diethyl Methylmalonate serve its purpose without turning into tomorrow’s safety headline. Every trained lab worker, teaching assistant, or hobby chemist learns soon enough—smart storage pays off, every single day.
Is Diethyl Methylmalonate hazardous or toxic?
Chemistry and Everyday Safety
Diethyl methylmalonate works quietly in the background of many chemical syntheses, especially those tied to pharmaceuticals and organic research. Not many people will find this compound outside a research lab, but its properties make understanding its risks important for those who handle it. This clear, oily liquid doesn’t look threatening. Despite its appearance, a closer look at its data sheet and practical handling advice shows this is far from just another chemical bottle in the cabinet.
What Science Tells Us
There's a simplicity to studying chemicals like diethyl methylmalonate: check how it behaves, how it interacts, and what it does inside a living system. The compound gives off fumes that can irritate the nose, throat, and eyes. Inhaling large amounts lets those fumes move into the lungs, causing coughing and breathing trouble. On the skin, direct contact starts with irritation—think redness, burning, or a stinging sensation that sometimes sticks around. Spills that go unchecked seep into the skin, potentially leading to systemic poisoning, though data in humans stays limited because most contact happens inside controlled environments.
What Toxicology Reports Warn
Animal studies fill the gaps left by the absence of large-scale human exposure. High doses have caused serious central nervous system depression in test rodents, bringing tremors and breathing complications. These effects came through injection or forced inhalation, which is more than what a lab worker would usually face. Still, patterns show up: repeated exposure can stress organs like the liver and kidneys. The compound’s physical properties mean it evaporates under normal lab conditions, putting unprotected workers at risk, especially if a fume hood fails or gets ignored out of habit.
Real World: Safety Shows Up in the Routine
In practice, anyone using diethyl methylmalonate ends up carrying out a balancing act. Whether accidentally splashing some on the bench or cleaning up a broken bottle, moments like these bring real hazards to the surface. Personal experience tells me accidents don’t announce themselves with a drumroll—they happen fast, and being casual with handling ramps up the danger. No one enjoys the burning feeling of a chemical on bare skin or coughing uncontrollably after a whiff of vapors. Such moments burn respect into a person for safety tricks that look simple on paper: keeping gloves and goggles on, using a good vent, sealing containers quickly, and always labeling clearly.
Finding Solutions in Training and Design
Risk drops when people learn from every incident, big or small. Basic lab safety makes a huge difference, but people tune out if they view these rules as mere formalities. Regular practice drills, sharp visual reminders, and sharing stories of what goes wrong keep awareness fresh. Institutionally, investing in better ventilation, clear signage, and accessible spill kits gives everyone tools to react quickly. Long-term, replacing hazardous reagents with safer versions, wherever practical, ends up improving safety and lowering risk for everyone involved.
The Importance of Responsible Handling
Diethyl methylmalonate isn’t something to fear, but it certainly isn’t something to ignore. My years in the lab taught one lesson above all: chemicals don’t care about intent, only about physics and chemistry. Facing the risk means respecting the facts, sharing experiences, and embracing good practices day in and day out.
What are the common applications of Diethyl Methylmalonate in organic synthesis?
What Makes Diethyl Methylmalonate Stand Out?
Diethyl methylmalonate pops up in so many labs you’d think it was a trusty screwdriver for organic chemists. Out of personal experience during a short stint in an academic organic lab, this compound stood as a frequent star for building bigger, more complex molecules. Its secret lies in its structure—two ethoxy groups hugging a methyl-substituted malonate backbone. The design opens doors to a world of custom chemical assembly.
Fueling the Synthesis of Pharmaceuticals
No shortage exists of demand for compounds that can be made with diethyl methylmalonate. The pharmaceutical industry leans on this molecule often. It provides a reliable way to stitch together new carbon-carbon bonds, paving pathways to a slew of drugs. As a student researcher, I modeled the synthesis of barbiturates, which gained attention as sedatives and anesthetics. Diethyl methylmalonate serves as a building block for these and other heterocycles, including pyrimidines, which play a big part in antiviral and anticancer agents.
Malonate esters also work as stepping stones for anti-inflammatory drugs, ACE inhibitors, and antiepileptics. Adding new groups to the active methylene position ends up surprisingly straightforward—a clean, efficient way to shape molecules into just what the body needs to respond to disease. Analytical studies show this pathway often leads to fewer byproducts compared to more complicated alternatives.
