Diethyl Malonate: Past, Present, and Where We're Headed
The Road Traveled: How Diethyl Malonate Entered the Scene
Diethyl malonate doesn’t show up in daily conversation, but its history tells us a lot about the way chemistry shapes industries far beyond the lab. Back in the latter half of the nineteenth century, as organic chemists dug deeper into ester compounds and began experimenting with condensation reactions, diethyl malonate emerged as a pivotal building block. Largely due to the pioneering work of chemists like Auguste Cahours and Frankland, chemists found a dependable ally in synthesizing complex molecules. For decades, this little ester has been featured in textbooks, labs, and factories, each time proving its worth to those who put their hands in the work of chemical synthesis or medicine development.
What Diethyl Malonate Brings to the Table
What we actually get with diethyl malonate is a clear, colorless liquid with a faintly fruity smell—a trait you’ll notice if you’ve spent time around esters. Chemically speaking, it shows the formula C7H12O4. Pour a small sample out in the lab, and you’ll find it’s slightly heavier than water, with a boiling point that lets you distill it under gentle conditions. Whether you’re an organic chemist looking for a starting material or a manufacturer seeking feedstock for more valuable products, the substance lives up to its reputation: stable under everyday conditions, not too quick to react except under the right prompts. These physical characteristics aren’t just academic observations—they’re directly tied to how chemists handle, store, and process this compound.
What Goes Into the Label and Tech Specs
Reading a label on a drum of diethyl malonate, you won’t find fancy marketing. You get information you need for safe handling: purity, boiling range, melting point, density, and refractive index. The purity tends to land at 99 percent and above—a necessity when downstream reactions count on every molecule behaving as expected. You’ll see standard hazard markings since diethyl malonate is flammable and should never find its way near a flame or an unventilated workspace. Some regulatory bodies demand extra labeling, setting clear lines for workplace exposure and environmental compliance.
How People Make It
Getting to diethyl malonate isn’t a feat left to chance. Large-scale production typically starts with malonic acid, which gets esterified with ethanol in the presence of an acid catalyst. Anyone who remembers their organic chemistry lab will recall how water must be drawn out to shift the reaction forward—a detail that seems small, but in industry, it’s vital to efficient conversion. The process benefits from solid engineering: continuous removal systems, efficient mixing, and careful temperature control. Years ago, purification often took a bigger bite out of the product yield; today’s refinements in distillation tech have made the process more competitive for companies that need consistent, reliable supply.
How Diethyl Malonate Changes the Game: Chemical Behavior and Reactions
What makes diethyl malonate a favorite among chemists isn’t just how easy it is to buy or store, but what it can do once it hits the reaction flask. The methylene group sandwiched between two carbonyls allows clean, reproducible alkylation, condensation, and hydrolysis steps. In my own grad-school research, we reached for this molecule because it could be coaxed into giving up its hydrogens under basic conditions—opening the door to making all sorts of new carbon-carbon bonds. In industry, this translates into tools for building flavors, vitamins, pharmaceuticals, and agrochemicals. The compound’s reactivity profile lets people dial in precise modifications without a lot of fuss or side products, so work doesn’t stall out over unpredictable yields.
Names and Aliases in the World of Chemistry
Most folks in the lab call it diethyl malonate, but depending on where you buy it or where you read about it, descriptors shift: DEM, malonic acid diethyl ester, or sometimes just malonic ester. Older literature, especially from Europe, sometimes swaps in slight variations. Navigating synonyms can quickly eat up time for those new to synthetic chemistry, but for seasoned hands, it’s just another fact of life—names change, reactivity doesn’t.
Safety and Good Practice in Handling Diethyl Malonate
Anyone with time on the bench knows not all clear liquids are harmless. Diethyl malonate is classified as flammable and irritating to skin, eyes, and airways. That matters not only for chemists but also for workers in blending, bottling, and shipping environments. Regulations like those from OSHA and REACH set out storage and handling norms: keep it cool, vent vapors, wear gloves and goggles, and don’t let the material get close to ignition sources. Over the years, I’ve seen what a careless moment can mean: ruined batches, workplace exposures, sometimes costly investigations. Safety isn’t just about ticking boxes. It’s about respecting the material in front of you and giving those around you peace of mind.
