Diethanolamine: More Than a Chemical Name
A Look Back: Diethanolamine Through History
Diethanolamine has a name that sounds more at home in a laboratory notebook than in a daily conversation, yet this compound has threaded its way into many areas of everyday life. Chemists started to pay closer attention to it early in the twentieth century, recognizing its usefulness beyond academic experiments. Its roots link back to a larger push for chemicals that solved practical problems—cleaning, conditioning, even helping to build up and tear down other molecules. Once industries realized just how versatile this amine could be, it didn't stay on the shelf for long. Factories producing detergents, textile treatments, and lubricants picked up the formula, pulling it out of textbooks and into mass production. This jump from academic curiosity to real-world workhorse spoke volumes about how innovation takes shape—one day a formula bears only a technical label, the next, it drives major pieces of industry.
Getting to Know Diethanolamine: What Sets It Apart
Anyone who works with chemicals recognizes the value of a product overview grounded in honest use, not just theory. Diethanolamine falls under the category of alkanolamines, with a foot in the world of alcohols and the world of amines. In plain language, that means it shows both a knack for bonding with water and a tendency to latch onto both acidic and basic molecules. That opens the door for surfactant properties, helping oily grime mix with water so it can wash away. Its ability to serve as a chemical middleman, acting as a buffer or stabilizer, brings value across fields from personal care to gas treatment facilities. With industries shifting toward sustainability, users look for what’s reliable and familiar, and diethanolamine still earns its place.
Physical and Chemical Characteristics: More Than Meets the Eye
Take a close look at pure diethanolamine and you'll see a colorless, sometimes faintly yellow, viscous liquid or solid, depending on the surrounding temperature. Its mild ammonia-like odor is unmistakable to anyone who’s worked on a plant floor or in a university lab. The compound mixes freely with water and alcohols, but not with hydrocarbons. Chemists appreciate its dual-function as a weak base and weak alcohol, a chemical "jack-of-all-trades," which allows it to act as both a reactant and a stabilizer in different contexts. Thermal stability stands out, making it popular in processes that see seasonal swings or demand steady supply lines. It also doesn't break down easily from sunlight or air, so leakage or spills stick around unless handled. This reality pushes operators to build their storage and disposal routines with longevity in mind.
The Technical Details: Labels and Standards Matter
In the real world, technical specifications and labeling shape how workers use any chemical, diethanolamine included. You won’t see this product leaving a certified facility without proper hazard warnings—corrosive to eyes and skin, not to mention the risks in mist or vapor form. Labeling on containers provides more than legal compliance; it carries the cumulative knowledge of generations who learned the hard way not to take shortcuts with amines. Every jug, drum, or bulk tank heading down the highway to a plant reflects a track record built from careful handling, well-written safety data, and clear hazard communication. Those standards keep communities safe and maintain the trust between producers and buyers. In an era of supply chain scrutiny, technical rigor can't be set aside.
Making Diethanolamine: Process Shapes Performance
Industrial methods to prepare diethanolamine draw from a chemical reaction between ethylene oxide and ammonia. By adjusting the temperature, pressure, and ratio of reactants, operators shift the product balance between monoethanolamine, diethanolamine, and triethanolamine. Mastery here doesn't come from textbooks alone—it develops on the plant floor, as crews learn how to avoid over-alkylation, manage exothermic spikes, and keep equipment free from fouling. Contemporary facilities use closed, continuous systems to minimize emissions and protect worker health. Equipment undergoes regular inspection not only for regulatory reasons but because these amine processes can bite back if left unchecked. Even as greener synthesis and bio-based feedstocks grab headlines, most commercial operations still fall back on this time-tested route, balancing efficiency and market demand.