Paving New Avenues in Agrochemicals
Folks who develop crop-protecting products and pesticides have another reason to reach for diethyl methylmalonate. The world depends on a steady food supply. Herbicides and fungicides made from this backbone keep harvests healthy. Malonate chemistry allows for the development of selective agents that leave key crops unharmed. As a gardener myself, I appreciate the ripple effect. Without safer molecules in plant protection, growers and consumers would have fewer, less affordable choices.
Key Role in Flavor and Fragrance Chemistry
Few people think of malonate esters ending up in perfumes or flavoring. Yet, this compound pops up there too. Its structure lets chemists create a wide variety of esters—some with fruity or floral notes, others with herbal aromas. During a summer job at a flavor company, I watched the team blend synthetic ingredients powered by malonate chemistry to mimic rare fruits and flowers. The variety on grocery store shelves owes a lot to this family of molecules.
A Tool for Making Unusual Amino Acids and Natural Products
Research keeps finding new reasons to use diethyl methylmalonate. One strong example involves making non-standard amino acids. These are valuable for biochemists studying proteins and enzymes, or for companies developing specialty peptides with improved properties. Synthesizing these unusual components can be a headache, but the active methylene group in diethyl methylmalonate lets chemists add all sorts of side chains with remarkable precision.
How to Address Safety and Sustainability
Greater use brings concerns. My colleagues and I ran into waste management issues when scaling up malonate reactions. The industry has started to focus on greener solvents, inventive recycling, and fine-tuning procedures to cut down on chemical byproducts. Regulatory data confirm that improved containment and responsible disposal reduce environmental hazards. Training makes a difference—every new chemist should learn smart handling and disposal right away.
Moving Forward
Diethyl methylmalonate’s versatility makes it a top pick for a lot of synthetic routes. Pharmas, agriculture, and flavor houses lean on this single molecule. Responsible use, backed by transparent research and hands-on training, helps share these benefits without risking health or the planet.
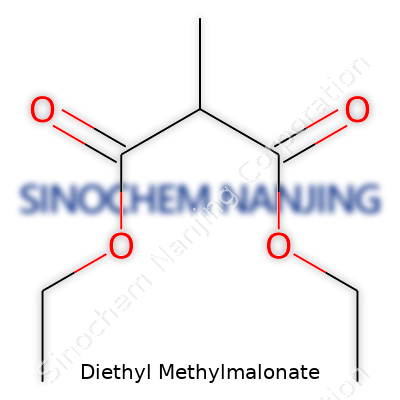
| Names | |
| Preferred IUPAC name | Diethyl 2-methylpropanedioate |
| Other names |
Diethyl methylpropanedioate Methylmalonic acid diethyl ester NSC 9706 |
| Pronunciation | /daɪˈɛθ.əl ˌmɛθ.ɪl.məˈloʊ.neɪt/ |
| Identifiers | |
| CAS Number | 607-81-8 |
| Beilstein Reference | 1208730 |
| ChEBI | CHEBI:37767 |
| ChEMBL | CHEMBL33847 |
| ChemSpider | 7595 |
| DrugBank | DB14047 |
| ECHA InfoCard | DTXSID4016992 |
| EC Number | 204-478-6 |
| Gmelin Reference | 137159 |
| KEGG | C06710 |
| MeSH | D02.241.223.211.112.173 |
| PubChem CID | 7841 |
| RTECS number | OA9625000 |
| UNII | Y5VD43009I |
| UN number | UN2529 |
| CompTox Dashboard (EPA) | DTXSID7023721 |
| Properties | |
| Chemical formula | C7H12O4 |
| Molar mass | 188.22 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Fruity |
| Density | 1.085 g/mL at 25 °C (lit.) |
| Solubility in water | Slightly soluble |
| log P | 0.93 |
| Vapor pressure | 0.05 mmHg (25°C) |
| Acidity (pKa) | 13.1 |
| Basicity (pKb) | 3.75 |
| Magnetic susceptibility (χ) | -43.5e-6 cm³/mol |
| Refractive index (nD) | nD 1.411 |
| Viscosity | 1.245 cP (20°C) |
| Dipole moment | 4.30 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 489.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -635.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1814 kJ mol-1 |
| Pharmacology | |
| ATC code | Diethyl Methylmalonate" does not have an ATC code. |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319 |
| Precautionary statements | P210, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-1-0 |
| Flash point | 83 °C |
| Autoignition temperature | 355 °C |
| Lethal dose or concentration | LD50 Oral Rat 3,373 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 2000 mg/kg |
| NIOSH | SN 1322000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 50 mg |
| Related compounds | |
| Related compounds |
Dimethyl malonate Diethyl malonate Diethyl ethylmalonate Diisopropyl methylmalonate Methylmalonic acid Ethyl acetoacetate |