Where Diethyl Malonate Actually Ends Up
One bottle of diethyl malonate can play a supporting role in more products than most people realize. The pharmaceutical sector leans heavily on it for barbiturates and certain vitamins, plus a range of intermediate compounds needed for bespoke drug molecules. Visit a flavor or fragrance house, and you’ll spot it in the background, used to stitch together more complex aroma compounds. Even the agrochemical world owes a debt to this little ester—herbicides, pesticides, fungicides—where precise synthetic steps matter for large-scale production. Research settings add another layer: Diethyl malonate is a launchpad for exploring new molecular scaffolds, especially for students learning the ropes of reaction design and molecular logic.
R&D: Tuning the Molecule, Pushing the Boundaries
Research in academic and industrial settings keeps diethyl malonate on the leading edge of synthetic innovation. More than once, I’ve watched as new derivative routes unlock previously inaccessible molecules, improving crop protection or sharpening the efficacy of medicines. As green chemistry grows, labs push for synthesis under milder conditions, or routes that draw on biomass-derived feedstocks. Researchers experiment with catalyst recycling and reduced solvent loading, not because regulations force a hand, but because these tweaks trim costs and environmental impact. Advances in continuous-flow systems and microreactors show real promise for safer, scalable transformations, especially when reactions need tight control.
Where Safety Meets Science: Looking Hard at Toxicity
Any chemical routinely used at scale deserves a close look at toxicology. For all its versatility, diethyl malonate still raises questions about long-term exposure, particularly for those spending years bottling or blending in industrial settings. Short-term exposure produces irritation but generally not much worse when handled with care. Some studies look towards chronic effects, but nothing so far points to severe long-term harm when workplaces follow safety rules. Researchers keep at it, updating their findings as better assays and deeper understanding of metabolic pathways emerge. This ongoing vigilance isn’t just habit—it’s a real-world necessity for those relying on diethyl malonate across their careers.
What Comes Next: The Future of Diethyl Malonate
Looking ahead, diethyl malonate seems set to claim a bigger role in the synthesis of next-generation drugs and advanced materials. The rise in green chemistry pushes producers to adopt cleaner methods, swapping old catalysts for greener alternatives, recovering solvents instead of dumping them, and sourcing feedstocks from less-polluting origins. Market demand for complex molecules—especially in pharmaceuticals and specialty chemicals—suggests that production volumes will likely tick upward. Researchers are already sketching pathways for higher selectivity, energy savings, and reactor designs that minimize waste. The world needs more tailored molecules, whether for new antibiotics or safer crop protection, and few intermediates have the track record or flexibility of diethyl malonate. Across all these developments, ongoing commitment to safe handling, clear labeling, and real transparency in reporting will determine just how widely and safely this compound serves the scientific community and the industries it supports.
What is Diethyl Malonate used for?
A Key Building Block in Chemistry
Diethyl malonate doesn’t attract much attention at first glance. It’s a clear, almost odorless liquid, usually handled by people in lab coats and factories. In practicality, its value shows up across many areas, from medicine to flavorings and even agricultural chemicals.
Helping to Make Medicine Safer and Cheaper
Pharmaceutical companies use diethyl malonate in the early stages of drug production. It’s a workhorse compound for chemists who need to build more complex molecules. Many common drugs—barbiturates, vitamins like B1, and even some heart medications—depend on diethyl malonate as part of their chemical “skeleton.” Production wouldn’t run smoothly without it. It’s not only about making new drugs but cutting costs. Diethyl malonate can lower the price tag on important medicines, which helps people get access to better healthcare.
Chemistry Classrooms and the Real World
Countless students have mixed diethyl malonate during organic chemistry labs in universities around the world. In those classrooms, the ideas being learned turn into jobs and real breakthroughs. It takes lots of hands-on work to develop new methods for making tough molecules, and diethyl malonate is often the starting point. For professors and industrial chemists alike, this simple liquid becomes part of a toolkit that drives the discoveries behind new drugs and other important products.