How It Reacts: Transforming and Tweaking the Molecule
Diethanolamine’s chemical structure provides reactive sites for both physical use and further modification. Manufacturers tailor its basic core by reacting it with fatty acids to form amides—key in the production of foaming agents and emulsifiers used in shampoos and dish soaps. Reacting it with carbon dioxide creates carbamates, known for capturing greenhouse gases in industrial scrubbing processes. Alkylation and acylation open additional pathways, while oxidation can split it into other amines or, in some cases, break down into potentially problematic byproducts. The way diethanolamine fits into these reactions speaks both to its flexibility and the need for skilled handling. Left unchecked, some reactions give rise to compounds linked to safety concerns, urging careful monitoring and continuous research.
Names You Might See: Synonyms in Use
Those using diethanolamine on the job or in academic research come across various synonyms: DEA, 2,2'-Iminodiethanol, and sometimes N,N-bis(2-hydroxyethyl)amine. In global trade and regulatory filings, these names crop up in documentation, adding complexity for anyone tracking inventory or researching toxicology data. Confusion over product naming doesn’t just slow shipments; it can muddle workplace training and safety drills as well. Workers need a clear connection between what’s on the label and what’s on the safety sheet, underlining the continuing need for shared language and open communication across borders.
Standards for Safety: Training Still Saves Lives
Regulatory agencies enforce rules for handling diethanolamine, calling for personal protective equipment, engineering controls, and strong ventilation in any facility where it’s stored or used. Over the years, tragic cases of eye and skin burns, or respiratory damage from inhaled mists, have pushed companies to invest in better shower stations, improved mixing tools, and routine health monitoring for workers. Industry protocols provide more than just hoop-jumping—they protect workers, the public, and company reputations. Regular drills and clear documentation reinforce what’s at stake when working with corrosives. Beyond the plant, downstream users—whether in a machine shop or on a cleaning crew—depend on upstream competence to ensure that what they handle day in and day out won’t leave them unsafe or facing unexpected exposure.
Where It Shows Up: Application in Modern Life
Applications for diethanolamine touch a surprising number of sectors. Personal care products, especially shampoos, conditioners, and bath gels, draw on its foaming and emulsifying properties—though public concern over ingredient safety has pushed both regulators and brands to scrutinize its use. Beyond the bathroom shelf, oil refineries rely on diethanolamine for gas purification, stripping away acidic contaminants from natural gas streams to prevent corrosion and safeguard downstream systems. Metalworking fluids, polishes, textile additives, and agricultural products fill out the list. Its uses persist because it bridges performance and cost, but tighter regulations and consumer trust demand more transparency in ingredient sourcing and disclosure.
Push for New Knowledge: Research and Development
Academic and industrial labs both dig into diethanolamine's chemistry, searching for safer formulations or eco-friendly substitutes. Contemporary research explores greener synthesis processes—possibly swapping out petrochemical feedstocks with renewable sources—and targets byproducts that might harm human health or the environment. As analytical technology improves, labs probe for ever-lower traces of nitrosamines, a group of toxic compounds that can form during certain chemical reactions with amines. Teams work not just to invent new uses, but to close gaps in understanding around risk. These efforts do more than line research journals; they regularly alter what makes its way into production or onto ingredient labels. The emphasis on science-based decisions pushes the field forward, ensuring production and regulation walk hand-in-hand with evidence.
Understanding Harm: Toxicity Research and Human Health
Toxicologists have been dissecting diethanolamine’s impact on humans and the environment for decades. Evidence shows that frequent, unprotected contact leads to skin and eye irritation—something shop workers and cleaners can't afford to ignore. Studies in rodents link long-term, high-dose exposure to tumors and developmental problems, stirring up concern about chronic workplace exposure and environmental releases. Regulators in Europe and North America frequently reassess standards, updating exposure limits based on the latest science. The push to understand nitrosamine formation—especially in personal care use—puts a spotlight on batch testing and trace contaminant analysis. It's one thing to promote productivity in manufacturing; it’s another to protect future generations from the ripple effect of short-sighted chemical management.
What Comes Next: Future Paths for Diethanolamine
The path forward for diethanolamine will be shaped by a balance of utility, health, and sustainability. Brands that once added it freely to cosmetics and cleaners now face calls for transparency, substitution, and full ingredient disclosure. Green chemistry movements push for less hazardous alternatives—but large-scale change still takes time and practical evidence. Shifts in policy could limit or ban diethanolamine in certain uses, especially where consumer exposure is possible. Research continues into alternative feedstocks, closed-loop production, and better end-of-life disposal, reflecting broader goals of reducing waste and minimizing environmental harm. As our scientific understanding evolves and regulatory frameworks adapt, the future of diethanolamine—like so many chemicals born in that early tide of industrial expansion—hinges on society’s commitment to marrying technological progress with genuine stewardship of human and environmental wellbeing.
What is diethanolamine used for?
From Industry to Home
Most folks haven’t heard of diethanolamine, but it shows up in some pretty common places. This chemical, a colorless, syrupy liquid, works as a jack-of-all-trades for manufacturers. It helps make shampoos nice and foamy, keeps paint smooth, and even finds its way into metal cleaning fluids. Industry loves it because it’s flexible and cheap. Soap companies and cleaning brands often blend it into their formulas so suds stick around and grease doesn’t stand a chance. Growing up, I remember reading shampoo labels and stumbling across this long name before moving on, never realizing how common it was.
Hidden in Personal Care Products
Diethanolamine plays a big role in personal care. It helps cut through oil, letting shampoos and body washes rinse dirt away easily. Toothpaste makers use it to keep their pastes creamy without separating in the tube. Even shaving creams lather up quicker with the help of this chemical. For decades, big brands in the cosmetics industry banked on its smoothing power and low price to win loyal customers. That’s why diethanolamine ended up on shelves around the world.
Health Concerns Stir Debate
Lately, questions popped up about whether frequent contact with diethanolamine is safe. Researchers have linked high doses in animal studies with liver and kidney changes, plus concerns about blending it with other ingredients that create carcinogenic byproducts. The European Union set strict limits on how much could go into products after these studies. The American Cancer Society and U.S. Environmental Protection Agency both flagged it for further review, though everyday exposure in consumer products seems much lower than risky test doses.
As a parent, reading these reports made me think twice before grabbing just any shampoo for my kids. I started reading labels more closely and looking for products that didn’t use it at all, though that search isn’t always simple. Some brands switched ingredients under pressure from watchdog groups, but others stuck to old formulas since official bans in the U.S. move slowly. My own experience is that customers have to do the legwork if they want to avoid chemicals like this—in the U.S., no rule forces companies to drop it completely.
The Struggle for Safer Alternatives
Alternatives do exist. Coconut-based cleansers and vegetable oils can replace diethanolamine in most soaps and shampoos. Some companies started using these safer ingredients because customers demanded it, but switching recipes takes time and investment. Larger chemical manufacturers argue that they’ve run their own safety tests and think the risks get overblown by headlines. As a consumer, it’s easy to feel stuck between catchy promises on bottles and scary stories in the news. Many shoppers, myself included, trust brands with strong track records and honest labeling. Retailers could push companies even faster by giving shelf space only to products that cut out questionable chemicals.
Moving Forward
Experience taught me that reading labels, keeping up with research, and supporting companies making real changes usually leads to the safest choices. Diethanolamine will probably stick around for a while since it still saves companies money and effort. At the same time, more folks are waking up to its risks and looking for softer, cleaner options. Consumer pressure, ongoing research, and honest conversations between brands and buyers have started to shift the market in the right direction—one bottle at a time.
Is diethanolamine hazardous to health?
What is Diethanolamine?
Diethanolamine, or DEA, shows up in a lot of everyday products. I've found it in shampoos, bubble baths, cleaning sprays, cutting fluids, and even some cosmetics. Companies like DEA because it helps ingredients blend and it produces foam.