Flavor and Fragrance Industry Staple
It’s not only about medicine. Most snacks, perfumes, and even cleaning sprays would smell a lot duller without the subtle effects of diethyl malonate. It’s essential in creating fruity or floral notes for artificial flavors and fragrances. During synthesis, it helps make compounds like ethyl maltol—a substance used to give everything from chewing gum to baked goods that sweet, caramelized ‘just right’ taste. The perfume industry favors it too, especially when blending unique scents.
Protection for Crops
In the field of agriculture, diethyl malonate contributes to products that protect crops against pests and diseases. It’s found in the background chemistry behind many modern pesticides and herbicides. Better yields and healthier plants often depend on these products, especially as the climate grows unpredictable and pests get tougher. The savings in crop losses speak for themselves.
Is It Safe?
Every chemical brings questions about safety. Diethyl malonate draws scrutiny because it’s widely used. Proper protocols help keep things safe for workers and end-users. Hazard assessments by organizations like the European Chemicals Agency and the U.S. EPA report that, when handled according to strict guidelines, diethyl malonate brings low risk to people and the environment. That said, ongoing reviews remain important. Everything from correct storage to effective cleanup measures matters, especially as global supply chains move bigger volumes of this compound.
Looking Ahead: Is There a Better Way?
Sustainability pops up often in conversations today. Producing diethyl malonate at scale still relies on oil-based starting materials. For people concerned about the future, more research into greener, bio-based routes could bring both economic benefits and reduced environmental impact. Investing in better recycling and safer processes would do good, not just for those working with it directly, but for everyone who ends up depending on the wider chemical industry.
What Matters
Diethyl malonate shows up quietly each day, woven into the background of everyday life. I’ve worked with it in labs—gloves on, goggles tightened. It’s not the flashiest molecule, but its influence reaches deep. Industries lean on it to keep shelves stocked, crops protected, and research moving forward. Progress usually comes from behind the scenes, in places few people notice.
What is the molecular formula and structure of Diethyl Malonate?
Breaking Down Diethyl Malonate
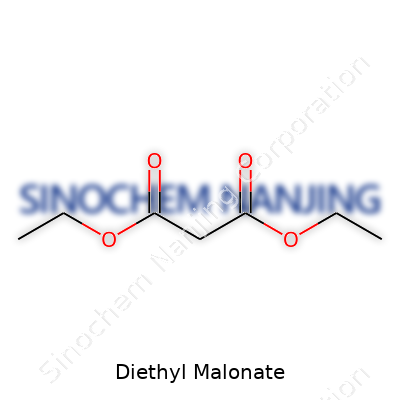
Chemists have a soft spot for diethyl malonate. On paper, it’s known by its molecular formula: C7H12O4. The shape and layout of this compound turn out to be pretty practical. At its core, diethyl malonate is an ester, carrying two ethyl groups on each side of a malonic acid backbone. Its structure looks like this:
CH2(COOC2H5)2
That means two ethyl ester arms (COOC2H5) branch off a central methylene group (CH2). This design lets the molecule take part in all sorts of useful reactions, especially for folks working in organic chemistry or the pharmaceutical field.
The Real-World Role of Diethyl Malonate
Anyone who’s spent time in a chemistry lab — whether as a student or professional — will recognize how often diethyl malonate comes up on reagent shelves. Back in college, I remember the satisfaction of watching its clear, faintly fruity-smelling liquid drip into a flask before a key reaction. It’s been a starting material for countless syntheses, from making vitamins to turning out barbiturates.
One reason chemists keep going back to diethyl malonate: its methylene group sits between two carbonyls, so the hydrogen atoms in this spot get pulled just enough to become pretty reactive. This 'active methylene' can be swapped easily with other groups, which makes diethyl malonate flexible and easy to use for building bigger, more complex molecules. Classic reactions like malonic ester synthesis build carboxylic acids, which slide into the manufacturing of pharmaceuticals or specialty flavors.
My own experience in research drove home how the accessibility of diethyl malonate smooths out big synthetic plans. In a world where every step adds cost and risk, a reliable building block truly shaves off hassle for companies and researchers.