Toxicity Concerns
I'm always suspicious of tongue-twister chemicals. With DEA, those suspicions have roots in real risks. Reports from respected sources—like the U.S. National Toxicology Program—have linked DEA to problems with skin and eye irritation. People who work in manufacturing often meet this chemical through direct contact, leading to red, itchy hands or sore eyes after a long day.
The bigger worry starts with long-term exposure. Studies on rats suggest DEA might interfere with choline absorption, which can affect brain health, especially during development. Some researchers saw possible links to liver tumors, raising eyebrows. These results pushed scientists and health agencies in the U.S. and Europe to keep a closer watch on products using DEA.
What the Regulators Say
During my research for personal care safety, I came across the International Agency for Research on Cancer (IARC) classifying DEA as “possibly carcinogenic to humans.” That means heavy users or production workers face higher-than-average risks. That doesn’t mean a single use of a shampoo containing DEA endangers health—risk increases with regular, repeated exposure.
The EU’s Scientific Committee on Consumer Safety suggests avoiding DEA and its compounds in cosmetics that remain on the skin. Health Canada goes a step further and restricts DEA in all personal care products. U.S. regulators recommend limiting concentrations in consumer goods.
Consumer Exposure and Bioaccumulation
Farmers and factory workers often face much greater exposure than someone washing their hair or scrubbing the kitchen. Home users usually don’t reach the threshold for acute toxicity, especially since rinse-off products—think shampoo—don’t sit long on the skin.
Still, most people aren’t told that DEA can build up a bit over time. If you use several personal care products containing DEA, exposure adds up. The same goes for children, whose developing bodies handle chemicals differently.
Safer Alternatives and Solutions
It only takes a walk down any store aisle to notice the growing number of “DEA-free” labels. Clean beauty advocates pushed manufacturers to swap out DEA for less controversial surfactants and foam boosters, many derived from plants. Many companies have dropped DEA due to shifts in consumer preferences. This trend addresses lingering concerns, even if science hasn’t declared absolute danger.
If you want to avoid DEA, check the ingredient list. Skip products with diethanolamine, cocamide DEA, or lauramide DEA. Sharing information and reading studies helps move manufacturers toward safer choices.
Health and safety often come down to asking questions—what am I using, how often, and what do credible experts say? Sticking with products from reputable brands with transparent ingredient lists helps keep your health in your own hands.
How should diethanolamine be stored and handled?
Real Risks Call for Real Solutions
Anyone who’s worked with chemicals like diethanolamine knows spills cause headaches far beyond a mess on the factory floor. I once managed a warehouse where a misunderstanding about loose caps ended up costing my crew a full afternoon. That situation served as a wake-up call. Diethanolamine isn’t only greasy and annoying—sloppy management can trigger everything from skin irritation to serious respiratory harm.
Why Storage Standards Matter
Diethanolamine, or DEA, pops up everywhere: shampoos, coolants, detergents. It remains liquid at room temperature and absorbs water fast from the air. This trait creates long-term headaches for container choice. Storing DEA in loose plastic barrels, for example, often leads to container decay and leaks. Polyethylene and stainless steel provide longer-lasting barriers. A well-sealed drum, away from sunlight and direct heat, keeps the chemical’s quality intact and helps protect anyone nearby.
If temperature control seems like an added hassle, it really isn’t. Keep DEA between 20 and 40°C. Lower storage temperatures invite crystallization and blockages in pipes. Leaving it exposed to warmth or sun can degrade product quality faster than expected—and make cleanup more toxic if things go sideways. Moisture sneaks in quickly if barrels sit open in humid rooms. Even a single forgotten lid can ruin the batch and threaten worker safety.