Why Purity and Sourcing Matter
Safety, reliability, and purity all sit front and center in the world of chemicals. Not every supplier treats these with respect. I’ve seen what happens when batches arrive with the wrong color, smell, or water content: ruined reactions, lost time, and wasted money. The stakes rise sharply when drug companies or food manufacturers use diethyl malonate. Residual solvents or impurities can sneak into finished products, raising health risks or regulatory headaches.
Industry looks for reputable manufacturers with strong track records. Companies publishing up-to-date certificates of analysis help establish trust. Batch consistency, traceability, and honest transparency with regulatory data tend to set apart the best suppliers. From my time speaking to purchasing managers and compliance officers, this is where conversations begin.
Carrying Responsibility in the Lab and Beyond
Diethyl malonate doesn’t attract much attention outside chemistry circles, but it shapes products that people count on daily. As research keeps moving forward and regulations become more robust, companies handling it have to stick to science-based practices and regular third-party testing. Simple steps, like airtight storage and good handling procedures, keep hazardous exposures to a minimum.
Researchers, teachers, and supply chain managers all have to take their roles seriously. Every clear bottle might look like another chemical, but inside, there’s a mix of opportunity and responsibility. That’s just a fact of working with compounds like diethyl malonate—molecular formula, practical structure, and all.
How should Diethyl Malonate be stored and handled safely?
Diethyl Malonate: Not Your Everyday Compound
I've spent years around chemical storage rooms, and I can say this—some bottles on those shelves raise more concern than others. Diethyl malonate is one of them. Used in flavors, pharmaceuticals, and chemical synthesis, it moves quietly in both big and small labs. Don't let its popularity trick you into taking risks with how it's stored or handled.
What Makes Diethyl Malonate Tricky?
Diethyl malonate smells fruity, sometimes misleading newcomers to underestimate what it can do. The liquid catches fire at temperatures many would call mild. It can also irritate the skin and eyes if you brush against it, and breathing its vapor over time shouldn't be taken lightly. Too much exposure has led to nasty headaches or worse for my coworkers. Working with this compound brings real risks.
Practical Storage Advice
On hot summer days, I've seen storage rooms reach higher temperatures that raise alarms. A bottle of diethyl malonate sitting in warmth could spell trouble, so a cool, dry cabinet always wins out. Away from heat sources and direct sunlight is key. Fireproof cabinets rated for flammables suit the purpose best—anyone using cardboard boxes or old shelves is learning the hard way.
You won't find diethyl malonate sitting out on an open bench in a well-run operation. Every container deserves tight seals. Even a slow leak lets fumes out, and those fumes drift through the room faster than expected. In one busy research lab, someone left a bottle loosely capped and it was shut down for an hour the next morning to clear the air. Don't stack bottles high or store them above eye level since bumps lead to broken glass and spills. With flexible shelving at chest height, the risks drop right away.
Safe Handling: Eyes Open, Gloves On
I always reach for gloves made to block solvents before uncapping diethyl malonate. Splash goggles get a lot of use too, because regular glasses don’t block the side spray from accidental sloshing. It takes more than one person in a lab to set a high standard. If even one person shrugs off protection, others tend to follow—next comes an accident.
Pouring diethyl malonate under a fume hood matters, especially in tightly packed spaces. These hoods aren't just fancy furniture; they grab the harmful vapors before you breathe them in. I learned from experience the value of good ventilation after working late one afternoon in a stuffy, windowless room. A slightly sweet odor meant excess vapor had escaped, and that led to headaches for everyone the next day.
Spill Response and Planning Ahead
Spills don't happen every week, but they do happen. Absorbent pads or sand kept nearby beat running around hunting for supplies. Never use sawdust or paper because these ignite easily when soaked with diethyl malonate. Used cleanup materials call for metal waste bins with tight lids—not the usual trash can.
Labeling every bottle matters. Scratched, faded, or missing labels confuse even those who know the lab by heart. Refreshed training every few months keeps everyone sharp and gives newer folks the confidence to speak up if something feels wrong.
Building a Safer Routine
Complacency becomes the biggest hazard. Diethyl malonate needs respect from everyone who encounters it. Start by treating storage and handling like important steps, not afterthoughts. Elevate your protocols and back them up with quality gear, regular walkthroughs, and honest conversations about safety gaps. Better routines catch small errors before they turn big.