Handling: More Than Just Gloves and Goggles
DEA gets a reputation for “mild” effects, but even small spills demand care. It absorbs directly into skin and eyes, burns throat tissue on inhalation, and can linger on clothes. Nitrile gloves and well-fitted splash goggles become non-negotiable in places I’ve worked. Keep emergency rinsing stations nearby—don’t bank on distant sinks. The stench alone in case of a spill can clear a room in seconds, long before you see visible damage.
Spills should never wait. Workers need clear clean-up protocols—absorbent pads and neutralizers must sit within arm’s reach, not locked away on another floor. General-purpose absorbents rarely handle DEA very well. Instead, go for those rated for organic chemicals, and replace them every quarter no matter what the inventory numbers say.
Transport Hazards: Learning from Mistakes
Trucking DEA across distances takes teamwork between shippers, drivers, and onsite staff. I’ve seen delivery drivers try shortcuts, rolling barrels instead of using forklifts. That wallops the bung and starts tiny leaks. Putting trust in training, not just obvious hazard labels, avoids costly insurance calls and hospital visits. Written logs help catch problems before they snowball: leaking seals, expired materials, or tanks that look fine during daylight but show rust under closer inspection.
Transportation always brings the risk of mixed loads. Don’t let DEA ride with oxidizers or acids. Cross-contamination fires up chemical reactions nobody wants to see. It’s all too easy for a mislabeled drum to slip onto a shipment.
Systems That Protect People First
Facilities that get storage and handling right do one thing differently—they treat safety routines like regular chores. Training keeps every handler sharper, even years after their first day. Investing in sealed drum pumps, automated fill lines, and regular medical checkups pays off. Injuries and contamination fall. Real-time air monitoring systems, with alerts for leaks, add a layer of protection that old-school inspection routines can’t match.
Safe storage and careful handling of diethanolamine makes the difference between smooth operations and accidents that haunt a team for years. It remains as much about paying attention to the little details—the right barrels, the well-placed goggles, the honest housekeeping logs—as about memorizing technical specs. Honoring these daily habits keeps workers healthy, protects the business, and keeps communities safe from drama and disaster alike.
What are common applications of diethanolamine in industry?
Cleaning Up: More Than a Simple Ingredient
Let’s talk about diethanolamine, often called DEA. It might sound like a mouthful, but you’ll find it behind the scenes in places you don’t expect. Take household cleaners or shampoos—DEA acts as a surfactant here, which means it helps water mix with oil and dirt. Think about washing greasy pans or scrubbing spilled soup off the counter. DEA breaks that stubborn barrier, making grime easier to rinse away. The same goes for foaming in shampoos—if you’ve ever wondered how those suds build up when you’re scrubbing your hair, DEA plays a key role.
Textile Industry: Getting Fabrics Just Right
Walk through any clothing store, and you’re seeing the results of a process that relies on chemistry. DEA helps soften fabric when mills finish cotton, denim, or blends. It loosens natural oils and waxes from fibers, so dye sticks better and colors come out vibrant. Factories value DEA for keeping equipment clean, too. In my time working with textile clients, I saw how essential it is for producing those softer shirts and vibrant colors. Without these processes, clothes would feel rougher and look duller straight from the loom.
Gas Treatment: Cleaning the Air We Breathe
DEA doesn’t just help in homes and garment plants; gas companies use it for sweetening natural gas. Before energy can flow through pipelines, refineries need to strip unwanted gases like hydrogen sulfide and carbon dioxide. DEA scrubs these out, which means fewer emissions escape and the gas that ends up heating homes burns cleaner. If you’ve ever smelled “rotten eggs” near a gas leak, that’s hydrogen sulfide—removing that isn’t just about comfort, it protects health and meets regulations. Modern refinery engineers rely on DEA for this workhorse task every day.
Metalworking Fluids: Extending Machine Life
Factories cutting, stamping, or grinding metal run into sticky problems—literally. Metal flakes and oils can gum up valuable machinery. Adding DEA to lubricants keeps things running smoother by stopping oils and bits from separating or caking onto surfaces. The mix creates a stable emulsion, which shields machine parts from rust and keeps operations moving. In shops where expensive lathes or presses grind away for hours, recipes for cutting fluids often call for DEA to prevent breakdowns and expensive downtime.
Cosmetics and Personal Care: The Gentle Side
DEA also lands in lotions, creams, and makeup. Manufacturers add it to balance acidity and improve texture. It helps deliver that silky, luxurious feel in moisturizers and keeps creams from separating in the jar. A neighbor who worked at a cosmetics plant once told me that small tweaks with compounds like DEA can mean the difference between a product people love and one they leave on the shelf.
Risks and Safer Practices
DEA comes with baggage. Researchers found that repeated exposure, especially at higher levels, can irritate skin or eyes. Some studies linked DEA derivatives to health risks, especially if used carelessly. The European Commission and American regulators stepped in with tougher labeling and limits, especially in personal care goods. Relying on quality control, using better alternatives in sensitive products, and following workplace safety protocols are simple ways to keep risks low. As new regulations come out, companies look for greener, safer substitutes where possible. Everybody deserves a clean floor, vivid clothes, and safe products—without health trade-offs.
What safety precautions should be taken when using diethanolamine?
Why Diethanolamine Matters for Workplace Safety
Diethanolamine, often called DEA, pops up in all sorts of workplaces. Cleaners, cutting fluids, even some cosmetics feature this chemical. Its flexibility as an emulsifier and corrosion inhibitor keeps operations smooth, but this same quality brings risks—both short-term and after years of handling it. My time on industrial work floors has made it clear: DEA is not an ingredient anyone should shrug off as harmless.
Understanding the Risks
When DEA touches skin, it irritates—leading to redness, itching, and sometimes rash. Splash some into your eye, and you’ll remember the sting for a long time. Breathing in vapors, especially during mixing or spraying, can cause sore throat and cough. The trickier aspect? Studies, including those by the National Toxicology Program, show that repeated contact links to liver and kidney trouble in animals, and possibly to human cancer risks over years. The European Union lists DEA as a substance of very high concern, and the American Conference of Governmental Industrial Hygienists sets a threshold limit value at just one mg/m³ for vapor and aerosol.
Personal Protection Starts with Basics
I never walk into an area where DEA is handled without checking for splash goggles and chemical-resistant gloves—nitrile and neoprene do the job. Regular work gloves don’t cut it. Apron and long sleeves come next if splashes could happen. Good lab practice keeps open bottles to a minimum, and transfer work in fume hoods goes a long way to keep vapor out of the breathing zone. I’ve seen burns and red eyes from simple mistakes like not wearing goggles or removing gloves too soon after cleaning.
Ventilation and Air Quality
Stale air makes working with DEA more risky. Local exhaust, such as fume hoods and at-source ventilation, helps stop vapor buildup before it starts. General ventilation fans aren’t enough; air should get pulled away from faces and out of the room. I’ve learned to check airflow by feeling with my hand or using a simple piece of paper. If it doesn’t move, neither should DEA containers.
Handling Spills and Emergencies
A spill kit, stocked and ready, isn’t optional. I go through the kit’s location and use whenever onboarding new colleagues. If DEA hits the floor, absorb it with inert material like sand or vermiculite. Never touch a chemical spill bare-handed. For eye or skin contact, the eyewash and shower must flow for 15 minutes. I make a habit of checking emergency stations monthly—they need to work before an accident, not after. Reporting and documenting every incident, no matter how minor, builds good habits and enforces accountability.
Storage Makes a Difference
Keep DEA drums and bottles in tight, well-labeled containers. Secure the area, away from heat, strong acids, and oxidizers. I’ve seen what happens when leaks mix with incompatible chemicals—unpleasant fumes, ruined floors, or worse. Double-checking seals and keeping containers closed between uses saves headaches and prevents danger.