Is Diethyl Malonate hazardous or toxic?
Understanding What We're Dealing With
Diethyl malonate shows up in labs, classrooms, and factories. People use it to make pharmaceuticals, fragrances, and specialty chemicals. The clear liquid looks harmless, and the faint fruity smell can even seem inviting. Looks can fool anyone. A bottle with a mild scent may not scream danger, but chemicals call for respect and caution, no matter how friendly they seem.
Diving Into the Risks
To talk about hazards, the first thing worth knowing is what actually happens if someone mishandles this compound. Inhaling large amounts of its vapor can cause headaches, dizziness, or nausea. Letting the liquid touch your skin won’t burn like strong acids, but it irritates. Splashing it in your eyes could set off a nasty sting, sometimes even a chemical burn if you don’t rinse quickly.
Taking it a step further, swallowing diethyl malonate wouldn’t play out quietly, either. Stomach pain, vomiting, and trouble breathing can kick in. No one’s cleaning their hands with it, at least not if they want to avoid a trip to the hospital.
Regulation and Classification
The Globally Harmonized System (GHS) calls diethyl malonate a category 4 acute oral toxicant, which puts it squarely in the ‘harmful’ bracket—not the worst, but not safe to shrug off. The European Chemicals Agency (ECHA) also points out its irritating effects on the skin and eyes. The U.S. Environmental Protection Agency (EPA) does not give it a high-priority hazardous ranking, but basic chemical handling standards matter.
This isn't a “silent killer” like some nerve agents or radioactive materials. It also won’t hang around in the body for decades or sneak its way up the food chain. Still, no worker wants to find out firsthand if guidelines for gloves, goggles, or fume hoods hold up under bad luck.
Respect and Preparation Fix a Lot
A lot of chemical work comes down to training and preparation. I remember my first chemistry class in college: A professor told us, “Trust your safety gear more than your logic.” Even if you think you understand a chemical, it can surprise you. Goggles, gloves, and a fume hood kept me and my classmates safe more than once. It’s tempting to skip goggles for a “quick transfer,” but those shortcuts lead straight to accidents people regret.
Having the right safety sheets at hand—Material Safety Data Sheets (MSDS) in particular—makes a real difference. They show first aid steps, storage tips, and disposal guidance. Knowing that water won’t always fix a chemical splash or spill can save someone long-term trouble.
The Big Picture
Some folks worry about long-term poisoning or environmental damage. Diethyl malonate breaks down fairly well in the environment. Labs and factories use closed systems, safe collection, and proper disposal, which limit leaks or pollution.
Education deserves the most attention. Science classes and workplaces benefit from honest risk discussions. “Not the most toxic” doesn’t mean “harmless.” If people know how exposure happens—breathing, swallowing, spills—real-world accidents go down. Teaching young students early helps future chemists and workers carry safety habits for life.
Diethyl malonate serves its purpose in industry and research, but taking it lightly invites problems. Respect, not fear, keeps everyone unharmed.
Can Diethyl Malonate be used in pharmaceutical or agrochemical synthesis?
Real Workhorse for Researchers
Walk through any fine chemicals lab, and one flask almost always keeps a spot on the shelf: diethyl malonate. This clear liquid, with a pleasant fruity smell, plays a basic but far-reaching role across pharmaceutical and agrochemical research. Chemistry teachers and synthetic chemists both know it as a reliable starting point. I remember in my organic chem class, diethyl malonate opened the door to experiments on chain extension and ring-building reactions—territory that still directs drug and pesticide discovery today.
Versatility Builds Blockbuster Molecules
There's a reason this compound remains a staple after more than a century of use. Diethyl malonate, with its pair of reactive ester groups and acidic methylene, allows scientists to build all sorts of carbon skeletons. You create new bonds in just a few steps, often leading to alpha, beta-unsaturated acids, barbiturates, or plant-protection molecules. Simply put, this one molecule unlocks a toolkit for assembling everything from antiviral drugs to synergist insecticides. Look at barbiturates. The malonic ester synthesis, with diethyl malonate at the heart, has enabled a whole branch of sedatives and anticonvulsants for decades.