Training and Culture Change the Game
No rule or label replaces real training. I’ve found short, repeated training sessions stick more than annual PowerPoints. Sharing stories about real-life accidents—without blame—makes people care about their own safety. Employees should quiz each other and walk through drills. It’s not fearmongering, it’s responsibility.
Improving Safety Day by Day
No company reaches perfection overnight. Setting up clear guidelines, keeping PPE handy, and encouraging open dialogue about safety concerns help build a workplace that respects both the usefulness and danger of chemicals like DEA. Safe handling depends on personal discipline, good ventilation, emergency readiness, and a culture that values every person’s health above speed or convenience. From my years in the field, skipping safety steps with DEA always backfires. Consistent respect for this chemical gets everyone home in the same shape they arrived.
| Names | |
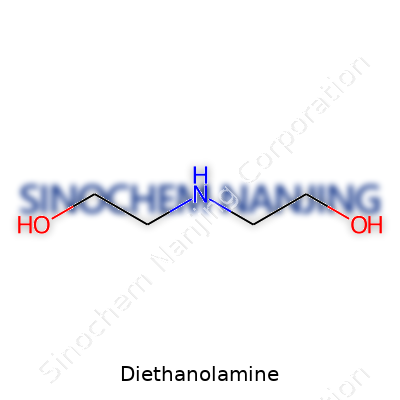
| Preferred IUPAC name | 2,2'-Iminodiethanol |
| Other names |
DEA di(2-hydroxyethyl)amine N,N-bis(2-hydroxyethyl)amine 2,2’-iminodiethanol |
| Pronunciation | /daɪ.iˌθɛn.əˈləʊ.ə.miːn/ |
| Identifiers | |
| CAS Number | 111-42-2 |
| Beilstein Reference | 1718736 |
| ChEBI | CHEBI:18036 |
| ChEMBL | CHEMBL715 |
| ChemSpider | 5737 |
| DrugBank | DB00173 |
| ECHA InfoCard | 03b3c465-ec36-4b93-80e3-98d8d80fb41b |
| EC Number | 205-218-2 |
| Gmelin Reference | 878 |
| KEGG | C00750 |
| MeSH | D02.241.081.569.262 |
| PubChem CID | 8030 |
| RTECS number | KL2975000 |
| UNII | J3R183A4U1 |
| UN number | UN 2055 |
| Properties | |
| Chemical formula | C4H11NO2 |
| Molar mass | 105.14 g/mol |
| Appearance | Clear, viscous, hygroscopic liquid |
| Odor | Ammonia-like odor |
| Density | 1.09 g/cm³ |
| Solubility in water | miscible |
| log P | -2.18 |
| Vapor pressure | 0.01 mmHg (20°C) |
| Acidity (pKa) | 8.88 |
| Basicity (pKb) | 1.43 |
| Magnetic susceptibility (χ) | -7.7×10⁻⁶ |
| Refractive index (nD) | 1.474 |
| Viscosity | 300 mPa·s (25 °C) |
| Dipole moment | 3.71 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 234.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -726.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3221.9 kJ/mol |
| Pharmacology | |
| ATC code | C04AX19 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause allergic skin reaction, suspected of causing cancer. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS05,GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H318, H373 |
| Precautionary statements | P260, P264, P280, P301+P312, P305+P351+P338, P308+P313, P405, P501 |
| Flash point | 215°C (closed cup) |
| Autoignition temperature | 363°C |
| Lethal dose or concentration | LD50 (oral, rat): 710 mg/kg |
| LD50 (median dose) | 2,200 mg/kg (oral, rat) |
| NIOSH | TN3500000 |
| PEL (Permissible) | 10 mg/m3 |
| REL (Recommended) | 50 mg/m³ |
| IDLH (Immediate danger) | 300 mg/m3 |
| Related compounds | |
| Related compounds |
Ethanolamine Triethanolamine Methyldiethanolamine Morpholine Diisopropanolamine |