Safe Handling, Responsible Use
Everything has drawbacks, and here, it's the need for careful handling and environmental awareness. Diethyl malonate doesn't rank as highly dangerous, but it can still irritate skin or eyes, and its solvents may raise flammability risks. Every scaling operation, from pharma pilot plant to tank-filling agrochemical setups, must respect these hazards and invest in smart ventilation, spill plans, and personal protection. My time on chemical plants taught me never to underestimate a spill—solvents seep everywhere, and the cleanup costs both time and trust.
Supply Pressures and Sustainability
Groups relying on diethyl malonate face supply disruptions and price swings tied to upstream ethyl acetate or ethanol markets. Global events, logistical delays, or new regulatory scrutiny often hit smaller buyers hardest. Labs faced tough choices a few years back when prices doubled and shipping stalled—many pushed more for domestic stocks and kept closer ties with trusted suppliers. As the world leans toward green chemistry, more folks consider bio-based synthesis routes for these simple esters, seeking to cut fossil fuel dependency and shrink carbon footprints.
Innovation and Regulation Set the Pace
Every few years, regulations on chemical residues, especially in food, tighten the rules for agrochemical actives and intermediates. Diethyl malonate-based pesticides, for example, must clear new hurdles around toxicity and breakdown rates in soil and water. On the other hand, creative researchers continue to publish new farming fungicides and herbi-safeners where this modest ester provides a launchpad. The push for new anti-infectives in human and animal health draws from the same well of chemistry.
Moving Forward with Smart Solutions
Education and training carry the next generation of makers forward—chemistry labs boost hands-on learning so future researchers deeply understand these workhorse molecules and their downstream impacts. Smarter process tech, such as flow chemistry setups, cut waste and improve material tracking. Steady collaboration with regulators keeps products on the market, and renewable sourcing can relieve pressure on oil-based feedstocks. Standing at the edge of synthesis and application, diethyl malonate proves that old compounds still shape the future of health and harvest.
| Names | |
| Preferred IUPAC name | Diethyl propanedioate |
| Other names |
DEM Diethyl propanedioate Malonic acid diethyl ester Propane-1,3-dioic acid diethyl ester |
| Pronunciation | /daɪˈɛθ.əl məˈləʊ.neɪt/ |
| Identifiers | |
| CAS Number | 105-53-3 |
| Beilstein Reference | Beilstein Reference: **1209226** |
| ChEBI | CHEBI:17617 |
| ChEMBL | CHEMBL14001 |
| ChemSpider | 5261 |
| DrugBank | DB14166 |
| ECHA InfoCard | 100.003.745 |
| EC Number | 203-305-9 |
| Gmelin Reference | Gmelin Reference: **80067** |
| KEGG | C00956 |
| MeSH | D02.241.223.211.590 |
| PubChem CID | 3121 |
| RTECS number | OW6650000 |
| UNII | YOW8V9698H |
| UN number | 1169 |
| Properties | |
| Chemical formula | C7H12O4 |
| Molar mass | 160.17 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Fruity |
| Density | 1.057 g/mL at 25 °C |
| Solubility in water | Soluble |
| log P | 0.52 |
| Vapor pressure | 0.2 mmHg (20°C) |
| Acidity (pKa) | 13.3 |
| Basicity (pKb) | pKb = 13.3 |
| Magnetic susceptibility (χ) | -7.41 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.415 |
| Viscosity | 1.48 cP (20°C) |
| Dipole moment | 2.77 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 248.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -604.5 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2059.9 kJ/mol |
| Pharmacology | |
| ATC code | D01AE12 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P243, P280, P301+P312, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | NFPA 704: 2-2-0 |
| Flash point | 87 °C |
| Autoignition temperature | 441 °C |
| Explosive limits | Explosive limits: 1.5–8.2% |
| Lethal dose or concentration | LD50 (oral, rat): 2820 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 2820 mg/kg |
| NIOSH | GM2100000 |
| REL (Recommended) | 1000 mg/L |
| IDLH (Immediate danger) | 150 ppm |
| Related compounds | |
| Related compounds |
Dimethyl malonate Diethyl methylmalonate Ethyl acetoacetate Malonic acid Diisopropyl malonate |

